Abstract
Introduction: Attention deficit and hyperactivity disorder (ADHD) is one of the most common developmental disorders in childhood which lasts lifelong. Sleep structure and sleep spindle features are disorganized in ADHD. In this study, we aimed to look for a new, simple, inexpensive, and an easily detectable electrographic marker in the diagnosis of ADHD by using electroencephalography (EEG). Method: We included treatment free 35 patients with ADHD and 32 healthy children (HC) who were examined by polysomnography (PSG) and EEG for sleep disorders. The ADHD group were separated into three groups according to predominant presentations of ADHD. We determined the sleep staging and slow and fast sleep spindles, calculated each spindle's amplitude, frequency, activity, duration and density at non rapid eye movement (REM) sleep stage 2. Results: Slow sleep spindle's amplitude, duration, density and activity are significantly higher in ADHD group (most significant in ADHD-I) than the HC group (p < 0,05). Sleep spindle's features are not statistically significant between in ADHD subgroups. Conclusions: In children with ADHD, slow sleep spindles showed higher amplitude, activity, density and duration in the frontal regions. These results indicate that slow sleep spindles in children with ADHD may reflect executive dysfunction and slow frontal spindles may be useful as a new electrographic marker in children with ADHD. This is the first study of its kind evaluating all aspects of sleep spindles in ADHD patients.
Introduction
Attention deficit hyperactivity disorder (ADHD) is one of the most common childhood onset neuropsychiatric disorder, consisting of three subtypes: predominantly inattentive type (ADHD–I), predominantly hyperactive–impulsive type (ADHD–H), and combined type (ADHD–C). 1 Symptoms begin at preschool ages and continue lifelong and negatively interfere with executive functioning, development, memory and learning.2–5 The neurobiological basis for the profound deficits in ADHD is not fully understood, but deterioration of prefrontal cortex circuits is thought to be involved. 4 Both sleep problems and ADHD are the major concerns in clinical practice, and in parental care.2,6
Sleep provides many functions within the spectrum of tissue restoration, synaptic plasticity, brain metabolite clearance and executive functions, memory consolidation, homeostasis which have been shown to be disrupted in many neuropsychiatric disorders including ADHD. 7 ADHD is commonly associated with disordered or disturbed sleep with a prevalence between 22-55% in various publications.4,8 However, till now there are no comprehensive electroclinical model explaining the genesis of sleep in ADHD. The absence of such research has hindered the detection of a reliable and clinically beneficial marker that could aid in early diagnosis in ADHD throughout the years.
Despite this well documented association, no clearly defined objective markers using polysomnography exist. Numerous previous polysomnographic studies showed inconsistent findings of increased nocturnal movements, reduced sleep efficiency, and decreased percentage of rapid eye movement (REM) sleep. Based on our current knowledge, sleep spindles have gained increasing interest in children with ADHD. Previous studies have shown that sleep spindle development is considered as an indicator of brain maturation due to its parallel progression with thalamocortical development and maturation. 9
Sleep spindles are one of the neural oscillations which have been identified as being an electroencephalographic fingerprint of human non-REM Stage 2 (NREM 2) sleep, occurring in short bursts of about 0.5–2 s and frequency range of 11 −16 Hz at the fronto-centro-parietal region, generated by the thalamic reticular nucleus, and relayed to the cortex by thalamo–thalamic and thalamo–cortical loops. 10 Sleep spindles appear in two types of electroencephalography (EEG) frequency, ie, the frontal-dominant 12-Hz slow spindles and the parietal-dominant 14-Hz fast spindles. 11 These two types showed distinctive courses of maturation throughout ages. Parietal dominant fast spindles changed little from 4 to 24 years of age, whereas the frontal slow spindles decreased remarkably in power and became stable at about 13 years of age, pointing out that the frontal spindle reflects biological maturation. The maturation period of frontal slow spindles is similar to that of frontal structural and functional development. Both types of spindle show age-related changes on the basis of density, duration, amplitude and frequency, these changes point to different developmental characteristics and they are supposed to be indicative of the maturation of thalamocortical structures and of the physiological system that generates spindles.
