Abstract
The purpose of this study was to assess the change in frequency and distribution of focal interictal epileptiform discharges (IEDs) as measured on scalp EEG after anti-seizure medications (ASMs) were weaned in the epilepsy monitoring unit. We retrospectively reviewed the EEG of patients with focal epilepsy on a single ASM. A two-hour EEG epoch was selected at sleep onset during the first day of admission and defined as the high-ASM epoch. This was compared to a two-hour low-ASM epoch at sleep onset after the ASM was weaned, at least 6 h before or after a seizure. IEDs were manually counted and characterized. A total of 115 patients were included. For those on levetiracetam, there was a significant increase in IED quantity when comparing the high-ASM to the low-ASM epoch (mean 40.6 to 71.4, p < 0.001). For those on sodium channel blockers, there was a non-significant trend towards a decrease in IED quantity as the ASM dose was decreased (p = 0.065). There was no statistically significant change found for other individual ASMs. For the cohort, 12 patients had IED observed only on the low-ASM epoch (which were not present on the high-ASM epoch), 6 of which were treated with levetiracetam. In summary, our findings showed weaning of levetiracetam was associated with a significant increase in IEDs whereas other ASMs were not. Some populations of IEDs were only seen after ASMs were weaned. These findings suggest that different ASMs may have unique effects on IEDs when weaned.
Keywords
Introduction
Interictal epileptiform discharges (IEDs) as measured on electroencephalography (EEG) are an electrographic hallmark of epilepsy. Although there is efficacy in the ability of anti-seizure medications (ASMs) to reduce seizure frequency, 1 the relationship between ASMs and IEDs in focal epilepsy is not fully understood. Some studies have demonstrated that the taper of ASMs is associated with an increase in IED prevalence.2–4 This finding could be consistent with the concept that IEDs are a marker of neuronal excitability, contributing to ictogenesis. On the other hand, it has also been observed that ASM taper may be accompanied by a decrease in IED rate.2,5–6 This finding may be consistent with the proposition that IEDs may have an anti-ictogenic effect and act to suppress seizures. The variable results may also be impacted by the different methods in which IEDs are quantified, the technique of EEG recording (intracranial, scalp, limited montages, etc), which ASMs were used, as well as other clinical factors.
The relationship between ASMs and IEDs is of clinical significance. In the work up for epilepsy, the presence of IEDs provides supportive evidence that the patient may have a seizure disorder and can further assist in its characterization. 7 Furthermore, it has been established that IEDs are not a benign finding or simply a bystander of epilepsy. IED occurrence during critical processing periods can have a negative impact on cognitive performance.8,9 This includes word finding difficulties, memory impairments and delayed reaction time.8,9 Thus, if certain ASMs can alter IED occurrence, frequency, or distribution, they may positively impact clinical outcomes independently of seizures.
ASM withdrawal in persons with epilepsy undergoing continuous video-EEG monitoring in the epilepsy monitoring unit (EMU) is one method in which the relationship between ASMs and IEDs can be elucidated. There have been few attempts to systematically evaluate the propensity of different ASMs to affect IEDs (individually or according to mechanism of action). Therefore, this study was undertaken to assess the change in IED characteristics as commonly used ASMs were weaned during long term EEG monitoring in the EMU.
Methods
Patient Selection
We retrospectively reviewed all patients who were admitted to the Vanderbilt University Medical Center EMU for scalp video-EEG recording from January 2014 to December 2023. Inclusion criteria required 1) patients were 18 years of age or older, 2) focal IEDs were present as interpreted by the treating epileptologist, 3) patients were taking a single ASM at the time of admission, 4) the ASM dose was weaned or stopped during admission, 5) on the first day of admission no seizures were recorded, and 6) on a subsequent admission day there was a second EEG epoch available to analyze IEDs based on criteria discussed below. At our center, patients are instructed to continue their ASM as prescribed up to and including the day of admission. The rate at which ASMs were subsequently tapered was at the discretion of the treating physician in the EMU and is generally individualized based on seizure frequency and other clinical factors. Chart review was performed to extract patient information including demographics, medical history, EEG data and seizure characteristics.
