Abstract
Introduction
Complex ventral hernia is an increasingly common challenge for general and plastic surgeons. Many patients with ventral hernia are medically complicated and present with multiple comorbidities that increase risk for hernia recurrence and surgical site occurrences (SSOs), such as infection. Although multiple approaches to the repair of ventral hernia have been described in the literature, no gold standard method has attained consensus.1 -3 Indeed, improved surgical techniques and materials are needed to further reduce risks for hernia recurrence and SSO.
One of the key causes of recurrence following ventral hernia repair is undue tension across the midline. Elevated tension on the abdominal wall caused by a poorly designed hernia repair may lead to tissue ischemia or mechanical slicing of suture through tissue, producing fascial dehiscence and hernia recurrence. 4 Several options for reducing tension and reinforcing the hernia repair have been described in the literature and applied clinically. These methods include the use of biologic or synthetic repair materials in various positions; autologous tissue transfers, such as latissimus dorsi and/or tensor fascia lata rotational flaps; and component separation. When large hernia cannot be closed primarily, bridging with repair material may be necessary. However, bridged repairs are associated with risk for laxity or bulging, visceral adhesions, bowel erosion, high rates of mesh extrusion, and subsequent reoperation for mesh removal.5,6 Autologous rotational flaps are denervated, leading to atrophy and laxity, and are associated with significant donor site morbidity.
In contrast, components separation is a local muscle flap advancement technique that allows for closure of even very large abdominal wall defects.5,7 First described by Ramirez et al, 5 component separation is recommended by many experts in ventral hernia repair for the reapproximation of the abdominal wall.5 -7 In this technique, advancement of the rectus muscle flap allows for fascial closure in the majority of hernias, even massive and complex deficits, and the rectus abdominus muscle remains innervated, which preserves abdominal wall dynamics and allows this type of repair to resist strain and stress better than grafted fascia.5,8 Furthermore, each well-constructed rectus flap remains vascularized, rendering it more resistant to infection than any graft material.9,10 Finally, advancement of muscle flaps through separation of components reconstructs the functional anatomy of the abdominal wall and allows for fascial closure without undue tension.
Based on the landmark study by Luijendijk et al, 4 the standard of repair in ventral hernia surgery includes reinforcement with prosthetic repair material. The use of prosthetic repair material and biologic grafts in conjunction with components separation have both been shown to reduce recurrence compared to unreinforced components separation repairs. 11 Therefore, as suggested by an expert panel, open repair of ventral hernia should include reapproximation of the fascia to the degree feasible using separation of components or other means and reinforcement of the repair with synthetic or biologic repair material, depending on the risk for surgical site infection. 6
In the current study, we report on the results of a novel refinement of the components separation technique that provides for the off-loading of tension from the fascial closure to the repair material and abdominal musculature. Briefly, a single sheet of noncrosslinked porcine acellular dermal matrix (PADM) is sutured to the edges of the laterally retracted external oblique muscles bilaterally to approximate physiologic tension across the repaired midline and reengage the oblique muscles released during separation of components. Thus, in a single position, the onlaid material functions to reinforce the midline, off-load tension on the herniorraphy, and reconstruct the lateral abdominal donor sites after component separation. This technique allows for the repair material and the external oblique muscles to form a separate myofascial layer, which acts as “load-sharing” support, thereby preventing undue tension on the repair closure and further restoring the functional anatomy of the abdominal wall.
This technique requires undermining and elevation of large adipocutaneous flaps for exposure of the lateral abdominal wall. Although this exposure allows for the release of the external oblique layer and placement of the onlaid reinforcement graft, it comes at the cost of relatively ischemic devascularized skin flaps, which can subsequently result in wound complications. To address a high rate of wound complications, our technique now includes other key elements for skin management after reinforcement, such as the use of progressive tension sutures to further off load skin tension and limit seroma formation.
The selection of noncrosslinked PADM over other repair materials is based on characteristics of PADM and the perceived advantages of biologic versus synthetic prostheses in surgical fields with or at risk for contamination. 6 These characteristics include intact extracellular matrix, low antigenic response, and the ability to support tissue regeneration through revascularization and cell repopulation, which may contribute to improved resistance to infection demonstrated by some biologic materials, such as noncrosslinked PADM.6,12 -16 An expert panel (The Ventral Hernia Working Group) has stratified hernia patients into 4 grades to assist surgeons in the prediction of a patient’s risk for SSO after hernia repair. 6 Patients in grades 3 and 4 have contamination or infection of their wounds, respectively; biologic repair materials are recommended for these patients, as biologic materials generally do not require removal in the setting of SSO such as infection. Grade 2 patients are deemed at risk for complications by virtue of predisposing comorbidities, such as smoking, obesity, diabetes, chronic obstructive pulmonary disease, and/or immunosuppression. The panel recommended that consideration be given to the use of biological repair materials to reinforce hernia repairs in grade 2 patients. Elevated risk of infection is a reasonable contraindication to the implantation of synthetic repair materials, whereas biological materials such as noncrosslinked PADM have been shown to develop a vascular supply and thus more reliably resist infection.9,13,15,17 -22
We hypothesized that noncrosslinked PADM would be safe and effective when used as an onlay reinforcement of components separation repair of ventral hernias in obese patients at high risk for hernia recurrence and/or SSO.
