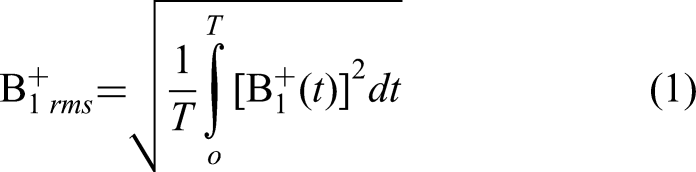Abstract
Magnetic resonance imaging (MRI) sequences commonly used in simultaneous electroencephalogram (EEG)-MRI studies include blood oxygenation level-dependent (BOLD) and anatomical T1-weighted MRI. Safety and electrode heating profiles for these sequences have been well-characterized. However, recent improvements in EEG design may allow for additional sequences to be performed with similar expectations of heating safety, which would expand the EEG-MRI infrastructure for quantitative physiological studies. We evaluated temperature changes ex vivo and in vivo over a wider range of preparation and readout modules with differing specific absorption rate (SAR). A 32-channel EEG cap was used at 3 T and ex vivo heating was assessed for 2D- and 3D-pseudo-continuous-arterial-spin-labeling, 2D-cine, 2D-phase-contrast, 2D T2-Relaxation-Under-Spin-Tagging, 32-direction b = 1000 s/mm2 and b = 2000 s/mm2 2D-diffusion tensor imaging, multiband-BOLD, 3D-T1 MPRAGE, 3D-FLAIR, and 3D-T2. Temperature was monitored with a fiberoptic probe system and plotted over six different electrodes, the amplifier, and battery pack. In vivo assessments were conducted in three participants with the same system. A further in vivo supplemental cohort (n = 10) was used to further evaluate qualitative self-reported heating. Device integrity was evaluated by the manufacturer following experiments. Peak temperature and maximum temperature increases were 23.0°C and 0.4°C respectively ex vivo, and 37.6°C and 0.7°C respectively in vivo. Temperatures did not approach the safety heating threshold of 40°C (defined as a conservative threshold based on manufacturer recommendations and burn injury data). Participants completed in vivo scans without adverse events. No manufacturer-reported device damage was identified. Overall, the tested scans induced heating below critical limits at the clinical field strength of 3 T.
Keywords
Introduction
Concurrent studies of electroencephalography (EEG) and functional magnetic resonance imaging (fMRI) using gradient echo blood oxygenation level-dependent (BOLD) contrast are frequently performed to evaluate neuronal and hemodynamic activity, respectively. The parallel use of these methodologies confers advantages in assessing brain activity, combining the high temporal resolution of EEG with the higher spatial resolution of BOLD-fMRI.1,2 As these methods report on different contrast mechanisms (i.e., electrical vs. hemodynamic activity), concurrent measurements provide a more complete perspective on brain function. Importantly, simultaneous EEG-fMRI using some commercially-available systems has been shown to be safe with a root-mean-square value of the MRI effective component of the RF transmit field (B1 + rms) of 1 μT or less3,4 and is being used in a growing number of evoked and resting state studies.5–7
Given the success of EEG-fMRI for mapping concurrent neuronal and hemodynamic activity in vivo, there is growing interest in how other MRI signal contrasts (including quantitative cerebral blood flow [CBF], diffusion, and most recently neurofluid circulation) adjust with behavioral state, or how quantitative measures from these contrasts are influenced by neuronal activity. Alternative imaging techniques such as arterial spin labeling (ASL), 8 high temporal resolution cine MRI,9,10 phase-contrast,11,12 and diffusion imaging may also have relevance in EEG-MRI protocols.13,14 Many of these sequences require higher radiofrequency (RF) transmission power, shorter repetition times with higher RF duty cycle, or multiple higher power RF pulses in repeated spin echo readouts, but heating of these sequences has not been previously evaluated in the setting of concurrent EEG.
The primary safety concern for EEG-MRI is excessive heating of the EEG electrodes or amplifier due to exposure to RF fields and B1 energy deposition in the scanner environment, increasing the risk of burn injury to the scalp or EEG equipment damage.3,4,15–17 This risk can be assessed with the specific absorption rate (SAR) of RF energy.18,19 The SAR value is defined as the amount of RF power absorbed per unit of mass of an object (indicated in W/kg), bearing a relationship with heating risk.
20
For EEG systems, important SAR variables are context-specific, including lead number, electrical properties of lead materials, tissue type in proximity, and RF field properties.21–24 The B1 + rms, or root-mean-square value of the effective component of the RF transmit field, is a summative measure of RF power for safety considerations.
20
It can be defined as:
However, limited temperature testing has been performed for the 1-1.5 µT range in EEG-MRI applications, mostly via private-sector internal evaluations. 25 Given the complex interplay between participant geometry, RF deposition, and heating, practical and in vivo empirical testing provides notable utility complementary to pure in silico simulation. In this work, we provide a protocol for assessing and systematically measuring temperature changes in EEG equipment, while evaluating emerging sequences in the context of EEG-MRI that are tailored to quantifying anatomy, dynamic neurofluid flow, and metabolism. We provide systematic temperature assessments across a range of sequences ex vivo and evaluate participant responses to these sequences through repeated measurements in vivo. Findings are intended to serve as a reference for the growing number of EEG-MRI studies that seek to utilize contrast beyond BOLD and structural imaging.
Methods
EEG and Ancillary Equipment
All tests used a 32-channel BrainCap MR system with a fronto-central cable bundle exit point (Brain Products GmbH, Gilching, Germany), which is a common commercially available MR-compatible EEG system and is analogous to many custom-built systems. This system contains ring-type sintered nonmagnetic Ag/AgCl electrodes. The default manufacturer electrode layout was used, with FCz as the reference, AFz as the ground, 26 EEG head electrodes, and four drop-down electrodes (one electrocardiogram [ECG] electrode attached at the left mid-thorax and three facial electromyogram [EMG] electrodes attached at the anterior mandible). Current-limiting resistors are built into the system, including 5 kΩ in scalp electrodes, 10 kΩ in reference/ground electrodes, 15 kΩ at drop-down electrodes, and an additive 5 kΩ at the amplifier box. Maximum B1 + rms is specified at 1.5 µT (3 T). The cap was selected based on object/head size (56 or 58 cm cap options). The cap cable tree was 31 cm, and a 10 cm cap-amplifier cable in a bundled format was used. EEG signal was amplified using a BrainAmp MR Plus amplifier, powered using a BrainVision PowerPack battery, and recorded with BrainVision Recorder software. The system was stabilized in the scanner using a plastic sled and sandbags (see Figure 1B for depiction of sled containing amplifier and battery pack). For experiments using temperature probes, all impedances were maintained below 5 kΩ for ground and reference electrodes and below 15 kΩ for all other electrodes via use of abrasive conductive gel (Abralyt HiCl [EasyCap GmbH, Wörthsee, Germany]). For human experiments without temperature probes, impedances were kept below 10 and 30 kΩ respectively, due to practical considerations.
