Abstract
Introduction. Gastroduodenal stenting is efficacious and safe in both benign and malignant foregut diseases. Transgastric duodenal stenting has been described and however requires a gastrostomy tube to remain in situ for 4 to 6 weeks post-procedure which can lead to complications. We present a technique for immediate gastric repair using a suture-mediated vascular closure device, without the need for a gastrostomy tube in porcine models. Methods. Percutaneous access into the stomach was achieved using fluoroscopy. Two or 3 Perclose Proglide devices were pre-deployed. The tract was dilated and a wire advanced into the distal duodenum. A 15.5 cm covered enteric stent was delivered through the gastrostomy, deployed and position confirmed. The gastrostomy was closed using Perclose Proglide sutures. Necropsy leak pressure measurement was performed to assess integrity of gastrostomy closure in the porcine models. Results. Two (n = 8) or 3 (n = 2) Perclose Proglide devices were deployed in ten porcine models, with 1 misfire (4.5%). Percutaneous transgastric access and stent delivery was successful in all porcine models. Mean leak pressure in the animals with adequately deployed devices was 219 mmHg (range 172 mmHg–270 mmHg). Conclusion. This study demonstrates percutaneous transgastric duodenal stenting with immediate gastric repair using suture-mediated vascular closure devices is a feasible procedure.
Introduction
Covered and uncovered stents have been proven to be efficacious, safe, and well tolerated in patients with either malignant or benign duodenal obstruction refractory to conventional treatment. 1 Duodenal stents can be inserted using a combination of endoscopic and fluoroscopic guidance (usually performed by the gastroenterologist or surgeon) or solely under fluoroscopic guidance (performed by the interventional radiologist [IR]). 2
The complex angles of the gastroduodenal tract, stent rigidity, and short deployment devices result in the loss of mechanical advantage as the operator attempts to manipulate these stents across the duodenum via the per oral route. When performed purely using fluoroscopy, a significant amount of length can be lost as a result of gastric looping of wires, increasing the difficulty of duodenal stent deployment. Transgastric percutaneous duodenal stenting performed by IR has been described. 2 However, the current technique requires a gastrostomy tube be left in place for 4 to 6 weeks post-procedure to allow for a mature tract to develop, with the potential for associated complications including gastric leak, infection, and pain. 3
Perclose Proglide (Abbott laboratories, Chicago, Illinois, US) is a suture-mediated vascular closure device routinely used in procedures such as endovascular aortic repair. In this proof of concept animal study, we assess the outcomes and safety of a novel method for percutaneous transgastric duodenal stenting, followed by immediate gastric closure using a vascular closure device, without the need for a gastrostomy tube.
Methods
Study Animals and Preparation
The institutional animal care and use committee approved this study (ICOMETH No. 38.2016.01.085) as well as the French Ministry of Superior Education and Research (APAFIS#8721-2017013010316298-v2). The feasibility and biomechanical principles of the technique were evaluated in ten live porcine models weighing between 25 and 35 kg. All animals were managed in accordance with French laws for animal use and care and also according to the directives of the European Community Council (2010/63/EU), ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines, and National Institute for Health guidelines for use of laboratory animals. 4
The animals were fasted for 24 hours with free access to water before surgery. Premedication was administered 10 minutes before surgery, using an intramuscular injection of ketamine (20 mg/kg) and azaperone (2 mg/kg) (Stresnil, Janssen-Cilag, Belgium). Intravenous propofol (3 mg/kg) combined with rocuronium (0.8 mg/kg) was used for induction. Anesthesia was maintained with 2% isoflurane.