Materials and Methods
Data Gathering
This is a retrospectively reviewed case series with institutional review board approval. Patients of 4 University of Maryland Medical Center plastic surgeons were identified using a search of CPT (Current Procedural Terminology) codes for PADM (Strattice, LifeCell Corp, Branchburg, NJ; code 15430) during the period from July 2008 through December 2009. Although this CPT code is general for all xenografts, Strattice was the only PADM used at this institution during the study period. Comorbidities associated with hernia formation and recurrence were noted, including obesity, smoking, diabetes mellitus, coronary artery disease, chronic obstructive pulmonary disease, anticoagulation, end-stage renal disease, concurrent use of immunosuppressive therapy, and concurrent active abdominal cancer.
The grade of the hernia repair was established using the Ventral Hernia Working Group grading system. 6 Primary outcomes of the study were hernia recurrence, reoperation, and death. Recurrences were identified through telephone contact with the patient and clinical examination. Reoperation was necessary in cases of abdominal hypertension, fistula formation, anastomotic leak, intra-abdominal abscess, small bowel obstruction, or debridement of exposed mesh. Postoperative SSO were defined as surgical site infection (SSI), seroma, hematoma, wound dehiscence, or development of enterocutaneous fistula(e), as described by the Ventral Hernia Working Group. 6
Inclusion criteria required patients to undergo either a unilateral or bilateral components separation reinforced with an onlay of noncrosslinked PADM. Patients eligible for inclusion in the study had hernia defects too large for laparoscopic repair. Eligible patients’ wounds were all graded as contaminated, potentially contaminated, or at high risk for infection according to Ventral Hernia Working Group definitions. 6 Therefore, eligible subjects were not considered candidates for repair with synthetic repair material. Exclusion criteria consisted of bridging with prosthetic repair material or having more than one type or piece of mesh present in the abdominal cavity at any time.
Surgical Technique
All steps preceding separation of components were performed by the general surgery service, including lysis of adhesions, any required visceral surgery, synthetic mesh removal, and enterocutaneous fistula(e) take down.
Components separation was then performed. If the tension required for midline closure was deemed too high, the contralateral side was separated as well. A 20 cm × 20 cm sheet of noncrosslinked PADM was placed centrally overlying the repair, trimmed if necessary, and sutured in place with either running or interrupted sutures along the periphery. To strengthen the lateral abdominal wall and repair the donor sites, the released lateral edges of the external oblique were sutured to the edges of the mesh; Figures 1-3 illustrate the steps of the technique in detail. In the case of a unilateral components separation, the mesh was sutured to the most lateral extent of the suprafascial dissection on the nonseparated side.
