Abstract
Background
Endoscopic tattooing of colorectal lesions has been performed employing several markers. The indocyanine green (ICG) that uses near infrared fluorescence technology, has been recently adopted in laparoscopic colorectal cancer surgery. This study aims to systematically review the international literature to validate the ICG in laparoscopic colorectal surgery, in order to include the ICG in the therapeutic protocol.
Methods
Following AMSTAR 2 criteria, we performed a systematic review to evaluate the use of green indocyanine as a marker for preoperative endoscopic tattooing and for lymph nodes mapping. The study selection was conducted using the PubMed database from January 1989 to July 2022.
Results
We identified 25 eligible studies. 13 based on fluorescent tumor localization in laparoscopic colorectal surgery using ICG while 12 of them reported the lymphatic road mapping and sentinel node identification by ICG using a near-infrared camera system. One study analyzed both topics.
Conclusions
In laparoscopic colorectal cancer surgery indocyanine green can be used to localize fluorescent tumors and mapping fluorescence lymph node. The use of ICG appears to be a valid and safe technique that helps the surgeon to achieve a better oncological radicality. However, the protocols need to be clarified by further studies.
Introduction
Endoscopic tattooing of colorectal lesions has been performed employing several markers so far. A new one, the indocyanine green (ICG) that uses near infrared fluorescence technology, has been recently adopted in laparoscopic colorectal cancer surgery. ICG has different applications such as fluorescent tumor localization, lymph node mapping and intraoperative angiography.1,2 Few studies have shown that ICG is feasible and safe, easy to handle because readily available and supplied in sterile package. It does not cause complications unlike other tattooing agents such as India ink that has some collateral effects: focal peritonitis, inflammation, abscesses, adhesions and pseudo-tumor.3-9 In laparoscopic surgery the preoperative colonoscopy tattooing with ICG allows to identify the site of the lesion carefully due to the fluorescence property of the marker and the development of the surgical NIR camera system. Accurate tumor localization will enable an adequate margin resection.10,11 Unlike other endoscopic markers, the preoperative ICG tattooing allows to visualize lymphatic drainage by submucosal injection around the tumor under the near-infrared (NIR) imaging systems. An adequate fluorescence lymph node mapping (FLNM) supports the surgeon when visualizing the lymphatic pathway and while performing an accurate lymph node dissection to reach greater oncological radicality. 2 The usefulness of ICG in this clinical field needs to be clarified.
This study aims to review systematically the international literature to validate the use of ICG in laparoscopic colorectal surgery, in order to include the ICG technique in the therapeutic protocol of this disease.
Methods
Following current AMSTAR 2 criteria, we performed an extensive systematic review of the English literature to evaluate the use of green indocyanine as a marker for preoperative endoscopic tattooing and for lymph nodes mapping. The research and its study selection were conducted using the PubMed database and articles in English language were selected from January 1989 to July 2022. Further searches were conducted using Web of Science (WOS), SCOPUS, and Google Scholar. We made the research using the following terms: “green indocyanine colonoscopy”, “preoperative colonoscopy tattoo”, “green indocyanine tattoo”, “lymph node mapping“, “green indocyanine and colon cancer”. To avoid data duplication, we only considered the most recent papers in front of articles written by the same research group with overlapping study periods. Forty-three articles were selected, but 18 of them were excluded because they were representative of animal or cadaveric studies, case reports, reviews and editorials. We included 25 articles. The checklist of PRISMA-ScR (Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews) was performed. 12 For the purposes of our review, the inclusion criteria were as follows: green indocyanine as an endoscopic marker and its use in lesion localization and during lymph node mapping. The exclusion criteria were animal and cadaveric studies, case reports, reviews and editorials. Data were extracted by one author (M.V.), based on the model of the CHARMS Checklist (Critical Appraisal and Data Extraction for Systematic Reviews of Prediction Modelling Studies). The discordances were solved by discussion with all coauthors and consensus. The analysis of the collected data was carried out in a double-blind fashion by two authors (S.P. and A.L.).
