Abstract
Keywords
Introduction
Pain as a unified percept is proposed to be related to an imbalance in pathways in the central nervous system: the lateral “painfulness” pathway, the medial “suffering” pathway, and the descending pain “inhibitory” pathway.1–5 The lateral pathway has the somatosensory cortex (SSC), the medial pathway has the dorsal anterior cingulate cortex (dACC), and the descending pain inhibitory pathway has the pregenual anterior cingulate cortex (pgACC) as their main cortical hubs.3,4,6 Whereas pain was initially interpreted as activity changes in specific brain areas, 7 it has become clear that pain may be an emergent property of pain networks in the brain, characterized by altered functional connectivity (FC)3,8,9 or a combination of activity and FC changes. The FC permits communication between different brain areas involved in pain-evoking (pain severity and its associated suffering) and pain-suppressing (descending inhibition of nociceptive inputs) areas. Thus, a balance of pain evoking and suppression may determine the pain perceived.3,4,6 Indeed, the pain has been shown to result from an imbalance between the activity in the dACC and SSC (pain evoking) and the pgACC (pain suppression).3,4,6,10,11 Furthermore, the bigger the imbalance between pain input and pain suppression, the more intense the pain is perceived. 6 Thus, modulating this imbalance should result in pain suppression and relief.
Non-invasive neuromodulatory approaches have been proposed as a potential intervention to modulate activity and FC between pain-processing brain regions with the potential to improve clinical pain outcomes.12,13 Electroencephalography (EEG) based brain–computer interface (BCI) training, also known as EEG neurofeedback (EEG-NF), is a form of intervention developed for augmenting or reducing brain activity patterns that are linked to disease states.13–15 Source-localized EEG-NF protocols can be designed to increase or decrease the intracortical neuronal activity at the targeted cortical networks or predefined regions of interest (ROIs) at a source level within the brain rather than only at a sensor level.16,17 A large body of literature is available from various EEG-NF protocols tested for different pain conditions, including both RCTs18–26 and non-RCTs.27–37 Those EEG-NF protocols upregulate or downregulate higher frequency oscillations [alpha (8-12 Hz), beta (13-30 Hz), and gamma (30.5-44 Hz)] at a single or two ROIs. While these studies were able to improve clinical pain outcomes, a recent systematic review and meta-analysis demonstrated inconsistencies across these studies in reporting measures of brain changes. Most studies failed to show frequency-specific EEG changes in the trained or other frequencies within the spectrum. 38
Alterations in the FC between pain-evoking and pain-inhibiting regions have been demonstrated in various chronic pain populations.39–44 It has been proposed that changes in the FC strength could be an objective marker of intervention efficacy alongside the changes reported in the subjective clinical measures. 41 Thus far, only a few studies have investigated FC changes in pain following EEG-NF training and demonstrated FC changes at the sensor level 45 and specific ROIs. 46 However, none of the studies thus far have investigated source-localized region-specific FC changes and their association with pain and physical function measures.
Although the findings from existing EEG-NF indicate positive results with pain modulation, increasing evidence implicates the role of infraslow fluctuations (ISF–frequency < 0.1 Hz) in modulating dynamic brain connections. 47 ISF underlies the excitability of cortical networks and is robustly correlated with the phase of the faster frequency oscillations. 48 Moreover, higher frequency bands are nested (phase-locked) on the ISF bands (0.0-0.1 Hz), synchronizing the higher frequency activity of the brain.49–53 Preclinical and empirical research shows neuropathic pain is associated with altered ISF within the dorsal horn, extending to the SSC.54,55 Additionally, evidence from neuroimaging studies demonstrates increased ISF activity within the pain-evoking brain regions, including dACC, and SSC, and decreased ISF activity within the antinociceptive network, including pgACC.56–66
Preliminary evidence suggests that training the ISF activity in pain processing regions can improve clinical outcomes such as pain and function.65–70 Yet, it remains unclear whether ISF training can alter activity within the ISF and higher EEG bands. Furthermore, it is unknown whether such shifts in EEG activity correlate with changes in clinical outcomes. An exploratory, secondary analysis was undertaken to examine the effects of ISF-NF on EEG measures in people with chronic painful knee osteoarthritis (OA). Therefore, the
Methods
Design
This study was designed as a two-arm, parallel-group, double-blinded (participants and outcome assessor) randomized sham-controlled feasibility clinical trial. 70 Methodological descriptions of this study followed the Consolidated Standards of Reporting Trials (CONSORT) extension for randomized pilot and feasibility trials. 71 The description of the study intervention was structured following the Template for Intervention Description and Replication (TIDieR) guide. 72 This study was approved by the Health & Disability Ethics Committee (HDEC), New Zealand (19CEN182), and the trial was registered in the Australian New Zealand Clinical Trials Registry (ACTRN12620000273987). The research was conducted in accordance with the Declaration of Helsinki. A detailed version of the study protocol and the feasibility results were published elsewhere.71,73
Participant Characteristics
Adults aged 44 to 75 years with a clinical diagnosis of knee OA; with pain (at least ≥ 4 on an 11-point numerical rating scale) for a minimum duration of 3 months were eligible to participate in the study. Participants were excluded if they had one of the following situations or conditions: (1) underwent surgery or other invasive procedures in the last six months and any surgical procedures scheduled within eight weeks after screening; (2) undertaken any steroid injections to the knee joint in the past three months or on oral steroids in the previous month; (3) current intake of centrally acting medications (eg, antidepressants, anticonvulsants, and neuropathic pain drugs) or intention of taking new medications in the next eight weeks; (4) neurological conditions or diseases (brain, spinal cord, or peripheral nerve injuries, radiculopathy, and neuropathies); (5) soft tissue injuries of the knee (eg, meniscus, muscle, tendon, or ligament injury) in the last three months; (6) cognitive impairments (dementia, posttraumatic stress disorders, and Alzheimer's disease); (7) difficulty or inability to read or understand English, or provide informed consent; and (8) pregnancy or six months postpartum.