Sleep spindles have major roles in maintenance of sleep and regulate learning, memory, motor skills, and general intellectual performance by providing plasticity, and these cognitive functions are also the primary domain of dysfunction in children with ADHD.12–14
Recently, Hoedlmoser reported that sleep spindles may be an important neurobiological marker in neural maturation and plasticity, namely cognitive development and spindle activity may reflect an abnormal neurodevelopmental trajectory in ADHD. 15
Considering the recent data, we hypothesized that frontal slow spindle activity may therefore be uniquely suited to shed light on the nosological position of ADHD and be used as a beneficial marker in the early diagnosis of ADHD. As such, investigating frontal slow spindles activity within a control group represents an important addition to the literature. Besides there is a lack of information about the comparisons of sleep spindle characteristics among the three ADHD subtypes.
The aim of this study to determine the density, frequency, duration, amplitude of slow sleep spindles in NREM stage 2 sleep in patients with ADHD in comparison with healthy control subjects free of any mental disorder. These questions are explored in a cohort of 35 children with ADHD and 32 healthy children who underwent overnight polysomnographic (PSG) recordings. First, we examined whether dimensional ADHD symptoms were associated with sleep spindle morphology, and whether frontal slow spindles may constitute an early diagnostic marker for children with ADHD. Furthermore, no detailed correlation of sleep spindles with clinical subtypes in specific clusters has thus far been consistently addressed. Therefore, we evaluated the additive contributive role of slow sleep spindle characteristics to characterize different ADHD phenotypical expressions to better achieve treatment and follow-up.
Material & Methods
Participants
Thirty-five children with ADHD were matched with thirty-two age and sex matched healthy controls (HC). All children were between the ages of 7–17 years and recruited from the Departments of Child and Adolescent Psychiatry, between September 2016 and April 2017. Children in the ADHD group were all medication naive and had been diagnosed according to the DSM-5 criteria by psychologists specializing in ADHD. Children with a total score less than 70 in the Wechsler Intelligence Scale for Children-fourth Edition (WISC-IV) or diagnosed with any severe neurologic disease or with sleep disorders were not included. None of the children were currently taking any medication (including psychostimulants) that may have interfere with their sleep. Figure 1 summarizes the study flow.
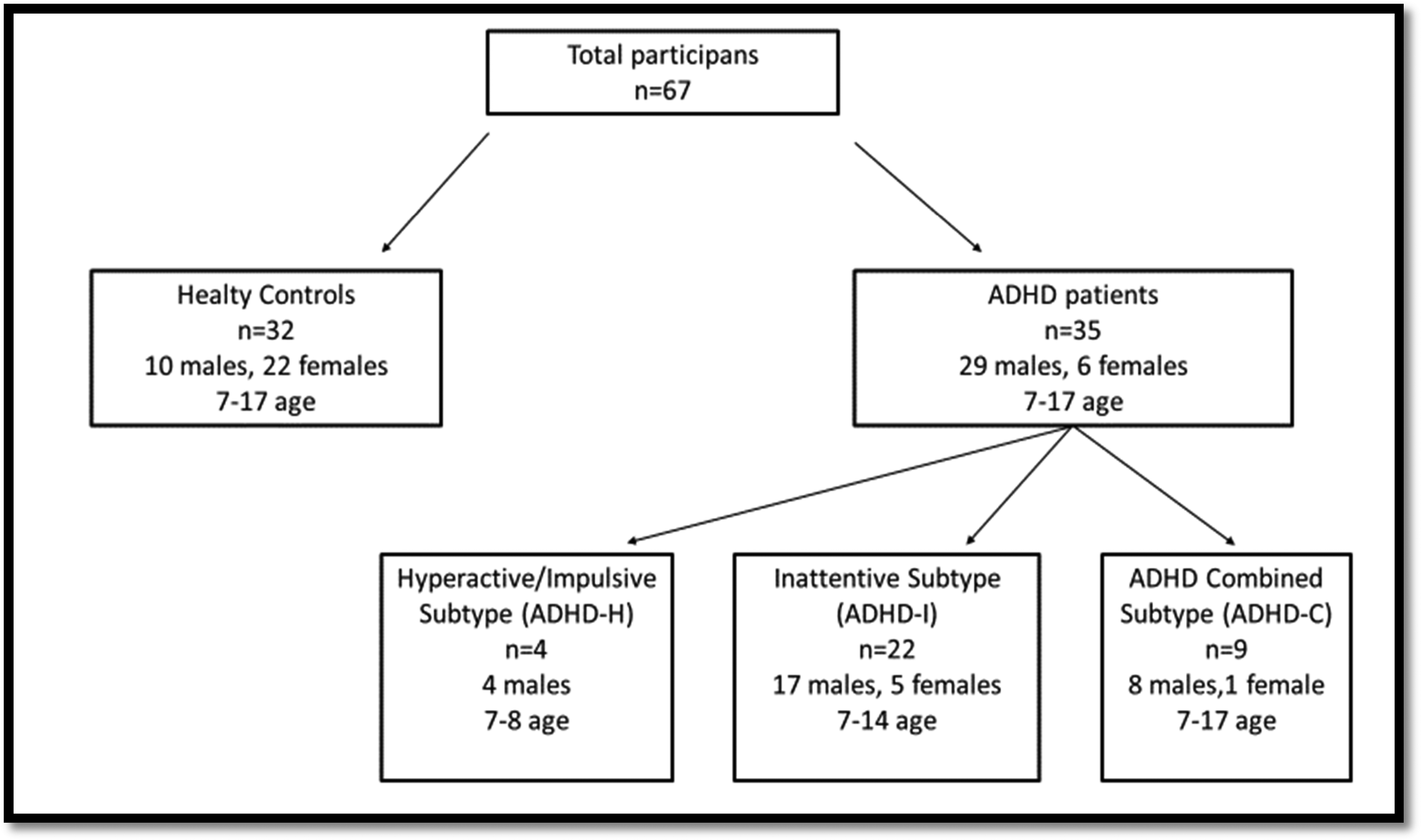
Summarizes tyhe study flow.
The local ethical review board declared no relevant ethical conflict for this anonymized retrospective analysis and informed assent and consent were obtained from each participant and from the children's parents after full explanation of the procedures involved (07/09/2020–547).
