Abstract
Keywords
Summary
We introduce an integrated Smart Sensor System (
Introduction
Background
Many of today’s surgical disciplines rely on drains to postoperatively evacuate fluid from the wound cavity. The evacuated fluid is then collected within the drain bowl for exudate storage and subsequent manual evaluation by visual inspection or laboratory analysis within larger time intervals. 1 In this way, quantity and quality of drainage output continue to be valuable proxies for monitoring the postoperative healing process following a large array of abdominal surgeries. 2 However, as important as this information may be, the practice of manual processing of drain fluids has evolved little to none over the last decades and continues to be associated with significant problems, such as clogging, 3 imprecise documentation, 4 delayed removal and analysis of drain fluid, 5 a high protocol workload for nursing staff 6 as well as, most importantly, missed or delayed recognition of changes in quality of drain output indicating serious complications such as infection, bleeding, pancreatic fistulas, 7 bile leaks, and intestinal perforations. 8
Aim
The aim of this study was to address and alleviate the clinical challenges of today’s drain management as stated above by developing a medical sensor device (
Material and Methods
Prototype Development
Since the quantity of drain output can easily be measured using an integrated flowmeter or a scale, this study focuses primarily on the characterization of the quality of the drain output using a sophisticated yet affordable and compact spectrometer. The spectrometer used in the prototype offers fast and non-obstructive measurement in the detection range between 280 nm (near-ultraviolet) until 940 nm (near-infrared) with values being captured by 19 discrete detection channels. It thus exceeds the perception of the human observer at both ends of the visible spectrum (roughly 380 nm until 700 nm). The spectrometer was combined with an array containing 17 different exposure configurations emitting light at wavelengths matching the respective detection channels. The
The prototype builds upon the existing workflow of drain insertion and management within the operating room.
Once surgery is completed, a sterile, disposable tubing with the connector for the Handling and clinical application of the 
Study Population
Study Population.

Measurements
Even though our prototype allows for direct integration into any standard drain tubing system (see Figure 1), in this pilot study, we carried out all measurements with drain fluid that was extracted from the drain container for this purpose. This was necessary in order to assure a control sample of the exact same composition as the sample analyzed by our Correlation of validated laboratory results with exposure values.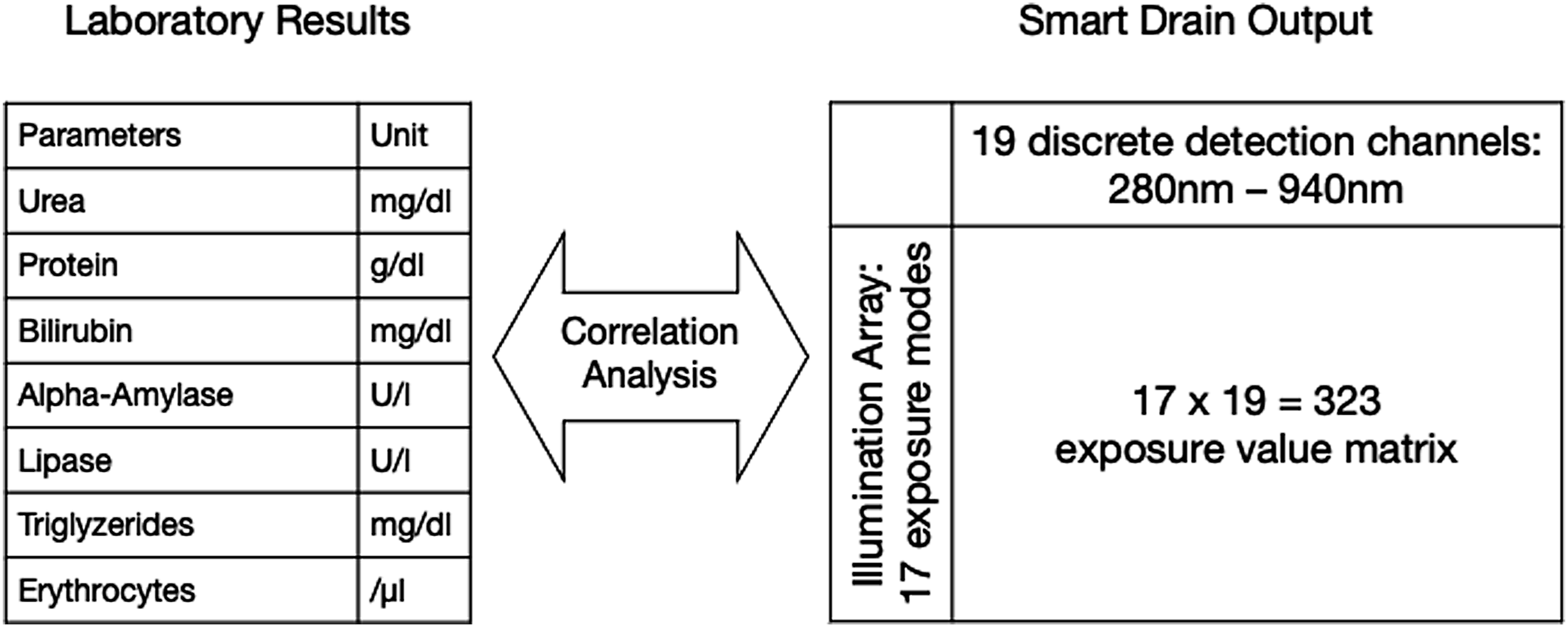
Results
Erythrocytes and Bilirubin
