Abstract
Background. Indocyanine green angiography (ICGA) offers the potential to provide objective data for evaluating tissue perfusion of flaps and reduce the incidence of postoperative necrosis. Consensus on ICGA protocols and information on factors that have an influence on fluorescence intensity is lacking. The aim of this article is to provide a comprehensive insight of in vivo and ex vivo evaluation of factors influencing the fluorescence intensity when using ICGA during reconstructive flap surgery. Methods. A systematic literature search was conducted to provide a comprehensive overview of currently used ICGA protocols in reconstructive flap surgery. Additionally, ex vivo experiments were performed to further investigate the practical influence of potentially relevant factors. Results. Factors that are considered important in ICGA protocols, as well as factors that might influence fluorescence intensity are scarcely reported. The ex vivo experiments demonstrated that fluorescence intensity was significantly related to dose, working distance, angle, penetration depth, and ambient light. Conclusions. This study identified factors that significantly influence the fluorescence intensity of ICGA. Applying a weight-adjusted ICG dose seems preferable over a fixed dose, recommended working distances are advocated, and the imaging head during ICGA should be positioned in an angle of 60° to 90° without significantly influencing the fluorescence intensity. All of these factors should be considered and reported when using ICGA for tissue perfusion assessment during reconstructive flap surgery.
Introduction
Postoperative (partial) flap necrosis is one of the most feared complications in reconstructive flap surgery for both the patient and the surgeon. With an incidence of 4 to 16% depending on flap type, it represents a significant problem.1-3 Flap necrosis can lead to slower recovery, infection, repeat surgery, delayed adjuvant therapy, and increased health care costs. Patients may even encounter psychological distress with a decline in their quality of life.1,4 In order to minimize the risk of necrosis, surgeons need to be able to objectively evaluate tissue perfusion during surgery, as partial or even total flap loss may be prevented by immediate intervention whenever perfusion appears to be insufficient. Likewise, consequences of insufficient flap edge circulation, including postoperative wound dehiscence and fat necrosis, could be prevented during surgery. 5
The current gold standard for evaluating tissue perfusion relies on the surgeon’s clinical judgement, that is, the subjective evaluation of tissue color, flap temperature, capillary refill, and assessment of dermal edge bleeding. Although this method is accurate in 84 to 96% of cases (depending on flap type), the accuracy depends highly on the surgeon’s experience and expertise, whereas (by definition) it is also restricted by the visual performance limits of the human eye. Evidence suggests that clinical judgement alone is an unreliable predictor of insufficient tissue perfusion. 6 Therefore, various medical imaging modalities are being developed to obtain real-time assessment of tissue perfusion in an objective and reproducible manner. One such innovative technique is near-infrared fluorescence (NIRF) imaging using indocyanine green (ICG), also known as indocyanine green angiography (ICGA). 5
Since Flower and Hochheimer developed an imaging technique to evaluate choroidal circulation routinely in 1976, 7 the principle has been adapted to currently available imaging devices. ICGA uses ICG, a water-soluble tricarbocyanine dye, as a contrast agent. Following intravenous administration, ICG is rapidly and extensively bound to plasma proteins, making it an ideal contrast agent to evaluate tissue perfusion. 8 When exposed to near-infrared excitation in the wavelength range of 750 to 810 nm, ICG reemits light (fluorescence) with a wavelength of approximately 840 nm. A dedicated digital video camera, which filters out the excitation light, allows the fluorescence of ICG to be recorded in real time. 9
ICGA offers the potential to provide objective data to support intraoperative decision-making regarding flap design and is a useful adjunct for evaluating tissue perfusion of flaps. Reported sensitivity and the accuracy of ICGA are 90.9% and 98.8%, respectively. 10
Clinical use of ICG has proved to be safe in humans. The incidence of adverse events is about 1 in 42 000 patients. 11 Furthermore, ICG has a plasma half-life of approximately 3 to 5 minutes, which allows multiple injections throughout a procedure, limited up to a safe maximum dose of 5 mg/kg. 8
ICGA is currently explored for multiple applications in surgery. 12 In plastic and reconstructive surgery, it is used predominantly to assess tissue perfusion in (free) flap surgery.5,9,13-18 Although recommendations for use of ICGA have been previously reported, 18 there is still no consensus about the technical use of ICGA during reconstructive surgery, including timing of evaluation and optimal intravenous dose of ICG. 5 This is important because ICG dosage influences fluorescence intensity, thereby influencing the adequacy of perfusion assessment. 19 On top of that, there are other factors that can have an impact on fluorescence intensity within the collected images, including the distance and angle between the camera and region of interest, and ambient light during perfusion evaluation. These are either not considered to be important or only briefly described in current literature. Since ICGA is rapidly being introduced in clinical practice worldwide, it is important that surgeons are aware of factors that potentially play a role with regard to the feasibility of this imaging modality. Therefore, this study aims to provide a comprehensive insight in potential factors influencing the fluorescence intensity when using ICGA by performing a systematic review, regarding all studies reporting on ICGA to assess tissue perfusion during reconstructive flap surgery, and ex vivo experiments.
Materials and Methods
This report is composed of 2 parts. First, a systematic search of the literature was conducted to provide a comprehensive overview of currently used ICGA protocols in reconstructive flap surgery, focusing on ICG dosage, timing of both application and assessment, working distance, and other possible influencing factors as discussed above. In the second part, ex vivo experiments were performed to further investigate the practical influence of potentially relevant factors.
Systematic Review
A systematic literature search was conducted in July 2018 in the following databases: National Library of Medicine (PubMed) database, EMBASE database (via OvidSP), and Cochrane Library CENTRAL, using the methodology described in the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement. 20 The following terms were used (including synonyms and closely related words) as index terms or free-text words: “flap surgery,” “indocyanine green,” “angiography,” “perfusion,” and “imaging.” The search syntax applied to each database, and the PRISMA 2009 checklist are attached as Supplementary Material (available online). First, all titles and abstracts derived from the search were screened and independently reviewed for eligibility by 2 researchers (TP and RMS). There were no restrictions on language in this review. Letters and comments on articles, conference abstracts, case reports including less than 10 flaps, studies conducting research other than on human subjects, reviews, and meta-analyses were excluded. Studies were considered eligible if they
Reported on ICGA in free flap, pedicled flap, or mastectomy skin flap surgery
Reported on ICGA to assess tissue perfusion
Described an ICGA protocol
In case of uncertainty, full-text reports were screened to determine eligibility. Any differences in the resulting derived articles were discussed by the 2 aforementioned researchers. If no consensus was reached, a third author (SSQ) decided after discussion. Other sources including the reference lists of included articles and recent review articles were screened for relevant articles not identified by the online databases based on previously described criteria. A data extraction sheet was developed containing items on the type of study included, the operated flap type, the applied imaging system, the dose of ICG, the working distance (eg, distance from imaging head to tissue), the timing of evaluation, the timing from administration of ICG to evaluation, the method to evaluate tissue perfusion, and the decision that was taken to excise tissue in the study. The data extraction sheet was completed for all eligible studies by 2 independent researchers (TP and RMS).
