Abstract
Background
Functional non-retentive fecal incontinence (FNRFI) is a psychologically upsetting and embarrassing issue and affects children's quality of life negatively.
Aim of this study
Evaluation of the short and long-term effect of Bilateral transcutaneous posterior tibial nerve stimulation (BTPTNS) in the treatment of FNRFI in children and its impact on the quality of life (QoL). Methodology: The current randomized controlled study included 94 Children with FNRFI who were randomly allocated into two equal groups. Group A received BTPTNS and Group B Received Sham BTPTNS. Follow-up was planned for 24 months for manometric findings, incontinence score, Incontinence episodes, and the QoL.
Results
The incontinence score was significantly decreases in Group A more than what was reported in Group B at 6, 12, 24 months follow up
Conclusion
BTPTNS can be a good modality in the treatment of FNRFI with favorable long-term maintenance of its effect together with a remarkable positive impact on all domains of QoL.
Keywords
Introduction
The international continence society classifies anal incontinence as any involuntary passage of stool and/or gas and divides it into two types, Fecal incontinence (FI) and gas incontinence at least once/month for two successive months.1,2 FI frequency ranges between .8% and 7.8% while
Intact muscles of the pelvic floor, the anal sphincter, local sensory and motor nerves, as well as cortical and spinal connections all contribute to the maintenance of Fecal continence. 6 Fecal incontinence can result from dysfunction of any of these structures so it is a more complicated and multifaceted illness. 2
Functional FI is classified into retentive (associated with stool retention and “overflow”) and non-retentive FI (FNRFI) based on Rome IV criteria and underlying pathophysiological mechanisms are still unclear. 7 the Rome III classification defines FNRFI as FI in a kid with a mental age > 4 years and without evidence of a metabolic, anatomical, or inflammatory etiology. 7
The diagnosis of FNRFI is made based on the clinical assessment. The only test that can aid in diagnosis is the Colonic Transit Time (CTT) measurement. And for the specific entity of FNRFI to be confirmed, CTT readings must be normal. 8
It is crucial to inform parents and children that the course of treatment may take a long time with variable responses. Child must informed about the value of using the restroom on a regular basis.2,9
Posterior tibial nerve stimulation (PTNS) for FI was initially reported on by Shafik and colleagues, 10 who also reported improved continence following a 4-week course. Percutaneous PTNS (PPTNS) and transcutaneous PTNS (TPTNS) were found to be effective treatments for FI in further investigations with a reduction in incontinence episode of 50%-80%. 11 Due to the stimulating electrode's proximity in (PPTNS), It exerted a larger effect than TPTNS. 12 TPTNS, however, is preferable PPTNS because to the worry that needle insertion could result in a stimulation strong enough to have a neuromodulatory impact. 13
However the grey area about the long term effect of TPTNS has motivated the authors to conduct this study.
Patients and Methods
Study Design
This prospective randomized controlled study was conducted at the Colorectal Surgery Unit, Benha University Egypt throughout the period from June 2020 December 2022 and was approved by ethical and research committees Benha Univerisity. A written informed consent was obtained from parents of all included children in the study Clinical trial registry at: https://clinicaltrials.gov/ct2/show/nct04478799.
The current study included 102 children among them 94 underwent randomization and allocation into one of 2 groups. Group A (n = 47) underwent Bilateral TPTNS and group B, Sham TPTNS (n = 47) and only 72 children completed 24 months follow up and analysis 41 in group A and 31 in group B. Inclusion criterion was children presented with FNRFI (ie FI and normal defecation frequency, bowel habits and stool consistency). The children were randomly divided into two equal groups. Group A, Received Bilateral TPTNS and group B, Sham TPTNS. Exclusion criteria were Children <4 years, Traumatic sphincter injury, anorectal malformation or spinal diseases causing FI.
Procedure
Before Consideration of enrollment of eligible patients, history about bowel habits and severity of incontinence was obtained. A physical examination together with Transanal ultrasonography using ( ) was performed to exclude sphincteric injury. CTT was the cornerstone in establishing the diagnosis of FNRFI. Radio opaque marker test with serial abdominal X-rays to determine both segmental and total colonic transit time was done.
14
) was performed to exclude sphincteric injury. CTT was the cornerstone in establishing the diagnosis of FNRFI. Radio opaque marker test with serial abdominal X-rays to determine both segmental and total colonic transit time was done.
14
Vaizey score, 15 ranging from (0 (perfect continence) with a maximum score of 24 indicating total incontinence) was used to document the initial incontinence score for all eligible children.
Baseline manometric assessment was done for all included children to determine resting pressure, squeeze pressure, first sensation, first urge, and intense urge using high resolution manometry ( ) with 24-channel water-perfused catheter with latex balloon following the same protocol describe by Banasiuk M et al.
16
) with 24-channel water-perfused catheter with latex balloon following the same protocol describe by Banasiuk M et al.