We first focussed our attention on the analysis of detection of blood and bile as the primary indicators of interest for two reasons: First, the majority of our patient population underwent abdominal surgery where blood and bile are important postoperative indicators. Second, blood and bile are both substances that are known to have distinct optical characteristics making them suitable for spectroscopic detection.
9
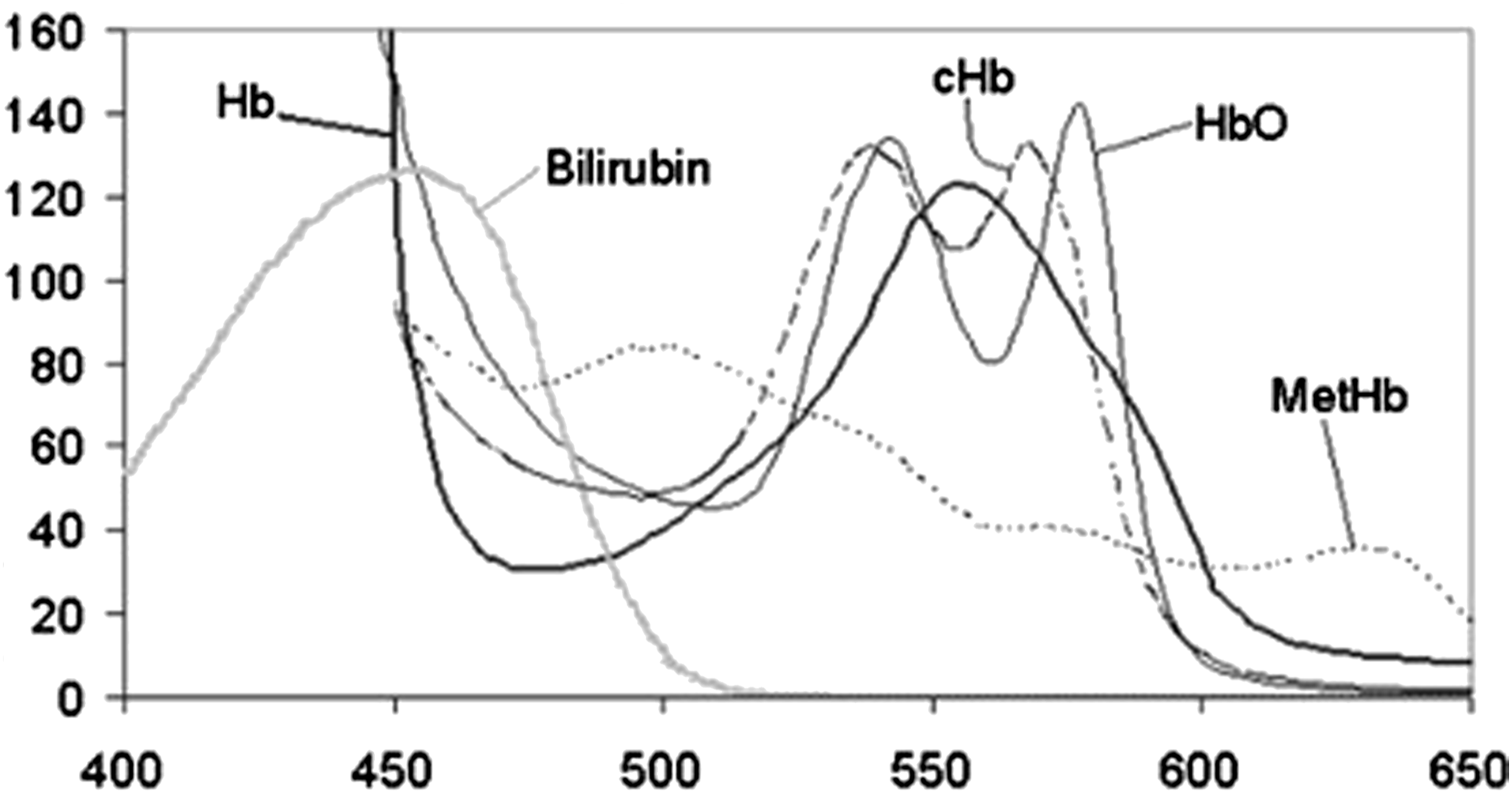
Hemoglobin present within the erythrocytes has absorption spectra varying depending on its carrier state (hemoglobin (Hb), oxyhemoglobin, carboxyhemoglobin, or methemoglobin). The absorption maxima are reported to occur between 525 nm and 575 nm as well as between 400 nm and 425 nm.
10
Bilirubin has its absorption maximum at 450.5 nm (see Figure 3). Absorption coefficients in mM–1cm–1 (Y-axis) for hemoglobin and bilirubin in the 400–650 nm range (X-Axis).
9
To detect the erythrocyte concentration, we choose the 535 nm and 560 nm detector channels as they are closest to the described absorption maxima. We observed statistically highly significant correlation coefficients at both channels (see Supplementary Figure S1). The stronger correlation coefficient (−.904; Pearson’s r) occurred at the 560 nm channel. A curve fitting with several models (quadratic, cubic, and logarithmic) was conducted and produced several viable models, with the logarithmic model finally yielding the best fit (see Figure 4). Logarithmic model predicting erythrocyte concentration (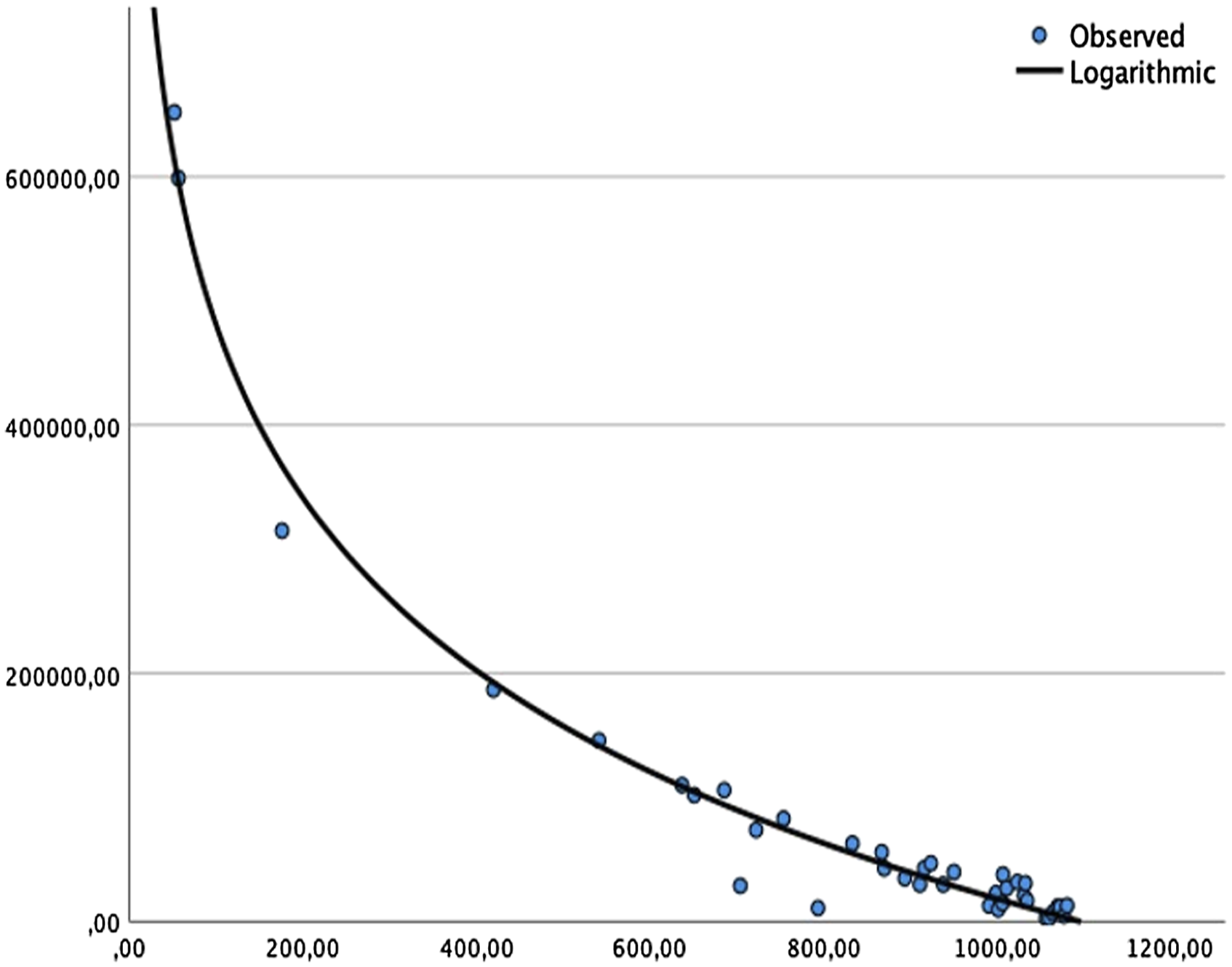