Results
We identified 25 eligible studies published between January 1989 and July 2022, 13 based on fluorescent tumor localization in laparoscopic colorectal surgery using ICG while 12 of them reported the lymphatic road mapping and sentinel node identification by ICG using a near-infrared camera system. One study analyzed both topics.
2
These 25 studies were evaluated and analyzed. The selection process is shown in Figure 1. Prisma flow chart of literature search.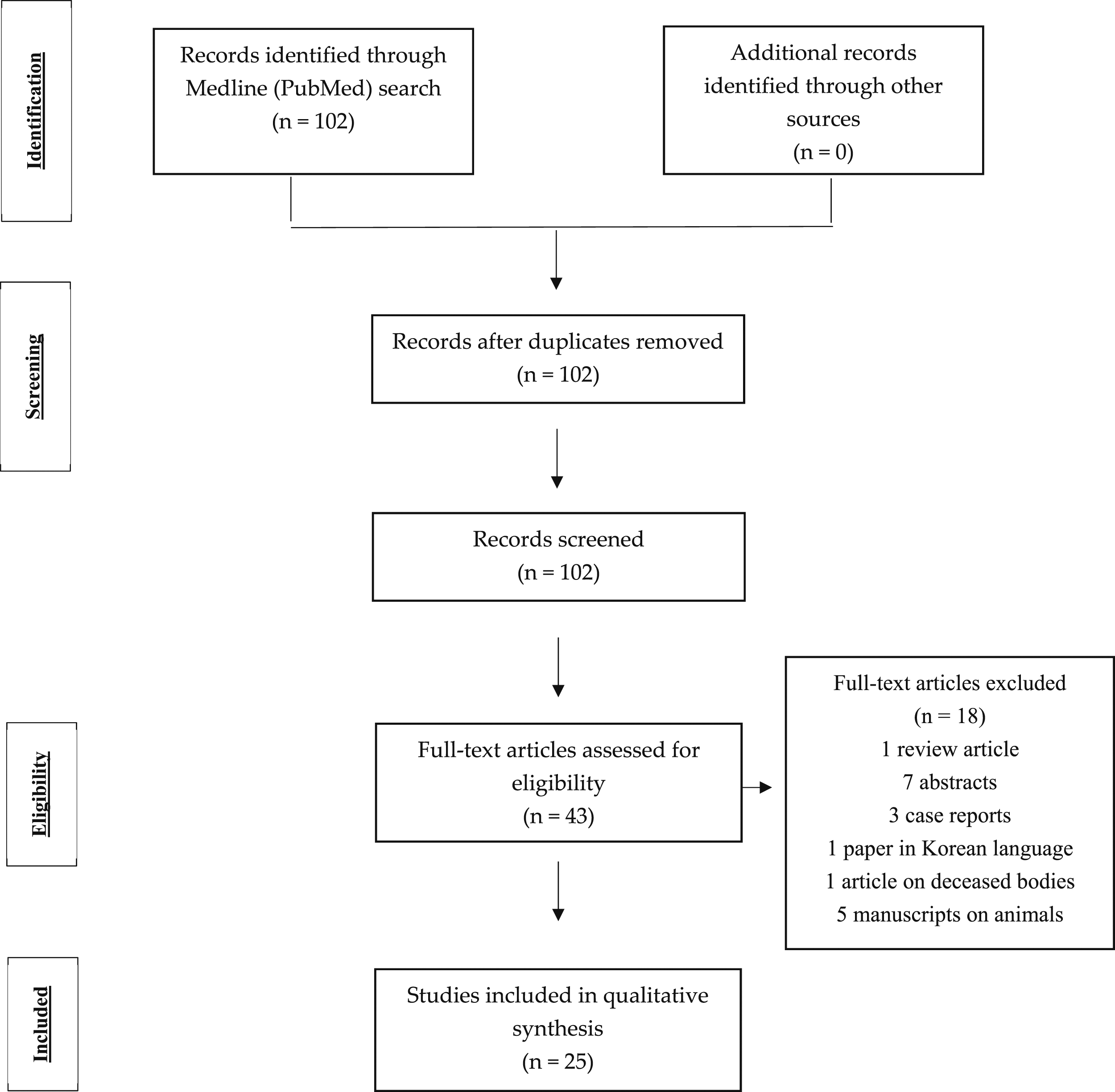
Indocyanine Tattooing
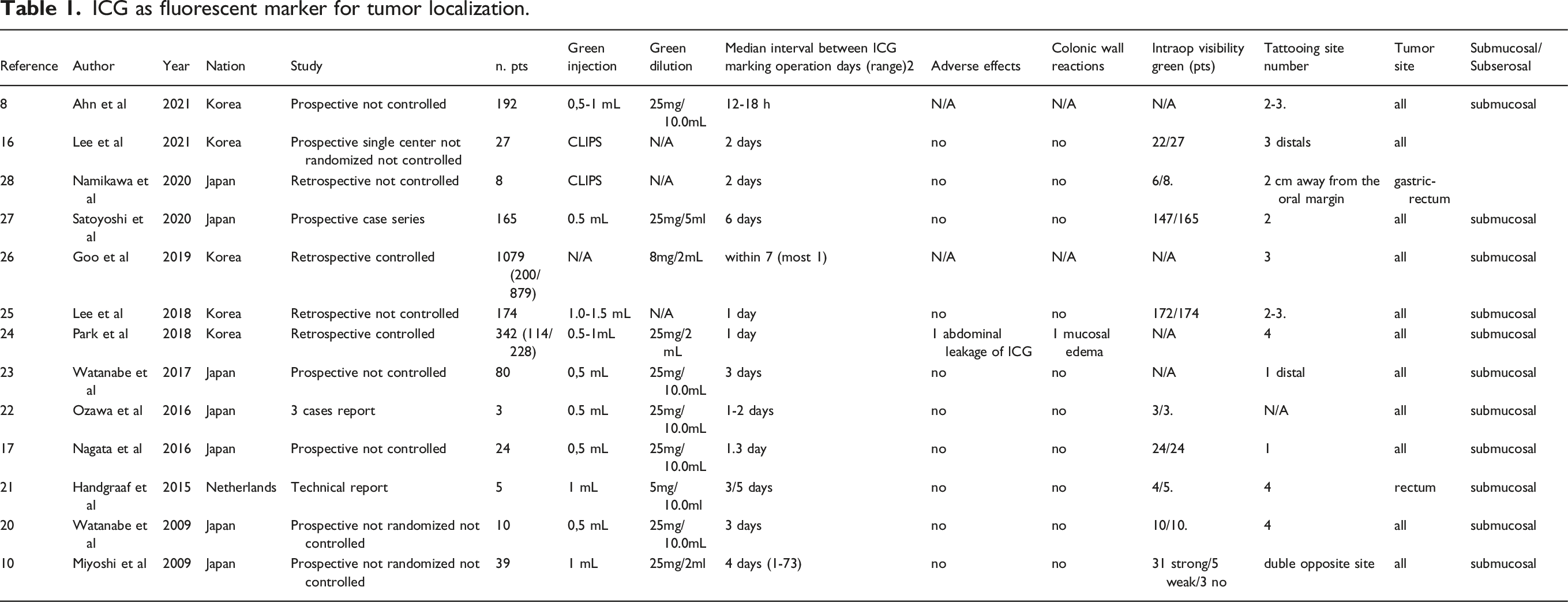
ICG as fluorescent marker for tumor localization.