Procedure
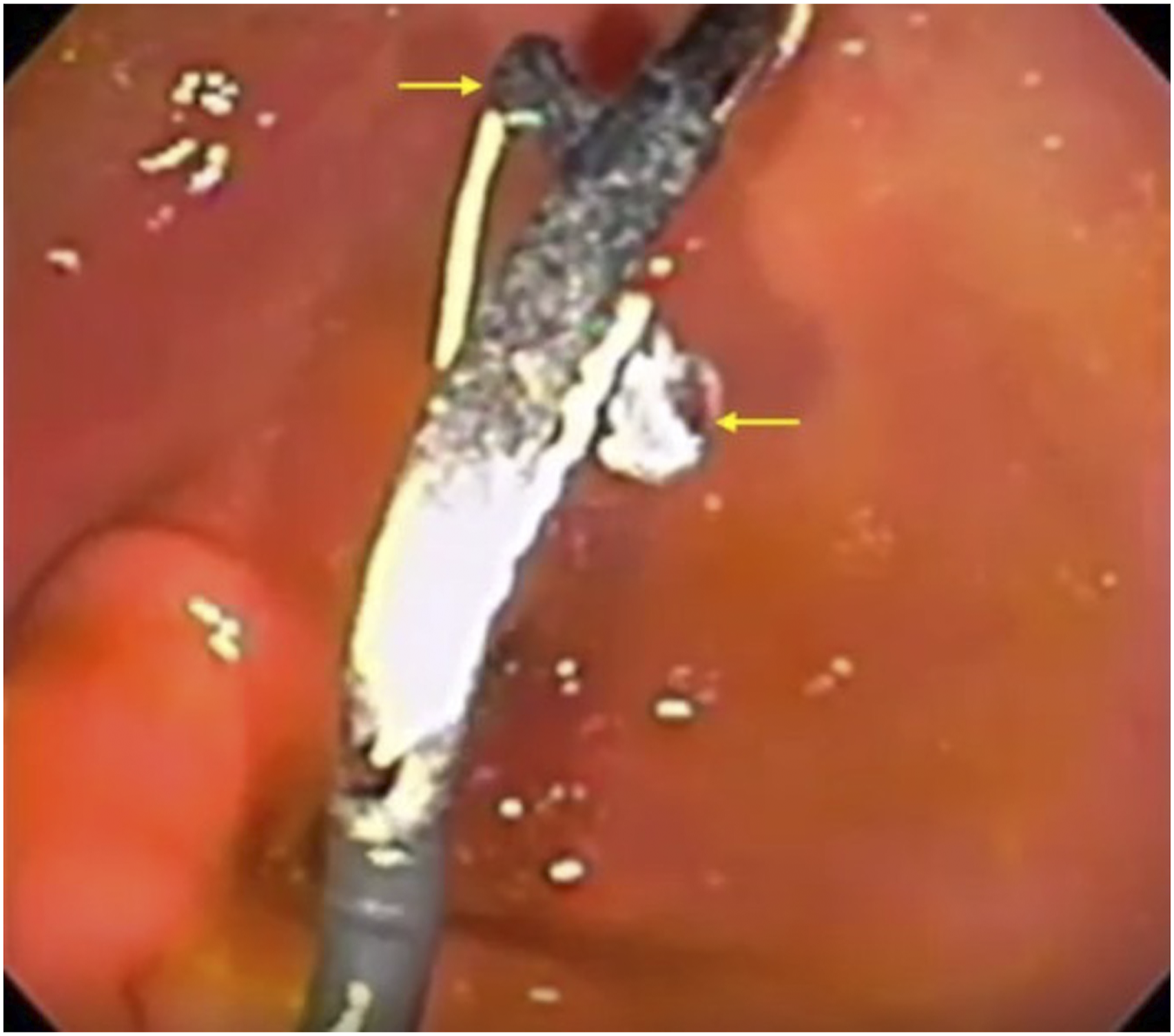
Endoscopy was only utilized in the first porcine model to confirm and document Perclose Proglide deployment but was not used for intervention; in subsequent procedures, endoscopy was not used. A nasogastric tube was passed into the stomach, which was subsequently insufflated with 500 cc of air. Percutaneous access into the stomach was achieved using an 18-gauge 20 cm trocar needle (Cook Medical, Indiana, US) under fluoroscopic guidance. Care was taken to puncture the stomach in a nearly perpendicular fashion to facilitate easy subsequent introduction and deployment of the closure device. The stylet was removed, and contrast was injected to confirm position. A 145 cm Amplatz wire (Cook Medical, Indiana, US) was advanced into the stomach (Figure 1). The tract was pre-dilated using an 8-French dilator (Cook Medical, Indiana, US). Once wire access was obtained, 2 or 3 Perclose Proglide devices were pre-deployed in turn