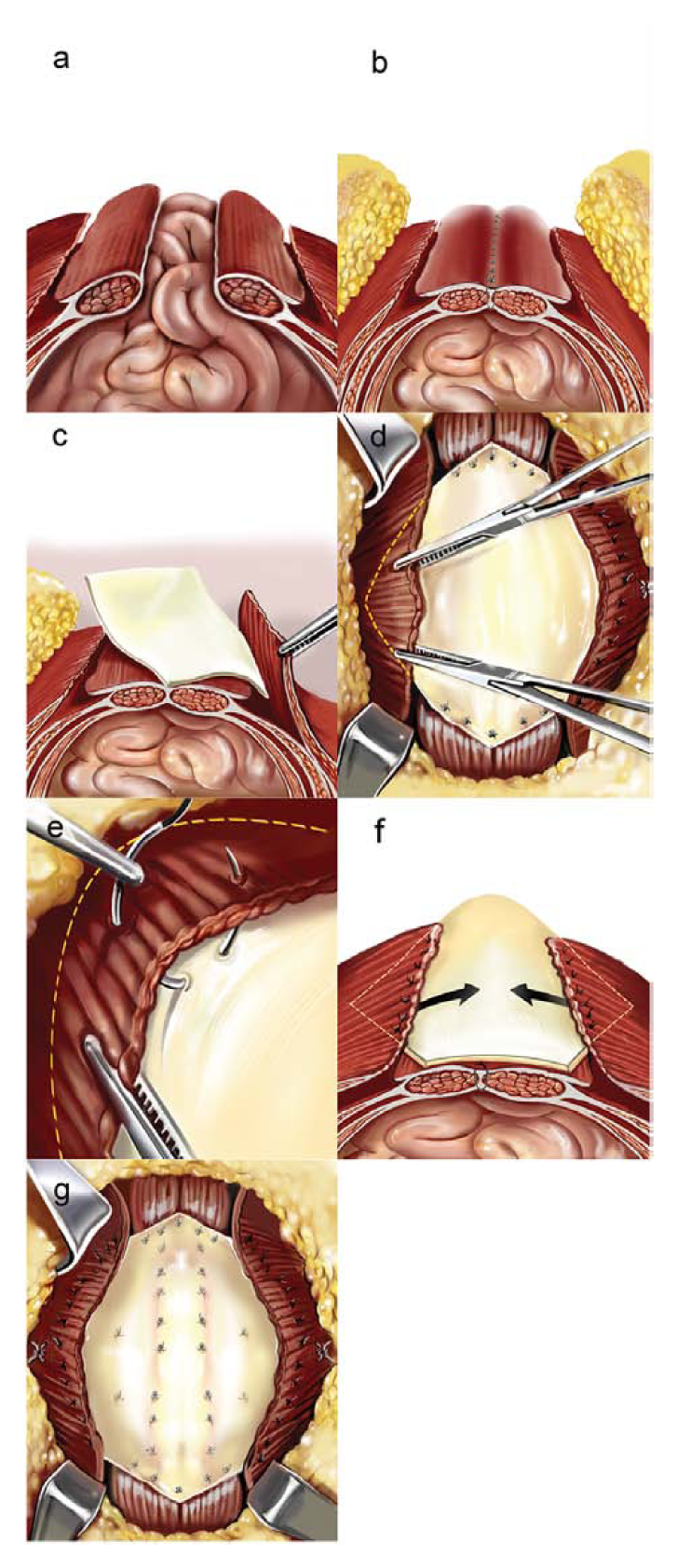
The technique of component separation with noncrosslinked porcine acellular dermal matrix (PADM) onlay reinforcement as described by the authors. (A) A ventral hernia defect prior to repair. In the component separation technique, the external oblique is released, allowing for advancement of the fascia and primary closure of the defect without undue tension (B). However, the released external oblique represents a potential site of weakness in the abdominal wall. To address this weakness, a 20 cm × 20 cm sheet of PADM is inserted in a diamond pattern over the rectus abdominus and laterally deep to the external oblique (C) and trimmed if necessary. The superior and inferior corners of the repair material are sutured onto the rectus abdominus and each lateral aspect of the material is attached deep to the external oblique (D) with interrupted sutures (E). After the external oblique is sutured to the PADM on one side, the contralateral side is sutured to the repair material to fully reincorporate the released external oblique muscles and restore the anatomy of the abdominal wall. This technique creates a load-sharing structure (F), displacing tension from the underlying fascial closure to the prosthesis and abdominal musculature. Mattress sutures (G) ensure tight, continuous contact between the prosthesis and the underlying tissue, reducing risk for seroma formation and fostering revascularization of the repair material.
