Abstract
Background
Photodynamic therapy (PDT) is a therapeutic modality that can be used to ablate tumors using the localized generation of reactive oxygen species by combining a photosensitizer, light, and molecular oxygen. This modality holds promise as an adjunctive therapy in the management of colorectal cancer and could be incorporated into neoadjuvant treatment plans under the auspices of prospective clinical trials.
Methods
We conducted a search of primary literature published until January 2021, based on PRISMA guidelines. Primary clinical studies of PDT for the management of colorectal cancer were included. Screening, inclusion, quality assessment, and data collection were performed in duplicate. Analyses were descriptive or thematic.
Results
Nineteen studies were included, most of which were case series. The total number of patients reported to have received PDT for colorectal cancer was 137, almost all of whom received PDT with palliative intent. The most common photosensitizer was hematoporphyin derivative or Photofrin. The light dose used varied from 32 J/cm2 to 500 J/cm2. Complete tumor response (cure) was reported in 40%, with partial response reported in 43.2%. Symptomatic improvement was reported in 51.9% of patients. In total, 32 complications were reported, the most common of which was a skin photosensitivity reaction.
Conclusions
PDT for the management of colorectal cancer has not been well studied, despite promising results in early clinical case series. New, well designed, prospective clinical trials are required to establish and define the role of PDT in the management of colorectal cancer.
Keywords
Background
Photodynamic therapy (PDT) is a therapeutic modality that destroys target cells using the generation of reactive oxygen species through the excitation of a photosensitizer. Photosensitizers can be administered topically or intravenously and subsequently excited by irradiation with a specific wavelength of light, typically using a laser. PDT is most commonly investigated for its ablative potential in the context of cancer and has been applied clinically to a large number of cancers, including non-melanoma skin cancer, various gastrointestinal cancers, non-small-cell lung cancer, brain cancer, breast cancer, head and neck cancer, genitourinary cancer, and more. 1 It is particularly attractive because the mechanism by which PDT ablates tumors spares connective tissues, affecting only living cells and resulting in less scarring and anatomic distortion compared with other surgical and ablative modalities. 2 PDT offers the opportunity to tightly target malignant tissues through a combination of localization of the photosensitizer and the directed delivery of light. Owing to the need to deliver light precisely, PDT is perhaps most readily deployed to easily accessible tumor sites, like the skin, lung, and gastrointestinal tract. A large quantity of pre-clinical data suggests that PDT can be used to ablate colorectal cancers; however, clinical translation of this data has been limited, and no photosensitizers are expressly approved, recommended, or used to treat colorectal cancer. 3 This gap may be due to confusion surrounding the ideal treatment patient population and treatment regimen as a result of the myriad of potential variables involved. We sought to synthesize the existing clinical data in a systematic fashion, particularly with a view to clarify which patients are most likely to benefit, and what regimen is most likely to succeed. This is the first systematic review of the clinical literature investigating the use of PDT for the management of colorectal cancer.
Methods
Review Protocol
Our review protocol was developed a priori and registered in the international prospective register of systematic reviews (PROSPERO, CRD42021233971) on February 28, 2021.
Search Strategy
We conducted a systematic literature search of MEDLINE (1946–present), Medline In-Process/ePubs (daily), Embase (1947–present), Cochrane Central Register of Controlled Trials (1991–present), Cochrane Database of Systematic Reviews (2005–present), and PsycINFO (1806–present). The Web of Science (Clarivate) database was searched (1900–present). Lastly, the Scopus (Elsevier, 1960–present) database was searched. All databases were searched on the same day, Monday January 4, 2021. An update of the search was conducted on May 1, 2021, which found no new eligible studies.
The searching process followed the Cochrane Handbook 4 and the Cochrane Methodological Expectations of Cochrane Intervention Reviews (MECIR) 5 for conducting the search, the PRISMA guideline 6 for reporting the search, and the PRESS guideline for peer-reviewing the search strategies 7 drawing on the PRESS 2015 Guideline Evidence-Based Checklist used to avoid potential search errors.