Ex Vivo Experiments
The methods for the ex vivo experiments have been previously reported by 2 of the authors (JVDB and FPW), using a laparoscopic NIRF imaging system. 19 In this study, a handheld NIRF camera (Fluobeam, Fluoptics, Grenoble, France) was applied, provided with integrated near-infrared light source with excitation between 750 and 800 nm and maximum fluorescein emission detection between 780 nm and 850 nm. In the author’s institution, this system is used in daily practice for perioperative tissue perfusion evaluation as well as for mapping of lymphatic collecting vessels in the outpatient clinic.21,22
Experiments were performed using ICG diluted within 40 mg/mL albumin in a 0.9% NaCl dilution. This was done accordingly as ICG is considered to bind to albumin in vivo, which modifies its optical properties; 40 mg/mL was chosen as a stable point within the normal reference range for serum albumin, which is 35 to 55 g/L. In all experiments, a total of 18 different concentrations of ICG were used, ranging between 0.01 and 0.0001 mg/mL, representing 50 to 0.5 mg of total dose of ICG administered intravenously in a female patient, weighing 77.0 kg, with a blood volume of 5000 mL, estimated using MedCalc300 (Medscape, 2018). A bodyweight of 77.0 kg was considered average after obtaining chart data on 25 consecutive patients, who had undergone a deep inferior epigastric artery flap in the authors’ institution. 2
Next to the ICG dilutions, wells plates and beeswax plates were used for this experiment. From each dilution, 9 times 3 mL of the ICG-containing mixture was placed on a wells plate in order to completely fill the wells with fluid, to minimize fluid-to-beeswax plate air layer. The influence of distance was measured fixating the imaging head at 12 distances varying from 50 to 5 cm from the surface of the dye. This was then repeated for all distances with, respectively, 1 and 2 beeswax plates of exactly 1 mm thickness, stacked to the wells plate. Beeswax plates (Stockmar, Kaltenkichen, Germany) were chosen because it approaches the scattering behavior and translucent light penetration of human tissue. 23 The experiments were performed in darkness (windows covered) with only one computer screen left on. The aforementioned experiment with 1 beeswax plate was repeated with uncovered windows to measure the influence of ambient light. The influence was measured at a distance of 15 cm for all ICG dilutions. The fluorescence intensity of the middle cup was measured at incident angles of 90°, 75°, 60°, and 45° between the imaging head and middle wells surface plane. The penetration depth was evaluated with the use of beeswax plates progressively stacked one by one to increase thickness until it was not possible anymore to distinguish the dilution-filled wells from its surroundings. In addition, the influence of beeswax plates themselves on fluorescence intensity was analyzed. The setup of the ex vivo experiment is illustrated in Figure 1. In all experiments, fluorescence intensity was measured on a grayscale from 0 to 255 using ImageJ software (Version 1.51, ImageJ, National Institutes of Health, Bethesda, MD). Zero is black and 255 is white on this scale. Values in between make up different shades of gray.

Setup of the ex vivo experiment.
Statistical Analysis
The association between dose and fluorescence intensity for different experimental conditions were first visualized using scatter plots with lines fitted using either linear spline regression, in case of an observed ceiling effect, or locally weighted scatterplot smoothing (LOESS), in case of an observed curvilinear association.
To quantify the associations between covariates of the experiment and fluorescence intensity, regression coefficients were estimated using multivariable, or adjusted, linear regression analysis. No univariate analyses were performed as the estimates would be too dependent on the setting of other covariates of the experiment. In case a ceiling effect was present, observations for which the intensity was >254 on the 0 to 255 scale were omitted to prevent biased estimates. Curvilinear associations were estimated using polynomial regression (ie, the linear regression model was extended with quadratic and cubic terms for continuous variables and tested for significance). All analyses were performed using R version 3.3.3 (R Foundation for Statistical Computing, Vienna, Austria) and the rms package version 5.1-0.
Results
Systematic Review
Forty-nine articles,24-72 including 1996 surgical flaps, were selected for the review. Study designs included prospective cohort studies (n = 27),31,34,35,38,39,41-43,47-50,56,58-60,62-71 retrospective cohort studies (n = 16),25-27,29,32,33,36,40,44,45,51,55,57,61,72 prospective pilot studies (n = 3),28,37,52 and retrospective case series (n = 3).46,53,54 No randomized controlled trials have been performed yet that concern ICGA in plastic and reconstructive flap surgery. A detailed overview of the study selection is presented in a PRISMA flow chart (see Figure 2). A summary of findings from the included articles is presented in Table 1.
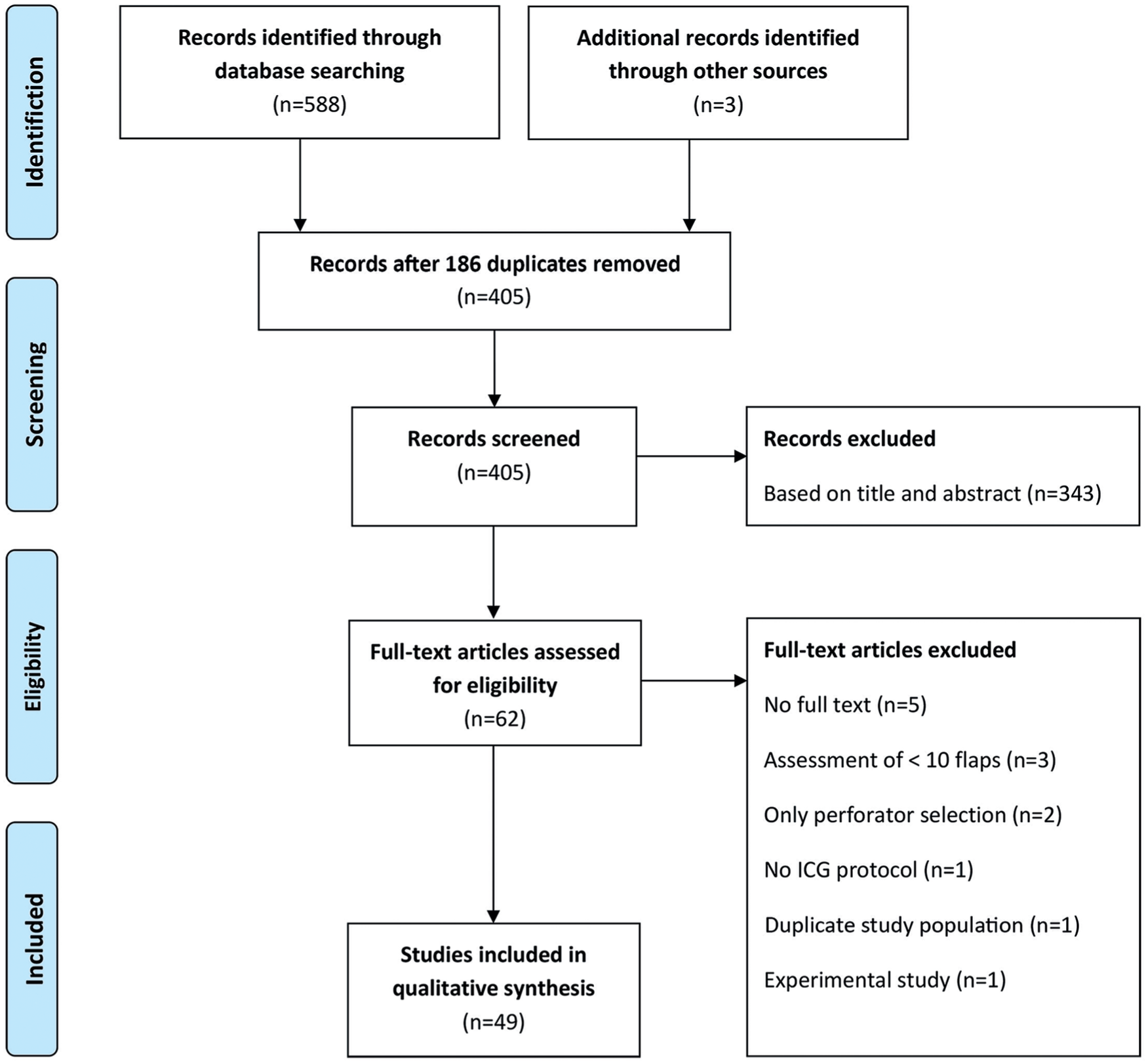
PRISMA flowchart showing selection of articles for review.
