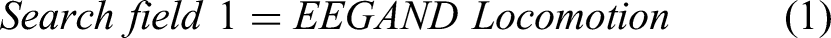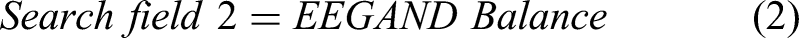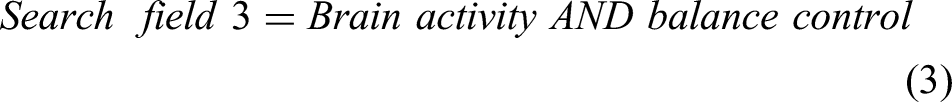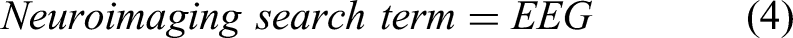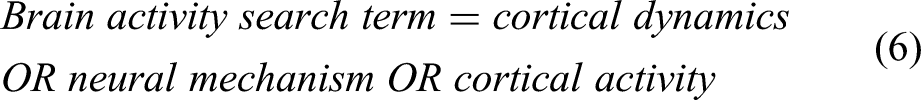Abstract
The past decade has witnessed tremendous growth in analyzing the cortical representation of human locomotion and balance using Electroencephalography (EEG). With the advanced developments in miniaturized electronics, wireless brain recording systems have been developed for mobile recordings, such as in locomotion. In this review, the cortical dynamics during locomotion are presented with extensive focus on motor imagery, and employing the treadmill as a tool for performing different locomotion tasks. Further, the studies that examine the cortical dynamics during balancing, focusing on two types of balancing tasks, ie, static and dynamic, with the challenges in sensory inputs and cognition (dual-task), are presented. Moreover, the current literature demonstrates the advancements in signal processing methods to detect and remove the artifacts from EEG signals. Prior studies show the electrocortical sources in the anterior cingulate, posterior parietal, and sensorimotor cortex was found to be activated during locomotion. The event-related potential has been observed to increase in the fronto-central region for a wide range of balance tasks. The advanced knowledge of cortical dynamics during mobility can benefit various application areas such as neuroprosthetics and gait/balance rehabilitation. This review will be beneficial for the development of neuroprostheses, and rehabilitation devices for patients suffering from movement or neurological disorders.
Introduction
Approximately 64, 6000 individuals die from falls all over the world, and out of which around 80% belong to low and middle-income countries (Statistics of World Health Organization, 2019) 1 . The adults aged 65 years or above suffer the largest number of falls. These days, lower-limb amputation is one of the most common problems in society. The primary causes for lower limb amputation are vascular diseases (54%), including diabetes and peripheral arterial disease, trauma (45%), and cancer (less than 2%), which result in the loss of balance and generally contributes to slips and falls (Limb loss statistics, 2017) 2 . Furthermore, there is an estimate of seven to ten million people worldwide that are living with neurological disorders like Parkinson’s, which also results in falls while walking (Statistics on Parkinson’s, 2015) 3 . Therefore, the analyses of balance and gait impairments are generally done for the benefit of these populations.
In the human body, balancing and locomotion are the biomechanical acts of maintaining the center of mass (COM) inside the support base. It involves a closed-loop control between the central nervous system and peripherals, leading to the required actuation of muscles. Sensory inputs like visual, vestibular, and somatosensory are essential for maintaining balance during standing and walking. Thus, it becomes necessary to understand the cortical dynamics of the coordination between the central nervous system and peripherals. The prior investigations related to human balance and locomotion control were mainly focused on quantifying the motor responses based on kinetic and kinematic measurements.1–7 However, this potential is currently limited by an incomplete understanding of cortical mechanisms involved in human balance and locomotion control. Such knowledge, if available, has the potential to provide gait rehabilitation to the population suffering from neurological disorders. Also, the intelligent prosthesis is another area that can undoubtedly benefit from this knowledge.