Preliminary searches were conducted, and full text literature was mined for potential keywords and appropriate controlled vocabulary terms (such as Medical Subject Headings for Medline and EMTREE descriptors for Embase). The search strategy concept blocks were built on the topics of: Photodynamic Therapy AND Colorectal Cancer AND Studies. Results were limited to English language, and human subjects.
Study Selection, Data Extraction, and Quality Assessment
Two trained reviewers (KG and LD) independently identified articles eligible for further review by performing an initial screen of identified abstracts. Articles were considered for inclusion if they reported results of human patients undergoing photodynamic therapy (i.e., administration of both a photosensitizer and a light dose) for the management of a primary colorectal cancer. Disagreement between reviewers was resolved in discussion between the two initial reviewers and a third trained reviewer (HY). Reviewers independently evaluated the quality of the studies and extracted the data. Quality assessment was performed using Joanna-Briggs Institute critical appraisal tools for use in systematic reviews, as appropriate for the study design.8,9
Summarization of Data
Due to generally poor study quality and a large degree of heterogeneity in the design, reported parameters, and reported outcomes of the study, no formal statistical analysis was conducted. Descriptive numerical analyses through frequency analysis were performed where appropriate. Thematic analyses were performed where appropriate to evaluate qualitative data.
Results
Literature Search and Selection Process
Our initial search resulted in 1651 citations. After the removal of duplicate citations (310), 1341 citations were screened for relevance, of which 1289 were excluded. Of the remaining 52 studies that underwent full-text assessment for eligibility, 19 were ultimately included in the study10-28 (see Figure 1). PRISMA flow diagram of citation inclusion.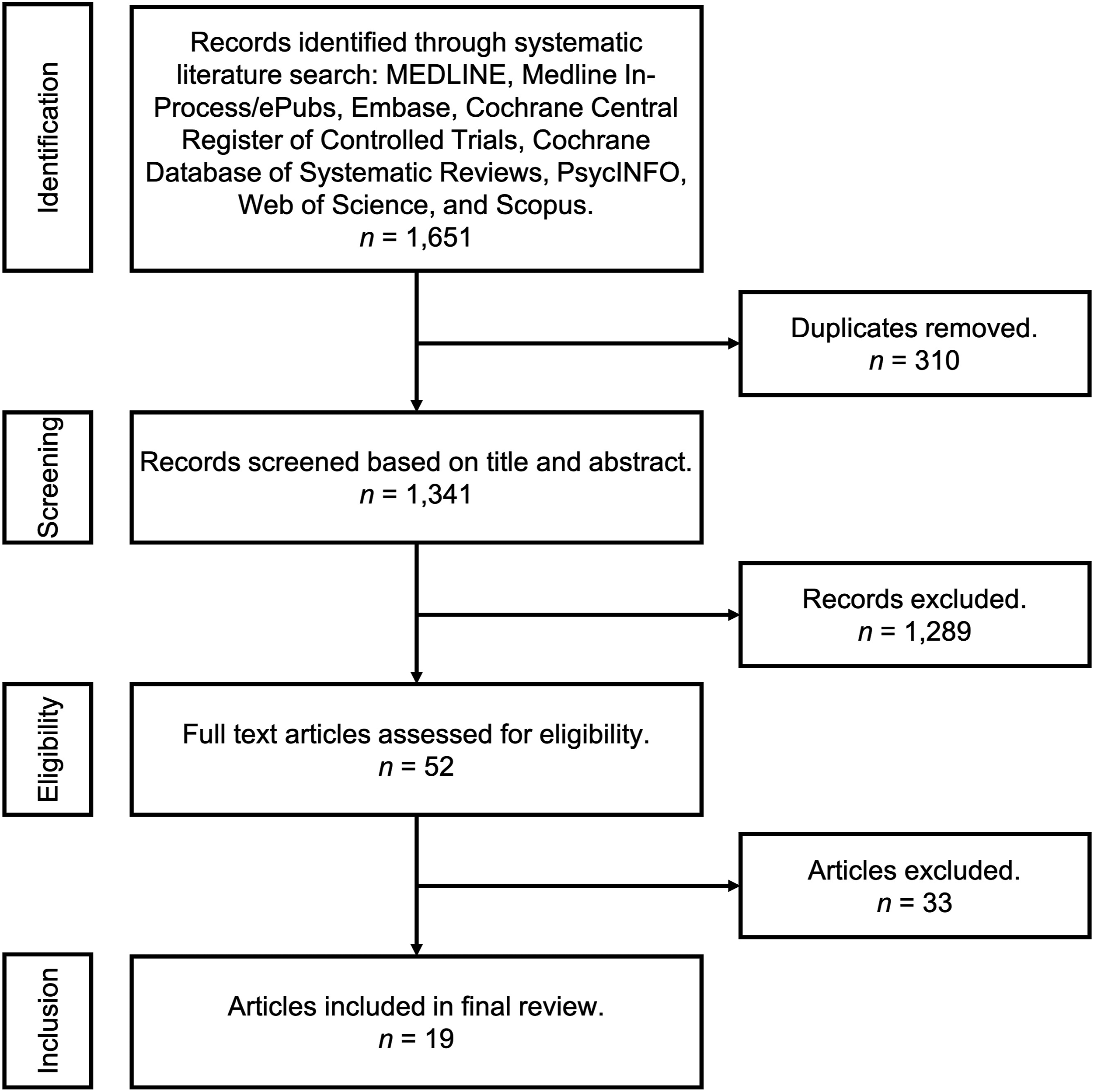
Study Characteristics
Demographic and study details. PDT = photodynamic therapy.
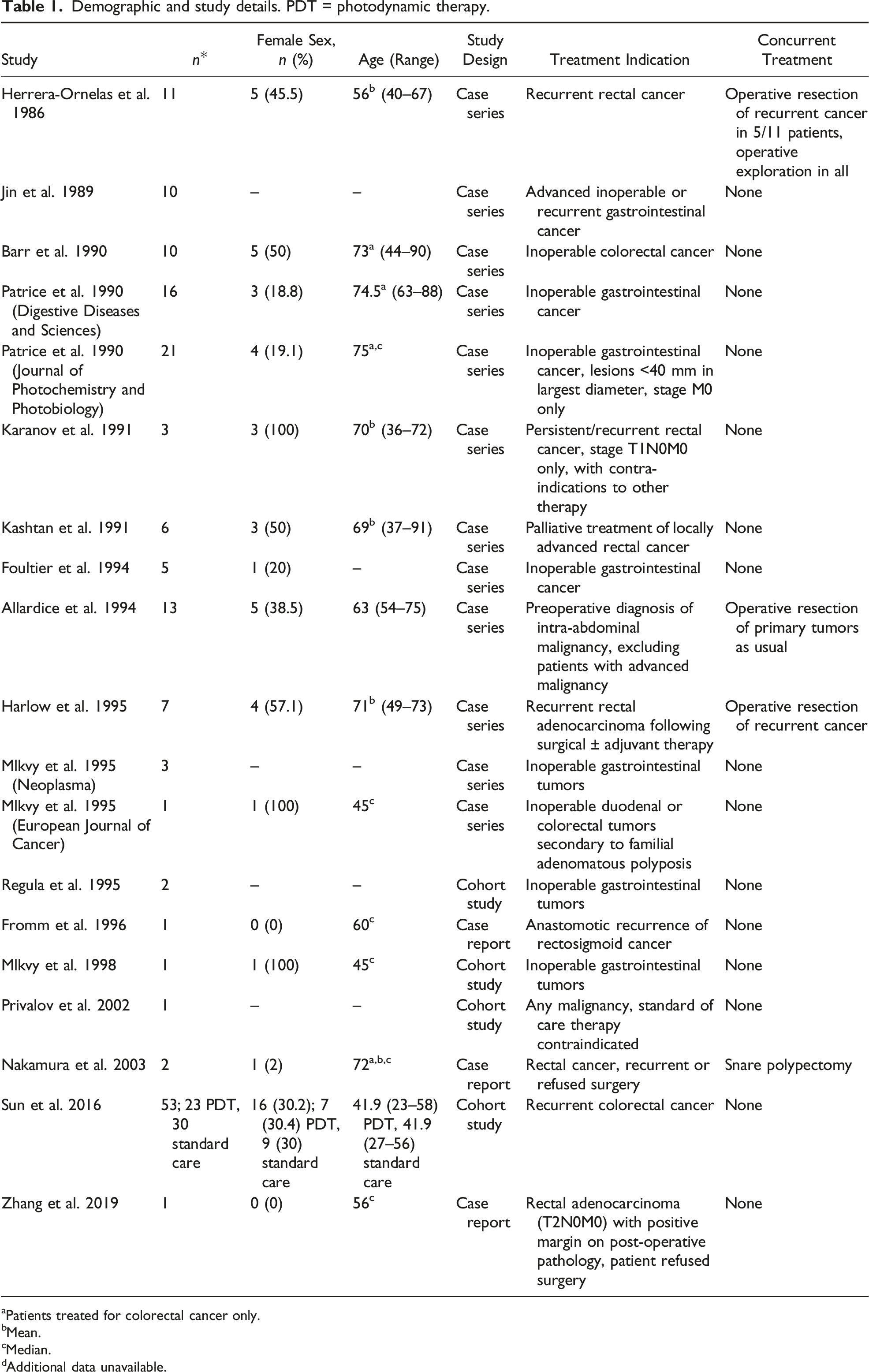
aPatients treated for colorectal cancer only.
bMean.
cMedian.
dAdditional data unavailable.