Summary of Findings From the Component Articles.
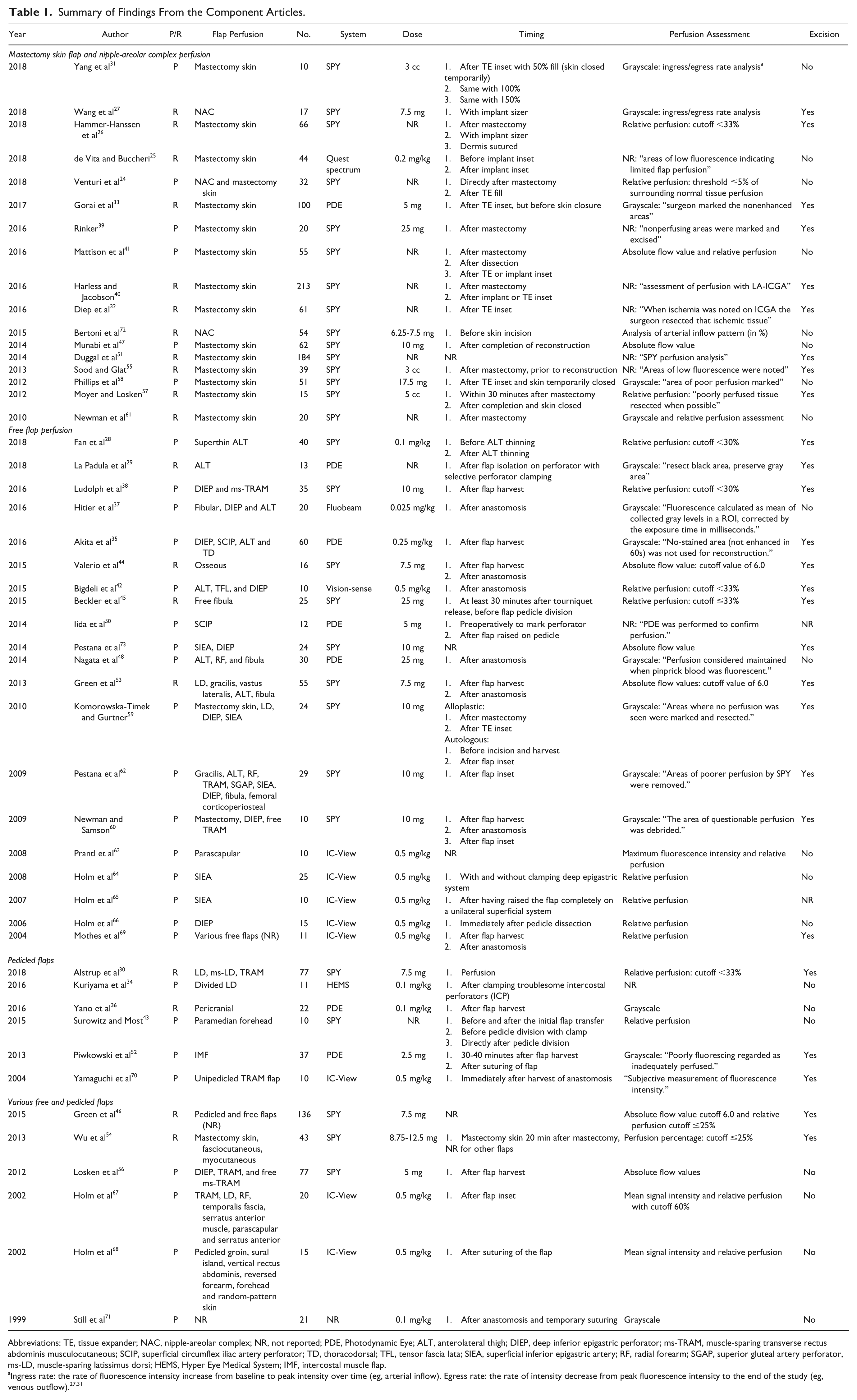
Abbreviations: TE, tissue expander; NAC, nipple-areolar complex; NR, not reported; PDE, Photodynamic Eye; ALT, anterolateral thigh; DIEP, deep inferior epigastric perforator; ms-TRAM, muscle-sparing transverse rectus abdominis musculocutaneous; SCIP, superficial circumflex iliac artery perforator; TD, thoracodorsal; TFL, tensor fascia lata; SIEA, superficial inferior epigastric artery; RF, radial forearm; SGAP, superior gluteal artery perforator, ms-LD, muscle-sparing latissimus dorsi; HEMS, Hyper Eye Medical System; IMF, intercostal muscle flap.
Flap Perfusion: Clinical Applications
Indocyanine green angiography has been reported for several types of flaps in plastic and reconstructive flap surgery in a clinical setting. The majority of included articles used ICGA to assess tissue perfusion of mastectomy skin flap (n = 14) intraoperatively25,26,31-33,39-41,47,51,55,57,58,61; the remainder focused on the intraoperative assessment of nipple-areolar complex perfusion,27,72 a combination of nipple-areolar complex and mastectomy skin flap perfusion, 24 anterolateral thigh flap,28,29 deep inferior epigastric perforator flap,38,56,66 transverse rectus abdominis myocutaneous flap,38,56,70 superficial inferior epigastric artery flap,64,65,73 superficial circrumflex iliac artery flap, 50 free pericranial flap, 36 latissimus dorsi flap, 34 free parascapular flaps, 63 paramedian forehead flap, 43 intercostal muscle flap, 52 osseus free flaps, 44 free fibula flap, 45 and various pedicled and/or free flaps.30,35,37,42,46,48,53,54,59,60,62,67,69,71 Two studies described preoperative perforator selection in addition to perfusion assessment50,73 (see also Table 1).
Imaging Systems
Several ICGA systems have been described in the literature. In the majority of included studies (n = 29),24,26-28,30-32,38-41,43-47,51,53-62,72,73 the SPY imaging system (Novadaq Technologies, Inc, Toronto, Canada) was used for tissue perfusion assessment; the remainder used the IC-View System (Pulsion Medical Systems, Munich, Germany; n = 8),63-70 the Photo Dynamic Eye (Hamamatsu Photonics KK, Hamamatsu, Japan; n = 7),29,33,35,36,48,50,52 the Visionsense 3D high-definition near-infrared-guided indocyanine green video angiography system (ICG-NIR-VA, Orangeburg, NY; n = 1), 42 Hyper Eye Medical System (Mizuho Medical Co, Ltd, Tokyo, Japan; n = 1), 34 the Fluobeam device (Fluoptics, Grenoble, France; n = 1), 37 and Quest spectrum TM (Quest Medical Imaging, Akron, OH; n = 1). 25 One study did not state what kind of device was used. 71
Dye and Dosing
In almost every study (n = 48), conventional ICG was used as fluorescent dye. In only one of the included articles, monopeak infracyanine green (Infracyanine, SERB Laboratory, Paris, France) was used. 37 The latter concerns an iodine-free preparation.