To detect the presence of bilirubin, we chose the 460 nm detector channel as it being closest to the absorption maximum. Subsequently, we observed a moderate negative correlation coefficient (r = −.507), which was highly significant (
When looking at the overlapping spectra of erythrocytes and bilirubin in Figure 3, mutual conflicting measurements seem likely. Indeed, the correlation table for both substances and its associated detector channels show that erythrocytes concentration is not only registered on the respective detector channels but also rather strongly on the 460 nm bilirubin detector channel (see Supplementary Figure S2). However, in turn bilirubin concentration is sensed by the 560 nm detector channel.
This indicates that the presence of erythrocytes in the drain fluid could interfere with the bilirubin measurement. Therefore, to improve our understanding of this effect, we conducted a partial correlation while specifying the laboratory erythrocyte concentration as a control variable. In this manner, we analyzed all samples and observed a rise of the correlation coefficient to −.666 (see Supplementary Figure S3).
Since the erythrocyte concentration from the laboratory as a control variable would not be known to the
Additional Findings
While correlating each of the 17 × 19 = 323 fields of the data matrix of the Data plots (scaled to fit for better visualization).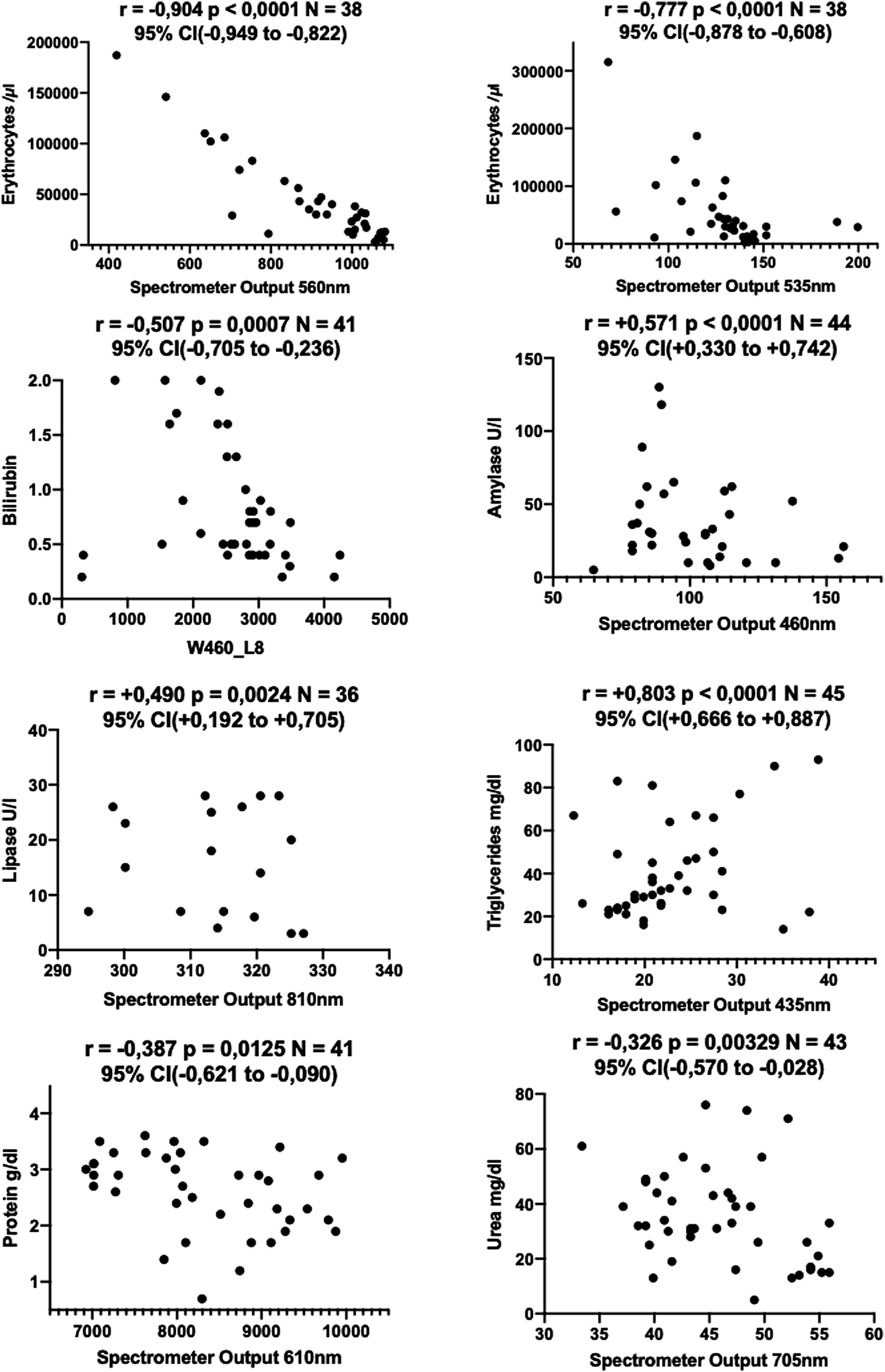
The correlation for amylase (r = +.571,
The correlation for lipase appeared in the near-infrared spectrum at 810 nm (r = +.490,
Furthermore, bivariate correlations for triglycerides at 435 nm (r = +.803,
Discussion
Despite all advances in the era of digitalization, to this day most if not all fluid samples collected in surgical drains around the world are being analyzed in an analogous fashion. This type of documentation is notoriously inaccurate, incomplete, labor-intense, and time consuming. Further, by design, results of charting or laboratory analysis are much delayed. We here demonstrate a functional prototype of an intelligent drain that can analyze fluid output from surgical drains using a spectrometer that delivers results continuously, in real-time and in a digitalized format. By analyzing 45 fluid samples from 19 patients, we demonstrated near perfect correlation coefficients for bilirubin, Hb, alpha-amylase/lipase, urea, protein, and triglycerides. The strength of the correlation coefficient in cases of bilirubin or alpha-amylase/lipase allows for a qualitative prediction and can safely state whether these compounds are present in the drain output or not. The strength of the correlation coefficient in case of the erythrocytes is most likely to allow for a quantitative prediction. This is underscored by the logarithmic modeling with an adjusted R-squared of 98.2%.