over the wire in the same fashion as described in the device instructions for use, and the sutures left loose (Figure 2). The devices were introduced under fluoroscopic guidance. Endoscopic image demonstrating percutaneous Amplatz wire access into stomach (arrows). Endoscopic image demonstrating deployment of first Perclose Proglide footplate within the stomach (arrows).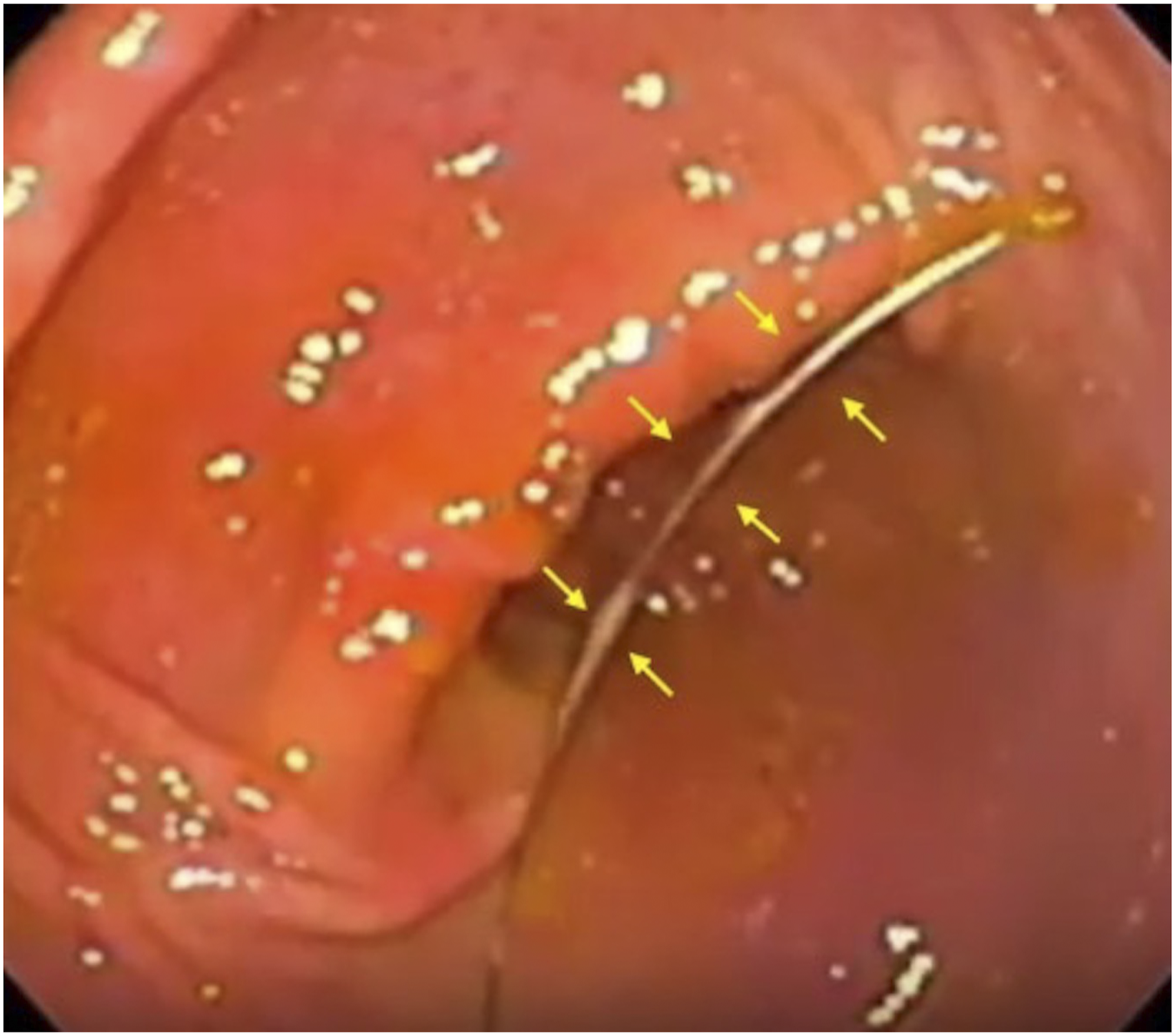
An 8-French Pinnacle sheath (Terumo, Shibuya, Japan) was inserted to prevent air escaping through the gastrostomy. Using a 5-French MPA catheter (Cook Medical, Indiana, US) and Glidewire (Terumo, Shibuya, Japan) system, the duodenum was cannulated under fluoroscopic guidance and the wire was advanced to the duodenojejunal flexure. The Glidewire was exchanged for a super-stiff Amplatz wire (Boston Scientific, Massachusetts, US). The tract was serially dilated to 18-French. A fully covered WallFlex enteric stent (Boston Scientific, Massachusetts, US) on an 