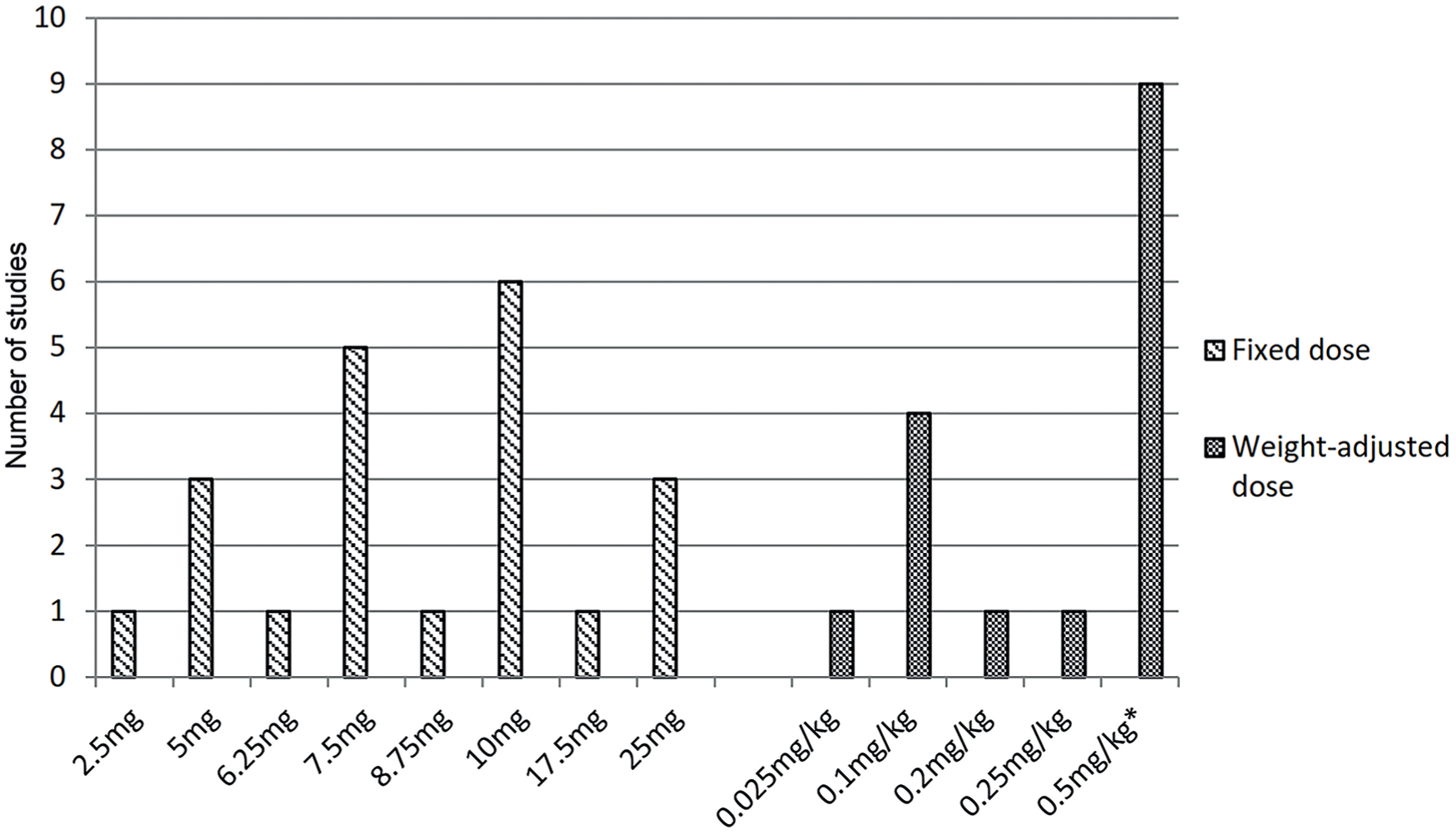
The majority (n = 37) of included articles reported a clear dosing regimen for ICGA, ranging from 2.5 to 25 mg or 0.025 to 0.5 mg/kg when a fixed dose (n = 21)27,30,33,38,39,44-48,50,52-54,56,58-60,62,72,73 or weight-adjusted dose (n = 16)25,28,34-37,42,63-69,71,74 was applied, respectively. Nine studies did not report any ICG dosing.24,26,29,32,40,41,43,51,61 Three reports described a dose of 3 and 5 cc but did not report the concentration of ICG55,57,75 (see Table 1). Ten milligrams and 0.5 mg/kg were the most frequently used fixed dose and weight-adjusted dose, respectively (see Figure 3). In addition, 9 articles report a flush of saline following ICG injection.25,27,35,47,55,56,59,72,75
Working Distance
The working distance, defined as the distance between the imaging head of the ICGA system and the area of interest (ie, skin of the flap), is reported in only 11 articles. Working distances of 20 cm,25,37,39,57 30 cm,33,42,60 30 to 100 cm,63,69 and 20 to 40 cm 48 have been reported. Valerio et al report a 2-dot laser-guided marker in the SPY system, which aids in identifying the optimal distance from soft tissue. 44
Time to and Duration of Assessment
The time to assessment, defined as the elapsed time from dye administration to perfusion assessment, is reported in 21 of the included studies. Most of the studies describe recording directly following ICG administration (n = 11),27,28,30,36,39,44,47,48,53,63,71 while other studies start recording 15 seconds (n = 3)29,49,59 or 60 seconds (n = 2) after intravenous administration, respectively.52,56 Five studies report to start recording when first fluorescence change is detected in the flap (n = 5).31,33,37,41,60 Total duration of ICGA from start of recording to the end is reported in 14 studies and is predominantly ranging between 60 and 120 seconds.27,29,30,33,39,41,47,50,54,62 Assessments of up to 20031,58 and 300 seconds36,71 have been reported as well.
Intraoperative Timing of ICGA
Timing, defined as the moment of perfusion assessment during operation, varies with the flap type to be assessed. In the included articles, no major difference exists in timing of assessment. For example, mastectomy skin flap perfusion is mainly assessed after mastectomy, prior to and after inset of an implant. 26 Free flaps are predominantly assessed after flap harvest when the flap is raised on its pedicle and/or after transplantation of the flap to the recipient site 44 (see also Table 1).