16
Procedure
Group A Bilateral Transcutaneous Posterior Tibial Nerve Stimulation
Using the method outlined in 2006 by Queralto et al. 17 1.5 inches above the medial malleolus, a positive auto-adhesive electrode was placed. In close proximity below the same malleolus, a second negative electrode was placed. Connected to each electrodes was an electrical stimulation device (EMS physio Ltd, OX129 F, England). The intensity of the low frequency (10 Amp) current delivered by this device was gradually increased until the big toe flexed or the other toes spread out in a fan-like pattern. The intensity was then reduced again to (10 AMP) in order to prevent the toes' motor response. For three months, the session was carried out for 20 to 30 minutes twice weekly.
Group B Sham Transcutaneous Posterior Tibial Nerve Stimulation
Patients also had twice-weekly 20-30-min sessions for 3 months. The same design of the electrodes In Group A was applied with a brief switch on for 30 s for induction of minor electrical sensation and then turned off for the rest of the session.
For both groups Kegal exercises, dietetic regulation, and local hygiene to the perianal skin were applied through out the whole course. 2
Randomization
Random Allocation Software 1.0, 2011 was used for block randomization by an independent investigator.
Evaluation and Followup
Primary End Point
It was a reduction in the number of incontinence episodes and an improvement in the QOL.
Secondary End Point
The secondary outcome was in the form of change in manometric findings.
The impact on the QoL was also assessed through fecal incontinence quality of life (FIQL) scale. 10 FIQL was modified where two items related to sexuality were removed and the word depressed was replaced with sad. So, the modified questionnaire is composed of 27 questions for four main domains: lifestyle, behavior, depression, and embarrassment. 11