Randomization and Allocation Concealment
Participants were randomized into either ISF-NF (active group) or sham NF (sham group) group using an open-access block randomization program by the department research administrator not involved in the assessments, allocation, or interventions. The allocation was concealed until after the initial assessment was completed. Participants and the outcome assessor were blinded to the group allocation. The flow of participants through the study phases is illustrated in Figure 1.70,73
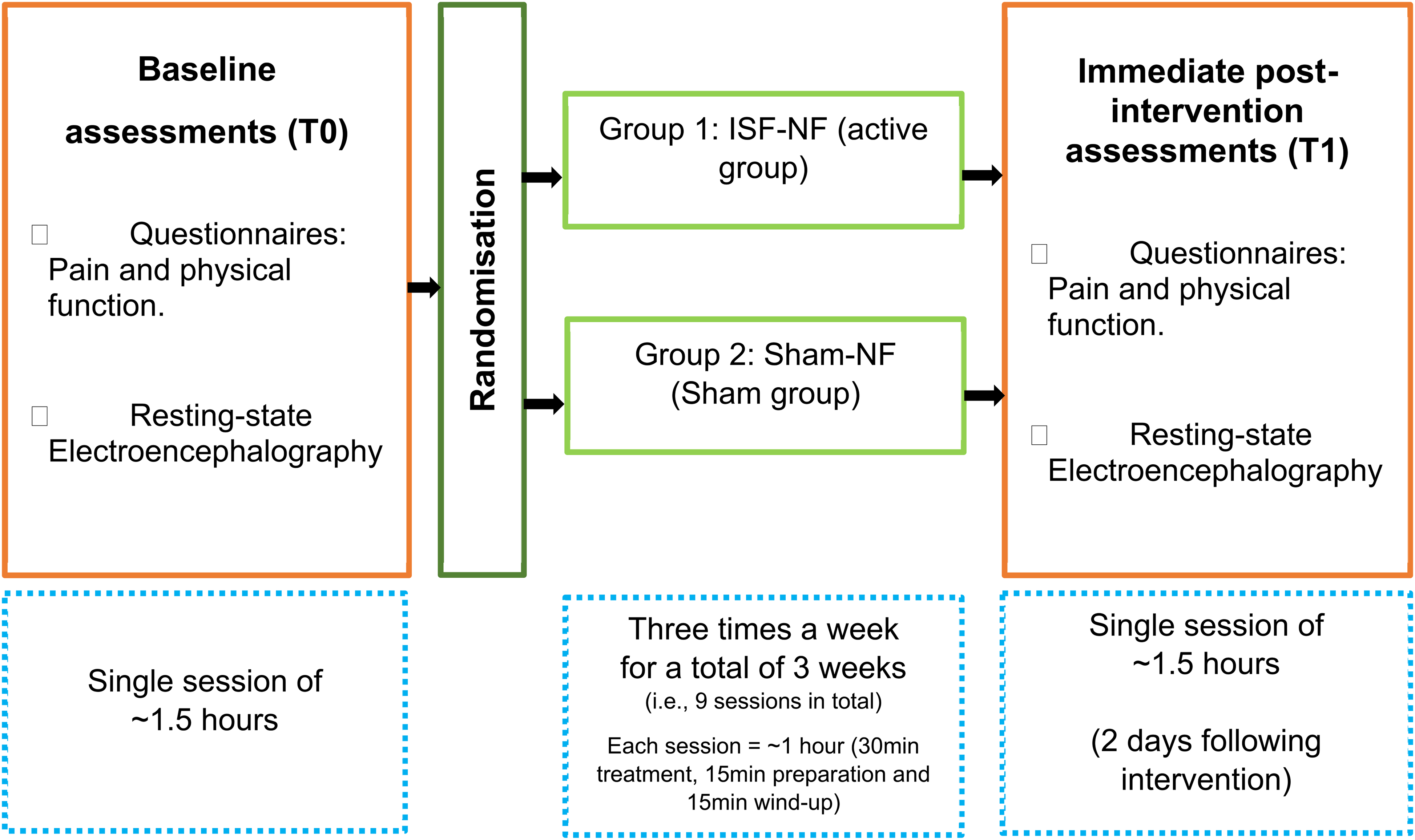
The flow of participants through the study phases.
Assessments and Outcome Measures
Summary of the Clinical Outcome Measures.