Resection of Tissue and Perfusion Assessment
A total of 27 studies used ICGA imaging to guide resection of insufficiently perfused tissue (see Table 1).26-30,32,33,35,38-40,42,44-46,51-55,57,59,60,62,69,70,73 The most applied methods to assess tissue perfusion were
Relative perfusion assessment (ie, “percentage of perfusion”)
Assessment of fluorescence intensity with grayscale imaging
Absolute flow value assessment, based on grayscale imaging
In 18 studies, quantitative software was used to calculate relative perfusion of the region of interest, compared with normal tissue quantified as a reference with 100% perfusion.24,26,28,30,38,42,43,45,54,57,64-69 Perfusion assessment using absolute flow value is reported in 9 studies41,44,46,47,53,56,61,63,73 and a combination of the aforementioned in 3 studies.41,46,63 In 9 of these studies, cutoff values ranging from 25 to 60% to excise flap tissue have been reported for relative perfusion.26,28,30,38,42,45,46,54,67 For absolute flow value assessment, in which a point value from 0 to 255 is based on a grayscale that corresponds to the signal intensity, higher values equate to superior perfusion. In the remaining 3 studies, a value of 6.0 was reported as the lower limit of acceptable perfusion44,46,53 (see Figure 4). For example, Green et al report that areas of poor flap perfusion with absolute flow value under 6.0, as objectively assessed with SPY Q analysis software, were excised before definitive inset of the flap. 53 Perfusion assessment according to grayscale imaging was performed in 12 studies.27,29,31,33,36,48,52,58-60,62,71 For example, Gorai et al marked the nonenhanced areas according to the grayscale image on the monitor. 33
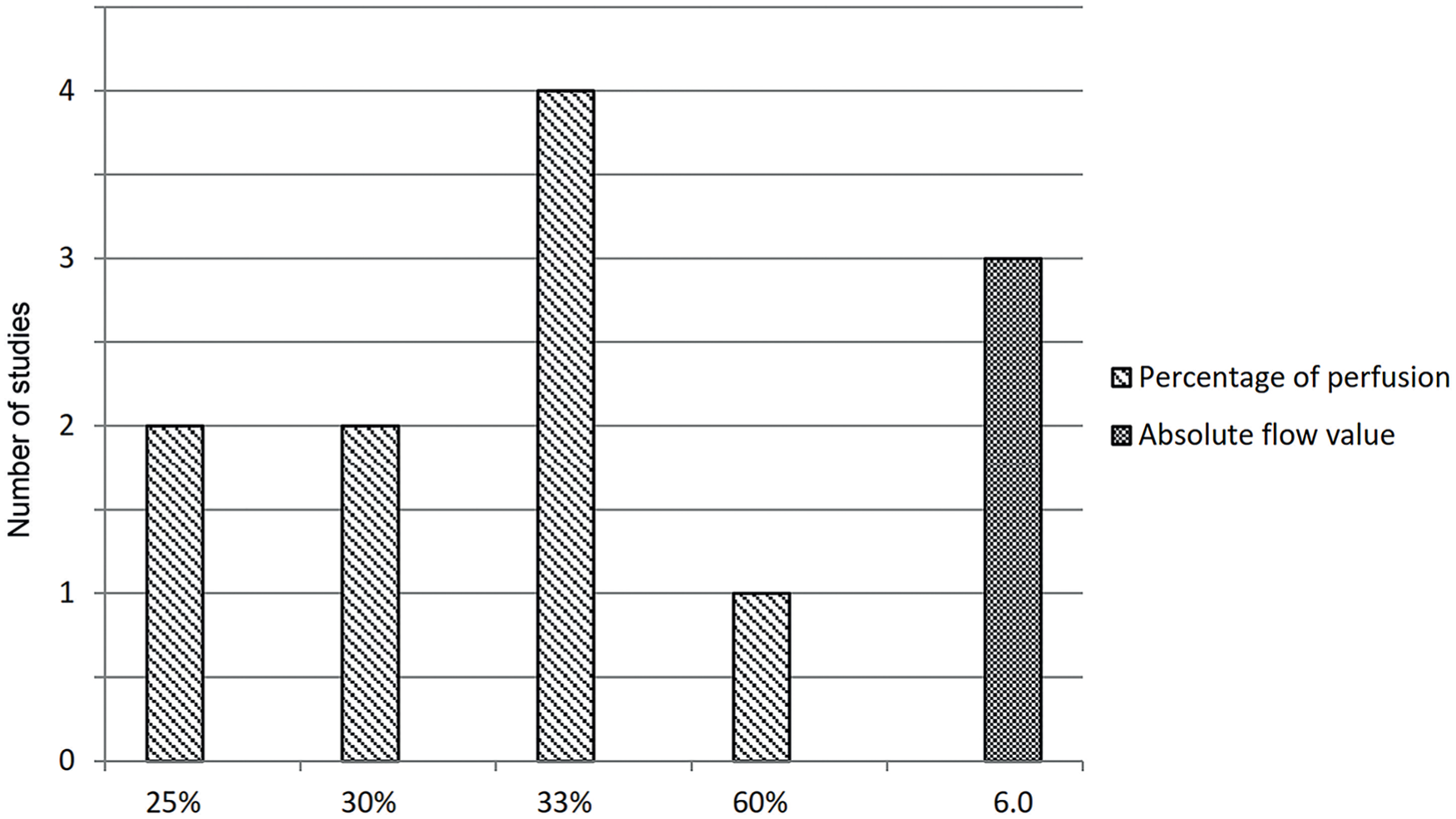
Frequencies of studies reporting a cutoff value for tissue excision using indocyanine green angiography to measure tissue perfusion as percentage of perfusion (scale 0 to 100%) or as absolute flow value (scale 0 to 255 based on a grayscale).
Factors of Influence During ICGA
Only 12 of the included studies report factors that might influence the assessment of tissue perfusion, including ambient light,31,35,39,42,47,70 the use of epinephrine containing injections,26,31-33,47,54,58 the use of papaverine or other vasodilating agents, 66 systolic blood pressure, 30 stretch level of the mastectomy skin flap, 39 use of absorbent compress surrounding or underneath the flap in order to reduce artifacts,28,42,52 use of the electric knife during assessment, 70 and the angle of the imaging head to the region of interest. 70
Of these studies, only 4 explicitly report that all operating room lights were turned off during the recording to avoid interference of ambient light with the detection of fluorescence.35,39,47,70 One study reported that ICGA was always performed under room light conditions. 42 None of these studies report the influence of ambient light on assessment.
Diep et al found that more patients developed severe flap necrosis when they received tumescence-containing epinephrine during their mastectomy. 32 Due to the difficulty in interpreting ICGA, the authors discontinued the use of tumescent solution. Munabi et al observed false-positive results in flap assessment due to the use of the tumescent technique, rendering ICGA less reliable to predict necrosis. 47 Hammer-Hansen et al report that no local anesthetics with adrenalin were used at any time during surgery in order to not impair visualization of the mastectomy flap perfusion when performing ICGA. 26 The other studies considering epinephrine as an influencing factor only describe that no epinephrine was injected into the surgical site.33,54,58,75
Alstrup et al performed ICGA measurements with a mean systolic blood pressure above 100 mm Hg and during the assessment, a Doppler confirmed flow through the pedicle. 30 Yamaguchi et al report that systolic and diastolic blood pressure is registered at the beginning of the analysis, without describing the purpose. 70
Rinker placed laparotomy sponges in the breast pocket after completion of the mastectomy, prior to ICGA, to fill the dead space and to allow the skin flaps to lie flat without areas of redundancy, but also without stretch. 39 Two reports described that flaps were surrounded with clean surgical towels during ICGA assessment to avoid background signal noise from the other vascular tissues.28,52 Another group used an absorbent compress to fill the dead space underneath the flap in order to reduce artifacts. 42
Yamaguchi et al reported that the electric knife had to be switched off to prevent artifacts in ICGA imaging, a commonly seen interference by electromagnetic interference. The authors also suggest that ICGA recording should be performed before flap reshaping, since the video camera must be perpendicular to the flap surface. 70
Ex Vivo Experiments
The results of the ex vivo experiments are depicted in Figures 5 and 6. These figures illustrate that fluorescence intensity is associated with each of the factors that were independently varied in the experiment, including positioning of the imaging head in various degrees (ie, 90°, 75°, 60°, and 45°), various distances, with or without ambient daylight and with stacking beeswax plate to mimic “penetration depth.”