EEG Acquisition
Each patient underwent continuous video-EEG monitoring with scalp electrodes using the international 10-20 electrode system as well as T1/T2 electrodes. In some cases, sphenoidal or zygomatic electrodes were used as well as inferior temporal chain electrodes of the 10-10 system. EEGs were interpreted by treating epileptologists at our center with the assistance of fellow physicians. Before October 2018, EEG was recorded on Nihon Koden (Irvine, CA) and thereafter Natus (Middleton, WI) was used.
IED Quantification
All patients who met inclusion criteria had EEG tracings manually reviewed by study authors CL and JF. CL is a board-certified neurologist and second year chief epilepsy fellow. JF is an attending epileptologist and board-certified in neurology, clinical neurophysiology and epilepsy. Two-hour EEG epochs were selected since it would not be feasible to perform manual scoring of the entire recording. To minimize the potential confounding effect of sleep, EEG epochs were chosen at the start of sleep onset. By nature, most epochs were recorded in stage 1 and 2 sleep (NREM sleep). If a patient briefly had an arousal, that segment was skipped until the resumption of NREM sleep was seen. A high-ASM epoch was defined as this two-hour period on the first day of admission. A low-ASM epoch was then chosen for comparison on a subsequent day of admission when the patient had been either off ASMs for the longest period or was being treated with the lowest dose for the longest period. Given that most ASMs included in this study have a relatively short half-life, the minimum time difference between the two epoch was 24 h, and most often several days. Both epochs for each patient were reviewed by the same study author for consistency. Both epochs were required to be at least 6 h either before or after a seizure. This period was selected since prior studies have demonstrated that IEDs can vary from baseline during the peri-ictal phase.3,5,6
IEDs were manually characterized based on quantity and distribution and compared between the high-ASM and low-ASM epochs. Researcher authors had a high specificity when counting epileptiform discharges based on standardized criteria, as to not include artifact, sharp transients, normal variants (such as wicket spikes), or sharply contoured slow waves. 10 Lateralized rhythmic delta activity (LRDA) and broad based lateralized periodic discharges (LPDs) with morphology that in isolation would not qualify as IEDs were not included. Short continuous bursts of epileptiform discharges with the appearance of focal rhythmic epileptiform discharges (REDs) were counted as a single IED. 11 If two or more independent IEDs overlapped in time, they were counted as separate unique events.
Statistical Analysis
All patients who met inclusion criteria were included in the analysis. Data were analyzed using Stata v15 (StataCorp, College Station, TX). Demographic data, where appropriate, were presented as median with first (Q1) and third (Q3) quartiles. Continuous variables (as with IED quantities) were analyzed by a Wilcoxon signed-rank test. ASMs were grouped into sodium channel blockers consisting of lamotrigine, oxcarbazepine, carbamazepine, eslicarbazepine, lacosamide, and phenytoin. A subset of patients had IEDs present on the low-ASM epoch only (and not on the high-ASM epoch), for which odds radios (ORs) and fisher's exact tests were calculated comparing the associations between individual ASMs. A p value ≤0.05 was considered significant.
Standard Protocol Approvals, Registrations, and Patient Consents
The study protocol was reviewed by the Vanderbilt University Institutional Review Board and exemption status was granted. Patient consent was waived for this retrospective review.
Data Availability
Anonymized data not published within this article will be shared by request from any qualified investigator for purposes of replicating procedures and results.
Results
A total of 115 patients met inclusion criteria. Demographics are summarized in Table 1. The median duration of video-EEG recording was 6 days (interquartile range 5-7 days). The median day used to calculate the low-ASM epoch was admission day 4 (interquartile range day 3-5). For the low-ASM epoch, 11 patients had received a lower dose of ASM than usual, while the remainder had their ASM discontinued. Interictal epileptiform discharges had a temporal lobe distribution in 102 patients (89%) and were extra-temporal in 13 patients (11%). The most common ASMs used in descending order were levetiracetam, lamotrigine, oxcarbazepine and lacosamide.
Patient Demographics.