18.5-French stent delivery system (Figure 3A) was then deployed over the wire (Figure 3B). Once deployed and confirmed to be in good position, all wires were removed and the gastrostomy closed using the previously placed Perclose Proglide sutures (Figure 4A and 4B). (A) Covered duodenal stent (18.5-French delivery system) passed over Amplatz wire. (B) Stent gradually deployed under fluoroscopic guidance, traversing the second and third parts of the duodenum. (A) Pre-deployed perclose proglide sutures are pulled gently forming a surgical knot on the gastrostomy. (B) The gastrostomy is eventually closed off with no residual gastrostomy defect.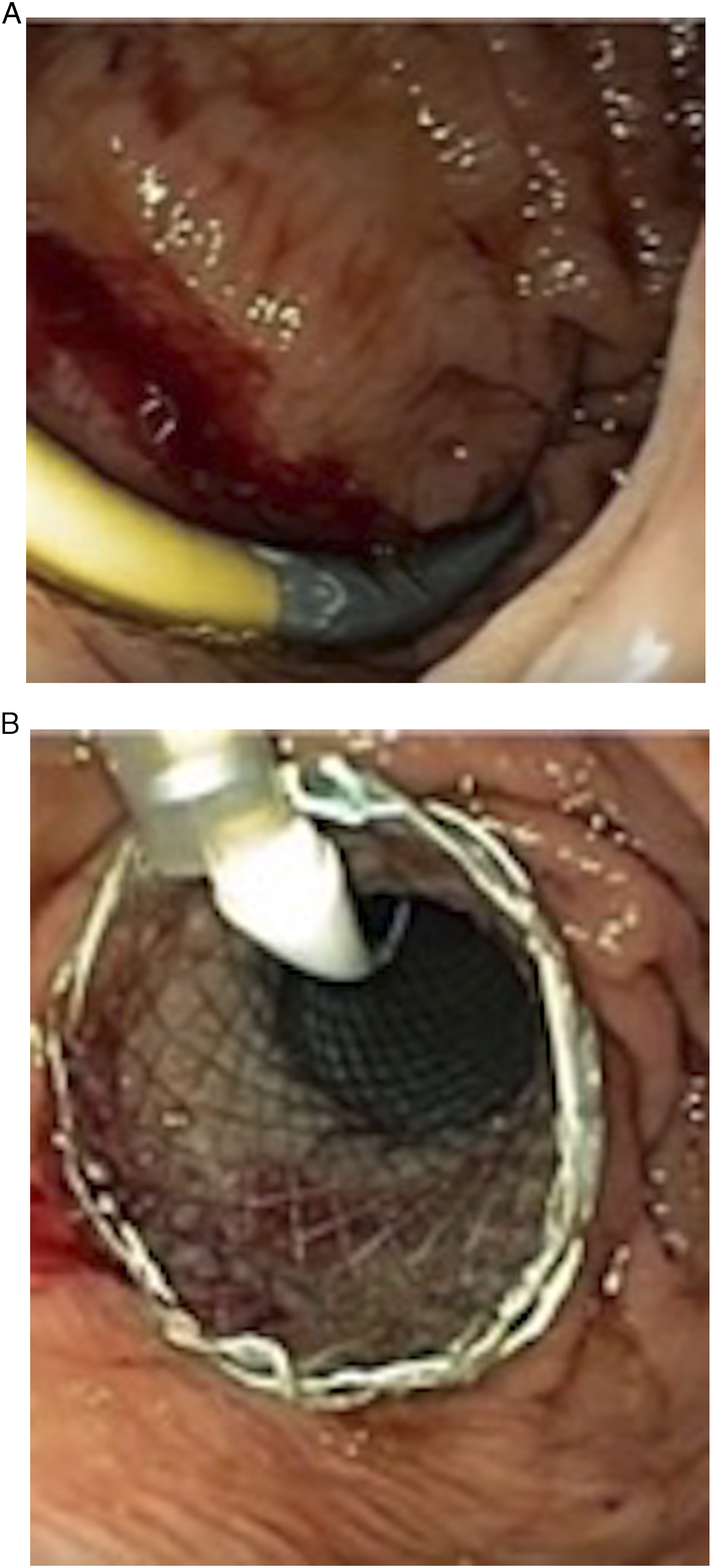
When performing the procedure in patients, the landing zones for the stent could be determined fluoroscopically, by inserting a long sheath distal to the obstruction subsequently slowly retracting under fluoroscopic guidance over the wire, while injecting contrast. This would allow the proximal