Children with ADHD were separated into three groups according to the predominant presentations of ADHD: ADHD predominantly inattentive type (ADHD–I), predominantly hyperactive–impulsive type (ADHD–H), and combined type (ADHD–C).
Exclusion criteria for both ADHD and control group were diagnosed or probable sleeping disorder (including sleep apnea), other disorders associated with impaired sleep (restless legs disorder, periodic limb movement disorder), substance use, chronic inflammatory conditions, neurological disorder, intellectual disability, using of any drug that could interfere with sleep.
Procedure and Overnight Sleep Assessment
Parental consent and child assent were collected for all children. Overnight PSG recordings were performed using Neurofax (Nihon Kohden Co, Tokyo, Japan), and data were recorded at 256 Hz. All participants were asked adhere to a regular sleep–wake schedule of rising before 8.00 am and going to bed before midnight on three days preceding PSG.
On the day of PSG, all children were required not to consume any caffeinated drinks or participate in any form of exercise in the afternoon. Each child underwent an overnight video -PSG (V-PSG) recording after one adaptation night in order to avoid the first-night effect. The V-PSG was performed in a dedicated sleep lab with a maintained comfortable temperature, and hypnogram for each case was obtained. PSG included electroencephalographic electrodes (EEG; Fp1, Fp2, F3, F4, C3, C4, P3, P4, O1, O2, F7, F8, T3, T4, T5, T6, Fz, Cz, Pz), bipolar submental electromyographic (EMG), and electrooculographic (EOG; E1-M2, E2-M1) recordings, nasal airflow, measures of thorax and abdominal effort, peripheral oxygen saturation, and pulse and position sensors recordings performed according to American Academy of Sleep Medicine (AASM) rules. Sleep stages (Wake, N1, N2, N3, rapid eye movement [REM] sleep) were scored in 30- sec epochs according to standard criteria. EEG, EMG, and EOG signals were sampled at 256 Hz; EEG and EOG were filtered at 0.3–35Hz; EMG was filtered at 10–100Hz. EEG channels were sampled at 256 Hz using Neurofax (Nihon Kohden Co, Tokyo, Japan) Nineteen electrodes were placed according to the international 10–20 standard with a 19-electrode montage referenced to the contralateral mastoid. All signals were digitally offline filtered with a passband of 0.5–40 Hz and were referenced to the average signal of A1 and A2 electrodes. Sleep was subdivided into 30-s epochs and sleep stages were scored according to the standard AASM criteria. 7 The following parameters were obtained: Time in bed, sleep period time, total sleep time (TST), sleep onset latency, sleep efficiency, wake time, proportion of the TST spent in each sleep stage, REM latency, arousal index, and index of periodic limb movements.