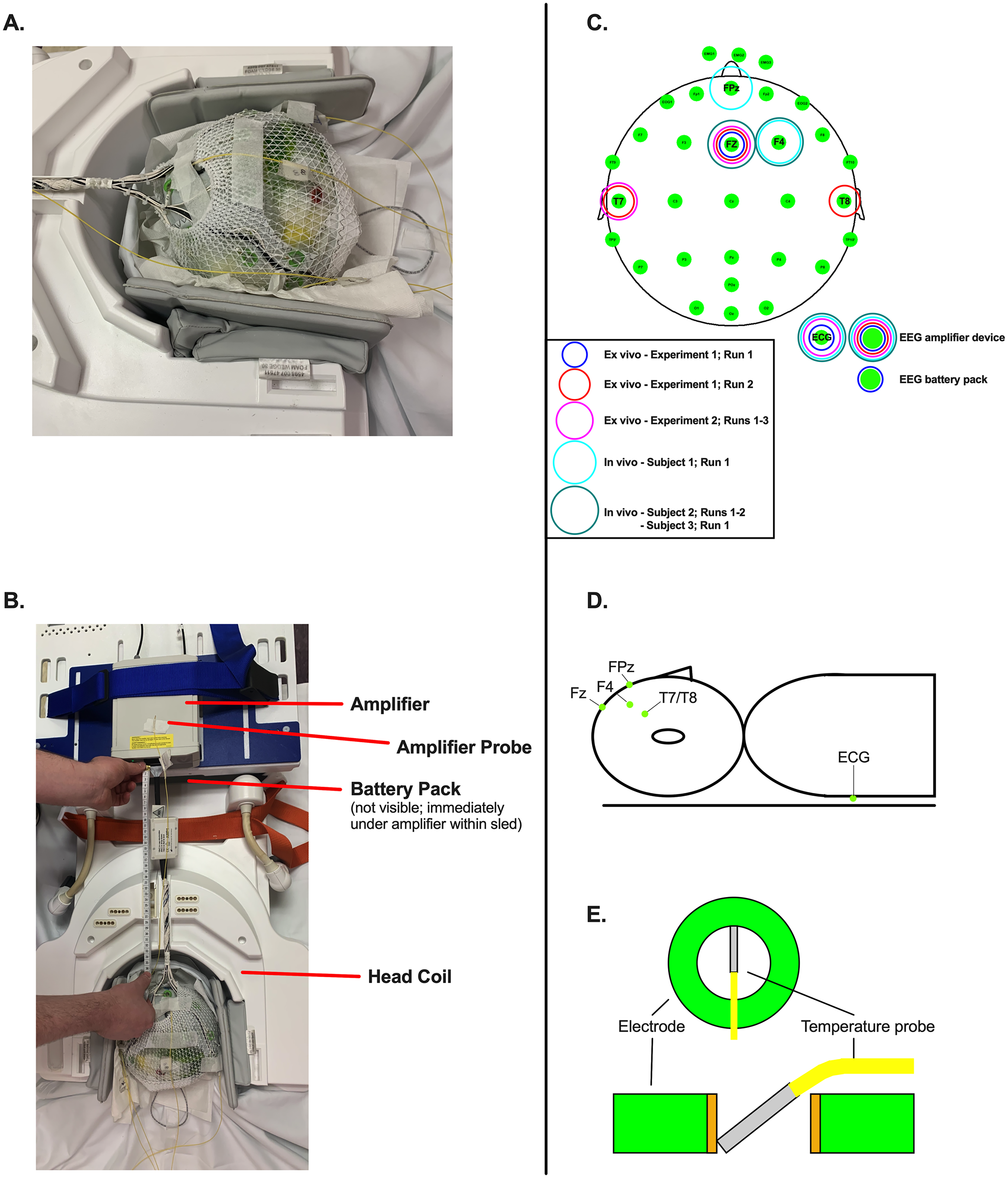
Temperatures were measured using four T1 fiberoptic probes (Neoptix, Inc., Québec, Canada) placed under electrodes connected to an outside interface device (Neoptix Reflex Signal Conditioner). Electrodes were composed of temperature gallium arsenide (GaAs) 0.4 mm crystals surrounded by a pure virgin PTFE Teflon™ standard 1.1 mm O.D. jacket, deriving temperature from the band-gap variation in the GaAs absorption spectrum. This system provides a maximum resolution of 0.1°C with an accuracy of the greater of 1°C and a response time of less than 500 milliseconds. 27 Probes were inserted over the top of the EEG cap, with the ends of the probes resting against the inner edge of the electrode rings and embedded in the layer of gel between the phantom surface and the EEG cap (see Figure 1 for depiction of probe placement). Temperatures were recorded at a 1 Hz sampling rate using OptiLink software (Neoptix, Inc., Québec, Canada).
Probe Validation
Prior to EEG-MRI experiments, the temperature probe system underwent evaluation for observation latency and accuracy (Supplementary Figure 1). The purpose of this preliminary assessment was to ensure that the temperature monitoring was accurate in a controlled environment of known, independently manipulated temperature. A 600 mL glass beaker was filled with 500 mL of tap water. Probes were secured at 2 cm water depth with tape. The beaker was placed on a Fisher Scientific hotplate, and a glass Thermo Fisher 0-100°C thermometer was inserted (Thermo Fisher Scientific Inc., Waltham, MA). The hotplate was set to 70°C and temperatures of both the thermometer and the probes were recorded at 30-s intervals. After 40 min, the hot plate was set to 200°C and temperatures were recorded at 15-s intervals for 15 additional minutes.
Ex vivo EEG-MRI
Images were collected on a Philips Ingenia system at 3 T with body coil RF transmission and 32-channel SENSE phased-array reception (Philips Healthcare, Best, The Netherlands). The use of body coil for RF transmission is an important consideration, as this selection could impact temperature effects; this method was selected due to EEG manufacturer recommendations for system operation. 28 A range of representative sequences with different readouts were tested, including functional sequences (ASL, cerebrospinal fluid [CSF] phase-contrast, cine, T2-relaxation-under-spin-tagging [TRUST], and BOLD) as well as structural sequences (T1-weighted, T2-weighted, T2-weighted fluid-attenuated inversion recovery [FLAIR], and diffusion tensor imaging [DTI]). Sequences were collected consecutively as rapidly as permitted by the scanner, and there were no breaks or cooling periods during a given run. Scan breaks were only indicated to reset probe positions and were defined as a new run. Notably, there are important variations in RF pulses for the different sequences which influence SAR and B1 +rms. For example, standard BOLD sequences have lower RF power with a single excitation pulse per slice per TR and gradient echo, while standard DTI sequences have an excitation plus a refocusing pulse per slice per TR, and pCASL is known to be particularly RF intensive due to the 1.5-2.0 s train of RF pulses with moderate-to-high duty cycle required for arterial blood water labeling. As a result, variation in heating is expected and assessed in this study. Some sequence parameter values differed slightly due to a scanner upgrade completed during data collection. Imaging parameters are summarized in Table 1.
Imaging Sequences.