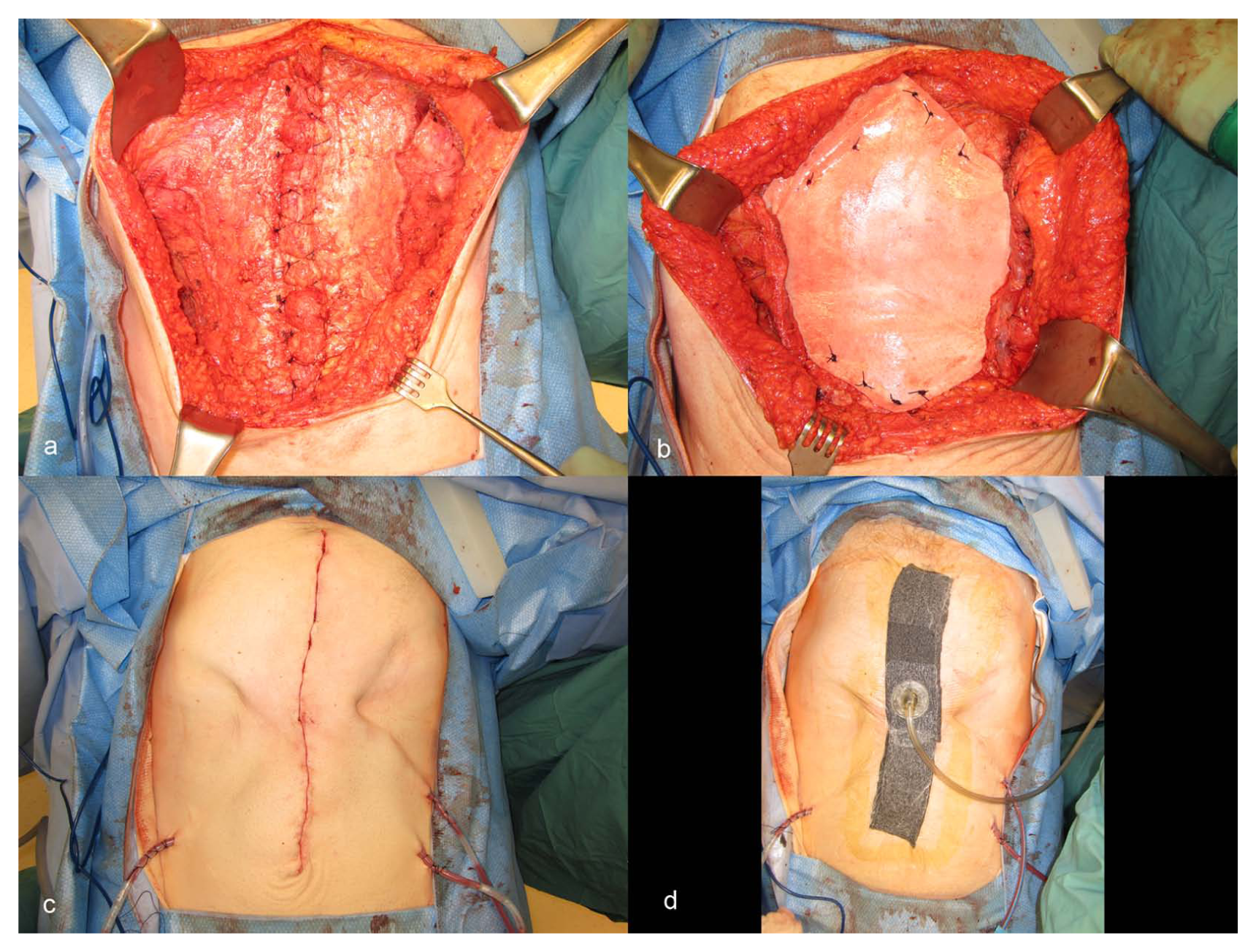
Digital photographs illustrating one case from the series reported by the authors. Bilateral release of the external oblique muscles (A) allowed for a tension-free fascial closure of this large deficit. A 20 cm × 20 cm sheet of porcine acellular dermal matrix (PADM) was placed as an overlay reinforcement over the rectus abdominus and laterally deep to the released external oblique muscles (B). The superior and inferior corners of the repair material have been sutured onto the rectus abdominus and each lateral aspect of the material has been sutured deep to the external oblique. Scarpa’s fascia was reapproximated and the skin was closed in layers; prophylactic drains were placed in the subcutaneous space to reduce risk for seroma formation (C). The incision was then dressed with a negative pressure wound therapy dressing (D).
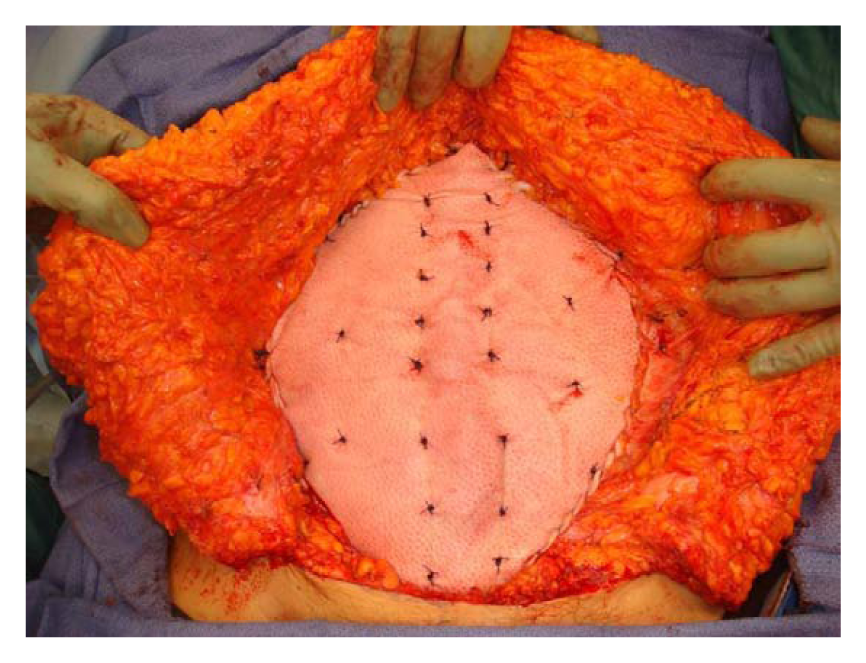
Example of porcine acellular dermal matrix (PADM) overlay illustrating load-sharing structure. The PADM is sutured onto the rectus abdominus and deep to the released external oblique with interrupted sutures. Mattress sutures ensure tight and continuous contact between the PADM and the underlying tissue.