EEG- Sleep Spindle Analysis
Sleep staging and sleep spindles in EEG were analyzed visually inspected by at least three pediatric neurologists. Spindles were extracted in artefact-free epochs and both on and off set marked by visual inspection by a blinded team member and verified by a trained somnologist in cases of uncertainty. An automated process was applied to reject outlier epochs from N2 and N3 sleep separately, in both the time and frequency domains. After the determination of NREM stage 2 periods in PSG, we obtained spindles from EEG signals during all NREM stage 2 sleep, with an impedance value equal to or lower than 10 kΩ during the corresponding 15-s epoch. Epochs containing arousals were excluded. The sleep spindles were analyzed in two frequency bands (10–13 Hz, and 13–16 Hz) to differentiate the slow and fast spindle characteristics. For all spindle analyses, C3 was used to identify and characterize fast spindles, and F3 was used for slow spindles. 16 For each sleep spindle, amplitude (peak-to-peak difference in spindle voltage, μV), frequency (number of waveforms per second), activity, duration of spindle bursts (s), and density (number of spindle bursts/minute of NREM sleep) on the reference montage of F3 channel were calculated. 17 Figure 2 shows sleep spindle features (duration, analysis, amplitude, spindle frequency).
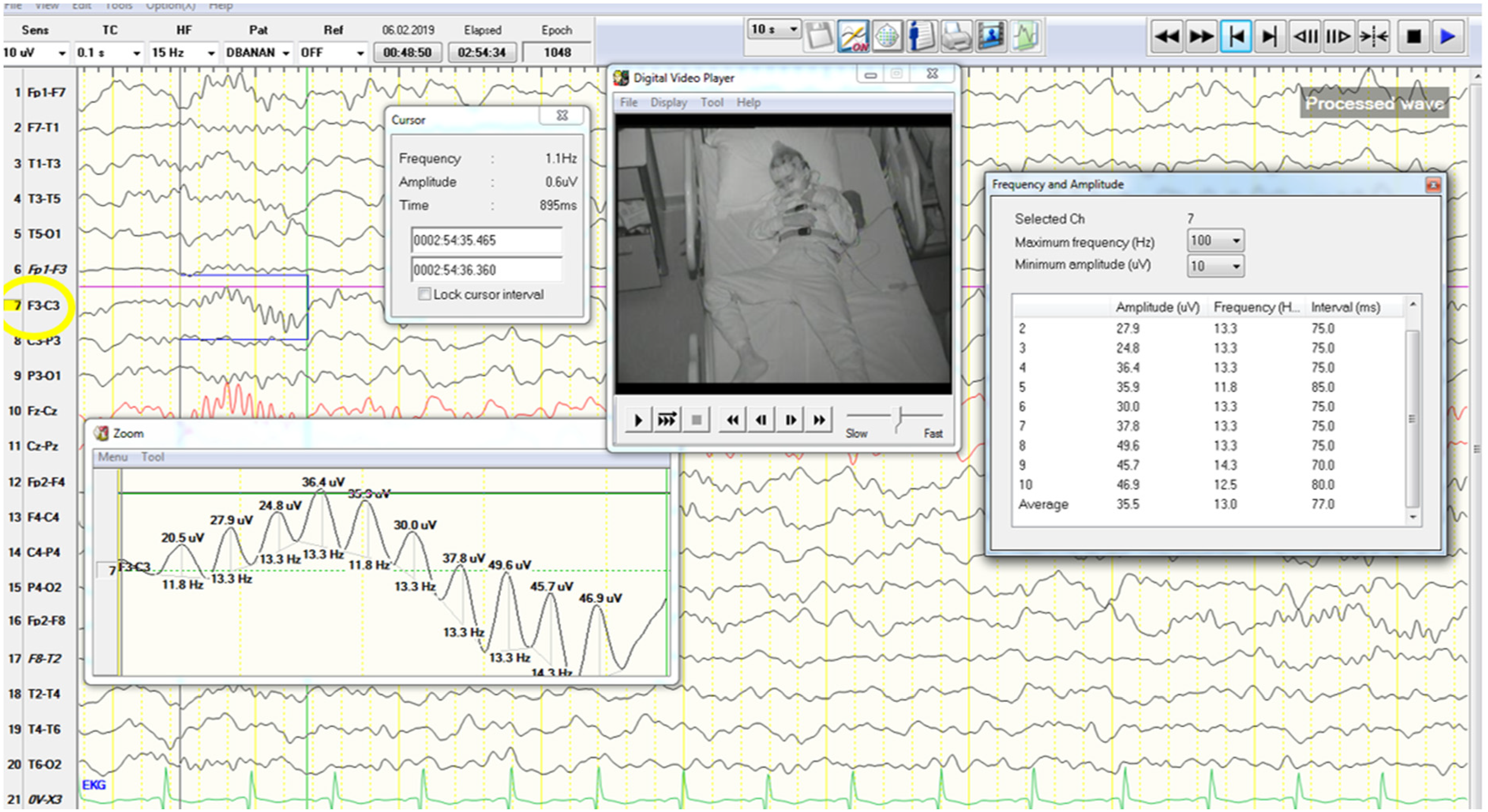
Shows sleep spindle features (duration, analysis, amplitude, spindle frequency).
Statistical Analysis
Data analyses were performed using the SPSS for Windows, version 23.0 software. For descriptive statistics, mean, standard deviation, median, frequency, and percentages were presented. The Chi-square test was used to compare qualitative data, and Fisher's exact test was used when necessary.
The Mann–Whitney test was used for comparisons between HC group and and the children with ADHD. The one-way ANOVA test was used to evaluate the differences between the denstiy (per min), frequency (Hz), amplitude (V), duration (s), and activity of spindles in HC and children with ADHD. In all cases, we considered a 95% confidence level (p < 0.05).
Results
Demographic Variables
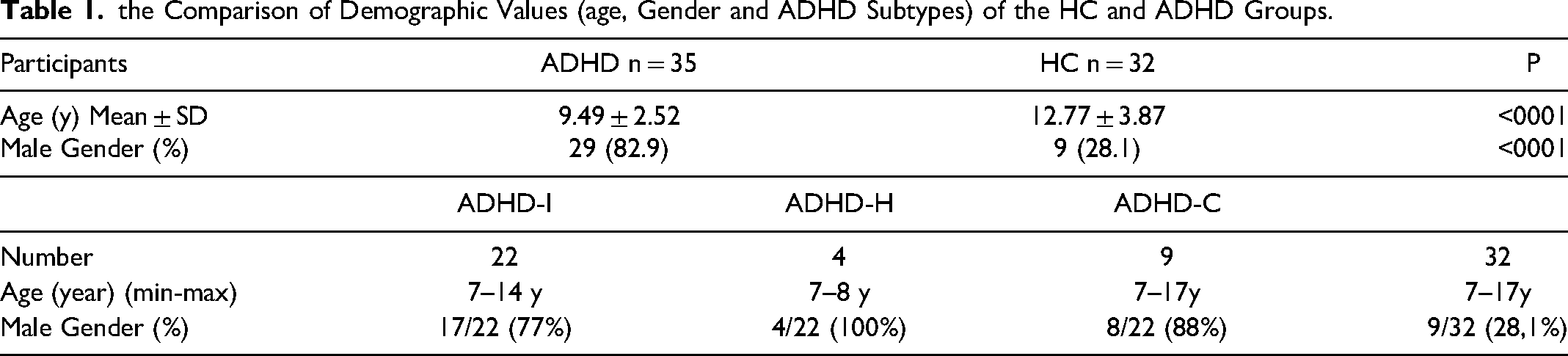
A total of 67 participants, thirty-five children with ADHD (six females and 29 males) aged 7–17 (mean: 9.49 ± 2.52) years, and 32 age and sex matched healthy control children (ten males and 22 females) aged 7–17 (mean: 12.77 ± 3.87) were included in the study. Table 1 summarizes the comparison of some demographic values (age, gender, number of subgroups participans) of the healthy control and ADHD groups.