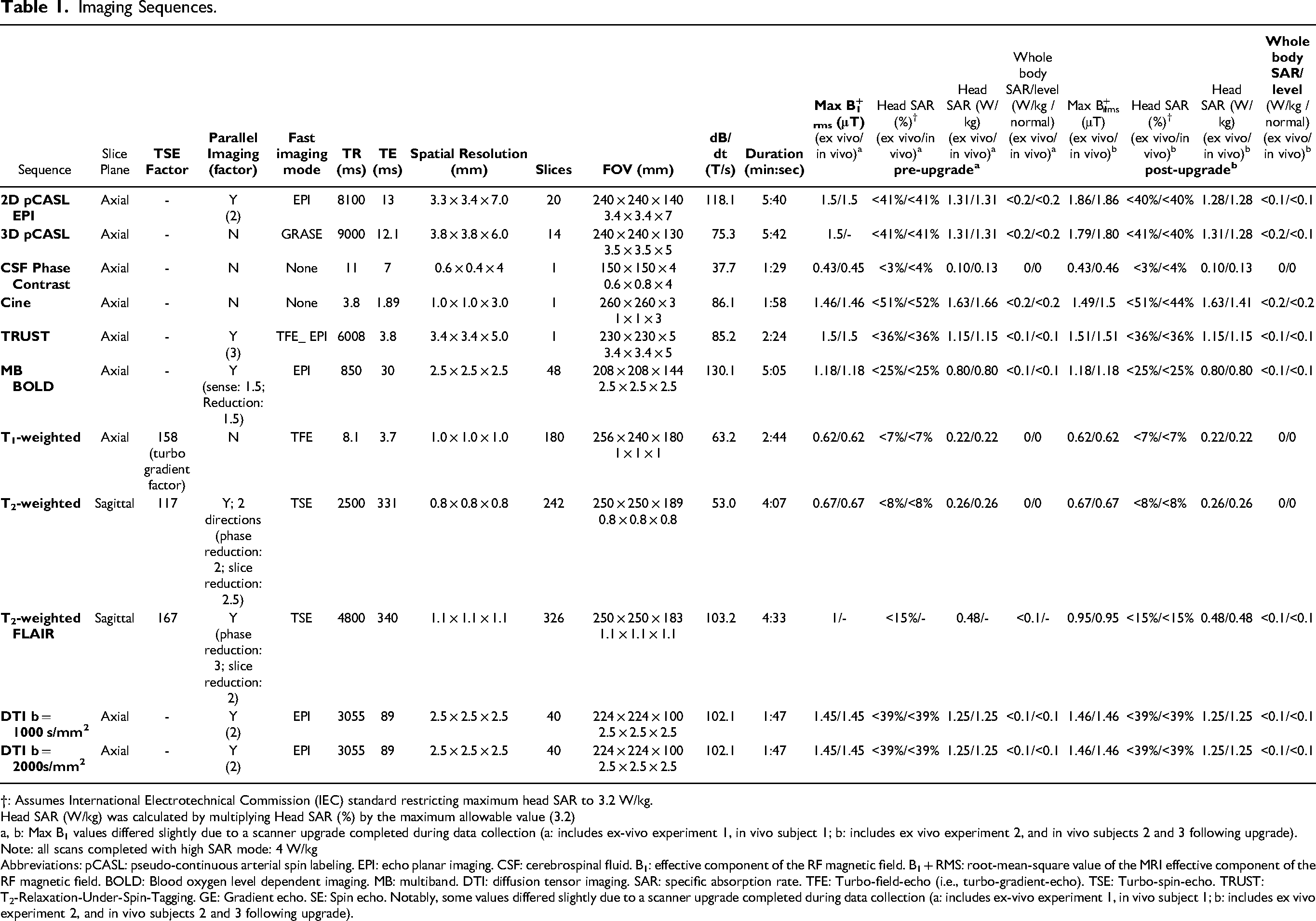
†: Assumes International Electrotechnical Commission (IEC) standard restricting maximum head SAR to 3.2 W/kg.
Head SAR (W/kg) was calculated by multiplying Head SAR (%) by the maximum allowable value (3.2)
a, b: Max B1 values differed slightly due to a scanner upgrade completed during data collection (a: includes ex-vivo experiment 1, in vivo subject 1; b: includes ex vivo experiment 2, and in vivo subjects 2 and 3 following upgrade).
Note: all scans completed with high SAR mode: 4 W/kg
Abbreviations: pCASL: pseudo-continuous arterial spin labeling. EPI: echo planar imaging. CSF: cerebrospinal fluid. B1: effective component of the RF magnetic field. B1 + RMS: root-mean-square value of the MRI effective component of the RF magnetic field. BOLD: Blood oxygen level dependent imaging. MB: multiband. DTI: diffusion tensor imaging. SAR: specific absorption rate. TFE: Turbo-field-echo (i.e., turbo-gradient-echo). TSE: Turbo-spin-echo. TRUST: T2-Relaxation-Under-Spin-Tagging. GE: Gradient echo. SE: Spin echo. Notably, some values differed slightly due to a scanner upgrade completed during data collection (a: includes ex-vivo experiment 1, in vivo subject 1; b: includes ex vivo experiment 2, and in vivo subjects 2 and 3 following upgrade).
Ex vivo testing of the EEG-MRI system was conducted on a watermelon phantom due to conductivity properties
16
and comparable size and shape to a human head (circumferences of 53 and 55 cm on the short and long axes respectively). The phantom underwent overnight temperature thermalisation, was abraded with silicon carbide sandpaper and covered with Abralyt gel to improve conductance, and was then fit with the 56 cm EEG cap. The ECG electrode was routed underneath and attached to the anterior surface with tape, while EMG electrodes were routed under the anterior lip of the cap, resting approximately 2 cm from the fabric border. Temperature probes were inserted over the top of the EEG cap, with the ends of the probes resting against the inner edge of the electrode rings and embedded in the layer of gel between the phantom surface and the EEG cap, and wires firmly secured. Amplifier and battery pack probes were placed in the center of the rectangular structures and secured with tape (Figure 1A-B
To evaluate spatial heating dependence and more fully sample local RF topography, the phantom was then removed from the scanner, and probes were moved to the following positions: Probe 1:amplifier; Probe 2:T7; Probe 3:Fz; Probe 4:T8. Figure 1C depicts evaluated electrodes. This set of parameters was defined as Ex Vivo Experiment 1; Run 2. The amplifier and Fz probes were monitored during both runs due to the value of the amplifier system and prior evidence that B1 fields may have the most significant effect near the superior pole of the head. 24
Following analysis and interpretation of the results of Ex Vivo Experiment 1; Runs 1 and 2, an additional ex vivo experiment was conducted. Ex Vivo Experiment 2 included three runs. Probes were set at the following positions for all runs, as Experiment 1 had captured data from a representative sample combined with aforementioned interest in assessing the amplifier system and superior pole of the head: Probe 1:amplifier; Probe 2:ECG; Probe 3:Fz; Probe 4:T7. During Ex Vivo Experiment 2; Run 1, no scanning was completed in order to assess temperature equilibration in the scanner environment. During Ex Vivo Experiment 2; Run 2; scanning was completed with a full set of sequences in the same order used in Ex Vivo Experiment 1; Runs 1 and 2 in order to provide additional replicate data. During Ex Vivo Experiment 2; Run 3, scanning was completed with a full set of sequences in a different order from prior which attempted to arrange high maximum B1 +rms sequences consecutively during scanning, in an attempt to eliminate scan order as a confound and assess the effect of consecutive high maximum B1 +rms sequences.