Indocyanine Mapping
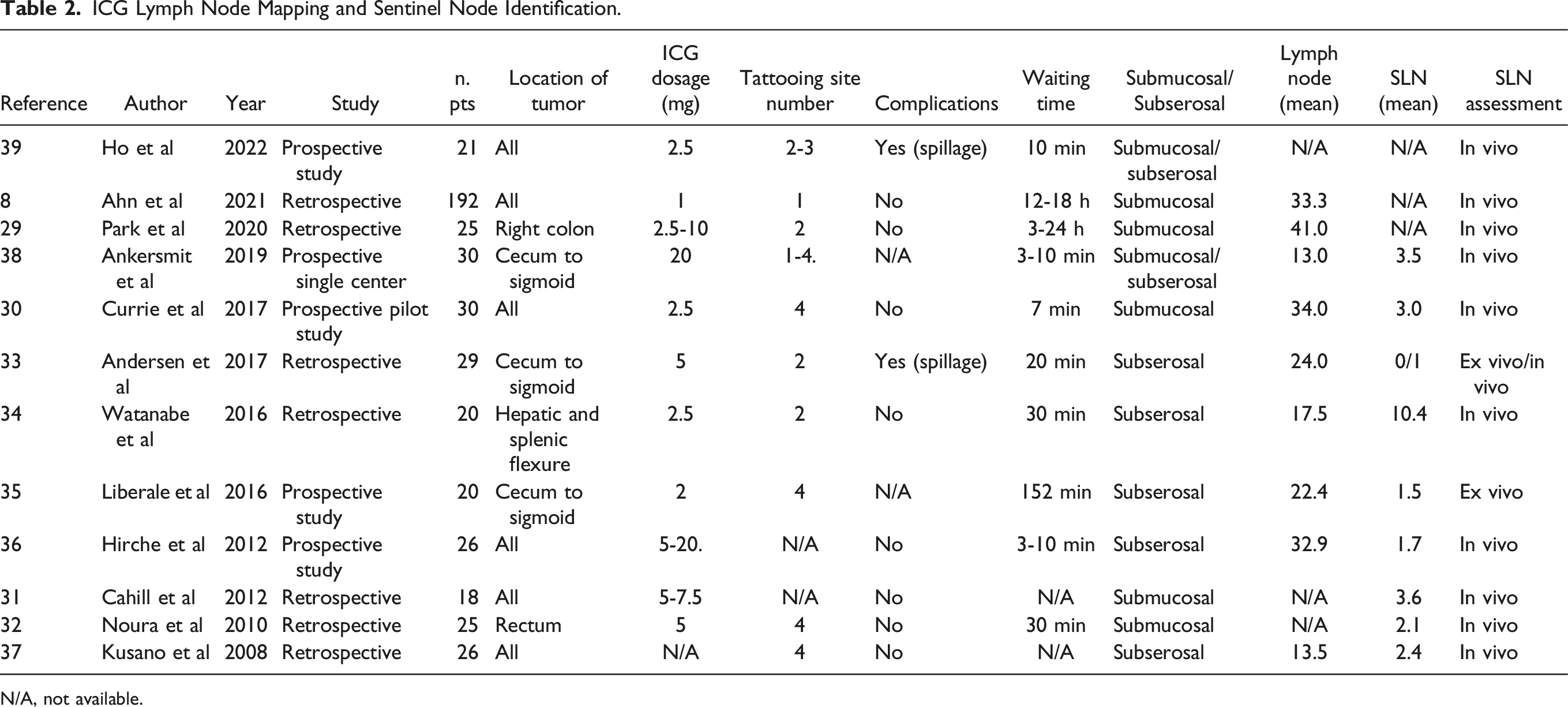
ICG Lymph Node Mapping and Sentinel Node Identification.
N/A, not available.