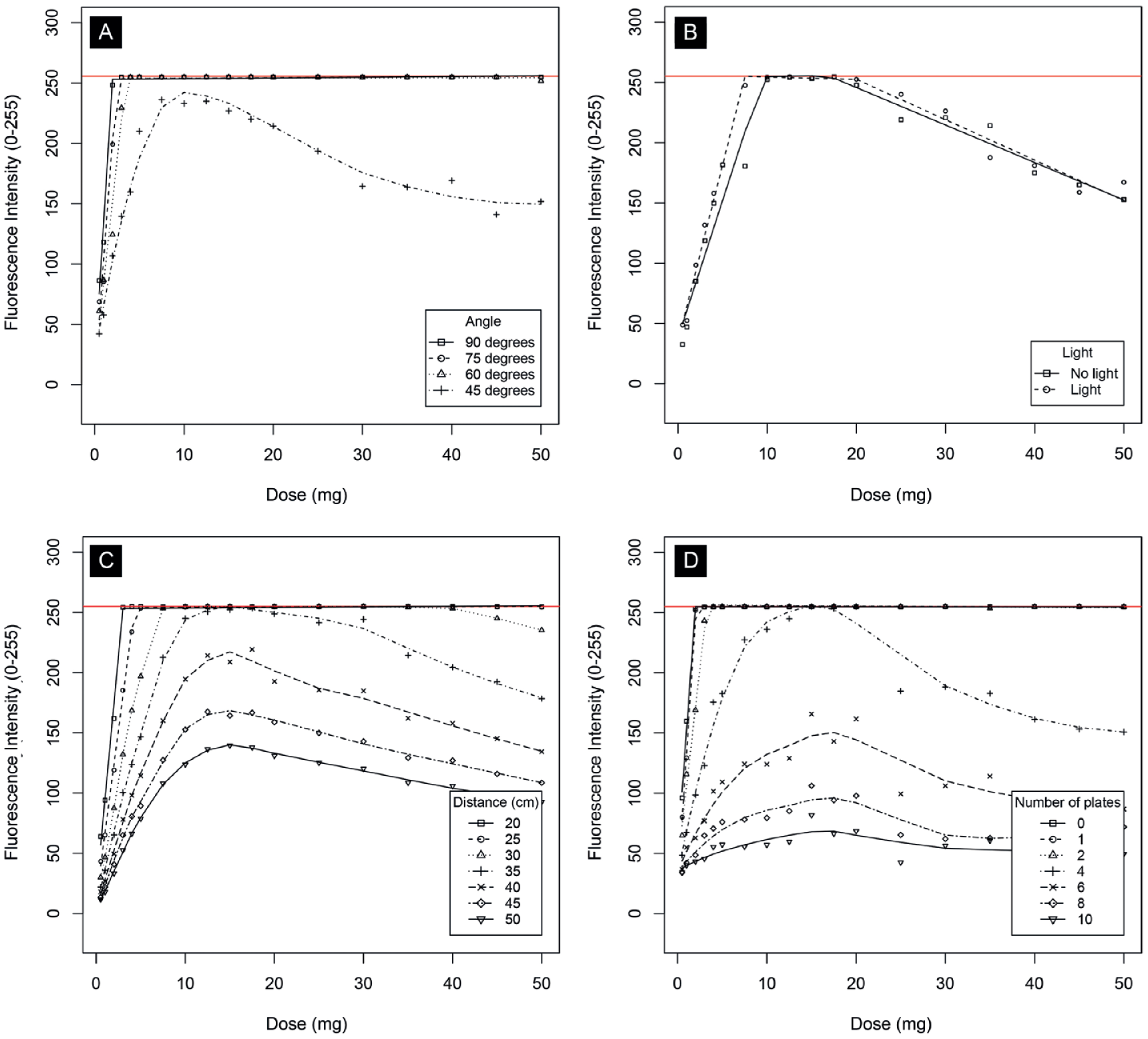
Results of ex vivo experiments. The graphs show fluorescence intensity for a range of doses. The different curves on each graph show differences between (A) various angles, (B) with and without ambient daylight at a distance of 30 cm, (C) various distances (in cm), (D) various penetration depths (in number of beeswax plates). Fluorescence intensity is measured on a grayscale from 0 to 255. Zero is black, 255 is white, and values in between make up different shades of gray.
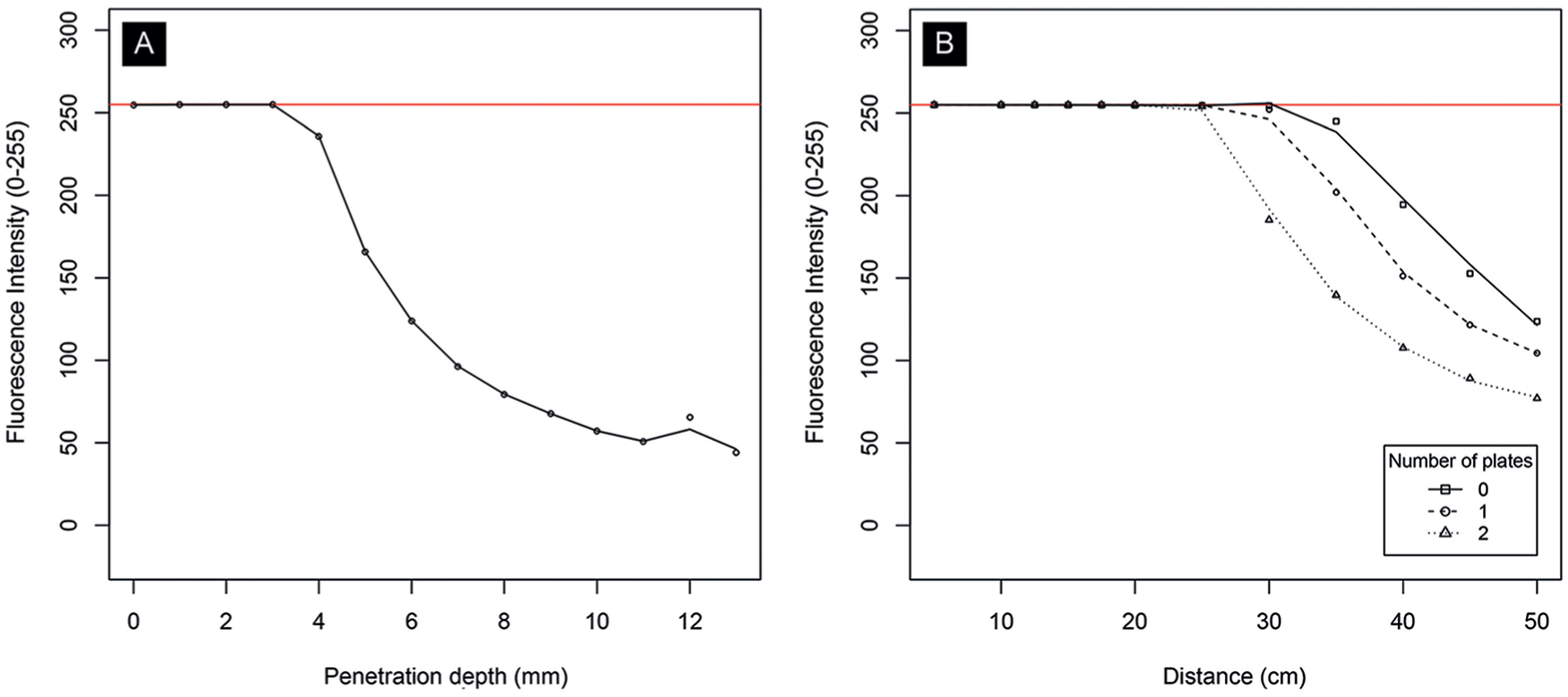
Results of ex vivo experiments. The graphs show fluorescence intensity for a range of (A) penetration depths (in mm) and (B) distances with various penetration depths (in number of beeswax plates). For both experiments, the indocyanine green dose was set to 10 mg. Fluorescence intensity is measured on a grayscale from 0 to 255. Zero is black, 255 is white, and values in between make up different shades of gray.
The curves in Figure 5A show that positioning the imaging head in an angle of 60° to 90° does not influence fluorescence intensity to any meaningful extent, exemplified by the fact they overlap almost completely. However, the fluorescence intensity for 45° does differ substantially from the other angles: the intensity is lower than any other angle over the whole range of doses. This was also substantiated by large negative regression coefficient for 45°. Compared with a 90° angle, the average difference for 75° was estimated to be −5.76 (95% confidence interval [CI] = −21.04 to 9.56; P = .460), for 60° −11.93 (95% CI = −27.21 to 3.36; P = .126), but for 45° −67.59 (95% CI = −82.88 to −52.31; P < .001).