An exploratory supplementary analysis was then completed based on preliminary results from ex vivo runs, defined as Ex Vivo Experiment 3. For three sequences that appeared to demonstrate heating indications (cine, TRUST, and MB BOLD), extended sequence recordings were designed with the goal of continuous scanning for a goal duration of approximately 15 min. Due to sequence design and scanner console limitations, extended durations were accomplished either by extending the scanning duration (MB BOLD) or repeating 2-3 instances of the normal scan sequence used in prior runs consecutively (cine, TRUST); this resulted in very brief scanning pauses for the latter sequences that could not be eliminated. Extended runs were completed in the following order: cine, TRUST, MB BOLD. Probes were set at the same positions as Ex Vivo Experiment 2: Probe 1:amplifier; Probe 2:ECG; Probe 3:Fz; Probe 4:T7.
In Vivo EEG-MRI
In vivo experiments were conducted in two phases. In phase one, we performed quantitative temperature mapping in three healthy adults while utilizing the same scans included in the ex vivo experiment. In phase two, we performed MRI sequences (T1-weighted, T2-weighted, DTI with b = 1000 s/mm2), 3D pCASL, CSF phase-contrast, and cine) in nine participants without quantitative temperature recording to determine whether qualitative heating was detectable by the participant.
For both phases, participants provided informed, written consent in accordance with the Vanderbilt University Institutional Review Board (IRB#202527) and the Declaration of Helsinki and its amendments. Specific counseling on the purposes and risks of this study was provided, and participants were instructed that study procedures would be immediately stopped if any heating was observed. The skin was prepared with 90% isopropyl alcohol, and the appropriate cap was fitted. Abralyt gel was liberally applied to electrodes, and the skin was abraded with a cotton swab; drop-down electrodes were appropriately applied as described previously. Cap fit was secured via cloth webbing.
In phase one, once the participant was resting comfortably on the scanner bed, temperature probes were placed at the following locations: Probe 1:ECG; Probe 2:amplifier; Probe 3:FPz (participant 1) or Fz (participants 2 and 3); Probe 4:F4. Altered placement of probe 3 at FPz vs Fz was due to participant head size and shape constraints while allowing comparable spatial sampling. Participants were instructed to pay close attention to any feeling of electrode heating and to promptly alert scanner staff via a squeeze-ball. Temperatures were closely monitored, with scan stoppage criteria of a rapid increase in temperature (greater than 3°C in a 30-s period), stable high temperature (>40°C), or heating observed by the subject. These stoppage criteria were based on prior evidence that exposure to 45°C or increases in skin temperature greater than 8°C for more than two hours can result in burns as well as the ICNIRP guidelines, and therefore were conservative.29–34 For participant 1, one run was completed with scanning, defined as In Vivo Subject 1; Run 1. For participant 2, two runs were completed: In Vivo Subject 2; Run 1 was completed without scanning but with a recording duration comparable to scanning runs, in order to assess temperature equilibration in the scanner environment. In Vivo Subject 2; Run 2 was subsequently completed with scanning immediately after equilibration, to delineate scanning effects with the possible equilibration confound removed. For participant 3, one run was completed with scanning, defined as In Vivo Subject 3; Run 1. In vivo scanning runs used slightly different sequence orders to minimize the effect of delayed responses to consecutive sequences.
In phase two of the in vivo study, no temperature probes were applied, and participants received the scans listed above; this was intended as a supplementary qualitative assessment of heating once scanning procedures appeared quantitatively within safety thresholds.
Analysis
Following all experiments, the EEG caps and amplifier were returned to the manufacturer for quality-control assessments to determine if any deficiencies in sensitivity or functionality arose. Mean and peak temperature changes for each sequence were recorded across ex vivo and in vivo runs in order to provide summative statistics. The relationship between B1 +rms and temperature change was evaluated with Pearson's correlation analyses. Separate analyses were conducted grouping measurements at scalp, ECG, amplifier, and battery pack electrodes, as well as for all electrodes combined. This was repeated for the following groupings: ex vivo, in vivo, and all runs combined. The relative effect of B1 +rms on temperature change was analyzed via general linear regression model (GLM) where temperature change was the dependent variable, and B1 +rms, sequence, electrode site category, and ex or in vivo status were dependent variables. False discovery rate (FDR) was controlled at 0.05 to correct for multiple comparisons in all analyses. All analyses were performed using Prism 10 (GraphPad, Boston, MA, USA) and the Microsoft Excel Analysis ToolPak (Redmond, WA, USA).
Results
Probe Validation
At the 200°C setting, although the thermometer appeared to have a slightly more rapid response to increasing temperature, the discrepancy between temperatures measured by the fiberoptic probe system and the gold-standard thermometer did not appear grossly distorted and the temperature changes detected from the fiberoptic probe system paralleled dynamic temperature manipulations. Mean differences and standard deviations were collected (probe 1: 0.84 ± 0.68°C; probe 2: 0.92 ± 0.83°C; probe 3: 1.02 ± 0.91°C; probe 4: 1.35 ± 1.05°C). The probe system was concluded to be effective for rapidly determining temperature changes within the range of possible RF-induced heating. Supplementary Figure 1 provides visualization of the experimental setup and temperature change.