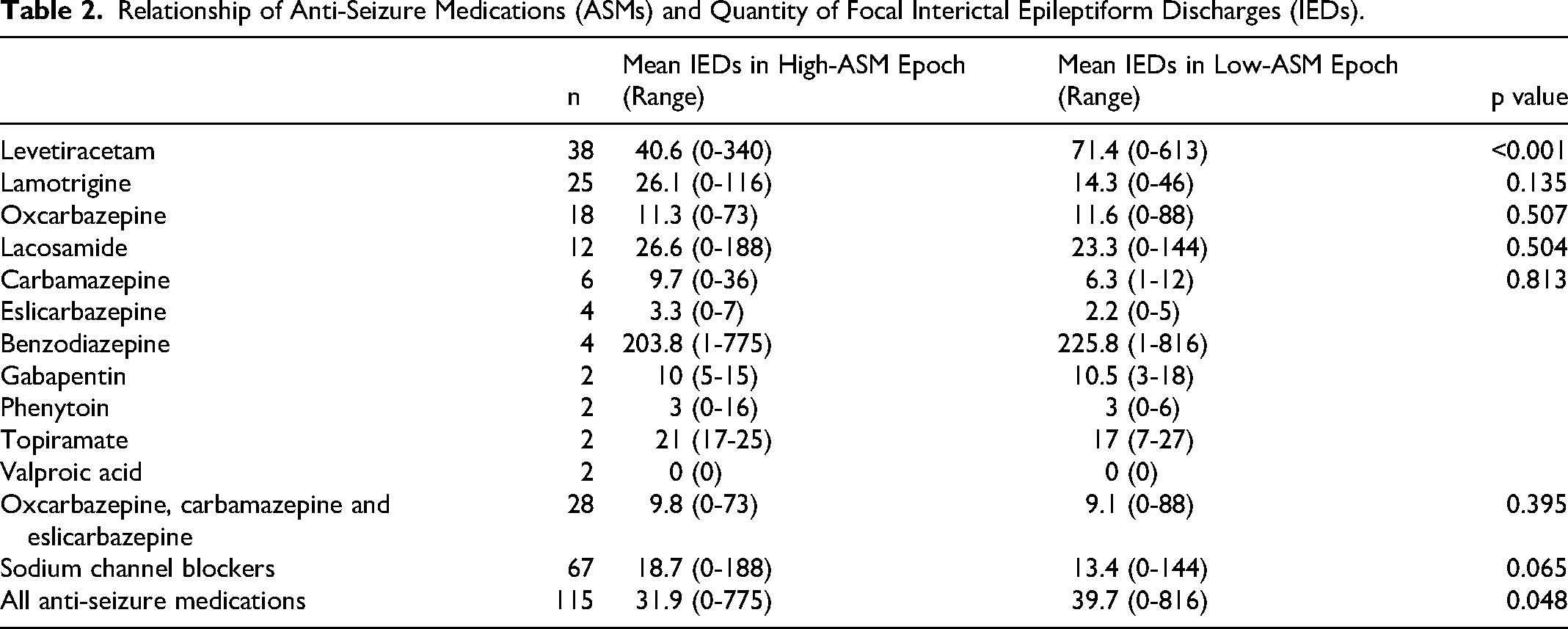
When comparing the high-ASM and low-ASM epochs, the relationship between change in ASM dose and quantities of focal IEDs are detailed in Table 2. For all patients (on any ASMs), the mean IED quantity increased from 31.9 in the high-ASM epoch to 39.7 in the low-ASM epoch (Figure 1, p = 0.048). However, based on subgroup analysis, this effect was influenced almost entirely by those patients on levetiracetam, who showed a significant increase in the quantity of IEDs (from 40.6 to 71.4, p < 0.001) as the ASM dose was decreased. On the other hand, although not statistically significant, for those on sodium channel blockers, there was a trend towards a decrease in IED quantity as ASM dose was decreased (p = 0.065). There was no significant change in IED quantity in the remaining ASM groups when analyzed individually.