In response to a high wound complication rate, we began an aggressive wound management protocol to decrease SSO during this case series. First, we minimized skin flap undermining to the extent possible while still allowing for release of the aponeurotic portion of the external oblique layer. Following reconstruction with rectus abdominus advancement flaps and placement of repair material, we debrided the undermined ischemic skin flaps to healthy bleeding tissue. To further decrease the risk of seroma formation, prophylactic drains were placed in the subcutaneous space. Skin flaps were typically quilted to the abdominal fascia using progressive tension suture technique. 23 Infraumbilical panniculectomy was performed if necessary. Scarpa’s fascia was re-approximated and the skin was closed in layers.
Later in the case series, incisions (midline vertical or inverted T) were dressed with a closed incisional negative-pressure wound therapy dressing (Wound VAC, Kinetic Concepts Inc, San Antonio, TX). A protective layer of semipermeable Vaseline gauze was placed over the closed incision, and negative pressure wound therapy was applied (see Figure 2). An abdominal binder was applied only after skin flaps were deemed viable after several days. Drains were removed once output was less than 25 mL per 24 hours for 2 consecutive days.
Data Analysis
Demographic data were compiled on Microsoft Excel. SPSS statistical software was used for statistical analysis.
Results
Fifty-eight patients met the inclusion criteria, resulting in 58 total procedures. The baseline characteristics of included subjects are listed in Table 1. The breakdown of comorbidities is listed in Table 2. Fourteen patients had a concurrent panniculectomy; the average body mass index of this subpopulation was 38.7 kg/m2.
Baseline Characteristics of Included Subjects.
In patients with prior repairs.
Frequency of Comorbidities Among Included Subjects.
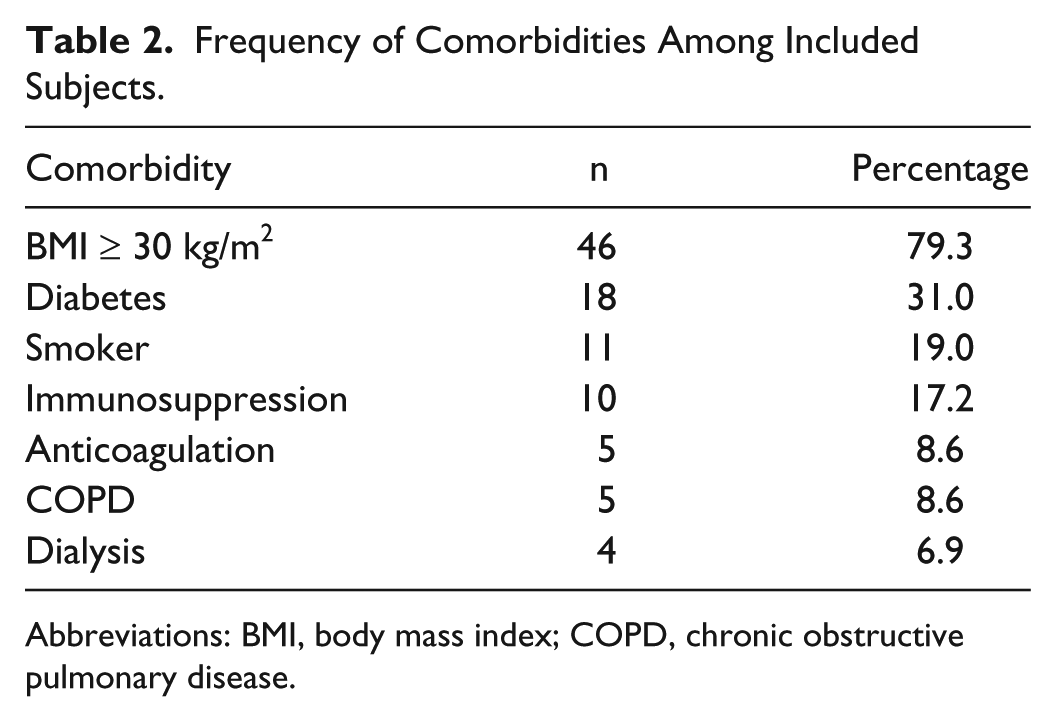
Abbreviations: BMI, body mass index; COPD, chronic obstructive pulmonary disease.