Moreover we analyzed the site of the lesion and we found that in six papers the whole intestinal tract was considered,2,22,23,28,29,31 three cases reported the tract from the cecum to sigmoid,25,27,30 in one paper the rectum, 24 in one article the right colon 21 and finally the hepatic and splenic flexure in a further study. 26 No complications related to ICG were reported except the study of Andersen et al and Ho et al26,32 in which 7 out of 29 patients experienced spillage. In two papers data were not available.27,30
We analyzed the mean number of sentinel lymph nodes (SLNs) that were identified by fluorescence, the mean number of resected lymph nodes (LNs) and the median time to detect the lymph nodes. In the prospective study of Liberale et al 27 20 patients were enrolled comparing indocyanine green fluorescence imaging (ICG-FI) with blue dye. The mean number of SLNs identified with ICG was 1.5 after 152 min, while the mean number of recovered LNs was 22.4. Ankersmith et al 30 in a prospective single center study identified on 30 patients a mean of 3.5 SLNs, 3-10 min after dye injection, and recovered approximately 13 LNs. Currie et al 22 performed a pilot study on 30 patients with ICG for intraoperative sentinel lymph node mapping in early colon cancer by identifying a mean of 3 mesocolic SLNs with a mean detection time of 7 minutes and a resection of 34 LNs. Andersen et al 25 compared, through a retrospective study on 29 patients, the use of ICG in vivo and methylene blue ex vivo to identify sentinel lymph nodes. The median number of analyzed lymph nodes was 24. The median number of SLNs identified by in vivo mapping was 1 and 0 in ex vivo mapping, the mean time to visualize the SLNs was 20 min. In the retrospective study by Watanabe et al 26 20 patients were analyzed and the tumor was located in the splenic or hepatic flexure, the mean number of resected lymph nodes was 17.5 with a mean SLNs of 10.4 while lymphatic flow was observed 30 min after injection. In the retrospective study of Kusano et al 29 22 patients with colorectal cancer were enrolled, the number of dissected lymph nodes was 13.5 while the number of SLNs was 2.4. In the retrospective study by Ahn et al 2 192 patients were analyzed and ICG injection was performed 12-18 h before surgery. The mean number of lymph nodes collected was 33.3. Park et al 21 enrolled 20 patients with advanced right colon cancer and examined the clinical value of an ICG D3 dissection compared to conventional laparoscopic surgery in a retrospective study. The median number of N3 lymph nodes collected by ICG was 41, greater than in the conventional group. The time interval between ICG injection and the operation was from 3 to 24 h. The retrospective study by Noura et al 24 analyzed on 25 patients the feasibility of sentinel lymph node biopsy of the lateral region in lower rectal cancer guided by ICG: the number of lateral SNs per patients was 2.1 and the mean time to visualize the SLNs was 30 min. The prospective study by Hirche et al 28 conducted on 26 patients revealed a mean number of 32.9 excised lymph nodes and showed a mean number of 1.7 SLNs. Cahill et al 23 conducted a retrospective study on 18 patients with early stage colorectal cancer identifying a mean number of 3.6 SNLs. Ho et al 31 compared, in a prospective study of 21 patients, the injection of ICG before or during surgery by either intraluminal submucosal or laparoscopic peritumoral injection. The timing to demonstrate lymphatic drainage was 10 min.
Discussion
Indocyanine Tattooing
Endoscopic marking of colonic lesions was firstly described in 1975 by Ponsky and King using India ink. 32 For many years India ink has been the standard colonic marking agent. 33 Different markers have been used over the years to locate the site of lesions including India ink, indigo carmine, metal clips, methylene blue and indocyanine green. The use of the ICG as a tattooing agent was initially described in few reports based on animal models by Hammond, Price and Lee34-36 as an alternative to India ink. In 1993 Hammond et al 37 conducted a study on 12 patients by using ICG as an endoscopic marker in order to allow an accurate localization of small or non-palpable lesions of the colon. Over the years several articles compared India ink and ICG as endoscopic markers. In 2009 a study by Miyoshi et al 4 evaluated the usefulness of indocyanine green as an alternative to India ink by examining the visibility, duration and adverse effects of ICG. India ink has higher visibility and longer duration compared to ICG whose visibility depends on: the intervals between endoscopic marking and surgery, omentum, pericolonic fat, mesentery and the posterior abdominal wall which can block the surgeon's view. Regarding the adverse effects, ICG is considered an excellent colonic marking because it does not present adverse reactions such as fever, abdominal pain or allergy symptoms differently from India ink where many articles3-9 report complications such as focal peritonitis, abscess, intestinal perforation, inflammatory pseudotumors, adhesions. This review of the literature has shown that an injection of indocyanine green for the tattoo of the tumor must be performed at an adequate dosage varying from .5 mg to 12.5 mg in 1 or more peritumor lesion sites to avoid incorrect identification of the tumor. The interval between the injection of ICG and surgery is on average 2.3 days and it is important to perform it within 7 days since Miyoshi et al 4 have shown that from the seventh day the detection with indocyanine green is reduced to 80%. An important difference between indocyanine green and India ink is the cost. In laparoscopic surgery, the preoperative tattoo using ICG identifies the site of the lesion thanks to the fluorescence properties of the marker and through the use of surgical NIR camera systems. Due to high costs these technologies are not currently widely available. 11 Recent studies have introduced a new innovative technique to localize the site of the lesions in laparoscopic surgery, especially in the early stages of cancer, through the use of ICG-coated endoscopic clips.10,16 This method is safe, easy, accurate, without complications but in few cases the clips are not visible due to fat tissue or mesorectum so it is necessary to perform an intraoperative colonoscopy. 10 Unlike endoscopic ICG tattooing, the use of clips does not allow the visualization of the lymphatic drainage during laparoscopic surgery. ICG-coated clipping is a promising technique but since it involves the use of surgical NIR camera systems has not been widely used so far.