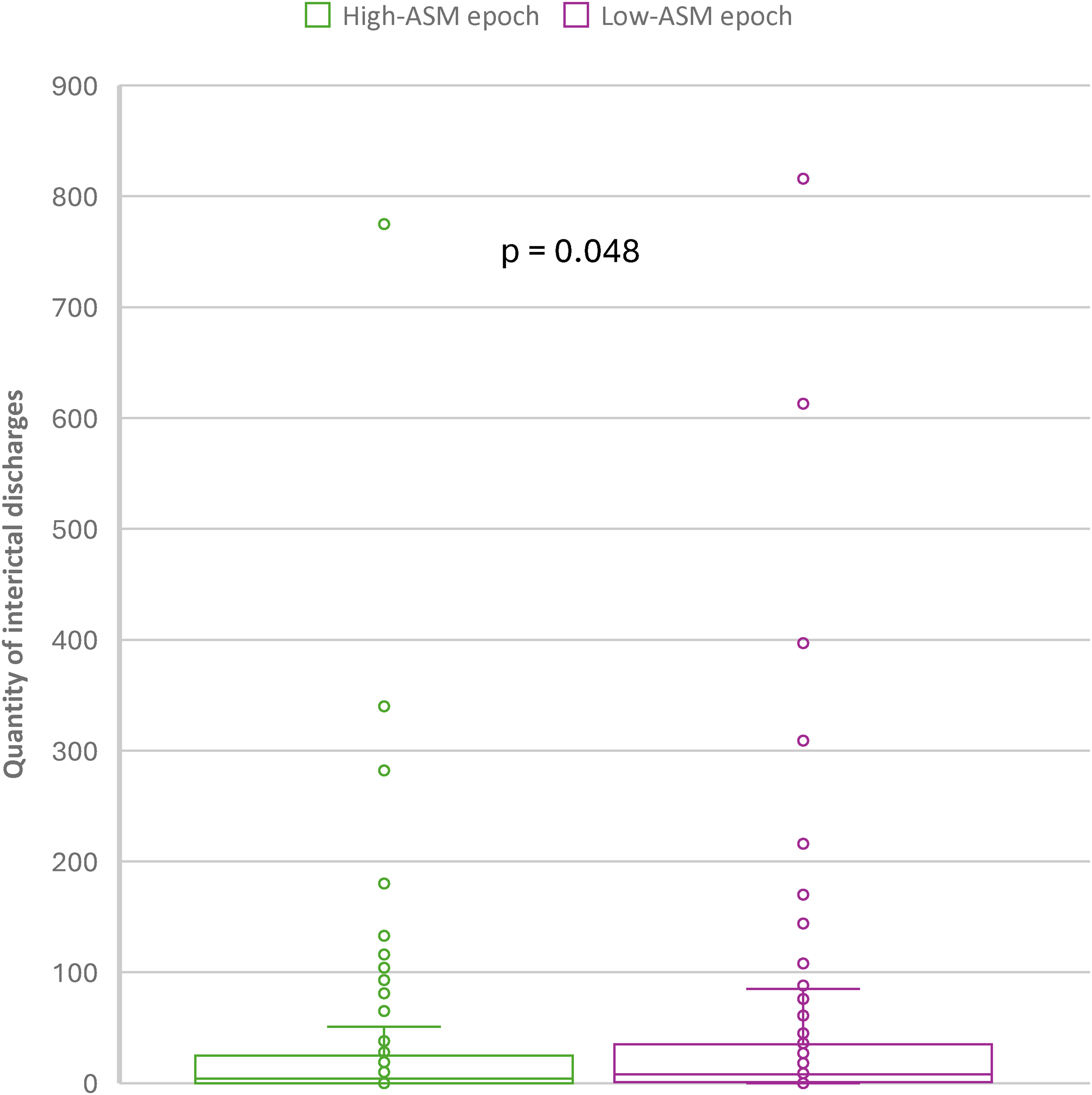
Quantity of interictal epileptiform discharges (IEDs) for each patient on any anti-seizure medication (ASM) when comparing the first day of admission (high-ASM epoch) to the day the patient had been either off ASMs or being treat with the lowest dose for the longest period (low-ASM epoch). The mean IED quantity increased from 31.9 in the high-ASM epoch to 39.7 in the low-ASM epoch (p=0.048).
Relationship of Anti-Seizure Medications (ASMs) and Quantity of Focal Interictal Epileptiform Discharges (IEDs).
Seventy patients (61%) had seizures recorded during the EMU admission. Forty-eight patients (42%) had seizures in between the high-ASM and low-ASM epochs. Twenty-four patients (21%) had seizures within 24 h of the low-ASM epoch. Excluding those patients who had recent seizures within 24 h of the low-ASM epoch, the change in IED quantity for those on levetiracetam was still statistically significant (p = 0.001).