The most common indication for abdominal wall reconstruction was incisional hernia (82.3%), followed by the presence of an open abdomen (11.8%) and primary ventral hernia (3.9%). Precise defect size was not recorded, but exceeded 100 cm2 in all cases. The most common hernia grade was grade 2, which was identified in 28 patients (48.3%), followed by grades 3 and 4 (36.2% and 15.5%, respectively). Eleven patients (19%) had a unilateral components separation whereas 47 patients (81%) required bilateral components separation. All patients achieved complete fascial closure.
The postoperative course was uneventful for 34 patients (58.6%). Of the cases with complications, 15 patients (25.9%) had 1 complication and 9 (15.5%) had 2 complications. The most common wound complication was SSI, with an incidence of 20.7% (12 patients). Seven patients (12%) required debridement and negative pressure wound therapy for skin and fat necrosis. Of the patients suffering wound breakdown, the average body mass index was 39.1 kg/m2; 57% were diabetic; and half of the diabetics were current smokers. There were 9 (15.5%) seromas and 3 (5.2%) hematomas that required intervention.
Three patients (5.2%) required exploratory laparotomy, and therefore the hernia repair was disrupted to provide intra-abdominal access. One patient developed enterocutaneous fistulae, one developed respiratory distress of unknown origin, and another developed intra-abdominal hypertension. There were 4 deaths in this series, all related to intraperitoneal complications rather than to the hernia repair proper. One patient died of cardiopulmonary arrest on postoperative day 3; one, of septic complications related to a peripherally inserted central catheter 4.5 months postoperation; one, of duodenal abscess and pancreatic fistula 3 months postoperation; and one, of septic complications related to a preoperative enterocutaneous fistula leading to renal failure 3 months postoperation. Except for the 3 cases requiring exploratory laparotomy, the mesh was not explanted in the remaining 55 cases.
Average length of follow-up was 384 ± 153.8 days (range 127-665 days). There were 4 recurrences at a mean 11.8 ± 3.1 months for a recurrence rate of 7.9%. All recurrences occurred in obese patients with bilateral components separations, and each of these patients developed a SSO before recurrence. All recurrences occurred at the midline fascial closure; there were no instances of lateral bulge among the study subjects. Two recurrences developed in patients with grade 2 hernias, both of whom suffered from diabetes and coronary artery disease. One recurrence developed in a patient with a grade 3 hernia. This patient developed a superficial abscess, which eventually necessitated partial debridement of the PADM graft. One recurrence happened in a patient with a grade 4 hernia and a history of 15 previous repairs. This patient also smoked, had diabetes, and was on therapeutic enoxaparin. After the initial recurrence, this patient required 2 subsequent repairs for recurrences. Of the 14 patients to undergo panniculectomy simultaneous with hernia repair, none developed a hernia recurrence.
Discussion
Surgeons faced with complex ventral hernia continue to search for refinements in technique and repair materials to reduce further the risk for hernia recurrence, as well as SSO, such as infection. In this study, we report a retrospective analysis of subjects with complex ventral hernia repaired using a modification of components separation in combination with onlay noncrosslinked PADM. Our results suggest that this approach leads to a low rate of hernia recurrence and rates of SSO comparable to those reported in previous studies involving similar high-risk patient populations.
The method described in this study employs several techniques designed to close large defects; reduce tension on the fascial repair; recreate an innervated, functional abdominal wall; and foster graft revascularization and wound healing. These techniques include separation of components, attachment of onlay PADM to the released external oblique muscles, use of progressive tension sutures, placement of drains above and within the repair material, and application of negative-pressure wound therapy to the incision site following closure.