According to the clinical response after 3 months, both groups were classified into four groups: Group A, fully continent Group B, decrease in incontinence episodes of more than 75% Group C, decrease in incontinence episodes of less than 75% Group D, no improvement or deterioration than before therapy.
Group D was excluded from the long-term follow-up as it failed to achieve the primary outcome goals. The remaining three groups were subjected to the long-term follow-up after 12 and 24 months from the initiation of the treatment program. Follow-up was done either during a clinical visit or by telephone. Successful treatment was considered if there were less than two incontinence episodes/month.
Statistical Analysis
The 1ry outcome of this study, incontinence episodes, was used to determine the sample size. Using the G*power 3.1 programme (Universities, Dusseldorf, Germany), a sample size of 31 at least in each group was taken into consideration with a power of 80%, P value of .05, and an effect size of .7.
The statistical analysis was performed using SPSS Statistics version 20. To compare measured variables in the 2 groups and during various time periods, a two-way mixed ANOVA was used. The ANOVA test was applied to the participant's demographic information. Mean and standard deviation was used to convey numerical data, whereas number and percentage were used to present nominal data. The threshold for significance was fixed at P ≤ .05.
Results
In the current study 102 children with FNRFI were tested for eligibility. Among them 94 children were randomly divided in to two equal groups (A and B). Group A underwent (n = 47) Bilateral TPTNS and group B, Sham TPTNS (n = 47) Figure 1. Consort flow chart.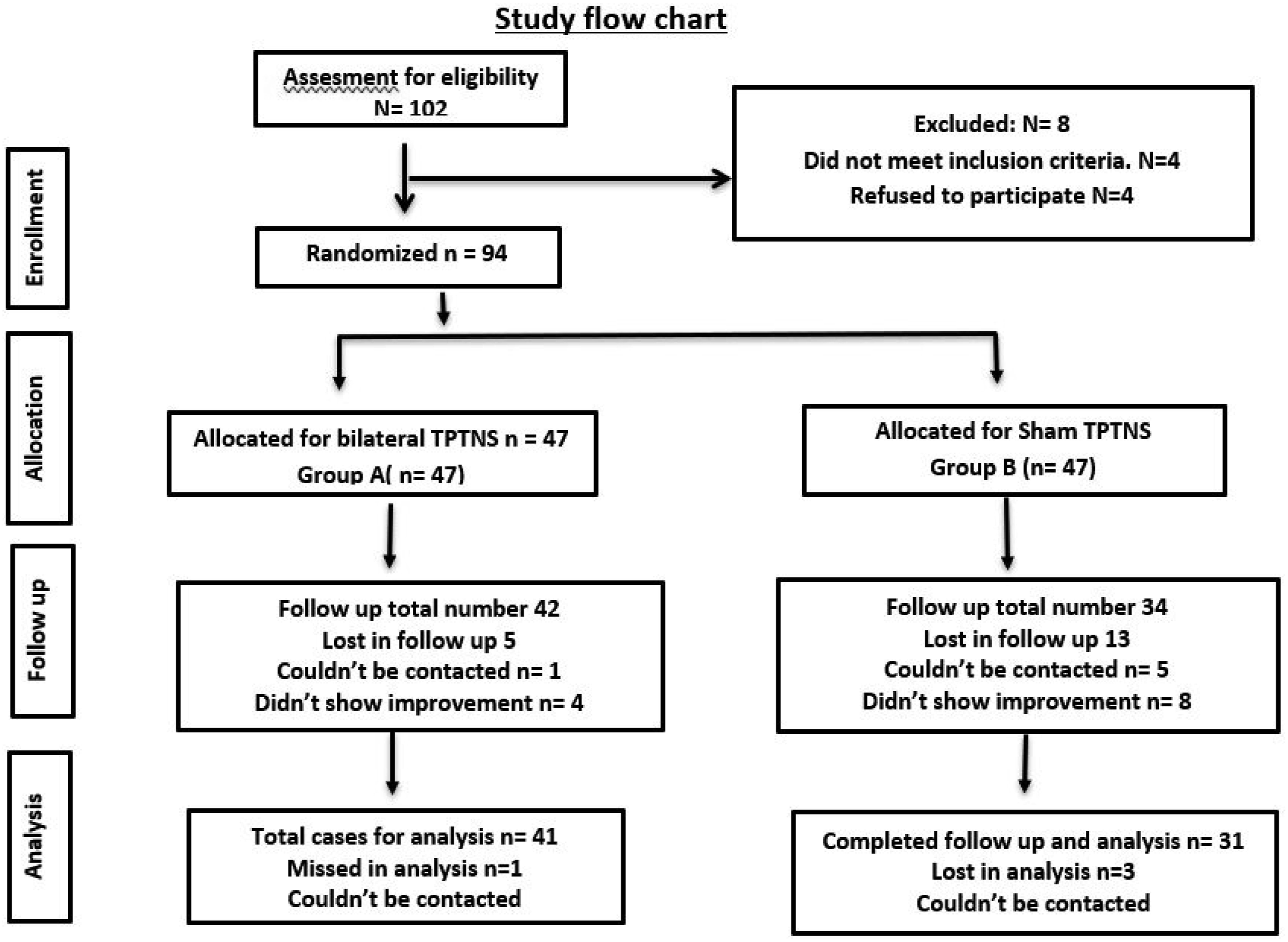
Socio-Demographic Data of the Two Groups.

M: mean, SD: stander deviation, n: number.
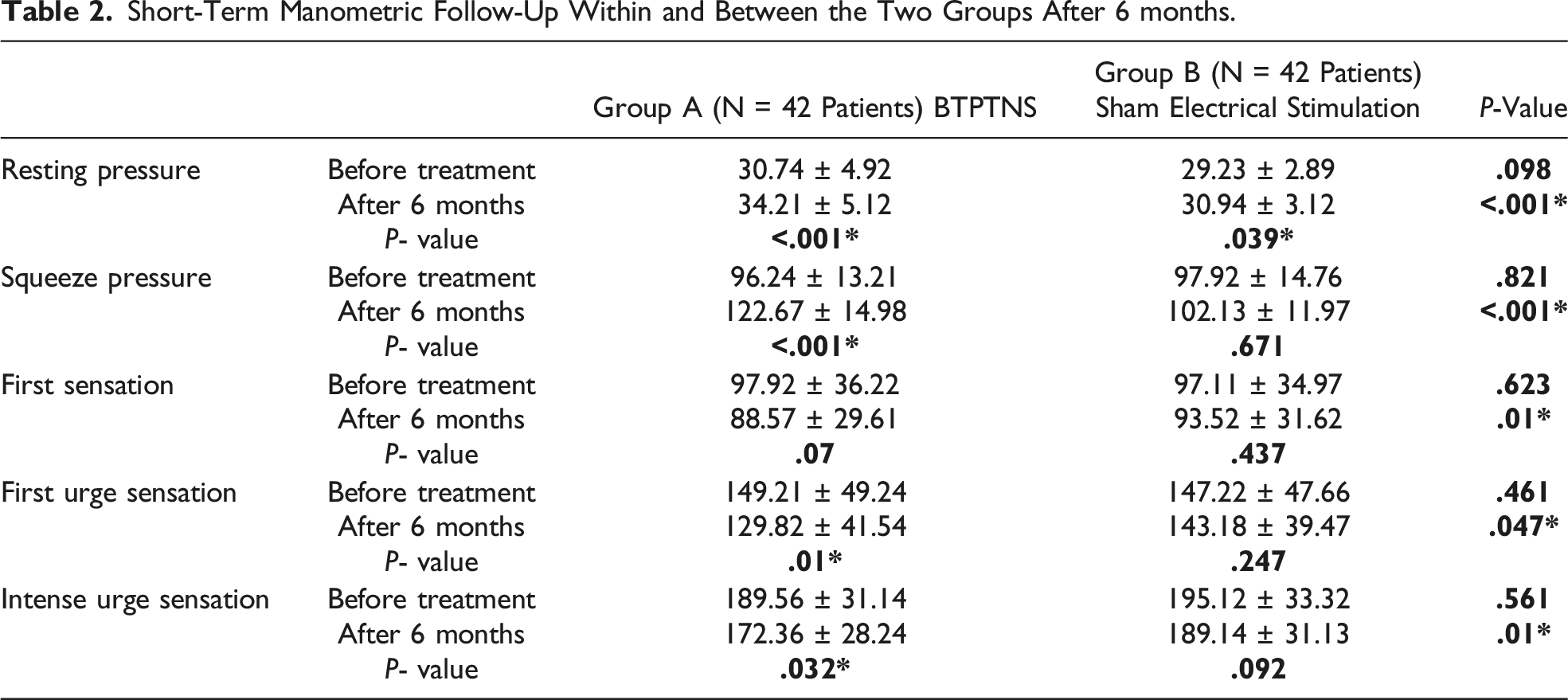
Short-Term Manometric Follow-Up Within and Between the Two Groups After 6 months.