EEG-Neurofeedback Protocol
Eligible participants were required to attend nine NF training sessions (30 min; 3 sessions/week) and two 90-min assessment sessions at T0 and T1. The experimental methodology and reporting of NF followed the consensus on the reporting and experimental design of clinical and cognitive-behavioral neurofeedback studies checklist (CRED-nf checklist-mandatory items). 88
During each session, participants were asked to sit on a chair with their backs supported and stay relaxed for 10 min, allowing the trainer to prepare them for the NF training. Both ISF-NF and sham-NF were administered using a 19-channel (ie, Fp1, Fp2, F3, F4, C3, C4, P3, P4, O1, O2, F7, F8, T3, T4, T5, T6, Fz, Cz, and Pz) DC-coupled amplifier produced by BrainMaster Technologies, Inc.69,70,73,89–91 The impedance of the active electrodes was monitored and kept below 5 kΩ. 67 Participants were instructed to minimize eye movements, movements of the head and neck, swallowing, and teeth clenching to reduce motion artifacts in the EEG recordings. Additionally, they were informed that the sound they heard indicated successful performance.92–97 Details of the intervention methodology and procedures can be found in previous publications.70,73
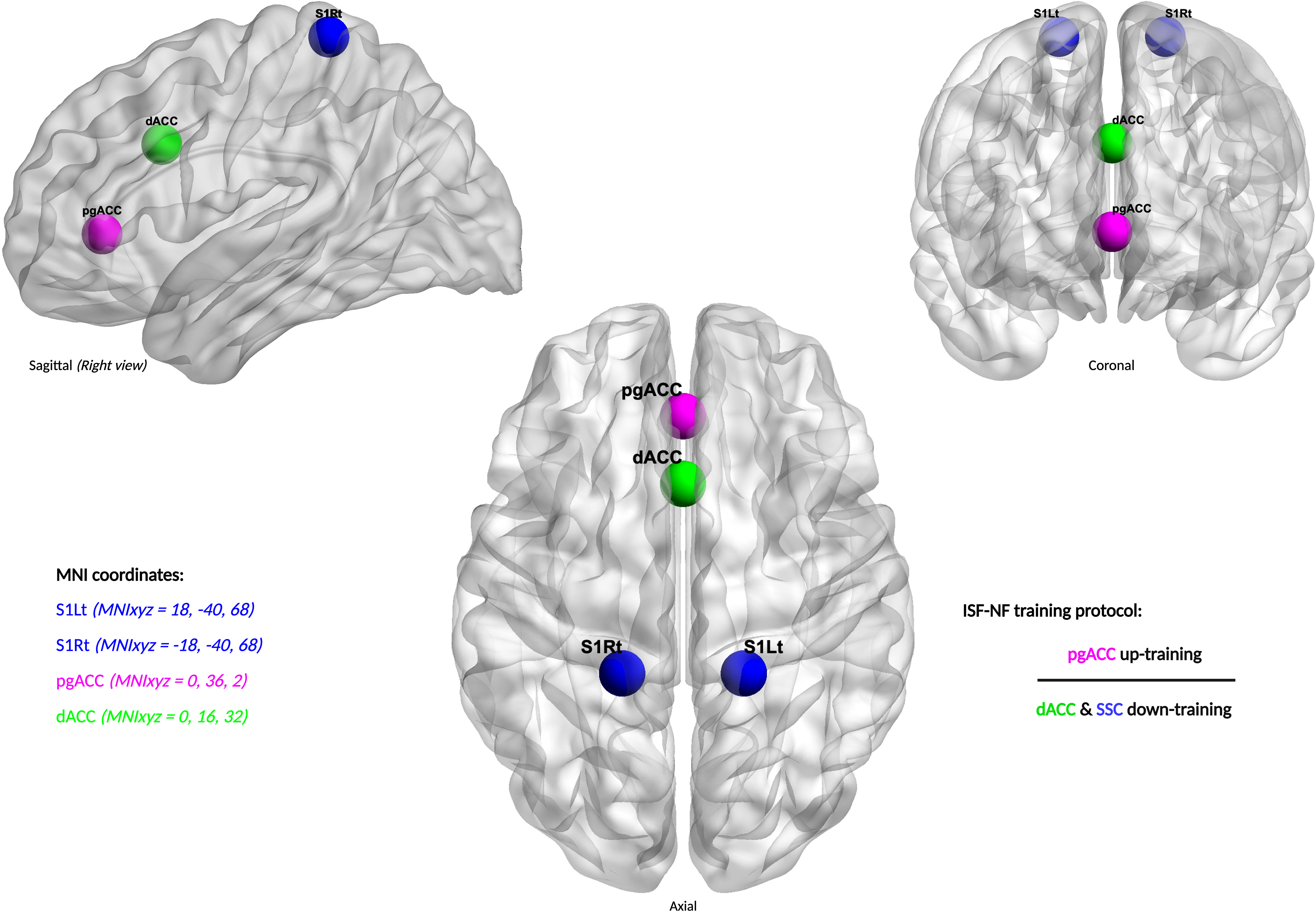
Regions of interest and ISF-NF training directions.
eLORETA source localization permits the selection of any region of the brain for feedback of the current density using voxels as ROI, which were selected based on the Montreal Neurological Institute (MNI) coordinate database,114,115 previous literature3,4,6,69,70,116,117 and based on a Neurosynth meta-analysis (https://neurosynth.org/) of pain. A complete list of targeted voxels used for defining each ROI is available from Supplemental Materials 1 to 3. Figure 2 provides the center voxel coordinate for each ROI and the details of the ISF-NF training program.
The BrainMaster Technologies Software
118
delivered sound feedback (reward) when the participant's brain activity met the desired ISF (0.0-0.1 Hz) threshold at the targeted ROIs via the laptop speakers. We utilized manual thresholding to adjust individual participant brain activity to meet the predetermined threshold if needed. This approach was selected based on insights from our prior study and the clinical experience of the authors.
67
Moreover, previous studies have reported better EEG learning with manual thresholding than with automated thresholding.93,95,119,120 The software calculated the ratio in real-time in the ISF band, and the feedback was given when the ratio was ≥ 1 based on satisfying the following equation: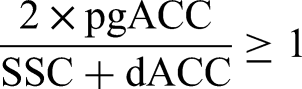
Data Analysis
The EEG data were preprocessed using MATLAB (R2020a) with the EEGLAB toolbox124,125 before the data were analyzed by eLORETA software. Each file was resampled to 128 Hz, bandpass filtered from 0.01 to 44 Hz, and EEG artifacts (eye blinks, muscle artifacts, perspiration, and body movements) were removed using IcoN Software Version 3.6,67,69,126
The EEG data were analyzed using the open eLORETA method using LORETA-Key software to estimate the current density of intracerebral electrical sources that generate scalp-recorded electrical activity in each of the following seven frequency bands: ISF (0.01-0.1 Hz), slow (0.2-1.5 Hz), delta (2-3.5 Hz), theta (4-7.5 Hz), alpha (8-12 Hz), beta (13-30 Hz), and gamma (30.5-44 Hz).127–129 We employed an average reference montage for localization. 130 eLORETA is one of the most robust methods for EEG source localization and can estimate current density over a grid of plausible cortical locations based on an inverse problem calculation.4,127,131–133
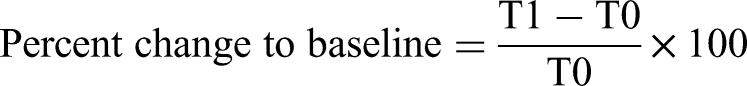
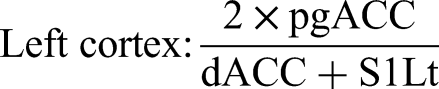
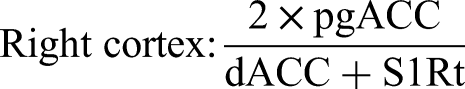
The final derived change scores were then exported to GraphPad Prism for statistical analysis. After conducting normality testing with the Shapiro–Wilk test and
Statistical Approach
The data processing and pooling for this study were done using either MATLAB 2020a124,125 and/or Microsoft Excel (2020).