Ex Vivo EEG-MRI Assessment
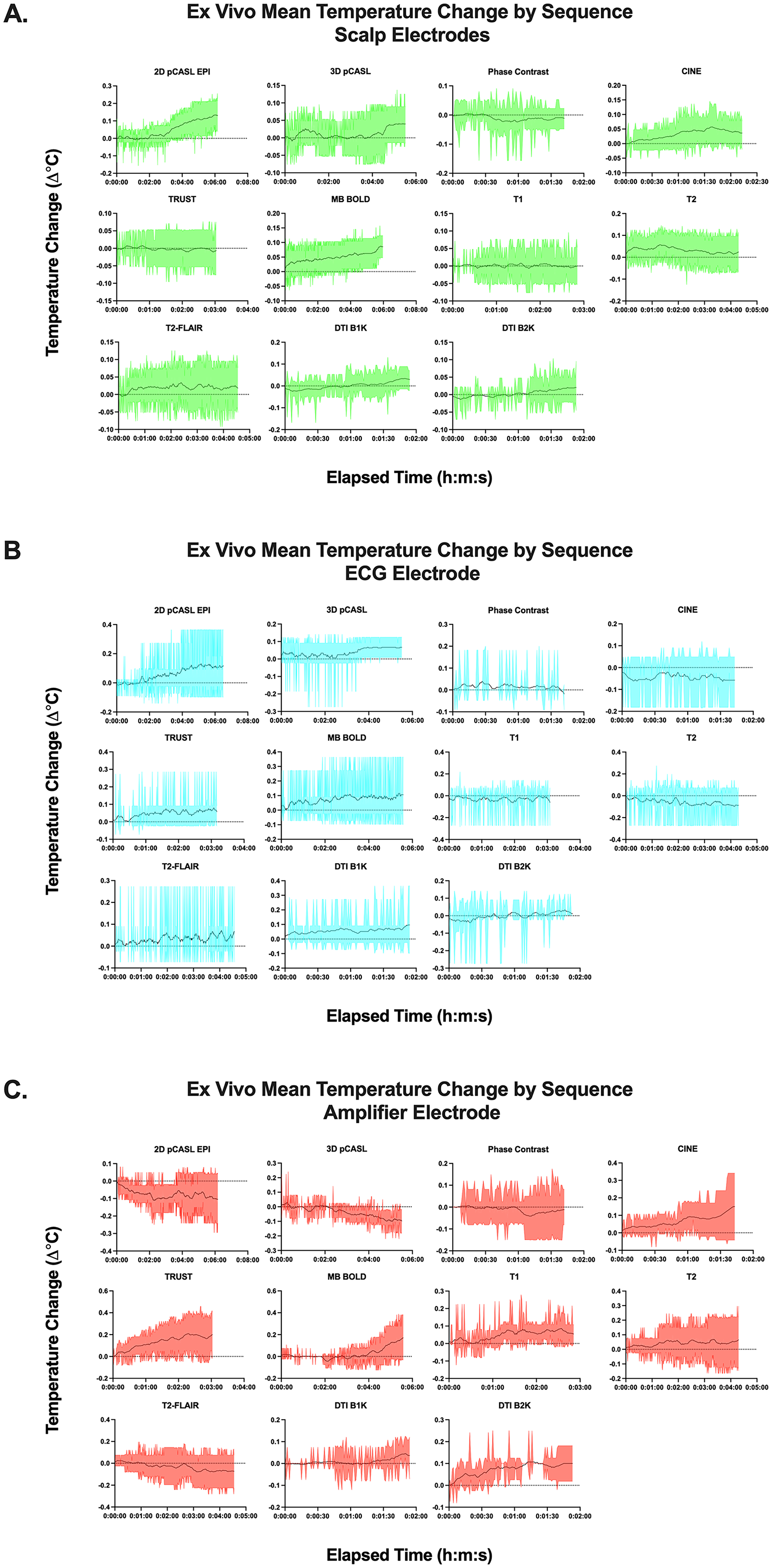
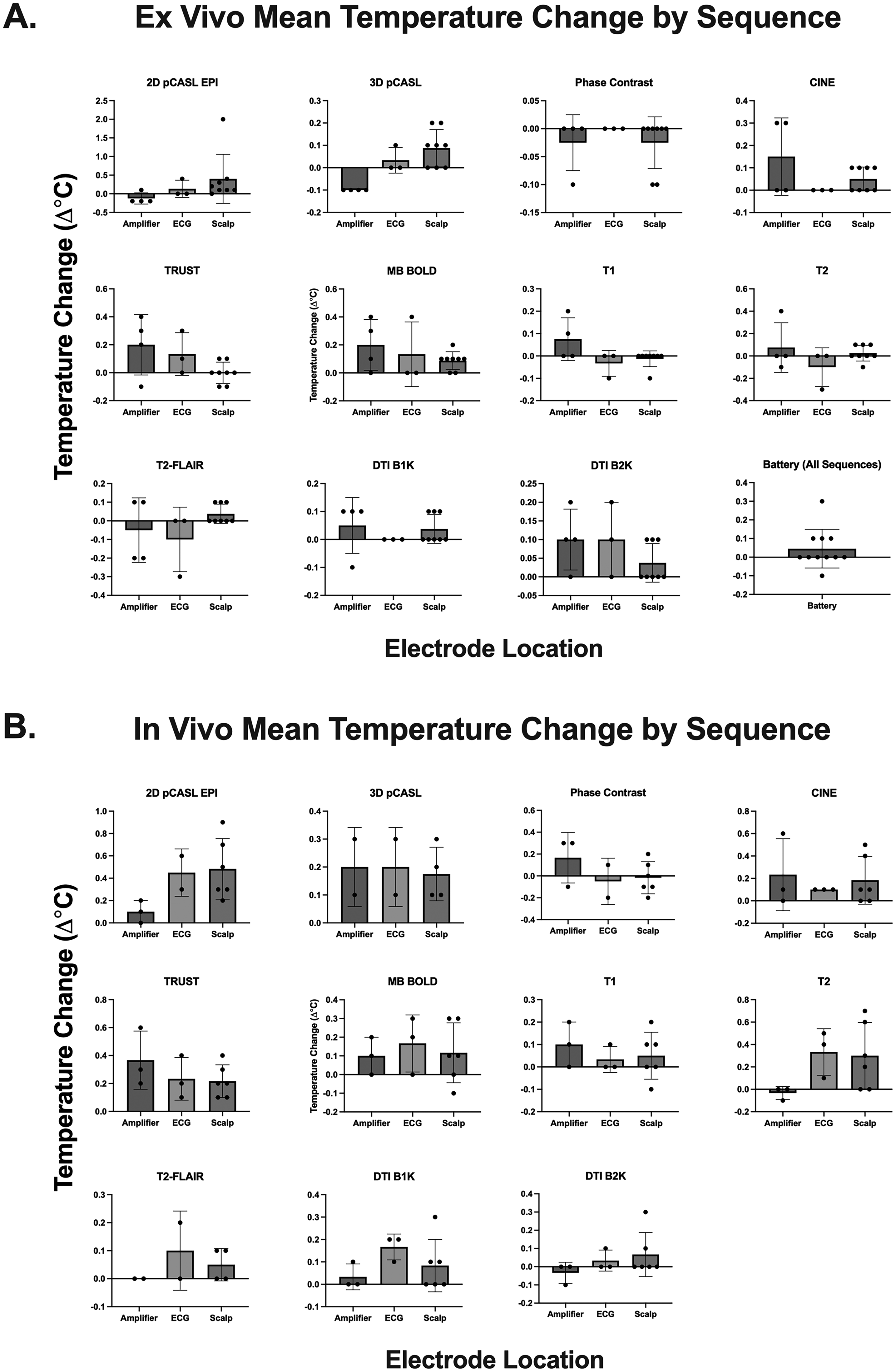
Temperature changes from the ex vivo study are summarized in Supplementary Table 1. Individual temperature and temperature change timecourses for ex vivo trials are displayed in Supplementary Figures 2 and 3. The maximum temperature across all probes and repetitions was 23.0°C, and there were no temperature increases greater than 3°C across the entirety of the scan protocol. A generalized pattern of increased temperature of less than 0.5°C was observed in all probes. More acute increases in temperature were observed in the cine, TRUST, and MB BOLD scans in the amplifier probe. However, the magnitude of these increases was not observed to approach the 40°C equipment damage threshold
28
or to be physiologically significant (greater than 3°C in a 30-s period, or stable high temperature greater than 40°C, as defined based on previous human and porcine studies32,33) with a maximum of 0.3°C (cine and TRUST) and 0.4°C (BOLD) across repeats. Equilibration to the scanner environment did not appear to be associated with considerable temperature change with a maximum increase of 0.3°C at Fz (Supplementary Table 2; Supplementary Figures 2C and 3C). In comparison to an ex vivo run completed immediately after setup, an ex vivo run preceded by a substantial equilibration period generally exhibited slightly lower temperatures (with a mean peak temperature difference of 0.8 ± 0.9°C lower in post-equilibration scans when evaluated in a pairwise manner
The extended supplementary runs for cine, TRUST, and MB BOLD indicated some heating (see Supplementary Table 3 and Supplementary Figure 4). Cine imaging exhibited notable temperature increases in the amplifier electrode (2.0°C increase with a 24.4°C peak) with smaller increases of 0.2-0.7°C in other electrodes. The pattern of heating appeared to parallel the scan structure in which 3 separate consecutive runs of approximately 5 min were used to compose the full 15 min scanning period, thereby defining two observable peaks and troughs. The subsequent extended TRUST scan exhibited falling temperatures, while the extended MB BOLD scan exhibited a 0.4°C increase in the amplifier electrode and no change for other electrodes.