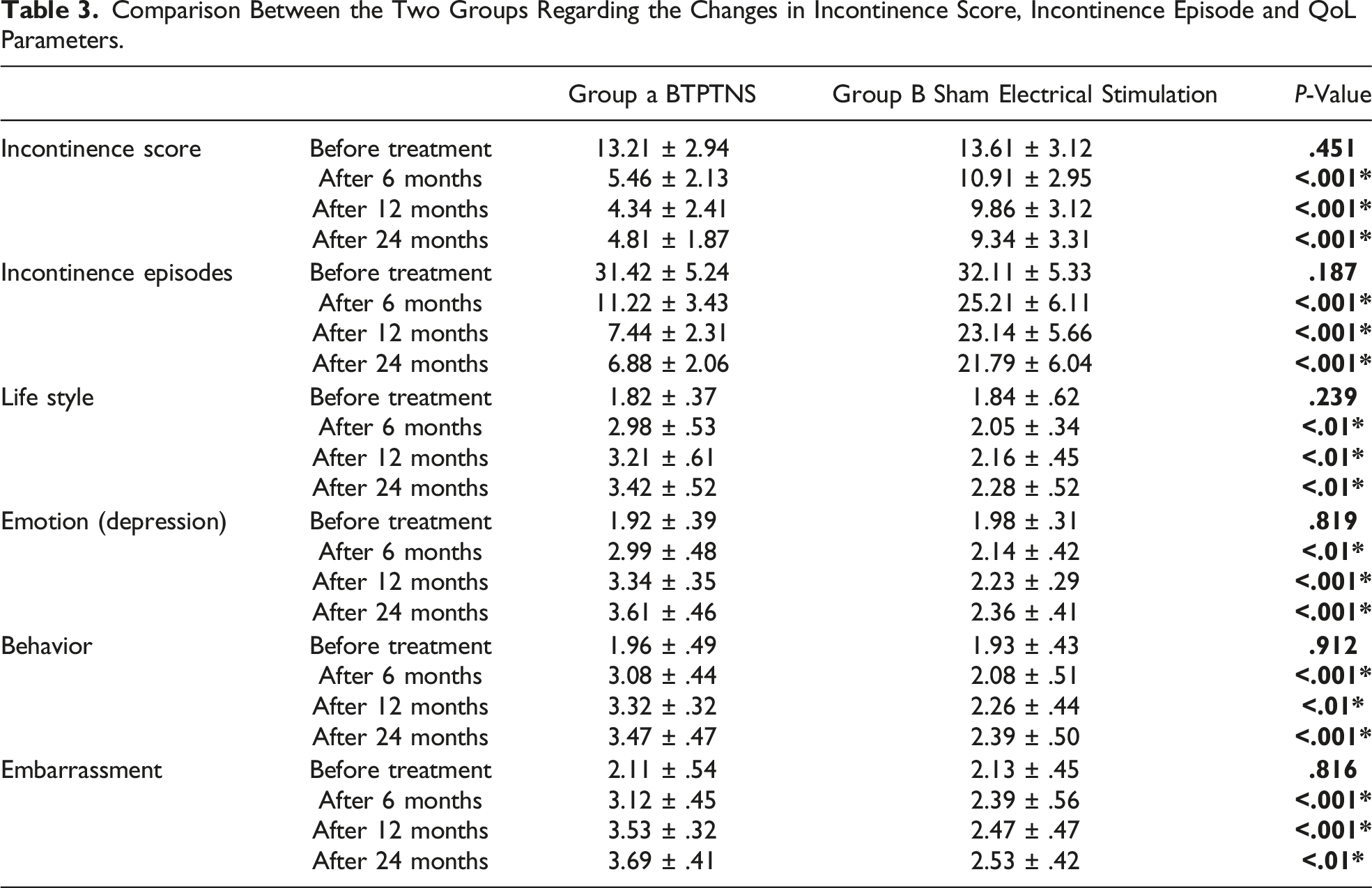
Comparison Between the Two Groups Regarding the Changes in Incontinence Score, Incontinence Episode and QoL Parameters.
Manometric findings
the squeeze and resting pressures were significantly increased in Group A as well as significant improvement of rectal sensation more than group B after 6 months follow up. Table 2.