139
All the graphs and statistical analysis were derived using GraphPad Prism software version 9.1.0 for Windows (GraphPad Software, San Diego, CA, USA).
140
The normality of the data distribution for all the variables was confirmed using the Shapiro–Wilk test and
Due to multiple ROIs and frequency bands in the analysis, a “Volcano plot” was created demonstrating the statistical summary for both ROI and FC analysis with Mann-Whitney mean rank difference on the
Results
Demographics
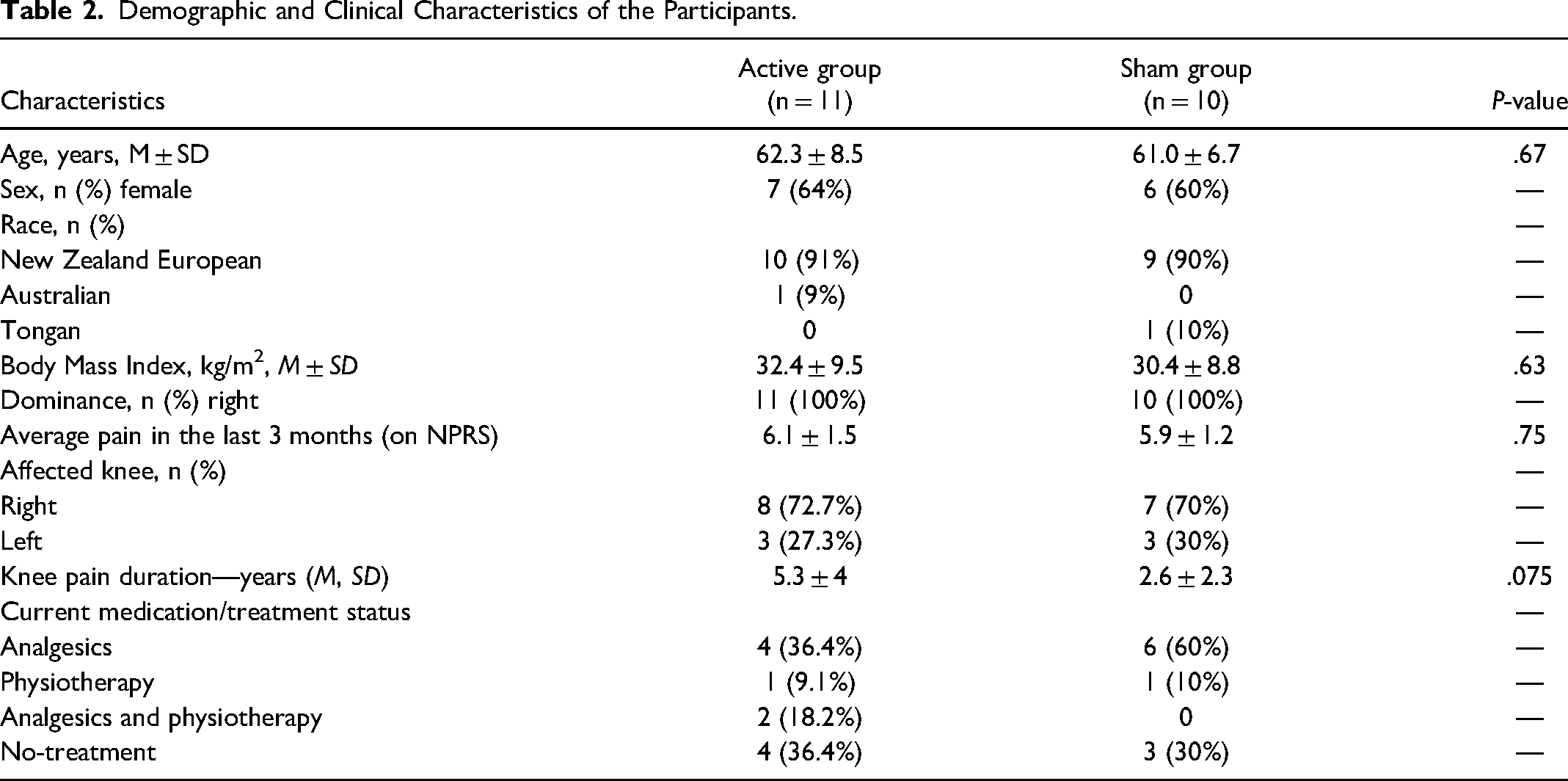
Twenty-one participants with knee OA underwent baseline assessment and were randomly assigned to receive either ISF-NF (n = 11) or sham-NF (n = 10) group. Participants were middle-aged (61.7 ± 7.6 years), New Zealand European (90.5%), and females (62%) with an average knee pain duration of 4.0 ± 3.4 years. The socio-demographic and clinical characteristics of the participants in both groups and the results of tests comparing averages and proportions for the two groups at baseline are summarized in Table 2. Between-group comparisons revealed that both groups were comparable in age and sex, and no significant differences were found between groups.
Demographic and Clinical Characteristics of the Participants.