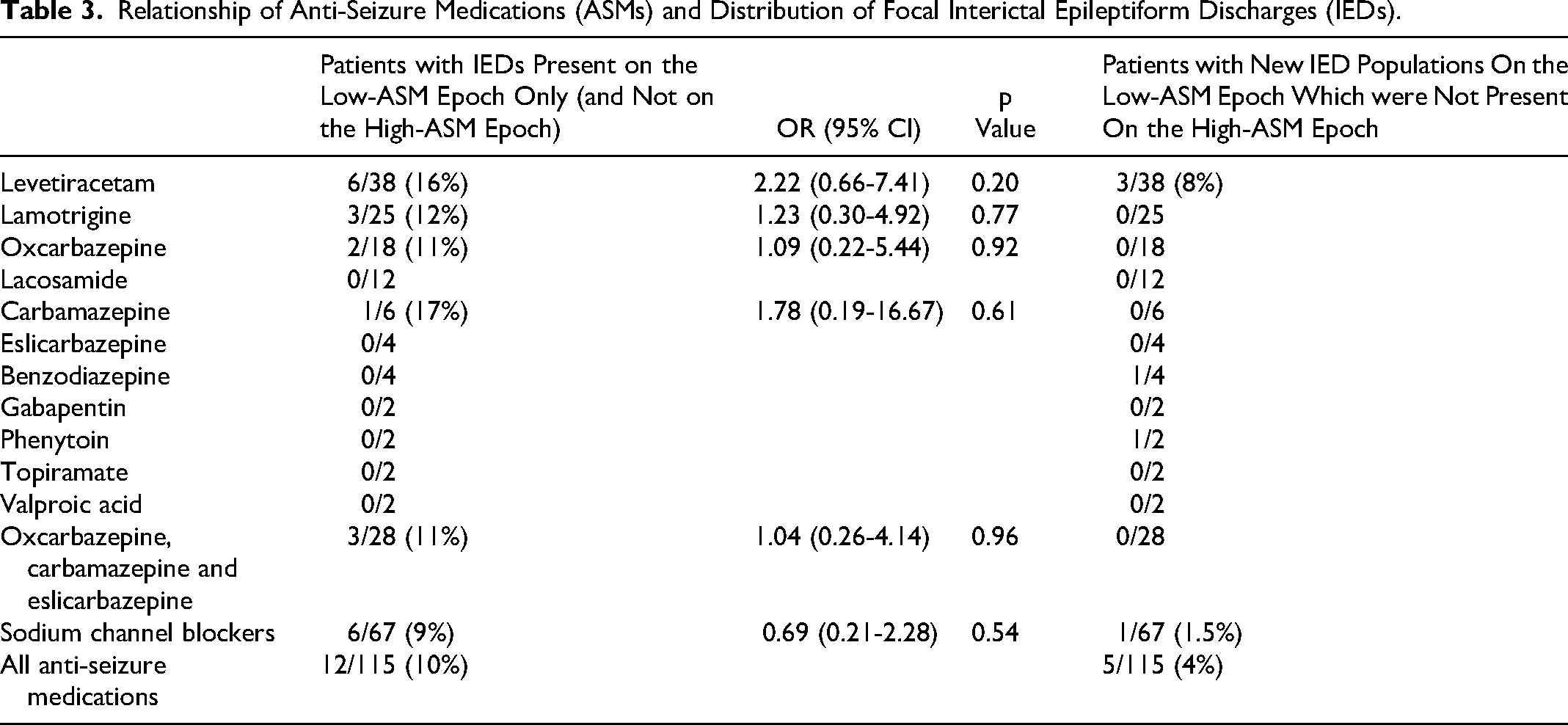
For the whole cohort, when ASM doses were tapered, 12 out of 115 (10%) patients had IED observed only on the low-ASM epoch which were not present on the high-ASM epoch (Table 3). Most of these patients were treated with levetiracetam, with 6 out of 38 patients (16%) found to have IEDs only on the low-ASM epoch. However, the odds ratio of finding new IEDs with the taper levetiracetam in comparison to other ASMs was not statistically significant (OR 2.22, 95% CI 0.66-7.41, p = 0.20). In addition, when ASM doses were decreased, a total of 5 out of 115 (4%) patients had new IED populations in different locations observed on the low-ASM epoch which were not originally present on the high-ASM epoch.