In Group A both resting and squeeze pressure were statistically increased (P
Incontinence Score and Incontinence Episodes
The incontinence score significantly decreased in group A more than what was reported in Group B at 6, 12, 24 months follow up (P < .001*). Table 3.
Mean Difference and 95% Confidence Interval and Pairwise Comparisons Values of the Incontinence Score, Incontinence Episodes, and Quality of Life Both Groups.
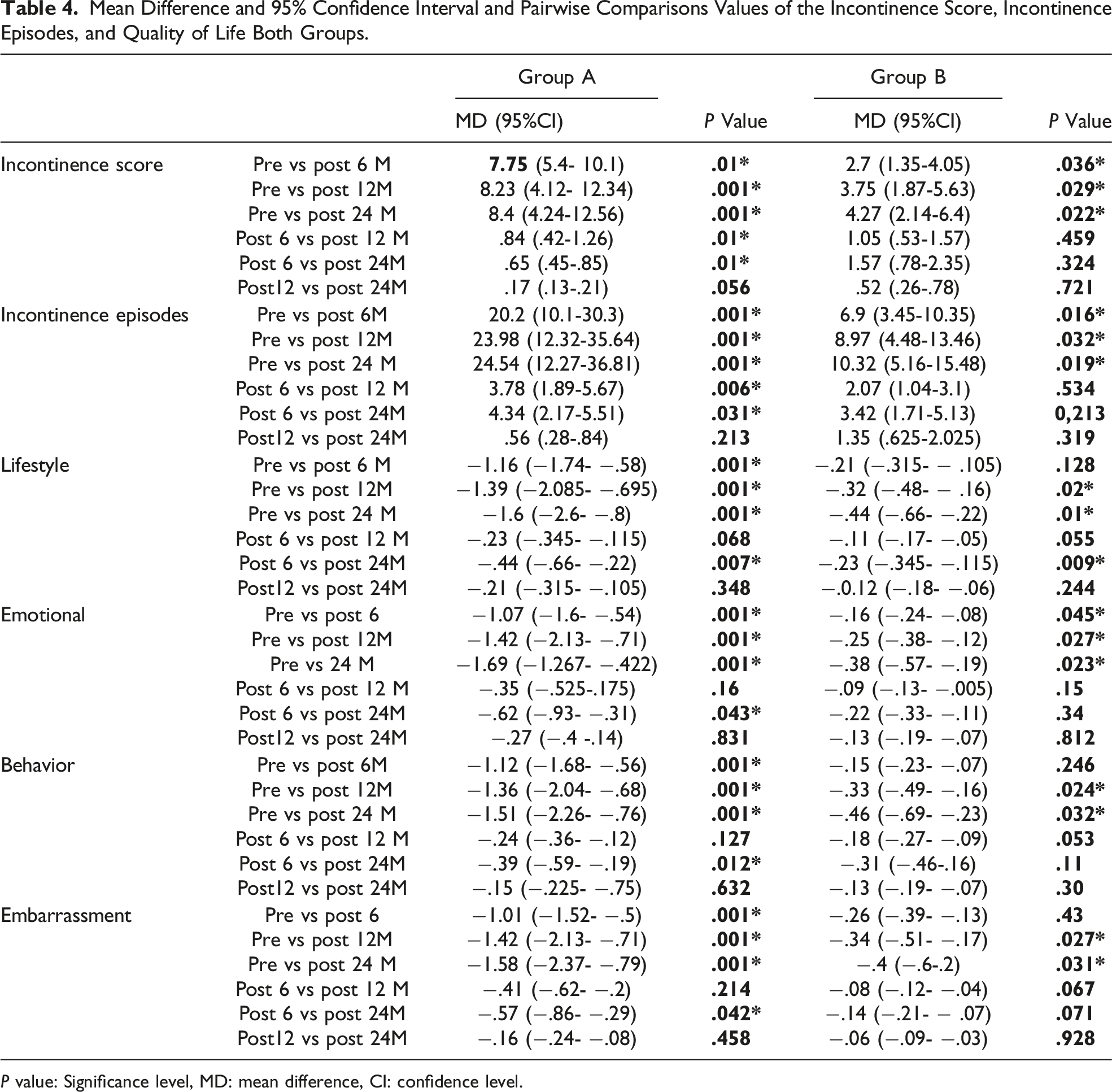
P value: Significance level, MD: mean difference, CI: confidence level.