The curves in Figure 5B show that fluorescence intensity is slightly higher in ambient light and suggest optimum fluorescence intensity reached with an ICG dose between the range of 10 and 20 mg, as the intensity is highest within this range. Figure 5C and D shows that fluorescence intensity decreases when distance increases or when penetration depth (ie, number of beeswax plates) increases, respectively. Both figures also suggest optimum fluorescence intensity reached with an ICG dose between the range of 10 and 20 mg as all curves are at their optimum in this range. When measured from a distance of 25 cm, maximum fluorescence intensity is reached with an ICG dose of approximately 5 mg or more.
Figure 6A illustrates fluorescence intensity for different penetration depths (in mm) and Figure 6B for different distances (in cm) stratified by various penetration depths (ie, number of beeswax plates). Both are measured with a constant dose of 10 mg of ICG. It is clear from these graphs that there is a negative association between number of plates or distance, and intensity.
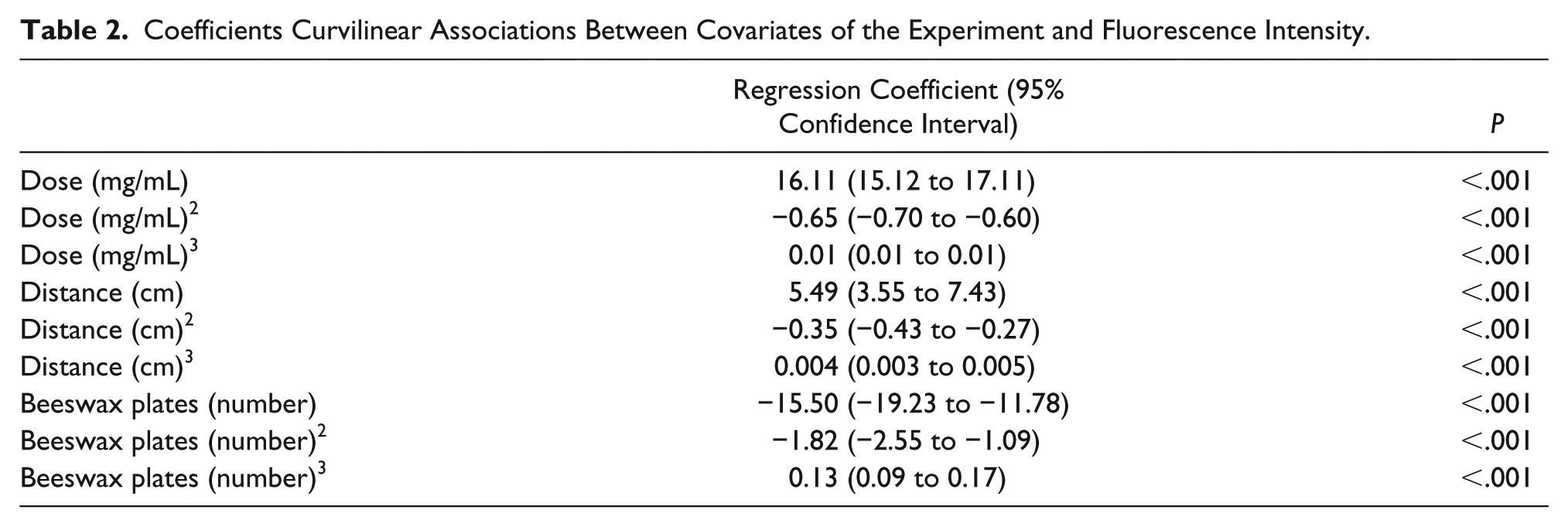
Table 2 shows the regression coefficients of the curvilinear associations between dose, distance, and penetration depth, and fluorescence intensity. All 3 associations could best be described using third degree polynomials, demonstrated by the highly significant coefficients. These strong curvilinear associations are in agreement with the figures presented above.
Coefficients Curvilinear Associations Between Covariates of the Experiment and Fluorescence Intensity.
Regression coefficient for ambient light was estimated 10.17 (95% CI = 5.23 to 15.10; P < .001).
Discussion
The aim of this study was to provide comprehensive insight in potential factors influencing the fluorescence intensity when using ICGA to assess tissue perfusion during reconstructive flap surgery. To the authors’ knowledge, the current study includes the first systematic review specifically regarding the use of ICGA protocols for evaluating tissue perfusion in reconstructive flap surgery and authors’ consideration of factors that might influence fluorescence intensity during ICGA assessment. Previous review articles mainly focused on the application and the effect of ICGA in flap surgery. For example, Smit et al recently published a systematic review and meta-analysis on intraoperative evaluation of perfusion in free flap surgery and concluded that ICGA is one of the most suitable methods to measure free flap tissue perfusion, resulting in improved flap survival. 5 Li et al recently published a review regarding the application of ICG in flap surgery and concluded that ICGA aids in the evaluation of flap microcirculation and perfusion. 9
In part 1 of the current study, factors that are considered important in ICGA protocols, as well as factors that might influence fluorescence intensity and are therefore considered to be important by the authors, have been reviewed based on a systematic literature search. Based on the results, it can be concluded that most ICGA protocols are insufficiently described, when concerning factors that might influence the outcome of NIRF imaging. When reported, there is no consensus on dosage of ICG, working distance, time to assessment, tissue resection and perfusion assessment, and time to and duration of assessment. Furthermore, only a few articles describe the actual consideration of potential factors of influence during ICGA.
Part 2 of this study comprises ex vivo experiments with a handheld ICGA system to identify and analyze factors that influence the fluorescence intensity. The methods for these ex vivo experiments have been previously reported using a laparoscopic NIRF imaging device with special emphasis on cholangiography. 19 In this study, these experiments were reproduced using a handheld imaging device with special emphasis on angiography; additionally, statistical analyses have been performed. Associations between dosage of ICG, working distance, angle, penetration depth, and ambient light, and fluorescence intensity have been quantified.
When concerning the dosage of ICG, fixed dose of ICG predominantly administered. Previous study by Li et al demonstrated that there is no consensus regarding the optimal intravenous dose in flap surgery and that different groups use their own experiences to determine the dosage. 9 Furthermore, it was reported that there is no evidence showing that multiple intravenous dosages will affect the result of the quality of ICGA. Although their review included animal studies and small series, the authors agree that there is no consensus on ICGA dosage in flap surgery. Nonetheless, the ex vivo experiments suggest an optimal ICG concentration of 0.002 mg/mL to 0.004 mg/mL and demonstrate that ICG dose significantly influences fluorescence intensity. Since patients with a higher body weight have a larger blood volume, the concentration of ICG can differ between patients. Since the estimated dose is based on ex vivo experiments, which is not comparable to in vivo conditions with unique circulating plasma volumes and cardiac outputs, 9 a recommendation regarding the optimal dose cannot be given. However, consistent with van den Bos et al, the authors conclude that applying a weight-adjusted dose seems preferable over a fixed dose. 19
With regard to working distance and ambient light, a minority of studies reported the importance of this factor. Moyer et al previously described that fluorescence intensity, emitted by ICG, can be dependent on the distance from the camera to the skin and ambient light in the room without reporting the relation between these factors. 57 The performed ex vivo experiments confirm that fluorescence intensity depends on distance and ambient light in the room. Analysis revealed that a higher distance significantly reduces fluorescence intensity. Since manufacturers recommend specific working distance for each available imaging device and fluorescence intensity is dependent on distance, the authors advocate not to deviate from this recommendation and to report working distances in studies regarding flap perfusion assessment.
When concerning ambient light, the observed fluorescence intensity was significantly higher when ICGA was performed in light, compared with total darkness. However, in these experiments, the surrounding objects outside the region of interest were also observed better (subjectively). Target-to-background ratios as previously reported by Schols et al were not determined to assess differences.76,77 However, subjective distinction between the region of interest (ie, wells plate) and surroundings did not differ in darkness or light. Yet, the assessment is preferred to be performed in the dark when possible, since there is no in vivo evidence on the exact influence of light.