Relationship of Anti-Seizure Medications (ASMs) and Distribution of Focal Interictal Epileptiform Discharges (IEDs).
Suspected epilepsy etiology (either based on MRI lesion or tissue pathology where applicable) was recognized in 38 patients (33%), with mesial temporal lobe sclerosis being the most common (Table 1). When analyzing this group (regardless of ASM choice), there was no statistically significant change in IED quantity between the high-ASM and low-ASM epochs (p = 0.26).
Discussion
We found that as ASMs were weaned and discontinued in the EMU, the total quantity of IEDs increased significantly, which occurred independently of seizure activity. However, based on subgroup analysis, it was apparent that this result was largely produced by patients taking levetiracetam (which was the most common medication), rather than as a result of all ASMs collectively. Levetiracetam was the only ASM in our group for which there was a significant increase in IEDs between the high-ASM and low-ASM epochs. We also found that a subset of patients on levetiracetam only had IEDs after tapering ASMs (with IEDs absent on the high-ASM epoch). However, the odds ratio of having new IEDs was not statistically significant when comparing levetiracetam with other ASMs in the cohort.
Our findings with levetiracetam are comparable to that of most previous studies which demonstrated that this medication could suppress focal IEDs with both acute and chronic use.12–17 Given this, we suggest that if the goal of EMU monitoring is to characterize a patient's epilepsy, then levetiracetam should be tapered if safe to do so since it may influence the findings on the interictal EEG. Furthermore, extrapolating this information into the outpatient setting, in some circumstances, we suggest it may be beneficial to delay administration of levetiracetam to increase the potential diagnostic yield of EEG, if safe to do so. For example, holding levetiracetam in patients who have infrequent seizure-like episodes, and a routine EEG is ordered to evaluate for interictal activity.
It has been suggested that both presence and number of IEDs were associated with a slower task reaction time and word finding difficulties.8,9 Prior studies also suggested that for ASMs which can reduce IEDs, the improvement in neurocognitive function from that effect may outweigh ASM related adverse effects. 8 Even though IEDs can have a negative impact on cognitive performance, the current consensus generally recommends against treating patients for the presence of IEDs alone. 18 Given our unique findings with levetiracetam, additional studies should be conducted to clarify what circumstances may justify treatment of IEDs specifically for potential neurocognitive benefits. One prospective pilot study found that in patients with Alzheimer disease and comorbid epilepsy, treatment with levetiracetam improved performance on spatial memory and executive function tasks. 19
The mechanism by which ASMs interact with IEDs is incompletely understood. Prior in vitro studies have proposed two types of IEDs with opposing characteristics. 20 Slow GABAergic interictal discharges can lead to hypersynchronization of neuronal networks and thus can be pro-ictogenic. Fast interictal activity on the other hand can produce desynchronizing effects that can be anti-ictogenic. Historically, these IEDs have also been classified as “red” and “green” spikes. 20 However, the application of these theories into clinical practice is challenging, as on surface EEG tracings these spikes all have the same morphology. Levetiracetam has a unique mechanism of action by acting on the synaptic vesicle glycoprotein 2A (SV2A) which modulates neurotransmitter release. In vitro studies have demonstrated that levetiracetam has a unique ability to inhibit neuronal hypersynchrony compared to traditional sodium channel blocking ASMs which have more of an effect on single neuron activity. 21 Based on our findings with levetiracetam, the overall increase of IEDs after the ASM was weaned can be interpreted as a possible manifestation of this unique mechanism of action relative to other medications.
In similar related recent studies,3,5,6 the most common method to identify IED was based on automated spike detection algorithms which may have inherent inaccuracies and a low specificity. In addition, the majority of these patients were taking multiple ASMs and conclusions drawn from these heterogenous groups can be challenging. Specifically, there was one prior study 5 which also retrospectively reviewed the frequency of IED as measured on scalp EEG as ASMs were tapered in the EMU. They found a decrease in spike rate that accompanied ASM taper. An important distinction between our studies was that we only included patients on monotherapy and a large proportion of our patients were on levetiracetam. On the contrary, 89% of patients in their study were on polytherapy and may have been taking ASMs with different mechanisms of action. Another distinction between our studies is that they used an automated spike detection algorithm with an average spike rate of over 500 per hour for their entire cohort. This value is significantly higher than our average rate of IEDs, which may be due to false positive detections by their algorithm. It is also possible that by only including patients on monotherapy, we selected patients with epilepsy who are less pharmacoresistant and may have less frequent IEDs.