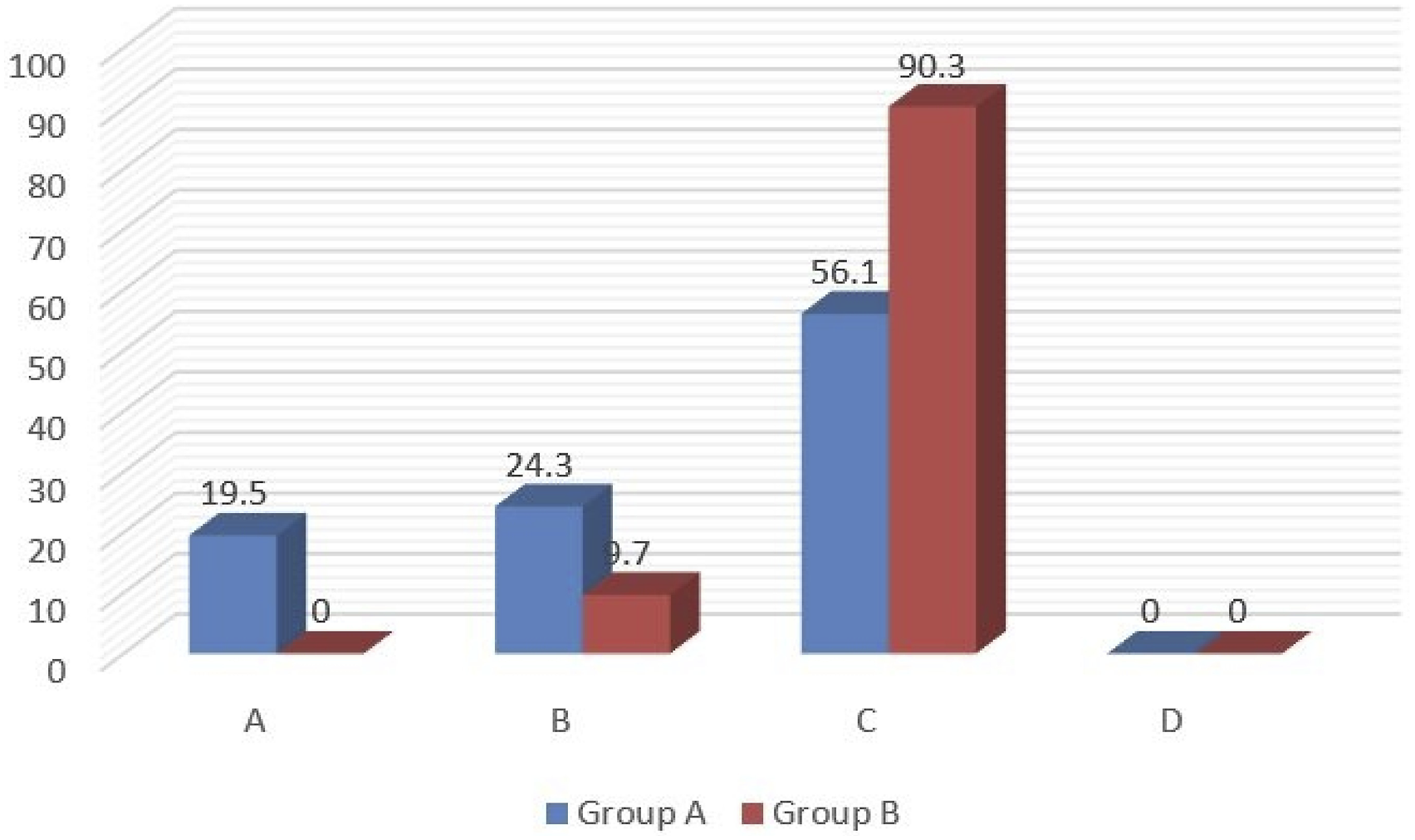
In group A 53.2% of the included children who received BTPTNS showed a decrease in the incontinence episodes more than 75% and among them, 23.4% were fully continent. The effect of BTPTNS after 12 and 24 months was maintained in 48.75% and 43.8% of children respectively who showed a decrease in the incontinence episodes of more than 75% and among them, 21.95% and 19.5% were fully continent after 12 and 24 months respectively. Figures 2 and 4. Short- and long-term results of Group A according to clinical outcome.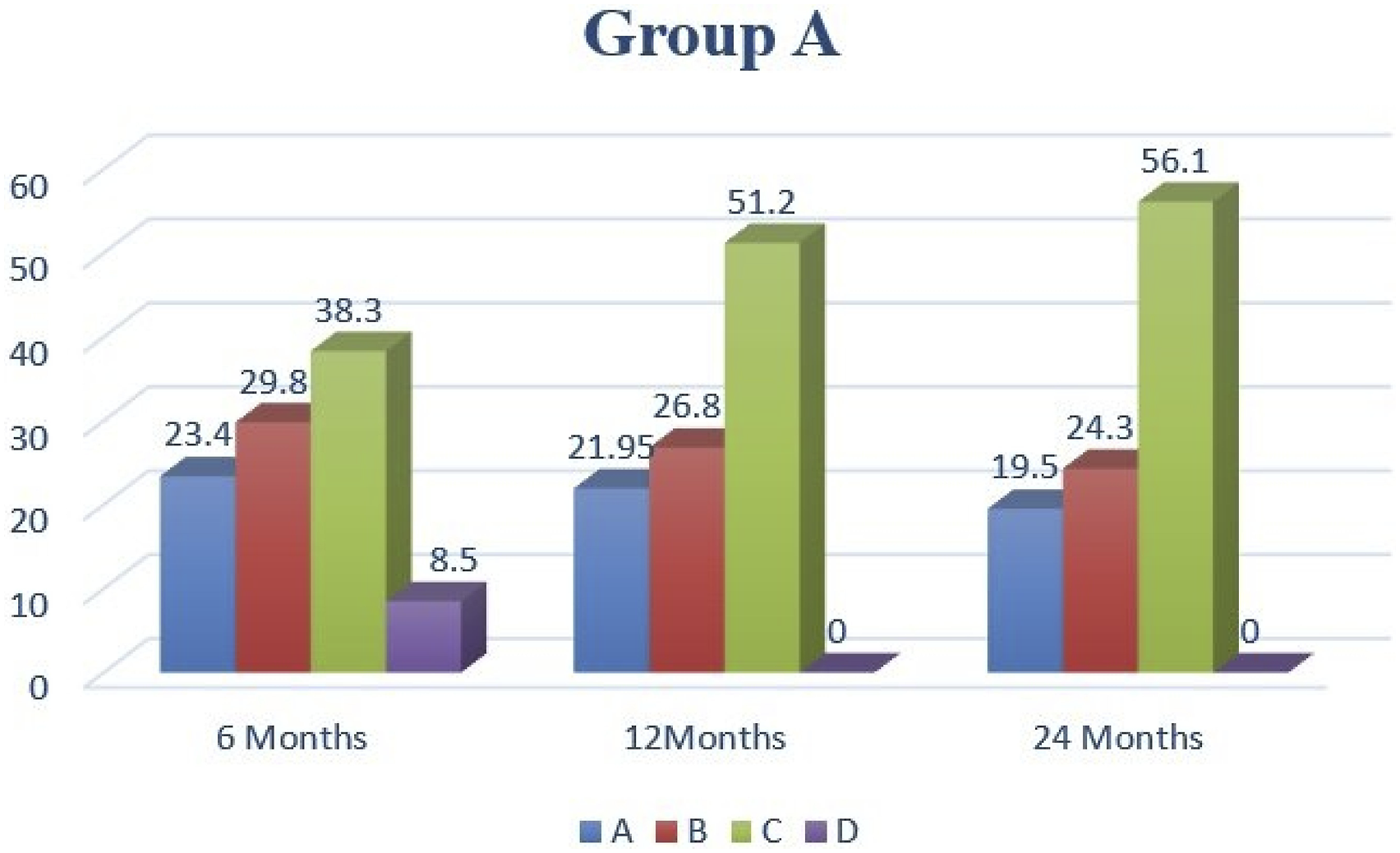
In Group B only 13% of the included children showed a decrease in the number of incontinence episodes by more than 75% after 12 months and 9.7% maintained effect after 24 months amd none of them was fully continent Figures 3 and 4. Short- and long-term results of Group B according to clinical outcome. Comparison of the clinical outcome in Group A and B by the end of study.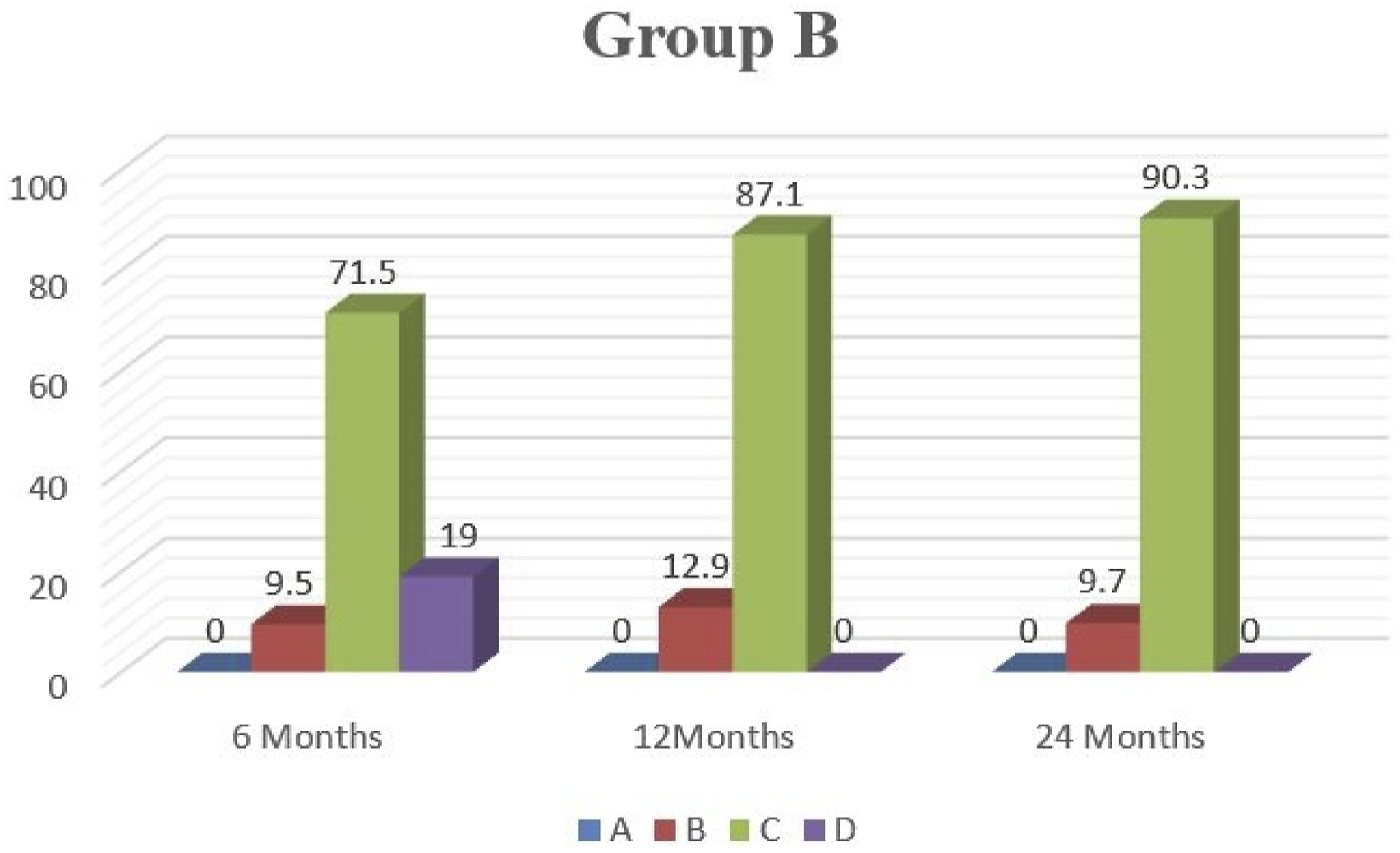
There was a stastically significant improvement in QoL domains at within Group A at 6, 12, 24 months follow-up when compared with the initial results. Tables 3 and 4.
Discussion
For many physicians, managing FNRFI in children had come to be seen as a grey area with many well-liked lines of treatment but with unexpected outcome. 18 Recent research has suggested electrical stimulation as an adjuvant therapy for FI, although the mechanism of electrical stimulation's beneficial effects on FNRFI needs more explanation. 19 The QOL of the affected child and family is reduced by FI with a bad impact on their social and personal lives.