The first technique is separation of components, which involves incisions to release the external oblique muscles on one or both sides of the defect to allow for advancement of the muscle flaps across the defect to achieve fascial closure without undue tension. The basic components of this technique have been previously described.5,7 The second element is the reinforcement of the repair with onlay PADM. The use of prosthetic repair material in conjunction with component separation was based on evidence describing reduced hernia recurrence rates compared to unreinforced component separation.11,18,24 -28
The third element of technique involved how this onlay material was applied. Traditional component separation leaves the external oblique muscle loose and permanently retracted, potentially weakening the lateral abdominal wall and predisposing the patient to lateral bulging. To correct this donor site defect, we developed a method of attaching the edges of the prosthesis to the released edge of the external oblique muscle on one side of the defect, then pulling the material taught and suturing to the edge of the opposite external oblique. This technique pulls the external oblique muscles toward the defect and restores the anatomy of the abdominal wall. The onlay mesh placed in this manner improves the hernia repair through three distinct mechanisms. First, the addition of an extra layer of mechanical support reinforces the midline fascial closure. Second, the reattachment of the released external oblique muscles repairs the bilateral donor sites and reduces the risk of lateral bulging at these areas of weakness. Third, and perhaps most important, we postulate that the prosthetic material used in this method of repair acts as a “load-sharing” construct, thereby off-loading tension from the underlying fascial closure. This function of the prosthetic material is analogous to the load-sharing role of plates in fracture fixation, where the plates are placed across a reduced fracture to neutralize axial and shear forces at the fracture site. 29 Together, the restoration of functional anatomy and use of a load-sharing prosthesis may decrease risk for hernia recurrence by reducing undue tension on the repair. With the variation in technique describe above, there were no instances of lateral bulging in any of the subjects in this study, and there was a low hernia recurrence rate (7.9%) after a mean of 384 days of follow up in a high-risk patient group.
The subjects in this study ranged from grades 2 to 4 according to Ventral Hernia Working Group definitions. 6 Therefore, all of the subjects had comorbidities that increased the risk for SSO or had contaminated or potentially contaminated fields. Given the complex and high-risk composition of these subjects, the overall SSI incidence of 20.7% may be expected. This infection rate is comparable to rates reported by similar studies of complex hernia repairs with high risk of infection, ranging from 16% to 25% in published reports.19,21 Interestingly, the infection rate increased little when stratified by grade: 17%, 24%, and 22% for grades 2, 3, and 4, respectively. Differences between grades were not statistically significant. Although the repair material became exposed in seven patients, none required explantation. The use of biologic repair material in this study, therefore, allowed for treatment of SSO without need for explantation, except in the 3 cases requiring exploratory laparotomy.
With regard to the position of prosthetic placement, it is important to separate hernia patients into bridged repairs or reinforced repairs. With respect to bridged defects, such as in laparoscopy, there are 3 positions available for mesh placement: inlay (or interpositional), underlay, and onlay. Interpositional placement of repair material for bridging a defect is not recommended. 6 When bridging a defect, an underlay placement is employed during laparoscopic hernia repair and similarly during open repairs, when complete fascial closure is not possible, despite component separation.
When reinforcing a hernia repair performed with component separation, there are only 2 available positions for mesh placement: overlay or underlay (either intraperitoneal or retrorectus). To date, there are no data that strongly support the superiority of one position over the other for placement of reinforcement. One recent analysis of a prospective study of contaminated ventral hernia repairs evaluated intraperitoneal versus retrorectus placement of PADM following component separation (N = 49).
30
Although there was no statistically significant difference between groups, recurrence at 1 year was identified in 10% of retrorectus repairs and 30% of intraperitoneal repairs (
Proponents of the underlay method of placement note that skin dehiscence will not lead to exposure of the repair material and that intra-abdominal pressures will push the repair material against the abdominal wall, rather than away from the repair.2,31 -35 However, underlay placement requires extensive lysis of adhesions and increases the risk for the formation of adhesions to the repair material and/or enterocutaneous fistulae. We have noted that during underlay placement with primary fascial closure, it is quite difficult to set repair material under the appropriate amount of tension, such that the mesh is not rippled or buckled on closure of the fascia. If the mesh is lax during underlay placement, then all forces pulling across the closure are borne by the fascia alone. Therefore, repair material placed in the underlay position typically does not act as a load-sharing structure to resist lateral stresses in the abdominal wall.
The onlay technique described in this study contributes to the restoration of an innervated, functional abdominal wall by reconstructing the lateral donor sites necessitated by separation of components. Therefore, in this method, the onlay biologic repair material reinforces the fascial closure, reincorporates the external oblique muscles into the abdominal wall, and acts as a load-sharing structure by displacing tension from the fascial closure to the prosthesis and the abdominal musculature. This onlay method also ensures tight, continuous contact between the biologic repair material and the underlying tissue, reducing risk for seroma formation and fostering revascularization and positive regeneration of the repair material. The potential benefits of this onlay load-sharing technique are summarized in Table 3.