Study quality was generally poor, with an enormous degree of heterogeneity in the design, conduct, and reporting of key methodological characteristics (Supplementary Table S1). Due to the relative dearth of evidence and similar study quality, no studies were excluded for reasons of poor quality.
Treatment Specifications
Summary of treatment parameters and outcomes by study. PDT = photodynamic therapy; HpD = hematoporphyrin derivative; ALA = aminolevulinic acid; IV = intravenous; LGIB = lower gastrointestinal bleed.
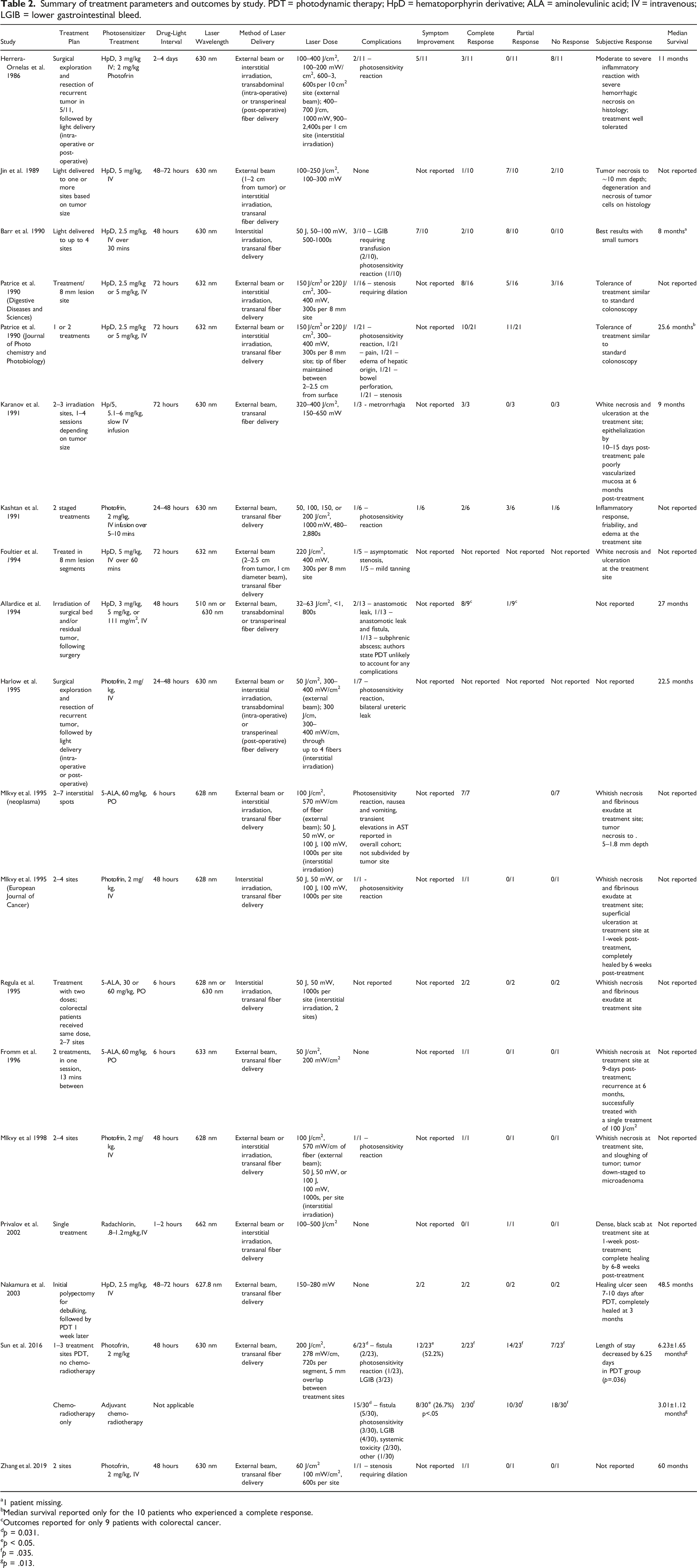
a1 patient missing.
bMedian survival reported only for the 10 patients who experienced a complete response.
cOutcomes reported for only 9 patients with colorectal cancer.
dp = 0.031.
ep < 0.05.
fp = .035.
gp = .013.
All studies except for two used a laser excitation wavelength around 630 nm (Allardice et al.
19
used 510 nm or 630 nm for HpD, and Privalov et al.
12
used 662 nm for their Radachlorin photosensitizer). Light was administered using one of two methods: either external beam irradiation (in which a beam of laser light is directed onto the