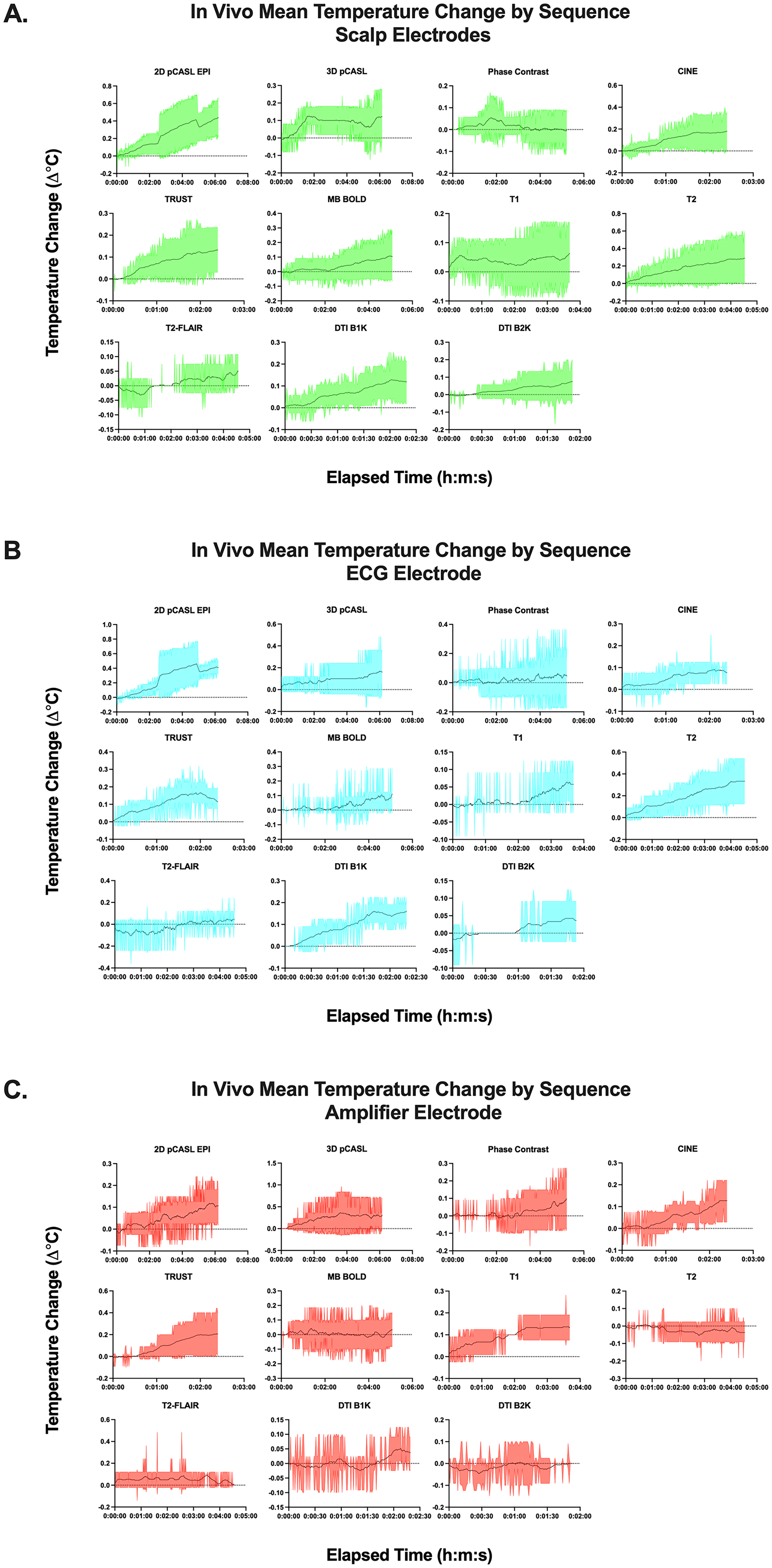
In Vivo EEG-MRI Assessment
Data from the first phase of the in vivo studies are summarized in Supplementary Table 1. Individual temperature and temperature change timecourses for ex vivo trials are displayed in Supplementary Figures 5 and 6; Supplementary Figure 7 provides comparative display following temperature equilibration in Subject 2; Run 2 relative to all runs combined
Equilibration to the subject-electrode complex and scanner environment appeared to be associated with temperature increases, with a maximum increase of 2.1°C at Fz/F4 (Supplementary Table 5; Supplementary Figures 5B and 6B). In comparison to an in vivo run completed immediately after setup, an in vivo run preceded by a substantial equilibration period generally exhibited slightly lower temperatures (with a mean peak temperature difference of 1.8 ± 2.0°C lower in post-equilibration scans when evaluated in a pairwise manner [Supplementary Table 4]); temperature change did not appear to be grossly different, with a maximum of 0.6Δ°C difference between the runs for 2D pCASL and smaller differences elsewhere (with a mean temperature change difference of 0.1 ± 0.2°C lower in post-equilibration scans when evaluated in a pairwise manner [Supplementary Table 1]). Individual timecourses following equilibration varied by sequence (Supplementary Figure 7).
For the second phase of the in vivo studies focused on qualitative assessment, subjects varied in demographic parameters (sex: 7M/3F; weight: 59-103 kg; age: 22-47 years). No participants noted temperature heating that prevented scan continuation. One participant noted a mild sensation of warmth on the face and head after entering the scanner bore, which was not restricted to the area in contact with the EEG cap; the participant elected to continue the scan and the sensation abated. No participants attributed sensations to cap heating.