and distal landing zones to be determined for stent deployment. As the animals in this study did not have duodenal lesions, this step was not required.
At the end of the procedures, pigs were sacrificed with an intravenous injection of pentobarbital sodium (40 mg/kg) (Exagon®, AXIENCE, France), under 5% isoflurane anesthesia and necropsy was performed. The upper tract including the stomach, esophagus, and duodenum in all animals was excised and examined followed by a leak test and pressure measurement.
Porcine Leak Test and Pressure Measurement
Intragastric pressure measurements of the excised stomachs were performed using an angiocath connected to a pressure transducer which was placed into the stomach. A nasogastric tube was used to introduce room air into the stomach. The tube was subsequently removed and the pylorus as well as distal esophagus was clamped to ensure the stomach was air-tight. Manual pressure was applied to the sealed stomach and underwater leak testing of the gastrostomy access and closure site was subsequently performed. Gas bubbles emanating from the gastrostomy site indicated leak and intragastric pressure measurements were obtained.
Results
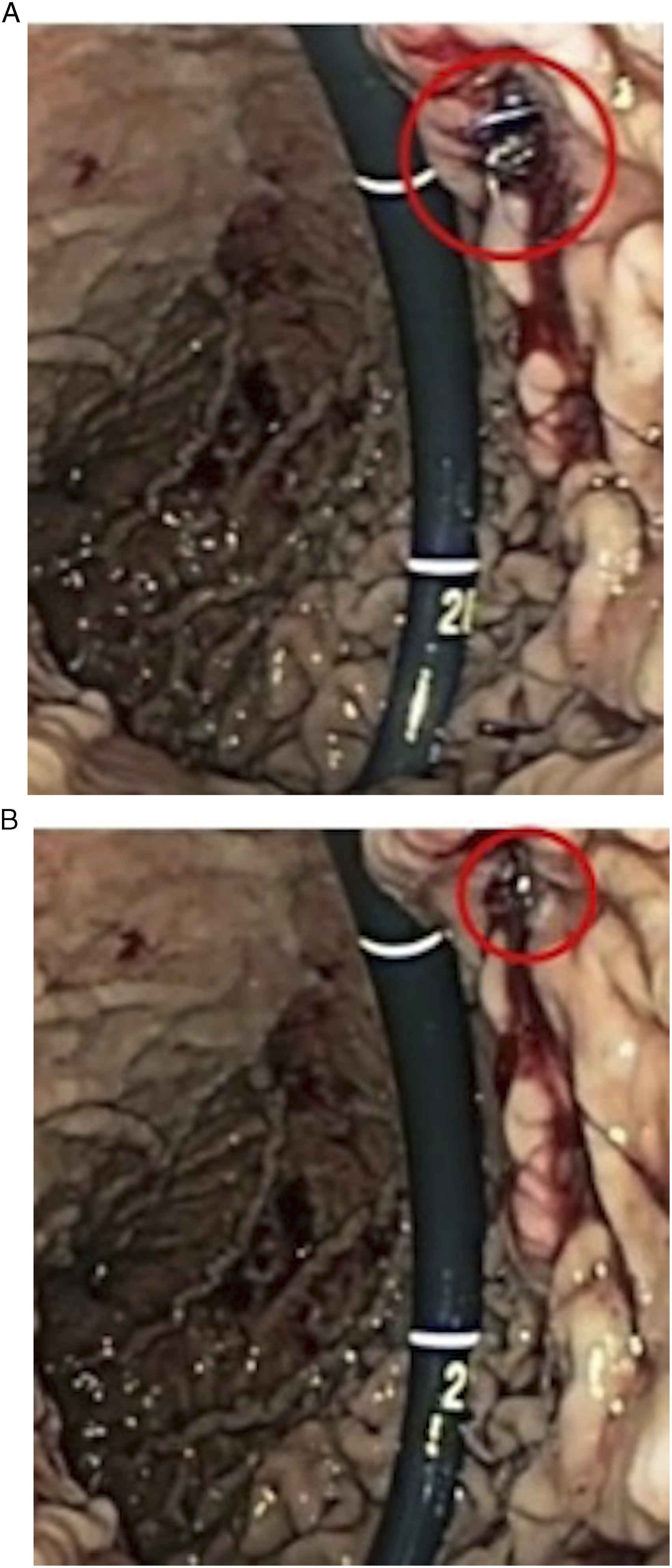
Two (n = 8) or 3 (n = 2) Perclose Proglide devices were deployed in the porcine models with 1 misfire noted (4.5%) which was immediately evident. No iatrogenic injury to adjacent structures occurred. Percutaneous transgastric access and stent delivery using an 18.5-French stent delivery system was subsequently successful in all porcine models. The stomach was noted to be attached to the peritoneum in all porcine cases (Figure 5). Gastrotomy leak pressures were measured in 7 of 10 animals. Leak pressure in the animals with adequately deployed devices was high, with a mean pressure of 219 mmHg (range 172 mmHg–270 mmHg). In the single animal with the mis-deployed device (1 Perclose Proglide suture in place for closure), the leak