tumor using a fiber optic) or interstitial irradiation (in which a fiber optic with a cylindrical diffuser is introduced into the tumor parenchyma). Overall, more studies used external beam irradiation compared with interstitial irradiation (15 vs 11); however, eight studies used a combination of both, with eight performing external beam irradiation only, and three performing interstitial irradiation only. All but three studies delivered the fiber optic transanally via an endoscope; the remaining studies administered light concurrently with transabdominal surgery and either delivered light intraoperatively, placed fiber optics during surgery that were later used to deliver light, or introduced fiber optics via the perineal wound following an abdominoperineal resection. The light dose delivered varied between studies from 32 J/cm(2)–500 J/cm(2), with the most common light dose falling around at ∼200 J/cm(2) (see Figure 2). The power varied from 50 mW/cm(2)–1000 mW/cm(2). Treatment time varied from 300s (5 mins) to 3,600s (60 mins). Forest plot of light energy doses used in the included studies.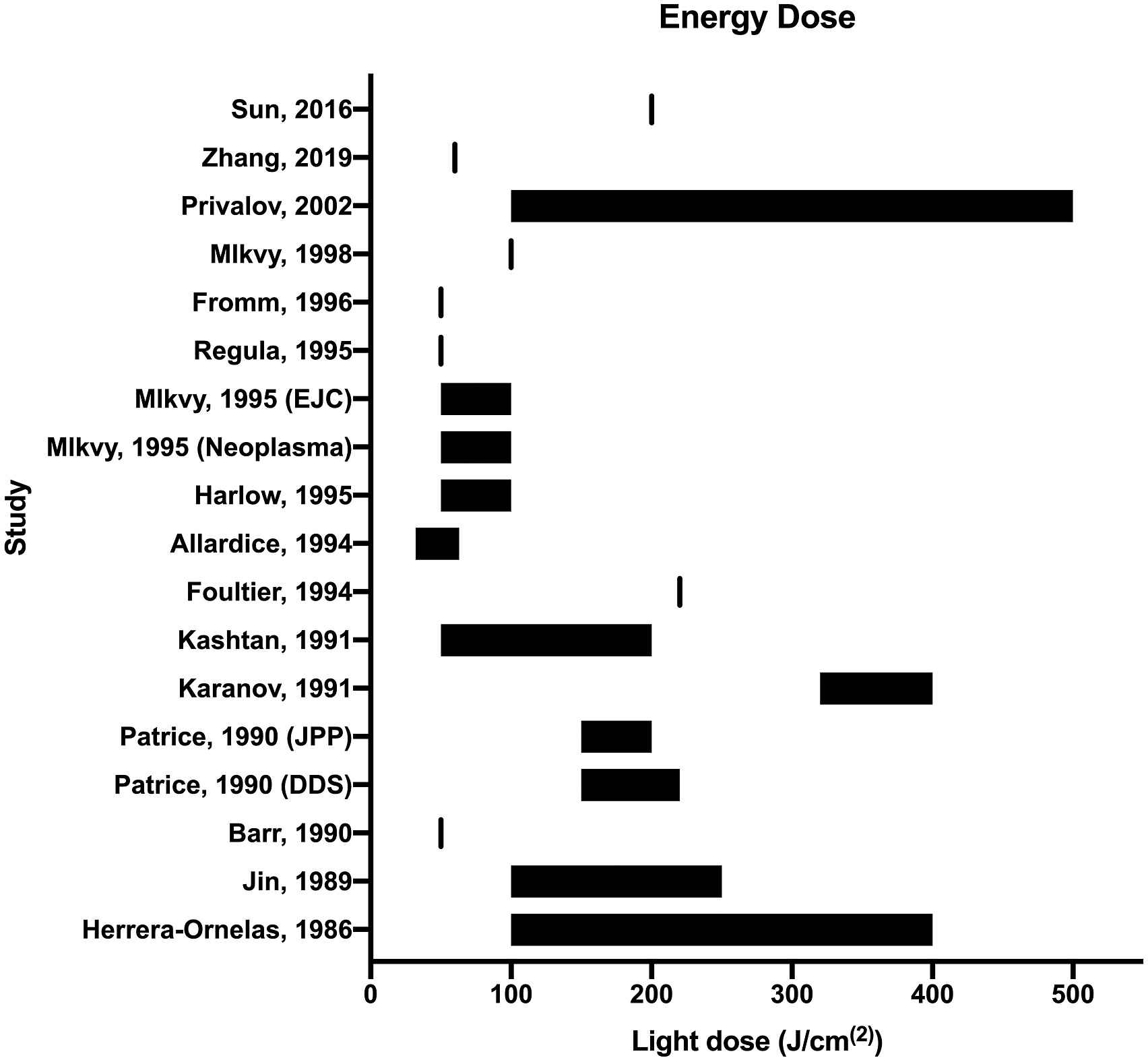
Outcomes
Complications of treatment were reported in 18 studies, with four reporting no complications at all. The most commonly reported complication was skin photosensitivity (usually manifested as a superficial burn upon exposure to sunlight, reported in at least nine patients); other common complications included lower gastrointestinal bleed (five patients), and stenosis (variably requiring dilation, four patients). In addition, five patients were reported to have suffered a fistula of some kind, one patient was reported to have suffered a bowel perforation, and another was reported to have suffered from a ureteric leak requiring ureteral stenting; however, this was in a patient who had received an abdominoperineal resection and received trans-perineal light delivery. In total, 32 complications were reported. The only study to compare a cohort of patients treated with PDT to a cohort treated without, was Sun et al. 10 who reported a complication rate of 26.1% in PDT treated patients compared with 50% in patients undergoing standard of care adjuvant chemo-radiotherapy (p = .031).