Indocyanine Mapping
Over the years the use of indocyanine green has been found to have different functions in laparoscopic colorectal cancer surgery based on different administration ways. When intravenously administered, it allows to perform an angiography of the colonic stumps to assess their adequate vascular perfusion. When injected into the subserosal or submucosal layer near the lesion it allows to visualize the lymphatic flow and lymph nodes by using the NIR light system.2,38 Indocyanine green was initially used in breast cancers and melanomas as a tool to identify sentinel lymph nodes, minimizing surgical procedures and complications. 39 In 2008 Kusano et al 29 conducted the first study to determine the feasibility of sentinel node mapping guided by ICG fluorescence imaging in gastrointestinal cancer. Since the injection of ICG can be performed closer to the lesion, in literature several studies have shown that the identification of the sentinel lymph node in colorectal cancer is becoming important in selected patients with early colorectal cancer but its efficacy in improving outcome is still questionable.2,22,23 In colorectal cancer an adequate fluorescence lymph node mapping supports the surgeon in visualizing the lymphatic pathway and in performing an accurate lymph node dissection thus reaching greater oncological radicality. 40 If the surgeon identifies a fluorescent lymph node outside the conventional dissection plane, he can extend the dissection for complete lymph node removal. 2 This review of the literature has shown that lymph node mapping should be performed at an optimal ICG dosage ranging from 1 to 20 mg. If the dosage is too high, there is a risk of activating accessory lymphatic pathways, not allowing the correct lymphatic drainage of the tumor to be visualized. The injection can be performed from 1 to 4 peritumor lesion sites in the submucosal or subserosal layer with an average waiting time of three hours. Patients with a high body mass index (BMI) can affect the visualization of the lymph node mapping due to the presence of a fatty mesocolon. 2 In a recent article dated 2020, Park et al 21 highlighted the efficacy and safety of the use of ICG for FLNM in right colon advanced cancers considered that in their study-group patients a higher number of lymph nodes were removed compared to the control-group ones, and in one third of cases the extent of the lymphadenectomy was modified to achieve a radical D3 dissection. In colon cancer, lymphadenectomy is not influenced by the positive or negative status of the sentinel lymph node. ICG in the intraoperative detection of lymph nodes during laparoscopic colon cancer surgery can be used during lymphadenectomy: this aspect could be important only if these nodes are not present in the usual dissection area because this information would change the surgical strategy. Conversely in laparoscopic rectal surgery, it could be of added value in assessing the need to remove the lateral lymph node chains: moreover, the preoperative identification by ICG of lymph nodes with possible invasion could be compared with TC findings after neoadjuvant chemotherapy, improving preoperative staging and subsequently guiding laparoscopic surgical management.
Conclusions
In laparoscopic colorectal cancer surgery indocyanine green can be used for various functions including the localization of fluorescent tumors and fluorescence lymph node mapping using the surgical NIR camera system. To date, the use of ICG appears to be a valid and safe technique that helps the surgeon to achieve a better oncological radicality but, however, the protocols regarding the adequate dosage and the time interval between tattooing and surgery need to be clarified by further studies.
Footnotes
Author Contributions
Conceptualization, S.P. and A.L.; formal analysis, M.V.; data curation, C.P, C.H. and R.P.; writing—original draft preparation, M.V.; writing—review and editing, D.M. and S.A.; supervision, A.L. and S.P. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