pressure was 24 mmHg. Successfully deployed Perclose Proglide sutures in the wall of the porcine stomach, fully closing the gastrostomy defect. The stomach was noted to be attached to the peritoneum (circle) in all cases.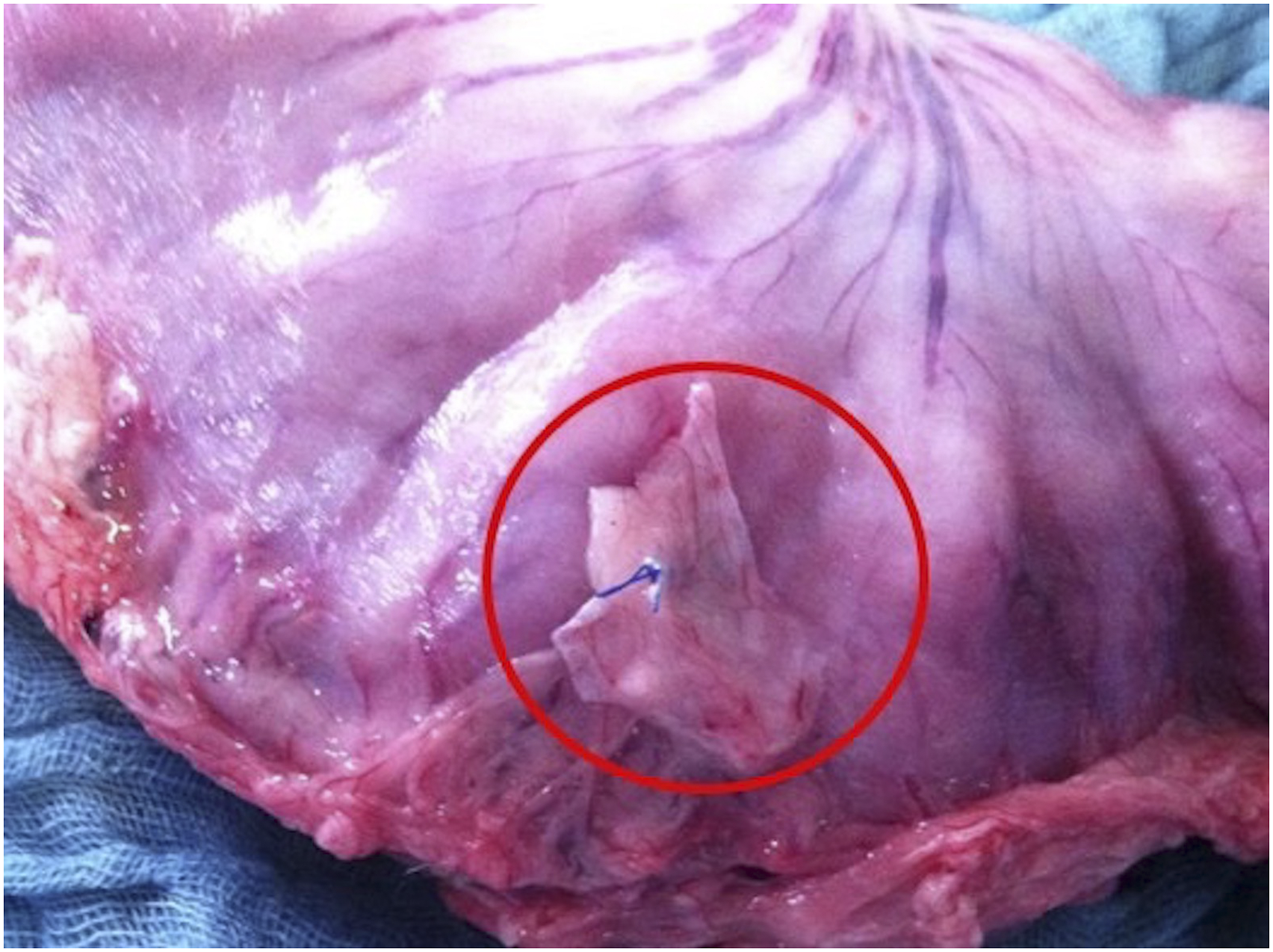
Discussion
This proof of concept pilot study demonstrates that percutaneous transgastric duodenal stenting with immediate gastric closure using a percutaneous vascular access closure device is a safe and feasible procedure. In select cases, the technique has the potential to reduce the difficulty of duodenal stent insertion when the per oral route may be problematic. Furthermore, for percutaneous transgastric procedures, the closure technique eliminates the morbidity and discomfort associated with indwelling gastrostomy tubes. Given radiologically inserted gastrostomy tubes are a relatively common procedure, the skills required to perform transgastric duodenal stent insertion would be possessed by most IRs.