the Comparison of Demographic Values (age, Gender and ADHD Subtypes) of the HC and ADHD Groups.
Sleep Spindles
Children with ADHD had significantly higher amplitude, duration, frequency, and activity of slow sleep spindles in NREM2 sleep in both frontal and central derivations (p < 0.05). Table 2 presents the comparison of total sleep spindles between the healthy controls and ADHD groups.
The Comparison of Sleep Spindles Between the HC and ADHD Groups on F3-A1 Derivation.
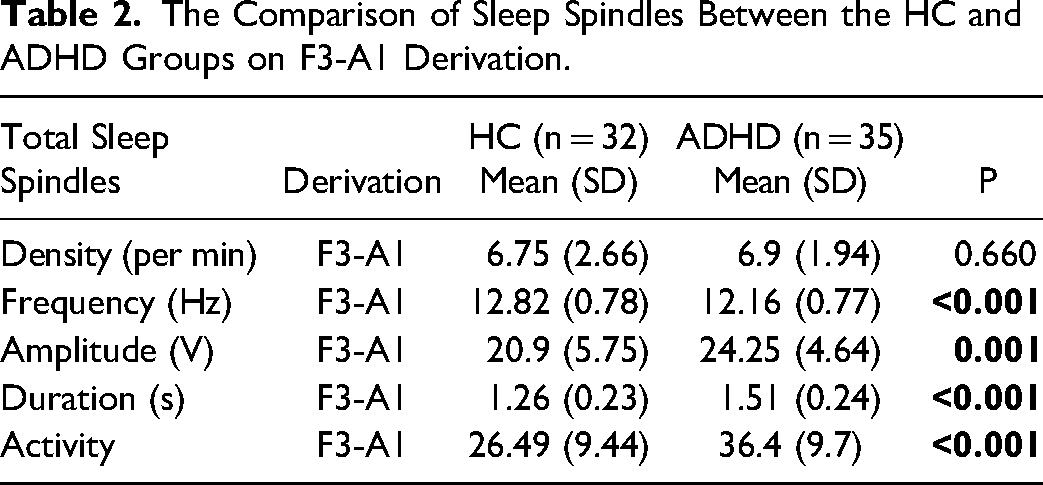
Patients with ADHD had a significantly lower frequence of sleep spindles within N2 sleep in both frontal and central derivations. But mean spindle amplitude,duration and activity for spindles were significantly increased in patients with ADHD in derivation F4 and both central derivations (Table 2).