Electroencephalography Measures
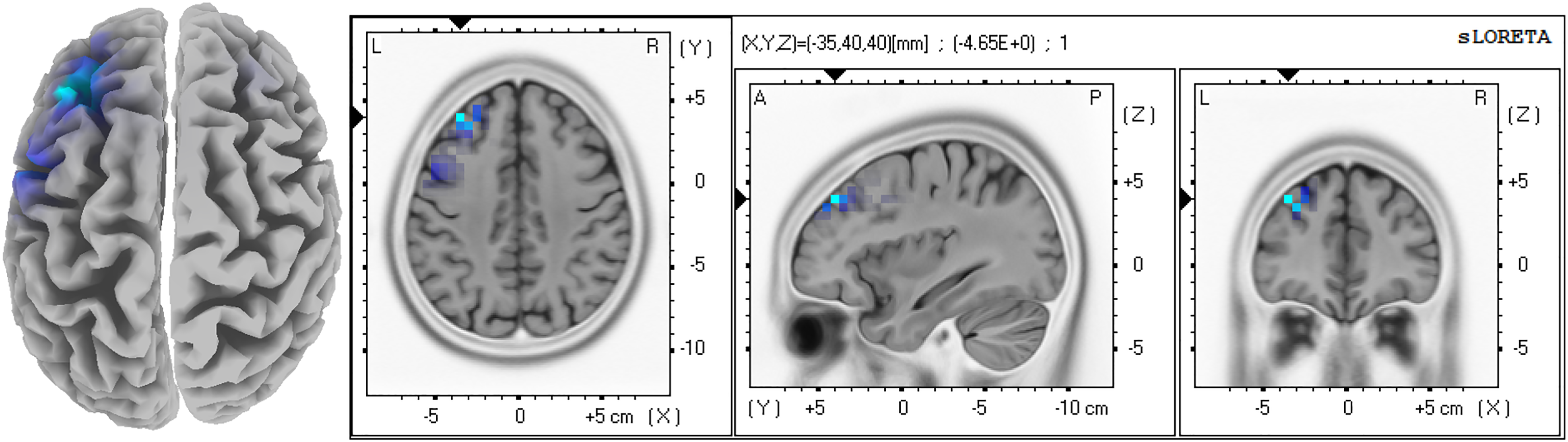
eLORETA statistical contrast maps: this figure is generated by the eLORETA after performing statistical non-parametric mapping on brain sources. The analysis demonstrates decreased activity in the ISF band in the LdlPFC (
We have included LdlPC as an ROI along with the “a priori” regions for further analyses (ROI and FC). The “a priori” ROIs in the present study are SSC left (S1Lt) (
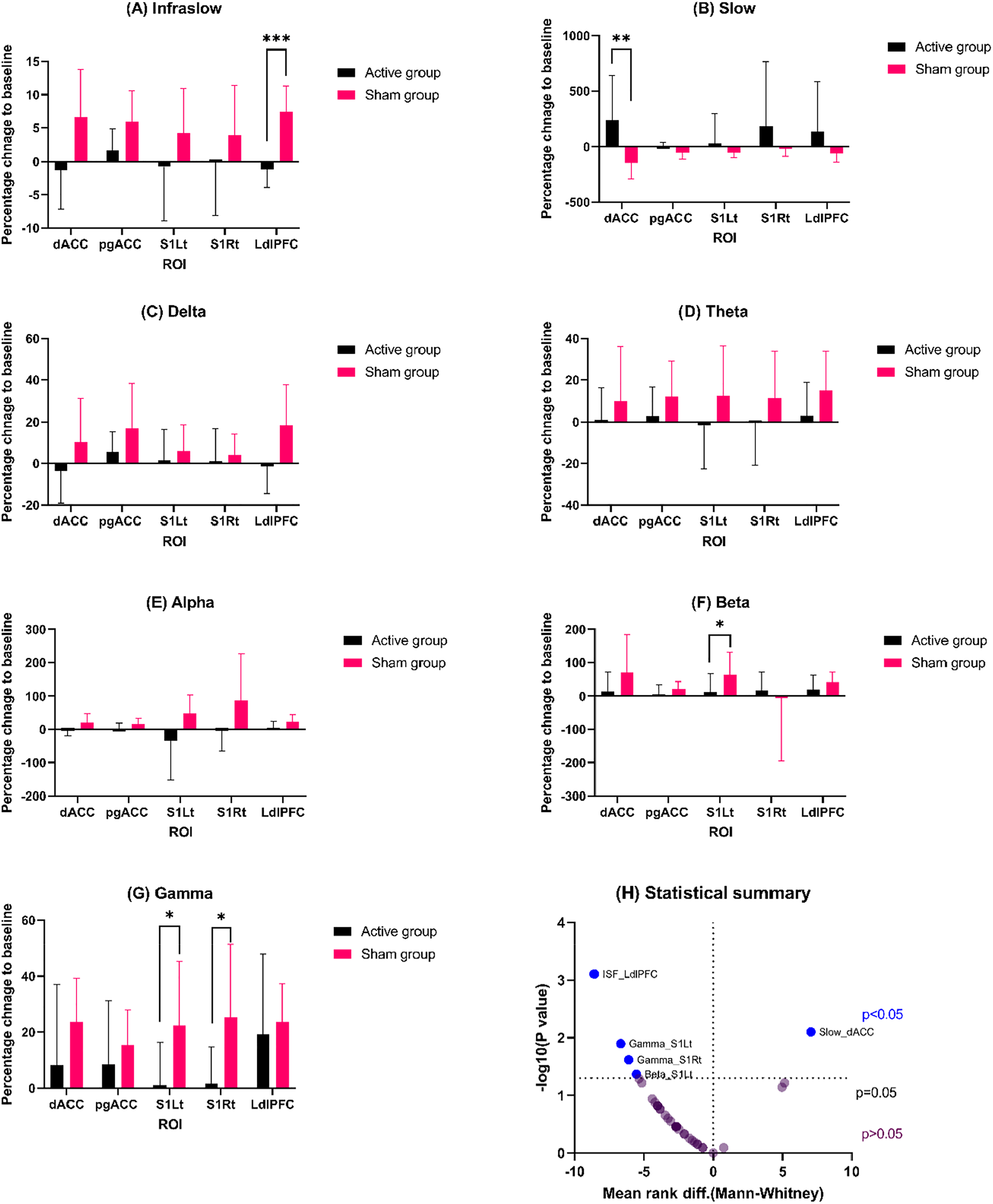
Displays the outcomes of the region of interest (ROI) analysis conducted with the Mann-Whitney U test: (A-G) Demonstrates the average current density of the percentage change to baseline (means with 95% CI) for each ROI and frequency band for both groups. (H) Provides a statistical summary of the ROI analysis based on the mean rank difference and
The study found no significant change in the ISF ratio for both the right and left cortical regions over the NF-targeted regions (Figure 5).