Potential Benefits of the Load-Sharing Application of Onlay Porcine Acellular Dermal Matrix in Complex Ventral Hernia Repair.

In the only published study comparing onlay mesh reinforcement to no reinforcement of components separation, Espinosa-de-los-Monteros et al 11 reported a reduction in recurrence rate from 20% with unreinforced repair to 5% with onlay repair material. The recurrence rate in the current study (7.9%) compares favorably with previously reported recurrence rates in studies of reinforced components separation, including studies of PADM in the onlay or underlay position, which range from 0% to 31%.11,36 -42 These results are especially encouraging in light of the high average grade and complexity of hernia in these subjects. Ultimately, the trade-off for better hernia outcomes using the onlay method described herein may be a higher risk of wound complications owing to the wide undermining of skin flaps required for exposure.
As traditionally employed, components separation requires take down of perforators during undermining of tissue flaps. This devascularization may increase the risk for tissue flap ischemia and other SSO. Minimally invasive and perforator-sparing methods of components separation have been described in the literature and may be employed if tissue flap perfusion must be maximized.43 -45 In the approach to components separation described in this study, most perforators were taken down during undermining of tissue. However, during the time period encompassed by this retrospective case review, we developed an aggressive wound management protocol in an effort to decrease SSO. Elements of the protocol include minimizing skin flap undermining to the extent possible, debriding undermined ischemic skin flaps back to healthy bleeding tissue, and advancing and insetting skin flaps using progressive tension sutures toward the midline. Progressive tension suturing is a powerful technique that helps to obliterate dead space and reduce tension on the midline incision, thereby decreasing skin and/or fat necrosis, reducing drain output, and promoting better long-term tissue restoration.23,46 Although not available for the subjects of the current study, the authors now employ intraoperative laser angiography with indocyanine green to identify tissue at risk for ischemia and guide debridement of skin flaps. 47
If necessary, we also routinely performed an infraumbilical panniculectomy and closed the defect with an inverted-T incision. Interestingly, none of the 14 patients who underwent panniculectomy experienced a recurrence. This finding may relate to 2 underlying mechanisms. First, removal of the pannus reduces mechanical burden on the abdomen and the fascial closure, thereby reducing tension across the repair. Second, removal of the pannus limits ischemic burden associated with fat, thereby reducing risk for SSO and improving hernia repair outcomes. Therefore, despite a larger inverted T incision, reducing pannus burden can reduce dead space and the risk for seroma, weight and tension on the closure, and the amount of potentially ischemic fat.48,49
Incisions were dressed with a negative pressure wound therapy dressing. By acting as a soft tissue splint, negative pressure reduces tension at the healing incision; furthermore, it may help wick fluid through the early incision which might otherwise percolate and disrupt healing.50,51 Investigators have reported significantly lower rates of wound complications and skin dehiscence with negative pressure wound therapy compared to conventional dressings following abdominal wall reconstruction.
52
Finally, there is some experimental evidence that negative pressure on closed incisions may promote angiogenesis.
49
Other practices we have adopted to reduce SSO are routine placement of drains above and below the repair material and
Study Limitations
The main limitations of this study are its retrospective design and lack of comparator group. Surgical techniques for the prevention of SSO evolved during the course of the study period and were not uniformly applied for all study subjects. Furthermore, recurrence was identified through telephone contact with subjects and clinical examination, rather than the gold standard of computed tomography scan. Future studies of this technique should employ a prospective design and intent-to-treat analysis, with recurrence determined by abdominal computed tomography at one year.
Conclusion
Large complex ventral hernias in high-risk patients can be treated with components separation and reinforced with prosthetic repair material. The presence of multiple comorbidities and certain surgical conditions that influence risk of postoperative SSI require appropriate judgment when selecting reinforcement materials. Using a dedicated wound management protocol, our data confirm that components separation in combination with the onlay position of noncrosslinked PADM is effective for treatment of high-risk ventral hernias while maintaining a reasonable SSO profile. An ongoing retrospective study will report on a subsequent series of patients with complex ventral hernia who were treated with the onlay load-sharing technique and full wound protocol described herein.
Footnotes
Acknowledgements
Editorial support was provided by Joshua Kilbridge of Kilbridge Associates, San Francisco, CA.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Devinder P. Singh is a consultant for LifeCell Corporation. Ronald P. Silverman is the Chief Medical Officer for Kinetic Concepts Incorporated.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by LifeCell Corporation, Branchburg, New Jersey.