ADHD Subtypes and Sleep Spindles
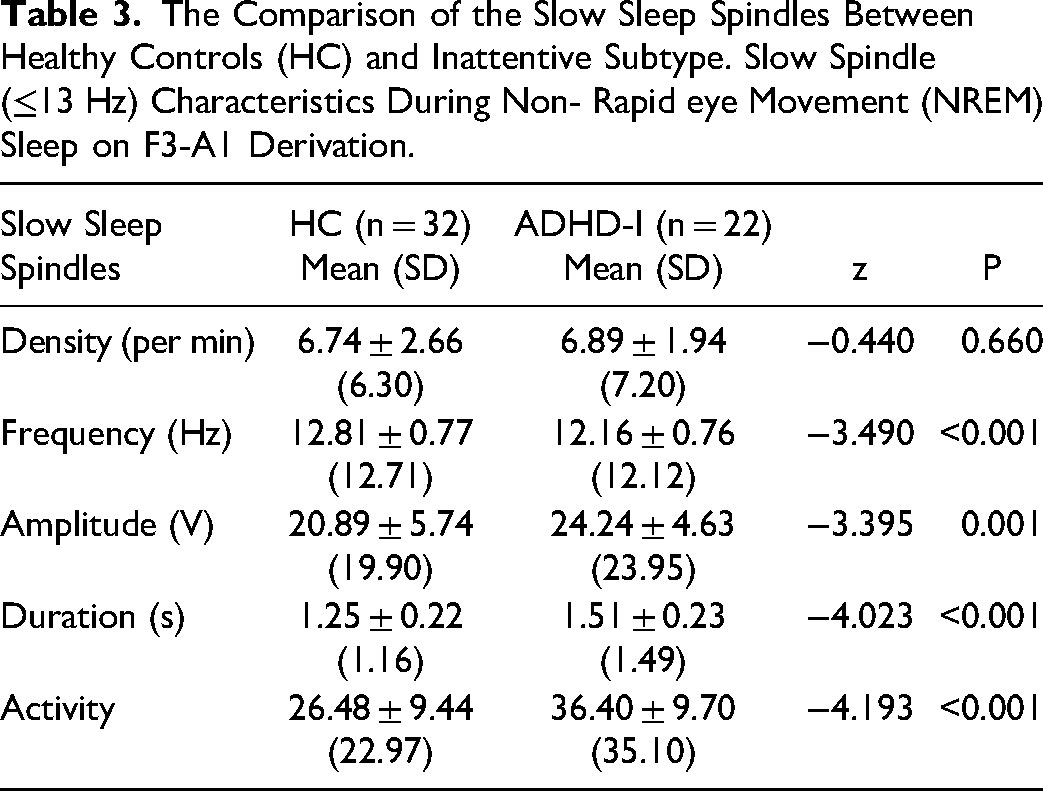
At the ADHD subgroup analysis, between ADHD-C and ADHD-I groups there were not statistically significant difference in sleep spindles features. The ADHD-H group was not included in this comparison because of small number of patients. But there was a significant difference in the density, duration, activity and amplitude of sleep spindles between HC and ADHD- I patients. Table 3 presents the comparison of the slow sleep spindles between HC and ADHD- I.
The Comparison of the Slow Sleep Spindles Between Healthy Controls (HC) and Inattentive Subtype. Slow Spindle (≤13 Hz) Characteristics During Non- Rapid eye Movement (NREM) Sleep on F3-A1 Derivation.
Discussion
There are a handful of studies examining sleep spindle characteristics in children with ADHD subgroups, However there is only one very recent study which has investigated slow sleep spindles in children with ADHD and compared this to HC children using all-night PSG. In line with this recent study, our results indicated that children with ADHD had significantly higher slow sleep spindle amplitude, duration, density, and activities at the frontal regions compared with healthy controls.
There may be two possible explanations for the presence of higher atypical spindles in ADHD. The first, and most likely, explanation may be the abnormalities in the thalamocortical (TC) network between the thalamic nuclei, which regulates attention and processes sensory information during wakefulness, and the frontal cortex related to the Na+ channel. The higher slow sleep spindle amplitudes, duration, density, and activities of frontal spindles in ADHD may reflect abnormality of the Na+ channel in the TC neuronal network. 18 In fact, Na+ channels are essential for neuronal excitability. They open in response to depolarizations increasing the membrane conductance to Na+ and thus generating the rising phase of action potentials. Dravet syndrome (DS) is the prototype of this abnormality. Dravet syndrome is a severe childhood-onset epilepsy largely due to heterozygous loss-of-function mutation of the gene SCN1A, which encodes the type 1 neuronal voltage-gated sodium (Na+) channel α subunit Nav1., is a prototype of Na+ channels abnormality. 19 Children with DS frequently have cognitive impairments such as inattention, hyperactivity, and mental retardation. Attention, hyperactivity and working memory impairments in DS was reported to be related to Na+ channel abnormality. 20 Thus, Na+ channel abnormality in the TC network, especially from the thalamus to the frontal may generate higher slow spindle activity.