Five studies reported on symptomatic improvement, with all studies reporting at least some degree of symptom improvement (specific symptoms varied, but included bleeding, obstruction, and pain) in patients who underwent PDT for colorectal cancer. In aggregate, 27/52 (51.9%) reported patients experienced symptomatic improvement. The only study to compare a cohort of patients treated with PDT to a cohort treated without, was Sun et al., 10 who found that 52.2% of patients treated with PDT experienced symptom improvement compared with 26.7% of patients treated with standard of care adjuvant chemo-radiotherapy (p < .05).
Tumor Response
Studies variably reported tumor response to PDT; two studies did not report any tumor response outcomes (Table 2). In those reporting tumor response, response was reported as complete, partial, or no response (though not all studies reported all of these categories). Among those reporting complete response (defined as complete regression of the tumor at any point during follow-up), the complete response rate was 46/115 (40%). Among those reporting partial response (variably defined as incomplete regression or temporary growth arrest of the tumor), the partial response rate was 38/88 (43.2%). Complete or partial response was reported in 82/103 (79.6%) of patients with reporting of such responses. No response was reported in 21/95 (22.1%) of patients with reporting of no response. The only study to compare a cohort of patients treated with PDT to a cohort treated without, was Sun et al., 10 who found a greater rate of complete and partial response in the PDT treated group as compared to the standard of care group (8.7% vs 6.7% and 60.9% vs 33.3%, respectively).
Sixteen studies reported subjective responses to PDT treatment, with eight reporting white necrosis, four reporting superficial ulceration, and three reporting fibrinous exudate at the treatment site within the first week of treatment. Necrosis was commonly seen on histological assessment of any lesion biopsies. Complete healing was reported as early as 6 weeks post-treatment in two studies. In addition, three studies reported that tolerance of treatment was comparable to that of endoscopy. Two studies reported on the effective treatment depth, with one reporting a range between 5 and 18 mm in depth of necrosis, 17 and the second reporting necrosis to ∼10 mm26; similarly, Barr et al. 25 reported that subjectively, smaller tumors were more likely to be ablated.
Median survival was reported in nine studies, with a median of 22.5 months (range 6.23–60). Sun et al. 10 directly compared a cohort receiving PDT to another not receiving PDT, finding a greater median survival in PDT treated patients compared to those receiving standard of care therapy (6.23 ± 1.65 months vs 3.01 ± 1.12 months, p = .013).
Discussion
Photodynamic therapy is a relatively novel treatment modality that has been thoroughly demonstrated in both pre-clinical and clinical studies to be capable of tumor ablation, yet it remains poorly utilized in clinical practice despite its promise for many modern and growing applications, including in the management of rectal cancer. Currently, almost all clinical PDT for cancer is conducted using Photofrin (Pinnacle Biologics) as a photosensitizer and using a laser assembly distributed by the same company for the management of certain endobronchial and esophageal tumors. Another significant clinical application of PDT is in the management of non-melanoma skin cancer, where the photosensitizer is applied topically.
Reasons for the generally poor uptake of PDT as a modality are frequently discussed in the PDT literature but can be summarized generally as challenges related to the complexity of the therapy. PDT relies upon delivery of the correct dose of a photosensitizing agent (usually administered intravenously) to a tumor, followed by irradiation at a specific time-point following drug administration, with a particular wavelength and power output light, for a specific period of time, via either external beam irradiation or interstitial irradiation, at one or more sites. This entire procedure may then be repeated any number of times. Even if all of these parameters can be achieved and consistently delivered to patients, the therapeutic effect may not be consistent between patients due to variation in the size and shape of both the tumor and the patient, as well as differences in tissue pigmentation.
All of these complexities in treatment plan are reflected in the vast heterogeneity of the treatment parameters used in the studies analyzed in this article. The various attempts of the authors to modify their protocol—either ad hoc or post hoc—can be seen in Table 2. For instance, Barr et al., 25 Mlkvy et al.,13,16,17 and Patrice et al.23,24 appear to have changed their light dose parameters mid-way through the study, and Kashtan et al. 21 designed a somewhat complex “step-up” protocol to increase their light dose depending upon the observed effect. All of this reflects the complexity involved in optimizing PDT for the management of colorectal cancer.