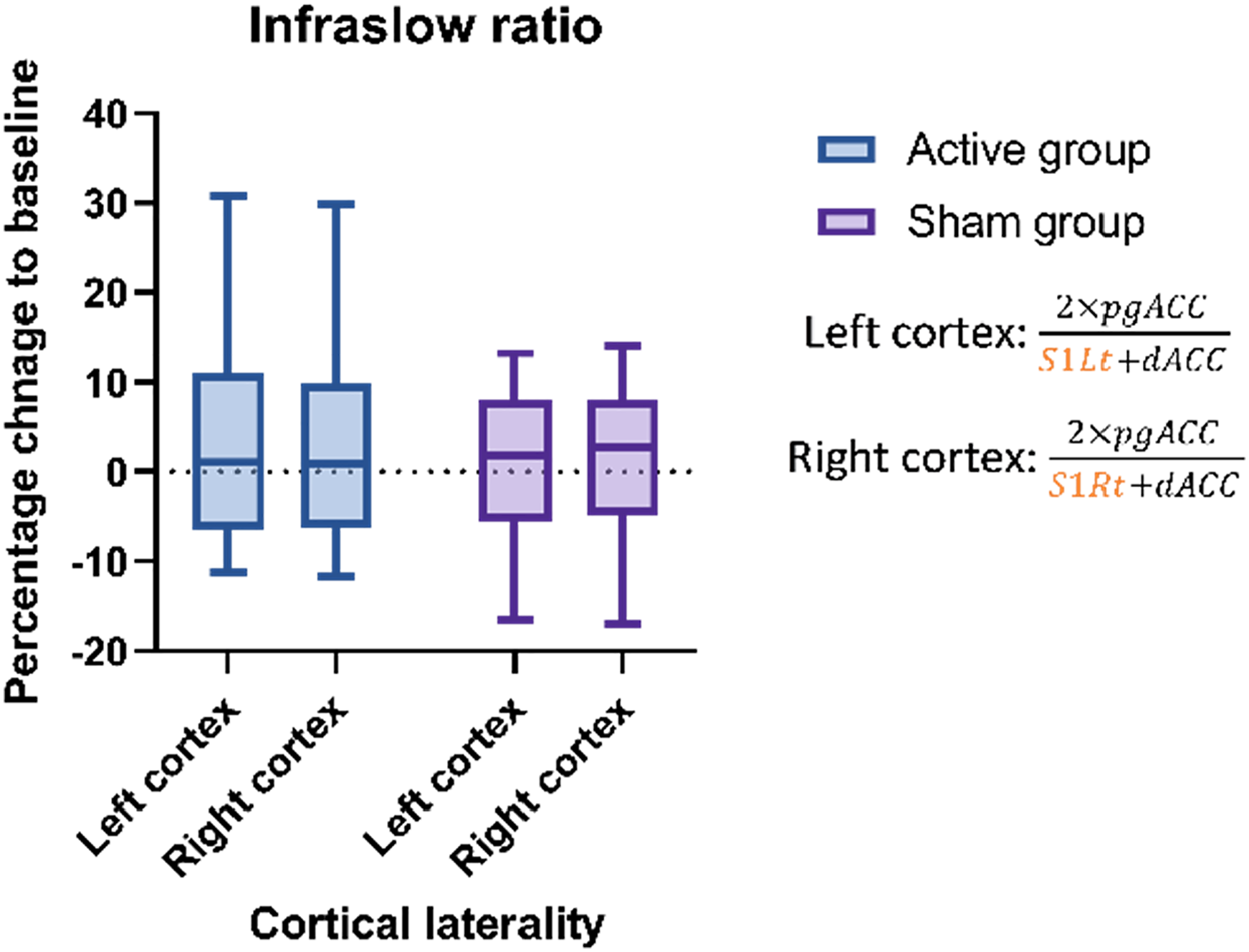
ISF ratio analysis: the box and whiskers plot demonstrates (means with 95% CI) that the ISF ratio changes for both groups.