The second possible reason is immaturity of the sleep/wake system in children with ADHD. The cortical maturation delay in ADHD was found to be most prominent in the lateral prefrontal cortex, particularly within the superior and dorsolateral prefrontal regions.21–23 The prefrontal cortex appears to be linked with cognitive functions such as the ability to suppress inappropriate responses and thoughts, working memory, information processing, behavioral organization, attention, judgment, and the ability to cope with novel experiences. Deficits in these cognitive functions have all been figured out in the pathogenesis of ADHD, and prefrontal cortical hypoactivation in children with ADHD during performance of many of these tasks is a relatively consistent finding.24,25 The activity of noradrenergic, cholinergic, orexinergic, histaminergic, and serotonergic neurons is strongly modulated by the sleep/wake cycle, being high during wake, low during NREM sleep and even lower during REM sleep. These functions all rely on prefrontal cortex. Thus, our results specifically suggest that the immature neural circuits and neuromodulatory systems of sleep/wake regulation system projecting to prefrontal cortex may be responsible for immature sleep spindles in children with ADHD. How such changes might influence the dynamics of cortical development remains unclear but would be an important area for future research.
The ADHD subtype comparisons in this study demonstrated significantly higher amplitude, duration, density and activities of frontal slow sleep spindles in the ADHD-I. To our knowledge, this finding is novel in children with ADHD-I. Attention is a complex neurobehavioral domain of cognition in which a specific target is selected for further processing, and neural resources directed toward that target. Recent functional neuroimaging investigations in patients with ADHD and experimental studies have lateralized attentional networks to thalamus and prefrontal cortex. 26 Higher slow spindle activity in the ADHD-I subgroup may suggest dysfunction and hyperfunction in the prefrontal cortex and interconnections between prefrontal cortex and thalamus. 15 In a previous study, slow-spindle activity in children with ADHD was found greater than the healthy control group. 27 They concluded that frontal slow spindles reflect attention, executive function and impulse suprression in patients with ADHD. In another study, higher slow-spindle activities in the frontal lobe were exhibited in children with higher IQ scores (mean age, 9.56 years). 28 In an adult study, fast spindle activities in the left frontal and parietal areas were enhanced while acquiring a new visuomotor skill. 29
On the other hand a prior study in healthy preschool children (mean age, 4.3 years), demonstrated greater slow-spindle activities to predict fast reaction times during cognitive tasks. 28 None of these findings can be considered as a single established mechanism in patients with ADHD. A key conclusion of our study is that slow spindle activity may be used as a new diagnostic biomarker in patients with ADHD, particularly in ADHD-I subgroup. To further understand these mechanisms, we need well conducted studies of spindles in children with ADHD.
Despite the new findings, there are some limitations of this study to acknowledge. First, the sample size is relatively small and that a larger sample size may add information on the differences between ADHD and healthy controls. Unfortunately, we were not able to perform a systematic cognitive assessment of these children, which would exactly be intriguing to evaluate in future studies. We also did not perform any correlation analyses regarding cognitive outcomes with sleep spindle activities in either group. Third and last, we used a visually scoring method for sleep spindles which did not allow measurement of other spindle characteristics, like power spectral analysis. We recommend specific software tools that can aid sleep specialists for elaborative sleep spindle analysis.
Conclusion
Children with ADHD and particularly ADHD-I subgroup exhibit significantly higher slow spindle activity in the frontal regions when compared to healthy children. If replicated together with systematic cognitive assessment, these findings would indicate dysfunction and hyperfunction in the prefrontal cortex and interconnections between prefrontal cortex and thalamus as a possible mechanism in ADHD. Sleep spindle characteristics may be used as a suitable tool in early recognition and diagnosis of ADHD and stratification of subgroups. To better assess the value of sleep spindle caharcteristics for ADHD and optimize the diagnosis, we recommend further research with improved study methodology to enhance.
Footnotes
Acknowledgments
We would like to thank all the participants.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