We found only one reasonably well-conducted study that makes a meaningful comparison between PDT and a control group 10 ; the remaining studies were extremely heterogenous in terms of study population, treatment parameters, and measured outcomes. In addition, they were generally smaller studies with limited statistical power. Despite these drawbacks, these studies provide compelling reasons to believe that PDT is a viable therapeutic modality that can be deployed to great effect in patients with colorectal cancer. We found that 79.6% of patients in these studies experienced at least a partial tumor response to therapy, with 40% experiencing a complete ablation of the tumor. In addition, 51.9% of patients reported symptom improvement following PDT, with a reasonable safety profile. All of these results must be understood while bearing in mind that all of these trials were conducted on patients who had no other viable treatment options, thereby underestimating the true therapeutic potential of PDT. These promising early results call for a more methodologically and statistically robust clinical study of PDT in a dedicated and well-defined colorectal cancer patient population.
Future clinical studies of colorectal PDT must look to previous work for guidance when determining the most scientifically robust methodology, and despite the heterogeneity seen in these studies, some common themes emerge. Firstly, the most commonly used photosensitizer used was HpD or Photofrin (largely identical), with Photofrin being the most readily available agent on the market. Secondly, a dose of 2 mg/kg, a laser wavelength of ∼630 nm, and a drug-light interval of 24–48 hours was universally used for Photofrin PDT. Light delivery is the most challenging and variable component of PDT; however, Photofrin trials typically deliver a light dose between 50 and 100 J/cm2 with a power between 100 and 500 mW/cm2. The optimal method of light delivery remains uncertain, with many studies employing both external beam and interstitial irradiation; this reflects the ongoing conflict between the perhaps more scientifically robust interstitial irradiation method and the more pragmatic external beam irradiation approach. The decision between these methods must be made based on the expertise and comfort of the local clinicians and medical biophysicists. These parameters can form the basis for the methodology of future studies seeking to perform PDT, particularly for colorectal cancer.
Clinicians’ interest in PDT for colorectal cancer was at a height two decades ago and has since waned, with a corresponding rapid advance in other non-surgical treatment options like chemotherapy and radiotherapy. However, given the recent interest in total neoadjuvant and sphincter-preserving therapy, it is no longer possible for oncologists to ignore the potential therapeutic benefits offered by PDT in good conscience. PDT has the potential to be used in combination with other neoadjuvant, adjuvant, and non-operative therapies to manage colorectal cancer. Further large-scale, prospective, randomized, clinical trials are required before PDT can be fully integrated into the treatment pathway for colorectal cancer; however, the ability to repeat PDT indefinitely and ablate tumors in an extremely precise and targeted fashion with limited off-target toxicity makes it an extremely attractive tool to add to the oncologist’s arsenal. We hope that this review can generate interest in PDT as an adjunctive ablative modality for the management of colorectal cancer and can help to guide future clinicians and researchers in the conduct of better-designed studies.
Supplemental Material
sj-pdf-1-sri-10.1177_15533506221083545 – Supplemental Material for Photodynamic Therapy for Colorectal Cancer: A Systematic Review of Clinical Research
Supplemental Material, sj-pdf-1-sri-10.1177_15533506221083545 for Photodynamic Therapy for Colorectal Cancer: A Systematic Review of Clinical Research by Keegan Guidolin, Lili Ding, Han Yan, Marina Englesakis HBA, Sami Chadi, Fayez Quereshy and Gang Zheng in Surgical Innovation
Footnotes
Acknowledgments
The authors would like to acknowledge the University of Toronto Surgeon-Scientist Training Program, the Hold’em for Life Oncology Fellowship Program, and the Banting and Best Doctoral Scholarship Program (KG), as well as the Canada Research Chairs Program (GZ).
Author Contributions
KG, GZ, SC, and FQ designed the study. ME conducted the literature search. KG, LD, and HY conducted citation screening, review, and extraction. The manuscript was written by KG and reviewed by LD, HY, ME, SC, FQ, and GZ.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