Functional Connectivity (Lagged Phase Synchronization)
The lagged linear connectivity analysis between groups revealed significant functional connectivity changes (Figure 6) in the slow band (
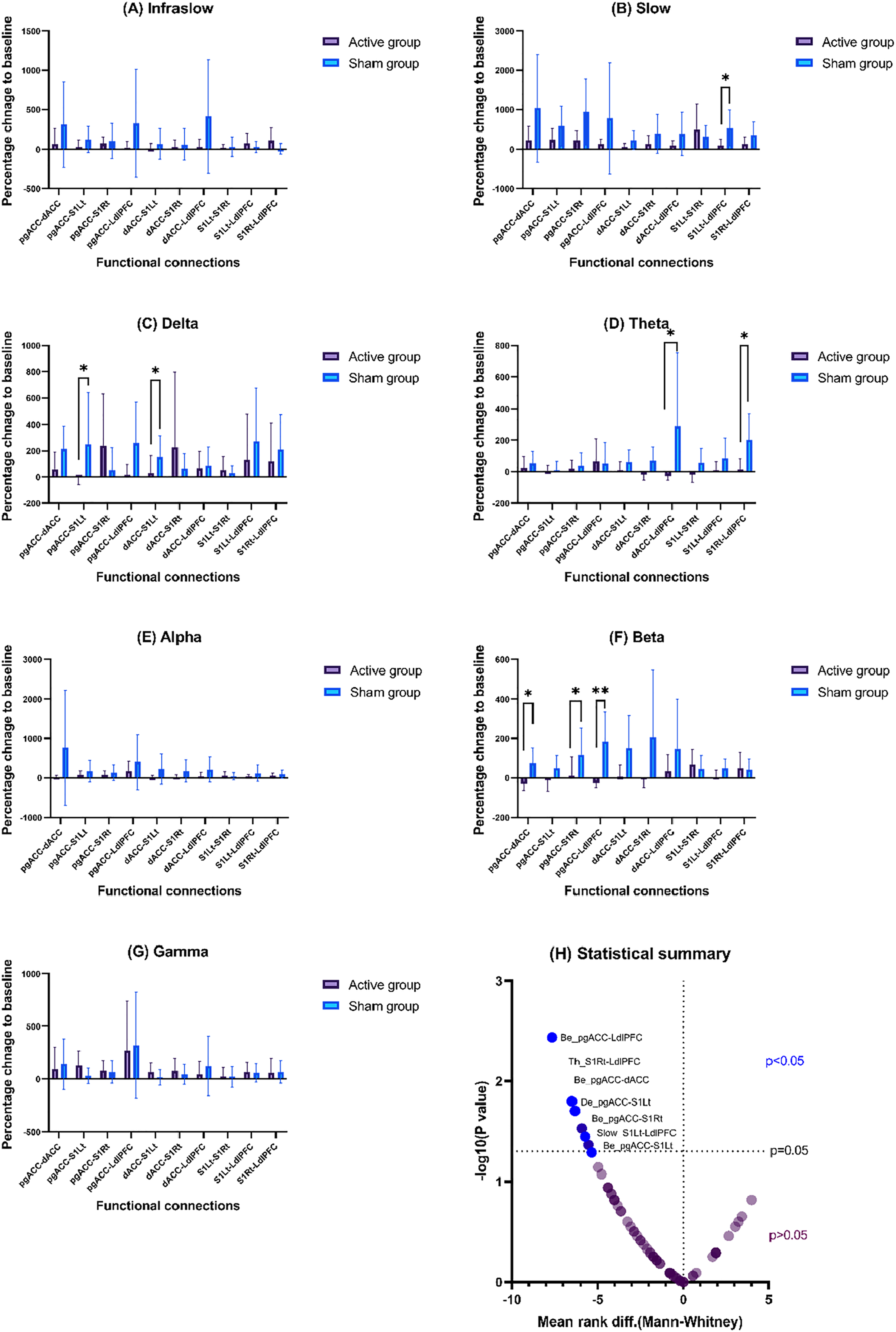
The results of functional connectivity analysis using the Mann-Whitney
Correlation Between Pain, Physical Function, and EEG Measures
The Spearman correlation yielded a moderate positive correlation between ISF band activity in the LdlPFC and pain unpleasantness (ρ = 0.6698,
No other significant correlations were found for other measures in both groups.
Discussion
Principal Findings
This study sought to determine whether the ISF-NF ratio training can result in measurable brain activity and FC changes in individuals suffering from chronic knee OA pain. The between-group source localization mapping demonstrated that the ISF-NF training significantly decreased activity at the left dlPFC compared to the sham group. ROI analysis also revealed a significant reduction of the ISF current density in the LdlPFC in the active group compared to the control group. Moreover, the decreased ISF power in the LdlPFC in the active group was positively correlated with the pain unpleasantness measure. FC changes were observed in the slow, delta, theta, and beta bands between various ROIs in both groups. Notably, the FC between pgACC and S1Lt in the delta band was negatively correlated with the pain bothersomeness change score in the ISF-NF group.
eLORETA Statistical Contrast Maps and Region of Interest Analysis
The eLORETA statistical contrast mapping method makes no “a priori” assumption about the brain regions. Instead, it performs a source-localized statistical parametric mapping to identify brain regions with significant changes compared to a control group.145,146 In our study, LdlPFC is the only region that demonstrated a significant change in the mapping with decreased ISF activity of the region in the active group. Additionally, the ROI analysis also showed a significant reduction of the ISF activity in the LdlPFC in the active group. The dlPFC is an important heterogeneous cortical region that mediates the antinociceptive, cognitive, and affective processing of pain.147–149 Importantly, dlPFC is a key node of several brain networks primarily implicated in the cognitive-affective and sensory processing of pain experience.147,148,150,151 Moreover, the dlPFC is functionally connected and positively correlated with the activity of the dACC, another important cortical hub modulating the affective dimensions of the pain experience.3,116,152,153 Previous studies have reported reduced functional and structural activity of the PFC in various chronic pain populations154–158 and have reported successful noninvasive stimulation of the PFC for treating chronic pain.45,159,160 In this study, we found no significant change in ISF neural activity over the targeted regions in both eLORETA brain mapping and ROI analysis. However, the training impacted the regions that are functionally connected with the targeted regions. The ratio ISF training down-regulated dACC may have resulted in the reduced ISF activity of the dlPFC in the active group. This evidence suggests an indirect effect of ISF-NF on other regions involved in the same functional network. This is of no surprise considering the profound role of ISF in modulating and synchronizing various cortical networks of the brain.50–53 In addition, the ISF activity of the LdlPFC was positively correlated with the unpleasantness of pain in the active group. Previous studies have described dACC and dlPFC as a region mediating unpleasant and affective aspects of pain.1,5,103,153,161,162 This is an important finding and supports the claim that the displayed ISF effects in the dlPFC are likely to be modulated by the downregulation of the dACC, thus improving clinical symptomatology linked to the regions.
However, it is worth mentioning that the training protocol was designed to increase the ISF activity of pgACC and simultaneously downregulate dACC and SSC (S1Lt and S1Rt). An unexpected but important finding was that we did not observe any activity changes in the targeted regions in the direction of training for the ISF bands. Previous studies in EEG-NF have also reported similar observations in tinnitus-related distress and cognitive NF training.76,163 The authors speculate that this could be due to the training of a relatively smaller cortical region to influence pain-related networks, where several cortical and subcortical circuits operate to modulate pain experience. 62 This could also explain why we could not find any significant changes in the ISF ratio. Additionally, the spatial specificity in localizing the targeted region using a 19-electrode head cap can be influenced by the activity of the nearby regions close to the targeted region, which could also have influenced the results.163,164 However, this needs to be investigated with an extended protocol influencing multiple brain regions using high-resolution EEG with more number of electrodes. 165 In summary, even though a ratio of activity (current density) was trained, only changes in FC are found and not limited to the trained frequency band.