Our study found that for ASMs other than levetiracetam, when analyzed separately, there was no statistically significant change in IEDs as ASMs were tapered. However, several medications were not well represented in our cohort, with very few patients treated with carbamazepine, lacosamide, phenytoin, topiramate or valproic acid monotherapy. Thus, our study was likely not sufficiently powered to draw conclusions on these medications individually. We therefore performed a subgroup analysis on a combination of sodium channel blocking ASMs (defined as lamotrigine, oxcarbazepine, carbamazepine, eslicarbazepine, lacosamide, and phenytoin). With this, we found a non-significant trend towards an overall decrease in IED quantity as the ASM dose was decreased. The mechanism by which sodium channel blocking ASMs effects IEDs is not completely understood. An in vitro study looking at carbamazepine found that despite its effectiveness at reducing seizure rates, it actually increased the spike rate in the amygdala. 22 This suggests that the mechanism underlying seizure suppression and interictal spiking are distinct.
Review of the literature suggests that use of lamotrigine2,23,24 and topiramate2,25 can reduce IED incidence compared to baseline EEGs. On the other hand, there is conflicting evidence regarding the effect of carbamazepine, phenytoin and valproic acid on IEDs. 2 There is insufficient data on lacosamide, zonisamide and newer anti-seizure medications to make any conclusions. Future research could elucidate how these newer medications with variable mechanisms of action exhibit effects on different subtypes of IEDs.
Our study has several limitations, primarily the small sample size (with several medications not being well represented as discussed above). In addition, because of the small sample size, this did not allow for subgroup analysis based on the type or location of IEDs. By including only those patients treated with monotherapy, we theoretically did not sample the EEG of patients who were more medically refractory and require multiple ASMs. Two-hour EEG epochs were chosen for practical reasons, as it would not be feasible to manually review the entire long term EEG tracing for the purpose of this study. Since IED exhibit natural fluctuations, it is possible they were not well exemplified during this two-hour sample. However, to mitigate the diurnal variability, we initiated EEG review at the start of sleep onset in the evening for every patient. However, it is possible that the proportion of time that patients spent in different stages of sleep was not consistent. IEDs were evaluated according to standardized criteria, 10 but nonetheless we acknowledge there may have been instances where counting (or not counting) a specific sharp transient was ultimately subjective, and there could have been differences between study authors CL and JF. For practical reasons, authors were not blinded when collecting data and thus subconscious bias cannot be excluded. Future studies could attempt to further evaluate the relationship between IEDs and ASMs using validated automated spike detectors which would help mitigate the potential for bias. Due to the retrospective nature of the study, it was not feasible to obtain plasma ASM levels to further assess the impact on IED frequency. It is possible that due to differences in medication half-life, the baseline dose taken, the time from the last dose until the start of the low dose epoch, and pharmacogenetic differences that the ASMs levels were not uniform among patients during the low ASM epoch. However, given that most of the ASMs had short and similar half-life durations and that the low-dose ASM epoch was on median day 4 of admission, it is likely that levels were similarly low at that time. Additional prospective studies including those with larger sample sizes and plasma ASM levels would be beneficial to further evaluate the interrelationship of ASMs and IEDs in focal epilepsy.
Conclusion
The detection of IEDs is critically important in the diagnosis and evaluation of epilepsy. We found that weaning of levetiracetam was associated with a significant increase in IED frequency, with some patients also demonstrating IEDs in new distributions. On the contrary, there was no significant change in IED quantity with taper of other individual ASMs analyzed in this study.
Footnotes
Ethical Considerations
Not applicable.
Consent to Participate
The study protocol was reviewed by the Vanderbilt University Institutional Review Board and exemption status was granted. Patient consent was waived.
Consent for Publication
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Anonymized data not published within this article will be shared by request from any qualified investigator for purposes of replicating procedures and results.