Time to and duration of assessment is only reported in a few studies as being an important factor. According to the authors’ clinical experience, assessing the flap perfusion in the first minute after ICG administration is the most important period of time to assess tissue perfusion adequately.
Regarding tissue resection and perfusion assessment, the assessment of absolute value together with assessment of grayscale imaging were more frequently used than relative perfusion assessment. These 2 perfusion assessment techniques are a direct measure of the fluorescence intensity, as well as “absolute value assessments.” With the results of the ex vivo experiments, the authors conclude that these measurements are influenced by dose, working distance, angle, and ambient light. Presumably, when using the relative perfusion assessment, the effects of these factors can be diminished since percentages of perfusion are compared with reference tissue during the same perfusion assessment. 57 Therefore, conclusions can be drawn that using “relative perfusion assessment” seems preferable over the other assessment methods. Furthermore, there is no consensus on cutoff values for tissue debridement in flap surgery when absolute value assessment or relative perfusion assessment is applied. Also, there is no consensus on debridement of tissue when applying grayscale imaging. For example, La Padula et al decided to respect the representing 0 fluorescence (ie, black area) and preserved “the hypovascularized gray area,” 29 whereas Pestana et al reported to remove all areas of “poorer perfusion.” 62 Further prospective trials are warranted to determine reliable cutoff values.
When considering other factors of influence during ICGA, these factors are only described briefly in the literature. Epinephrine is the only factor that is demonstrated to negatively influence assessment of tissue perfusion when using ICGA. 47
The described ex vivo experiments have refuted the importance of the imaging head positioned perpendicular to the skin, as suggested by Yamaguchi et al. 70 The imaging head can be positioned in an angle of 60° to 90° without influencing the observed fluorescence intensity to any meaningful extent.
Furthermore, penetration depth was analyzed in ex vivo experiments. The ex vivo experiments confirm that fluorescence intensity is significantly reduced when penetration depth increases. Reported penetration depth ranges from 3 mm to 1 cm.39,57,58 In the experiments, optimum fluorescence intensity was observed up to 4 mm of depth and up to approximately 8 mm of depth was observed subjectively to distinct fluorescence intensity from the surroundings. However, this experiment is limited by beeswax plates that were used to measure penetration depth. Although the spectral scattering properties are similar to human tissue, the spectral absorption differs, so it is to be expected that the penetration depth in human tissue is different. Another possible limitation is revealed by the observation of lower fluorescence intensity with higher doses of ICG. This phenomenon was also observed by van den Bos et al. 19 Since the concentrations of ICG were diluted within 40 mg/mL of albumin in 0.9% NaCl dilution and lower concentrations were obtained by adding 40 mg/mL solution, it is possible that the absolute quantity of albumin is higher in lower concentrations of ICG. Therefore, an optimum dose cannot be given based on these ex vivo experiments.
In addition to ICGA, other imaging techniques with the ability to assess intraoperative perfusion in free flap surgery have been described in a recent systematic review. 5 These methods include the use of laser Doppler, oxygen saturation (SO2) measurements, ultrasound, dynamic infrared thermography, venous pressure measurement, and microdialysis. Of these methods, ICGA and laser Doppler have currently been the most objective and reliable methods to directly assess tissue perfusion, leading to improvement of flap survival. 5 Furthermore, a new imaging technique titled hyperspectral imaging has already shown promising results for physiologic tissue parameters. 78 The technique can be used in precision surgery and is already applied to guide flap reconstruction. 79 Preliminary results show a high capability for a camera to be used in perfusion measurements. 80 Hyperspectral imaging is a noninvasive technique with no risk of adverse events.
With regard to adverse events, Li et al described potential adverse reactions to ICG, preoperative allergy testing, and contraindications of ICG in their systematic review. 9 The authors found a lack of reported preoperative ICG allergy tests and concluded that this may be due to the acceptance that ICG has a very low rate of allergy (1 out of 42 000 to 60 000) and does not damage blood composition and the coagulation system.9,11 Since 2 cases of fatal ICG anaphylaxis have been previously reported, Li et al consider preoperative iodine allergy testing a necessary precaution as iodine allergies are the most probable source of an adverse reaction to ICG. In addition to hypersensitivity to iodine, several contraindications for applying ICG are mentioned, including closed-angle glaucoma, allergic asthma, severe hypertension, hepatic and renal function failure, and pregnancy. 9
One of the previously mentioned imaging technologies may ultimately replace ICGA for flap assessment, since ICGA is an invasive procedure. However, at this time ICGA is one of the most suitable methods to directly assess tissue perfusion in free flap reconstructive surgery. 5
In vivo studies would have been preferable over ex vivo experiments, including the effect on human tissue. On the other hand, measurements of ICG are confounded by in vivo fluorescence quenching, which makes it difficult to predict the precise working dose of ICG needed in the flap. 9 In addition, a half-time life of 3 to 5 minutes hinders to assess different factors (eg, distance) in a limited time frame. Nevertheless, the current ex vivo setup offered a simple and objective method to assess factors of influence when using ICGA.
In future studies, the optimal ICG dose should be standardized through large series and clinical trials. In addition, authors should consider reporting ICGA protocols comprehensively to provide reproducibility and enable comparison of used methods between studies. Fortunately, the American Association of Physicist in Medicine recently established a Task Group working toward consensus around guidelines and standards for advancing the field of fluorescence-guided surgery, by inventorying the key parameters, stakeholders, impacts, and outcomes of clinical fluorescence-guided surgery technology and its applications, to come to objective benchmarking and standardization within this field, and is expected to stimulate innovation. 81
Conclusion
In conclusion, this study identified factors that significantly influence the fluorescence intensity of ICGA, including dose, working distance, angle, penetration depth, and ambient light. Consequently, conclusions can be drawn that applying a weight-adjusted ICG dose seems preferable over a fixed dose when using ICGA for tissue perfusion assessment during reconstructive flap surgery. In addition, “relative perfusion assessment” seems preferable over other assessment methods. It is advocated to use recommended working distances. Furthermore, the imaging head during ICGA can be positioned in an angle of 60° to 90° without significantly influencing the observed fluorescence intensity. All of these factors should be considered when using ICGA for tissue perfusion assessment during reconstructive flap surgery. To work toward consensus and construct uniform guidelines, more transparency in methods in future studies is advocated.
Supplemental Material
SUPPLEMENTARY_SECTION – Supplemental material for Optimizing Indocyanine Green Fluorescence Angiography in Reconstructive Flap Surgery: A Systematic Review and Ex Vivo Experiments
Supplemental material, SUPPLEMENTARY_SECTION for Optimizing Indocyanine Green Fluorescence Angiography in Reconstructive Flap Surgery: A Systematic Review and Ex Vivo Experiments by Tim Pruimboom, Sander M. J. van Kuijk, Shan S. Qiu, Jacqueline van den Bos, Fokko P. Wieringa, René R. W. J. van der Hulst and Rutger M. Schols in Surgical Innovation
Footnotes
Author Contributions
Study concept and design: Tim Pruimboom, Shan S. Qiu, Jacqueline van den Bos, René R. W. J. van der Hulst, Rutger M. Schols
Acquisition of data: Tim Pruimboom, Sander M. J. van Kuijk, Shan S. Qiu, Jacqueline van den Bos, Rutger M. Schols
Analysis and interpretation: Tim Pruimboom, Sander M. J. van Kuijk, Fokko P. Wieringa, René R. W. J. van der Hulst, Rutger M. Schols
Study supervision: Tim Pruimboom, Shan S. Qiu, René R. W. J. van der Hulst, Rutger M. Schols
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