Summative Analyses
Upon re-evaluation of the cap and amplifier functionality by the manufacturer, the equipment was determined to be in normal working order with no observed occurrences of malfunction. Summative average temperature change data is displayed in Supplementary Table 6 and Figures 2-4; broadly, the largest average temperature increases were 0.4 ± 0.7°C for 2D pCASL for ex vivo trials, and 0.5 ± 0.3°C for 2D pCASL for in vivo trials. There were significant associations between temperature change and B1 + rms with the following relationships passing multiple comparisons correction: all electrodes/in vivo (r = 0.26; p = 0.0034), scalp electrodes/all runs (r = 0.25; p = 0.002), and all electrodes/all runs (r = 0.20; p = 0.0005) (Supplementary Table 7). GLM analysis indicated a significant effect of B1 + rms (t statistic = 0.03, p = 0.009) and ex or in vivo status (t statistic = 4.3, p < 0.0001) on temperature change which passed multiple comparisons correction, but no significant effect for electrode category or sequence (Supplementary Table 8). Summative graphics for ex vivo trials (Figures 2-4) indicated broad patterns of increasing temperature trajectory for some electrodes with an approximate 0.2°C (scalp/ECG: 2D pCASL, MB BOLD; amplifier: CINE, TRUST, and MB BOLD); measurements at other electrodes appeared broadly stable. Similar patterns were exhibited for in vivo trials (Figures 2-4) with some electrodes appearing to exhibit a mildly increasing temperature trajectory with an approximate 0.2°C increase (scalp/ECG: 2D pCASL, T2; amplifier: CINE, TRUST); measurements at other electrodes appeared broadly stable.
Discussion
We evaluated temperature changes in an EEG-MRI system using emerging functional and conventional anatomical imaging sequences at the clinical field strength of 3 T. Generalized temperature increases (approximately 0.5°C in all probes during phantom runs and 2-3°C in skin electrodes during human runs) were not observed to be physiologically significant, to approach concerning levels for equipment damage, to affect participant comfort, or to affect equipment functionality. This observation was substantiated by the gradual timecourse of changes and a slower rate of increase as the scan progressed. Extended trials indicated potential heating of the amplifier during cine and MB BOLD imaging. Significant relationships between B1 + rms and temperature change were observed which appeared to be more prominent with larger sample sizes that combined data from all electrodes and all runs. Variation in temperature change appeared to be best explained by B1 + rms and ex or in vivo status, rather than electrode type or sequence. Temperature equilibration appeared to exert a notable effect during in vivo trials which interacted with scan order, but scan order did not otherwise appear to impact heating.
Range of Temperature Changes Observed in the Context of Physiological Skin Temperature
Prior recommendations from the International Electrotechnical Commission have specified a maximum 1°C increase in core body temperature during MRI scans. 30 However, the limit for skin temperature increases is less conservative, and modified by both exposure time and temperature. Human skin temperature does not exceed 35°C under healthy physiological conditions, where it lies between core body and ambient temperatures.29,30 A previous study of thermal injury on porcine skin, which is the closest animal surrogate of human skin tissue, 35 indicated 2nd-3rd degree burns from a 3-h exposure to 45°C; however, reducing the exposure time to 2.5 h decreased the severity to 1st degree burns, and reducing the temperature to 44°C increased the exposure time required to produce 2nd-3rd degree burns to 7 h. 31 Evidence in human subjects suggests that the perception of pain begins just above 43°C, and burn injury begins when the dermal-epidermal junction reaches approximately 44°C. 33 Overall, this indicates that increased skin temperature greater than 8°C and sustained for two hours would likely be required to produce 1st degree burns. 16 Our tests did not approach this threshold on skin-contact electrodes, with temperature maxima of 23.0°C for phantom scans (3D pCASL, T1-weighted, DTI b = 1000 s/mm2) and 37.6°C for human scans (TRUST), and temperature change maxima of 0.4°C for phantom scans (BOLD and 2D pCASL) and 0.7°C for human scans (2D pCASL).
During the in vivo test, a consistent gradual temperature increase was observed across electrodes in a majority of sequences, with more notable effects in skin-contact electrodes. Summative data across in vivo trials indicated more notable temperature increases of approximately 0.3°C and 0.5°C respectively for 2D pCASL and T2. The acute 1°C increase observed in the 2D pCASL of subject 1; run 1 was most likely consistent with data artifact or patient movement due the manner in which the temperature suddenly increased and decreased by the same magnitude, and the lack of similar observations in other trials. This effect could be due to equilibration of temperature probes to participant skin temperature, which might be elevated in the context of intra-scan conditions (including occupying an enclosed space with the majority of the body surface covered by a blanket). However, this component could also be attributed to scanner-induced heating. Alternatively, other unexpected factors (such as the potential for heat insulating characteristics of the EEG mesh cap material) might also play a role. The single run allowing an approximately 40-min equilibration period prior to scan initiation (In Vivo Subject 2; Run 2) was conducted to assess the relative contribution of these components. During this trial, there was a less notable temperature increase, indicating a likely contribution from temperature equilibration to the scanner environment in other trials. However, further temperature increases of approximately 2°C total (and 0.3-0.9°C individually) were observed for one skin contact electrode (Fz) during 2D pCASL, DTI (b = 1000 s/mm2), CINE, and TRUST scans. This may be more indicative of scanner-induced heating, especially given prior evidence on the significant effect of B1 fields near the superior pole of the head. 24 This trial also complicates interpretation of aforementioned summative group data: heating during T2 scans apparent in the aggregate data was not exhibited after equilibration, and conversely, temperature increases were observed for DTI (b = 1000 s/mm2), CINE, and TRUST scans that were not apparent in aggregate. Only 2D pCASL appeared to exhibit an effect across trials. T2 scan temperatures may have been unduly influenced by inclusion near the beginning of the protocol when thermalisation effects may have been most prominent. Sequence order did not otherwise appear to broadly affect heating in trials with varied orders. However, the overall pattern did not approach the threshold for stoppage criteria or known safety concerns. These data, therefore, do not indicate that these sequences cause obvious unsafe temperature changes in human participants for scan lengths of approximately 1 h.
For electrodes on EEG equipment (amplifier and battery), temperature maxima were 23.0°C for phantom scans (DTI) and 23.9°C for human scans (TRUST), and temperature change maxima were 0.4°C for phantom scans (BOLD) and 0.6°C for human scans (cine). Acute temperature increases consistently occurred at the amplifier during cine, TRUST, and BOLD scans. Extended supplementary runs indicated heating for cine (approximately 2°C for amplifier and 0.5°C for scalp/ECG electrodes) and MB BOLD (approximately 0.4°C for amplifier electrodes). Cine equilibrium temperature did not appear to be achieved after approximately 15 min of scanning, with a pattern of continued heating. However, overall temperatures did not approach the manufacturer-recommended 40°C threshold for equipment damage, and independent equipment re-evaluation by the manufacturer did not observe functionality changes or damage in our non-consecutive acquisition schema. Reasons for divergent heating patterns between EEG cap components and the amplifier are not clear; the RF field is known to vary throughout the scanner bore which could result in a difference in RF field experienced by the scanner. However, future studies might more directly assess the cause of this phenomenon.