In this study, there was a statistically significant increase in both resting and squeeze pressure, as well as the rectal sensation in children who received TPTNS more than those in group B who received Sham electrical stimulation and this can be explained by proper orientation about the pathophysiological changes associated with PTNS that will inhibit the preganglionic neurons that are in direct action with the sacral roots with overall up-regulation of the afferent rectal sensory perception and improved muscle function.
20
One main point in the outcome in treatment of FNRFI is the decrease in the FI severity throughout decrease the FI episodes and improvement of FI score. For the effectiveness of TPTNS, several studies21-25 reported statistically significant improvement of FI score after 3 months or after 6 months as reported by
For FI episodes,
Pairwise comparison between both groups in this study revealed a statistically significant decrease in the FI episodes and FI Score in children who received TPTNS in comparison to those who received Sham electrical stimulation and this is assumed to be due to the more improvement of the anal pressure and rectal sensation as previously reported in Group A and this comes with the results of
Several studies described the effectiveness of BTPTNS for short-term follow-up for 6 months23-25 and reported a statistically improved the quality of life domains and total score following a decrease in the Frequency of FI episode and this matched the results of the current study that reported that Lifestyle, Depression, Behavior and Embarrassment domains were significantly improved in both groups with much improvement in children received BTPTNS than those received Sham electrical stimulation and this simply can be explained by the more decrease in the FI episodes in Group A in comparison with Group B.
The assessment of QOL domains is one of the cornerstones In the current study and the lack of follow-up data in the literature about the long-term effect of BTPTNS was a great obstacle. To our knowledge, only
Although there was decay in the effectiveness of BTPTNS over time and the effectiveness was maintained in 49% and 44% at 12 and 24 months follow-up in comparison with the initial improvement that occurred in 53% of children in group A who reported a decrease in the FI episodes more than 75% but still significant improvement in the QOl domains matching the results of
Conclusion
TPTNS can be a Good Modality in Treatment of FNRFI in Children with Favorable Long-Term Maintenance of its Effect Together with a Remarkable Positive Impact on all Domains of QoL.
Limitations of the study
Long time for follow up was faced by obstacles in the commitment of the included children.
Recommendations
Combination TPTNS to Biofeedback in the treatment of FNRFI in children.
Footnotes
Acknowledgements
Sincere gratitude is expressed to Dr Mohammed Abuelnasr, the independent investigator in randomization
Author Contributions
Study conception and design:
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