The clinical implications of the technique here are obvious. A recent study by Lyons et al. in 2015 revealed significant shortcomings in drain documentation. For example, their analysis showed that the 24 h flowcharting was missing in 35% of cases analyzed and a clear indication of the drain’s location at the patient’s body in 45%. Importantly, the recording of the type of the fluid output was missing in a 100% of the observed cases. Furthermore, events like flushing a clogged drain were not documented properly, which leads to misleading readings if the washing fluid is mistaken for regular drain output. While these numbers certainly cannot be generalized to other hospitals, it is obvious that documentation of drain output in an analogous fashion is a setup for inaccuracy and inconsistency. It is easily perceivable that the inability to appropriately monitor the output of surgical drains can lead to serious harm of the patient. This is especially true if changes in drain output quality are missed or delayed but would otherwise indicate an early pick up of a postoperative bleed or another serious complication. 8 Also, improper documentation of surgical drain output may delay the removal of the drain and therefore the patient’s discharge from the hospital. 5 The latter point is important to consider because it indicates how a type of intelligent drain that analyzes drain output digitally may improve the postoperative care not only of the few patients at risk for complications, but the much larger group of patients whose postoperative course will be uneventful. For this patient population, reassuring drain output analysis could steer medical decision-making toward early drainage removal and its associated benefits such as reduced infection risk and early discharge. 7
One of the biggest advantages of an intelligent drain as we describe here is the provision of real-time, continuous and digital data. This type of analysis allows for easy integration of results into existing medical records of the patients and can be used for charting, documentation, billing, capacity planning, and in order to create “red flags” or another type of alerts provided in a digital fashion in order to indicate trouble. The
Further, in a larger vision, digitalized assessment and documentation of surgical drain output should be one additional data point incorporated into what is becoming the digitalized patient, along with vital signs and other patient characteristics. In the near future and using large sets of data points, artificial intelligence–based deep learning tools will allow for the creation of algorithms that can predict specific risk profiles of individual patients. An intelligent drain system as the one presented here takes into account these revolutionary developments. In our case, acquiring larger amounts of data enables the application of machine learning algorithms.
There are several limitations to our study. Importantly, the optical characteristics of some of the substances analyzed here are not yet described in the literature. We observed a strikingly high correlation for most substances, which is enormously encouraging. However, given the shortage of literature describing the optical characteristics of some substances in a setting as we describe it here, we cannot fully attain with certainty why these correlations occurred in the way they did. Furthermore, the limited number of samples used in this study cautions to make generalizations regarding the drain output in surgical fields other than abdominal surgery. Especially considering that drain fluid with its many possible fractions and enzymatic interactions is from a biochemical standpoint, a highly complex compound fluid. Also, at this point in our research and despite the obvious advantages that an intelligent drain system would have for the patient and within a health provider system, we cannot claim to what extend this will positively influence outcome. Therefore, further research with larger sample sizes and a better biochemical understanding are needed to validate the observed effects in specific clinical scenarios.
Having said that, we have successfully shown that a prototype whose detector chip’s cost amount to a mere US$12 is capable of reliably detecting the presence of bilirubin and other important markers in abdominal surgery and predict the concentration of erythrocytes in an artificial yet realistic laboratory setup. Given that our results are reported in real time allow for continuous assessment in a digitalized format, this development has the potential to substantially improve patient outcome.
Supplemental Material
sj-tiff-1-sri-10.1177_15533506211031459 – Supplemental Material for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study
Supplemental Material, sj-tiff-1-sri-10.1177_15533506211031459 for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study by Mario V. Roser, Alexander H. R. Frank, Lea Henrichs, Christian Heiliger, Dorian Andrade, Laura A Ritz, Jan Sabo, Andreas Rauschmayr, Oliver Muensterer, Jens Werner, Wojciech Konrad Karcz and Michael F. Berger in Surgical Innovation
Supplemental Material