Prior comparable studies mostly evaluated the effects of MB BOLD with reported temperature increases of approximately 1.0°C or less,3,4,36 with one of these studies also assessing temperature. Our ex vivo results were in line with this figure while our in vivo results exhibited relatively larger increases in temperature, although still within what is reported to be a safe range. This could be due to differences in temperature equipment, subject-specific differences in a small sample size, and differences in scan paradigm.
Range of Sequences Evaluated
The motivation for this work arose from the need to implement sequences with growing relevance to characterizing brain health in multimodal paradigms. While BOLD-fMRI, the most common neuroimaging approach for assessing human cerebral hemodynamics, has been well-characterized in terms of EEG-MRI safety, it provides only a surrogate marker of hemodynamic activity secondary to changes in intravascular and extravascular water relaxation in capillaries and draining veins. ASL, which provides a direct marker of perfusion (ml/100 g/min) has now been applied in more than 5,000 technical and application studies, with both 2D echo-planar imaging (EPI) and 3D gradient- and spin-echo (GRASE) readouts, as well as pulsed and pseudocontinuous arterial spin labeling. 8 Additionally, both cine and phase contrast scans are being applied with increasing frequency to evaluate blood and neurofluid flow, both of which are known to be modulated by neuronal activity, aging, and behavioral state.37,38 Multiband and multiplexed imaging is being used with increasing frequency to reduce scan times and increase temporal resolution in both functional and structural connectivity studies, 39 however, historical EEG-MRI temperature assessments have generally been made for older 2D EPI readouts only. Finally, while 3D T1-weighted scans are useful for localization and co-registration, they provide an incomplete perspective on tissue health, which requires additional contrasts from T2-weighted and T2-weighted FLAIR MRI. The findings from this study, which report on temperature changes over the wide range of these popular sequences, magnetization preparations, and readouts, should provide a basis for incorporating these sequences into future concurrent EEG-MRI studies.
Limitations
Limitations of the study include short scanning periods without consecutive repeats, potentially preventing full understanding of temperature change over longer durations. We attempted to mitigate this concern by performing additional exploratory extended runs of cine, MB BOLD, and TRUST imaging (selected for signs of heating during early trials). Furthermore, although these trials did not find concerning levels of heating, a notable increase in amplifier temperature was observed for cine that did not appear to reach equilibrium during a 15-min scan period, indicating the potential for further heating during longer scans. However, because most functional studies are approximately 1 h given attention to participant comfort, cine imaging greater than 15 min is uncommon, and some of these effects would likely be reduced by varying sequences or brief scan breaks, shorter timelines are likely not inappropriate as examples for practical functional studies. Secondly, the in vivo cohort was limited to healthy subjects; however, given the small temperature changes observed, increased heating vulnerability in a more varied cohort would not be expected. Thirdly, no direct evaluation was made between older caps with 1.0 µT limits, and newer designs with 1.5 µT limits; furthermore, no evaluation was completed with different cap cable lengths or without the amplifier connected due to manufacturer recommendations, making it challenging to fully assess the form of RF current that may be inducing heating. Fourthly, the reason for the gradual increase in temperature across probes during the in vivo test is not perfectly clear. Future studies might collect additional data on the equilibration effect. Fifth, our study found that sequence order may play a role when combined with the equilibration effect, as sequences placed near the beginning of a scanning run may experience a larger effect; future work could more systematically vary sequence order to further eliminate this as a confound. Sixth, our study was not able to completely assess differences in temperature change experienced by scalp electrodes compared to the amplifier; future studies might more deeply interrogate this. Finally, this study focuses only on careful measurements of temperature changes for common sequences, which we record quantitatively. Our results apply only to the MRI system, hardware configuration, and pulse sequence parameters tested. We make no recommendations with regards to absolute safety, and this determination should be made by each investigator based on information available, participant population, and in the context of oversight from Human Research Protections committees.
Conclusion
We performed ex vivo and in vivo temperature measurements with MRI-compatible EEG equipment across a wide range of popular MRI sequences at the high clinical field strength of 3 Tesla. We did not observe adverse events related to heating, and temperature changes for prescribed B1 + rms≤1.5 μT and SAR≤1.79 W/kg were approximately 0.5°C or less.
Supplemental Material
sj-docx-1-eeg-10.1177_15500594251320294 - Supplemental material for Electroencephalogram Electrode and Amplifier Temperature Changes During Routine Anatomical and Functional Magnetic Resonance Imaging Sequences at 3 Tesla
Supplemental material, sj-docx-1-eeg-10.1177_15500594251320294 for Electroencephalogram Electrode and Amplifier Temperature Changes During Routine Anatomical and Functional Magnetic Resonance Imaging Sequences at 3 Tesla by Adam J Stark, Caleb J Han, Jarrod J Eisma, Alexander K Song, Maria E Garza, Leah G Mann, Daniel O Claassen and Manus J Donahue in Clinical EEG and Neuroscience
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by NIH/NIA grant 5R01AG062574, NIH/NINR grant 5R01NR015079, NIH/NCCIH grant 1R01AT011456, and the Vanderbilt Medical Scholars program, and was approved by the Vanderbilt University Medical Center Institutional Review Board. The authors offer their sincerest thanks to the volunteers who participated in this study. Manus J. Donahue receives research related support from the National Institutes of Health (NINDS, NCI, NIA, NCCIH, NINR, and NHLBI), Philips Healthcare and is a paid consultant for Graphite Bio, Pfizer Inc, Global Blood Therapeutics, and LymphaTouch. He is a paid advisory board member for Novartis and bluebird bio and receives research funding from the National Institutes of Health and Pfizer Inc. Manus J. Donahue is also the CEO of Biosight Inc which operates as a clinical research organization and provides healthcare technology vendor services. Daniel O. Claassen has received research support from the NIH/NINDS/NIA/NICHD/NCCIH, Department of Defense, Griffin Family Foundation, and Huntington Disease Society of America; he has received pharmaceutical grant support from AbbVie, Alterity, Acadia, Biogen, BMS, Cerecour, Eli Lilly, Genentech-Roche, Lundbeck, Jazz Pharmaceuticals, Neurocrine, Teva Neuroscience, Wave Life Sciences, UniQure, and Vaccinex. He has received personal fees for consulting from Acadia, Alterity, Adamas, Anexon, Ceruvel, Lundbeck, Neurocrine, Spark, Uniqure, and Teva Neuroscience.
Prepared for submission as an Original Manuscript in Clinical EEG and Neuroscience
For both phases, participants provided informed, written consent in accordance with the Vanderbilt University Institutional Review Board (IRB#202527) and the Declaration of Helsinki and its amendments. Specific counseling on the purposes and risks of this study was provided, and participants were instructed that study procedures would be immediately stopped if any heating was observed.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.