Palliative gastroduodenal stenting is an accepted alternative to surgical gastrojejunostomy formation in patients with malignant gastric outlet obstruction secondary to gastric, duodenal, and pancreatobiliary cancers. Palliative surgery carries significant peri- and post-operative morbidity, exacerbated by the poor nutritional state in this patient group.5-7 Furthermore, the complication rate of gastroduodenal stenting is low, can be organized as an outpatient procedure, and is well tolerated by patients. 8 Less commonly, duodenal stenting may be performed for benign conditions. Benign duodenal strictures caused by peptic ulcer disease have traditionally been treated surgically. Studies have promoted the role of stenting in patients with inoperable benign duodenal stenosis and however have sited the risk of stent migration.9,10
The debate over the superiority of covered vs uncovered stents is also ongoing; next generation devices attempt to overcome both stent migration and tumor overgrowth, which reflect the major disadvantages of currently available covered and uncovered stents, respectively.7,11-13 Regardless of the type of stent chosen, the major drawback of performing gastroduodenal stenting via the per oral route is the challenging anatomical tortuosity of the upper gastrointestinal tract. When attempting to deliver the stent orally under fluoroscopic guidance alone, this tortuosity combined with the rigidity of the stent delivery system results in the loss of mechanical advantage as the operator attempts to manipulate the stent into the duodenum. Despite these disadvantages, the quoted success rate of duodenal stenting performed under fluoroscopic guidance by interventional radiology remains high, with studies quoting successful outcomes in between 94% and 100% of cases. 14 Therefore, the transgastric route for duodenal stenting is usually reserved as a secondary option in cases where the duodenal lesion cannot be crossed via the per oral route. 15
Although transgastric duodenal stenting has previously been described, which overcomes the aforementioned technical disadvantages, a gastrostomy tube is required to be left in situ until a mature tract has formed.2,15,16 As a result, potential complications associated with gastrostomy placement may arise including leakage of gastric contents, peritonitis or local infection, and abscess formation, with incidence rates ranging from 4% to 30%.17,18
Perclose Proglide is a suture-mediated vascular closure device routinely used in vascular procedures. When using the pre-closure technique, Perclose Proglide devices can be used to close arteriotomies and venotomies up to a maximum outer sheath diameter of 26-French and 29-French, respectively. 19 The use of Perclose Proglide for gastrostomy closure has recently been described in both in vitro and in vivo porcine model experiments by a South Korean group.20,21 One in vitro study concluded the use of Perclose Proglide was as effective as manual suturing in successfully closing gastrostomy openings. 20 Furthermore, histological analysis of the percutaneously closed gastrostomy site performed 2 weeks post-procedure in a second experiment demonstrated complete wound healing in all animals. The current study builds upon this work by successfully deploying duodenal stents in all animals and subsequently performing in vivo percutaneous duodenal stent delivery and gastrostomy closure with Perclose Proglide devices. Previously, various devices have been deployed successfully with regard to gastrostomy closure after natural orifice transluminal endoscopic surgery (NOTES) including standard endoscopic clips, T-bars, tip-mounted sutures, and padlock G-clips.22-26 Such devices however primarily require endoscopic control and are designed for larger gastrostomy openings when compared with percutaneous gastrostomy closure with Perclose Proglide. The technique described in the current study is advantageous in situations where endoscopic duodenal stent insertion may not be technically feasible.
Percutaneous gastric access with immediate closure using the Perclose Proglide device has the potential to be adapted for use in a variety of other percutaneous transluminal interventions. Novak et al. 27 demonstrated the use of transgastric esophageal stent placement in patients who were unable to undergo antegrade per oral stent placement due to anatomical and technical reasons. The ability to provide durable and immediate gastric closure negates the need to leave an indwelling gastrostomy tube, thus reducing patient discomfort and making percutaneous duodenal stenting a viable alternative to the endoscopic and fluoroscopic per oral technique.
Limitations of this study include a lack of control for comparison. Future human studies should be performed to compare outcomes related to primary percutaneous closure of gastrostomy opening, with leaving a gastrostomy feeding tube in situ. Studies should also compare outcomes of percutaneous gastrostomy closure with and without gastropexy, the former technique not utilized in this study. Finally, leak testing and pressure measurements were performed in vitro which do not account for peristalsis, blood flow, and fasting which may also affect the integrity of gastrostomy closure. 20
This pilot animal study demonstrates percutaneous transgastric duodenal stenting with immediate gastric repair using suture-mediated vascular access closure devices is a feasible procedure and avoids a gastrostomy feeding tube remaining in situ. The technique may be an alternative for radiologic duodenal stenting performed by IR when the per oral route is unsuccessful or not possible and also in centeres where interventional endoscopy is not available.
Footnotes
Author Contribution
Study concept and design: Eran Shlomovitz, Michele Diana, Radu Pescarus, Lee L. Swanström
Acquisition of data: Eran Shlomovitz, Neeral R. Patel, Michele Diana, Radu Pescarus, Lee L. Swanström Analysis and interpretation: Eran Shlomovitz, Neeral R. Patel, Michele Diana, Radu Pescarus, Lee L. Swanström
Study supervision: Eran Shlomovitz, Lee L. Swanström
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