sj-tiff-2-sri-10.1177_15533506211031459 – Supplemental Material for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study
Supplemental Material, sj-tiff-2-sri-10.1177_15533506211031459 for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study by Mario V. Roser, Alexander H. R. Frank, Lea Henrichs, Christian Heiliger, Dorian Andrade, Laura A Ritz, Jan Sabo, Andreas Rauschmayr, Oliver Muensterer, Jens Werner, Wojciech Konrad Karcz and Michael F. Berger in Surgical Innovation
Supplemental Material
sj-tiff-3-sri-10.1177_15533506211031459 – Supplemental Material for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study
Supplemental Material, sj-tiff-3-sri-10.1177_15533506211031459 for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study by Mario V. Roser, Alexander H. R. Frank, Lea Henrichs, Christian Heiliger, Dorian Andrade, Laura A Ritz, Jan Sabo, Andreas Rauschmayr, Oliver Muensterer, Jens Werner, Wojciech Konrad Karcz and Michael F. Berger in Surgical Innovation
Supplemental Material
sj-tiff-4-sri-10.1177_15533506211031459 – Supplemental Material for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study
Supplemental Material, sj-tiff-4-sri-10.1177_15533506211031459 for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study by Mario V. Roser, Alexander H. R. Frank, Lea Henrichs, Christian Heiliger, Dorian Andrade, Laura A Ritz, Jan Sabo, Andreas Rauschmayr, Oliver Muensterer, Jens Werner, Wojciech Konrad Karcz and Michael F. Berger in Surgical Innovation
Supplemental Material
sj-tiff-5-sri-10.1177_15533506211031459 – Supplemental Material for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study
Supplemental Material, sj-tiff-5-sri-10.1177_15533506211031459 for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study by Mario V. Roser, Alexander H. R. Frank, Lea Henrichs, Christian Heiliger, Dorian Andrade, Laura A Ritz, Jan Sabo, Andreas Rauschmayr, Oliver Muensterer, Jens Werner, Wojciech Konrad Karcz and Michael F. Berger in Surgical Innovation
Supplemental Material
sj-tiff-6-sri-10.1177_15533506211031459 – Supplemental Material for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study
Supplemental Material, sj-tiff-6-sri-10.1177_15533506211031459 for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study by Mario V. Roser, Alexander H. R. Frank, Lea Henrichs, Christian Heiliger, Dorian Andrade, Laura A Ritz, Jan Sabo, Andreas Rauschmayr, Oliver Muensterer, Jens Werner, Wojciech Konrad Karcz and Michael F. Berger in Surgical Innovation
Supplemental Material
sj-tiff-7-sri-10.1177_15533506211031459 – Supplemental Material for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study
Supplemental Material, sj-tiff-7-sri-10.1177_15533506211031459 for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study by Mario V. Roser, Alexander H. R. Frank, Lea Henrichs, Christian Heiliger, Dorian Andrade, Laura A Ritz, Jan Sabo, Andreas Rauschmayr, Oliver Muensterer, Jens Werner, Wojciech Konrad Karcz and Michael F. Berger in Surgical Innovation
Supplemental Material
sj-tiff-8-sri-10.1177_15533506211031459 – Supplemental Material for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study
Supplemental Material, sj-tiff-8-sri-10.1177_15533506211031459 for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study by Mario V. Roser, Alexander H. R. Frank, Lea Henrichs, Christian Heiliger, Dorian Andrade, Laura A Ritz, Jan Sabo, Andreas Rauschmayr, Oliver Muensterer, Jens Werner, Wojciech Konrad Karcz and Michael F. Berger in Surgical Innovation
Supplemental Material
sj-tiff-9-sri-10.1177_15533506211031459 – Supplemental Material for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study
Supplemental Material, sj-tiff-9-sri-10.1177_15533506211031459 for Evaluation of an Integrated Smart Sensor System for Real-Time Characterization and Digitalization of Postoperative Abdominal Drain Output: A Pilot Study by Mario V. Roser, Alexander H. R. Frank, Lea Henrichs, Christian Heiliger, Dorian Andrade, Laura A Ritz, Jan Sabo, Andreas Rauschmayr, Oliver Muensterer, Jens Werner, Wojciech Konrad Karcz and Michael F. Berger in Surgical Innovation
Footnotes
Acknowledgments
Furthermore, we would like to thank Prof. Nassir Navab, Dr. Christoph Hennersperger, MSc Maria Tirindelli, MSc Benjamin Busam, MSc Rüdiger Göbel at the
Author Contributions
Study concept and design: Mario V. Roser and Michael F. Berger Acquisition of data: Laura A. Ritz, Jan Sabo, Christian Heiliger, Dorian Andrade, and Andreas RauschmayrAnalysis and interpretation: Lea Henrichs and Mario V. RoserStudy supervision: Michael F. Berger and Alexander H. R. FrankReview of manuscript: Wojciech Konrad Karcz, Jens Werner, and Oliver Muensterer
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is funded by the German Federal Ministry of Economics and Technology (ZIM-BMWI, Grant Number: ZF4592703AW9) Michael F. Berger and Mario V. Roser received a hardware grant of UnternehmerTUM Industrial Innovators LEC GmbH (Initiative for Industrial Innovators) which ended prior to the beginning of funding by German Federal Ministry of Education and Research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