Functional Connectivity Changes and Potential Clinical Benefits of Ratio ISF-NF Training
In chronic pain, decreased communication (ie, FC) has been demonstrated between pgACC and SSC. 6 Moreover, FC alterations are observed in chronic pain in the regions or associated networks tested in this investigation.166–170 Supporting the literature, the study demonstrated FC changes between ROIs in the slow band (S1Lt-LdlPFC), delta (pgACC-S1Lt; dACC-S1Lt), theta (S1Rt-LdlPFC; dACC-LdlPFC), and beta (pgACC-LdlPFC; pgACC-dACC). Most of these changes were also observed in the sham group. However, the FC changes exhibited a negative correlation between the FC of pgACC and S1Lt in the delta band, with pain bothersomeness only in the active group. One of the ISF-NF ratio components was to uptrain the ISF activity of the pgACC. We believe the training must have influenced the functional communication from pgACC to SSC, thus influencing the clinical pain outcomes.
Limitations and Future Research Recommendations
Given that this is the first ISF-NF ratio study, all the analyses performed were explorative, informing the importance of a full clinical trial. We recruited a small sample of individuals with knee OA with a homogeneous sex and race distribution, which restricts the generalizability of the study findings. Furthermore, all correlation analyses conducted in the study were exploratory. Consequently, the study refrained from applying corrections to the p values for multiple bivariate correlations. However, it is important to acknowledge that employing the Bonferroni correction method might enhance the risk of type II errors, potentially overlooking real relationships. 171 Despite the limitations, this study provides an essential foundation for designing and implementing future clinical trials investigating ISF-NF protocols.
Ratio training protocols are more functional and reduce the hassle of separately training individual ROIs. However, further investigations are required to determine the efficiency of the ratio training protocol against the training of cortical areas separately on EEG measures at prespecified ROI and network-level changes. Similarly, it is also worth investigating whether NF training of higher EEG bands can influence the ISF band and comparing the effectiveness of ISF-NF against higher band NF training protocols on clinical outcomes in people with chronic pain.
Conclusions
The ratio ISF-NF training can result in measurable changes in brain activity and functional connectivity in individuals suffering from chronic knee OA pain. However, ISF-NF training did not change the EEG activity of the regions exposed to training; instead, it exerted its effect on a functional network level. Caution is warranted in interpreting these findings since it is explorative analyses on an unpowered sample, and the observations could be due to chance.
Supplemental Material
sj-csv-1-eeg-10.1177_15500594241264892 - Supplemental material for Closed-Loop Infraslow Brain–Computer Interface can Modulate Cortical Activity and Connectivity in Individuals With Chronic Painful Knee Osteoarthritis: A Secondary Analysis of a Randomized Placebo-Controlled Clinical Trial
Supplemental material, sj-csv-1-eeg-10.1177_15500594241264892 for Closed-Loop Infraslow Brain–Computer Interface can Modulate Cortical Activity and Connectivity in Individuals With Chronic Painful Knee Osteoarthritis: A Secondary Analysis of a Randomized Placebo-Controlled Clinical Trial by Jerin Mathew, Divya Bharatkumar Adhia, Mark Llewellyn Smith, Dirk De Ridder and Ramakrishnan Mani in Clinical EEG and Neuroscience
Supplemental Material
sj-csv-2-eeg-10.1177_15500594241264892 - Supplemental material for Closed-Loop Infraslow Brain–Computer Interface can Modulate Cortical Activity and Connectivity in Individuals With Chronic Painful Knee Osteoarthritis: A Secondary Analysis of a Randomized Placebo-Controlled Clinical Trial
Supplemental material, sj-csv-2-eeg-10.1177_15500594241264892 for Closed-Loop Infraslow Brain–Computer Interface can Modulate Cortical Activity and Connectivity in Individuals With Chronic Painful Knee Osteoarthritis: A Secondary Analysis of a Randomized Placebo-Controlled Clinical Trial by Jerin Mathew, Divya Bharatkumar Adhia, Mark Llewellyn Smith, Dirk De Ridder and Ramakrishnan Mani in Clinical EEG and Neuroscience
Supplemental Material
sj-csv-3-eeg-10.1177_15500594241264892 - Supplemental material for Closed-Loop Infraslow Brain–Computer Interface can Modulate Cortical Activity and Connectivity in Individuals With Chronic Painful Knee Osteoarthritis: A Secondary Analysis of a Randomized Placebo-Controlled Clinical Trial
Supplemental material, sj-csv-3-eeg-10.1177_15500594241264892 for Closed-Loop Infraslow Brain–Computer Interface can Modulate Cortical Activity and Connectivity in Individuals With Chronic Painful Knee Osteoarthritis: A Secondary Analysis of a Randomized Placebo-Controlled Clinical Trial by Jerin Mathew, Divya Bharatkumar Adhia, Mark Llewellyn Smith, Dirk De Ridder and Ramakrishnan Mani in Clinical EEG and Neuroscience
Footnotes
Acknowledgments
We acknowledge the International Society for Neuroregulation and Research (ISNR) and the International QEEG Certification Board for supporting the first author through monetary awards and scholarships in support of this study. The authors would like to thank Hazel Potterton, Tyson Michael Perez, and Matt Hall for their active support and help throughout this study. Also, the authors thank all the study participants for their time and support in completing this study during the COVID-19 pandemic in 2020.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge the International Society for Neuroregulation and Research (ISNR) and the International QEEG Certification Board for supporting the first author through monetary awards and scholarships in support of this study. This study was also supported by the Centre for Health, Activity, and Rehabilitation Research (CHARR) yearly graduate research fund.
Ethical Statement
This study was approved by the Health & Disability Ethics Committee (HDEC), New Zealand (19CEN182), and the trial was registered in the Australian New Zealand Clinical Trials Registry (ACTRN12620000273987).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