There are several neuroimaging techniques such as positron emission tomography (PET- based on injecting radioactive tracers into the bloodstream), functional near-infrared Spectroscopy (fNIRS- based on using near-infrared light to estimate cortical hemodynamic activity which occur in response to neural activity), functional magnetic resonance imaging (fMRI- based on detecting the blood-oxygen-level-dependent contrast to map out the neural activity in terms of energy used by brain cells) which have provided the evidence of cortical involvement while maintaining balance with some extent of subject movement. Using fNIRS, sensorimotor cortices, supplementary motor area (SMA) and prefrontal cortex has been observed to be activated during the gait initiation. 8 While applying various external loads in an fMRI paradigm during stepping movements to stimulate ground reaction forces experienced during real walking, SMA was again found to be activated. 9 Using PET to reveal the spatial characteristics of balance control, it was found that, following unipedal stance, cerebral anterior vermis and posterior lobe lateral were activated. 10 However, during bipedal stance, cerebellar anterior lobe and right visual cortex were found to be activated. These findings provide spatial insights into the neural correlates of human balance control. In spite of this, these modalities are extremely sensitive to motion, particularly in voxels that have a large difference in signal intensity relative to adjacent voxels. During signal acquisition, even a motion of a fraction of a voxel can induce large fractional signal change, leading to incorrect results. Although the high spatial resolution of fNIRS, PET and fMRI neuroimaging modalities is valuable for clarifying the neural sources included in human balance control. However, with a blurred intrinsic hemodynamic response and a finite signal-to-noise ratio, these modalities lack the temporal resolution from spatially distinct brain regions as provided by Electroencephalogram (EEG). 11 The use of EEG offers the best time resolution of the brain activity and has the potential for applications where portability is required.12–14 Previously, the cortical activity during lower limb tasks (locomotion and balancing) was not well understood due to the presence of substantial movement artifacts. However, the recent development of advanced signal processing in EEG and analytical methods such as independent component analysis (ICA), has made it possible to dissociate cerebral activities and non-cerebral artifacts. Additionally, advances in hardware system of EEG have allowed in free movement in space with wireless data transmission which is quite affordable in comparison to fMRI and PET. Therefore, the EEG seems a better and practical choice for the detailed study of the cortical activities during various locomotion and balance tasks. EEG based brain computer interface (BCI) is one of the most rapidly developing fields and has the potential to expand more. The potential of EEG based BCI system in medical applications has also been used in the rehabilitation of patients suffering from neuromuscular diseases, such as amyotrophic lateral sclerosis, motor disabilities, spinal cord injuries (SCI), or stroke, etc. 15
Traditionally, EEG was used to evaluate neurological disorders, but now it is frequently used to quantify the cortical response with event-related changes occurring during balance and gait. The event-related potential has been observed to increase in the fronto-central region for a wide range of balance tasks.16–18 It was also reported that the theta (4-8 Hz) power increased in the frontal, central and parietal regions of the cortex during challenging postural conditions. 19 Moreover, the electrocortical sources in the brain areas, namely the anterior cingulate, posterior parietal, and sensorimotor cortex, remain active during walking.20,21 It is found that the frontoparietal region controls the postural stability following a transcranial magnetic stimulation (TMS) over the SMA during a continuous balance task in healthy individuals. 22 These studies directly indicate the cortical structures involved in the adjustment of postural equilibrium. The previous review studies on the neuroimaging of human balance and locomotion control, focus on all the neuroimaging modalities, including fNIRS, PET, fMRI, EEG, etc.23,24 Here in this paper, the primary objective is to present a detailed study on the findings of cortical activities involved in two human activities, ie, balancing and locomotion, using the EEG methodology.
Review Objectives
In this section, the significant objectives designed for this study are presented. The main reason behind this study is to address the findings, limitations and future possibilities of EEG technique, mainly for two human activities, ie, balancing and locomotion. The basic diagram of an EEG system is presented in Figure 1, in which there is an EEG cap to be worn by the subject. The electrodes are generally placed as per the standard 10-20 international electrodes placement system. The EEG datasets for particular experimental protocols are acquired by the EEG acquisition system, which is then processed using signal processing techniques, to make it in a usable form. Further, the processed data is analyzed and used for various applications such as brain-computer interface, prosthetic design, exoskeleton, and rehabilitation of patients suffering from movement or neurological disorders.
The basic setup for EEG acquisition during locomotion and balancing.
The analysis of cortical activity in human balance and locomotion control is noteworthy to address slips, falls, and uncoordinated walking in lower limb amputees, elderly subjects, and individuals suffering from neurological disorders. Here, we present a detailed review of the EEG modalities to investigate the cortical correlates of human balance and locomotion control, which could address the following three research questions:
Q1: How do the cortical dynamics modulate while envisioning or executing different locomotion tasks?
Q2: How does the cortical activity changes while executing different balancing tasks with or without the challenges in sensory inputs and cognition?
Q3: Which techniques have been used to detect and remove the artifacts from EEG signals during locomotion and balancing tasks?
Review Methodology
In this Section, the search strategy and search terms used for this study are presented. Further, the method for the search process and inclusion-exclusion criteria are described.
Search Strategy and Search Terms
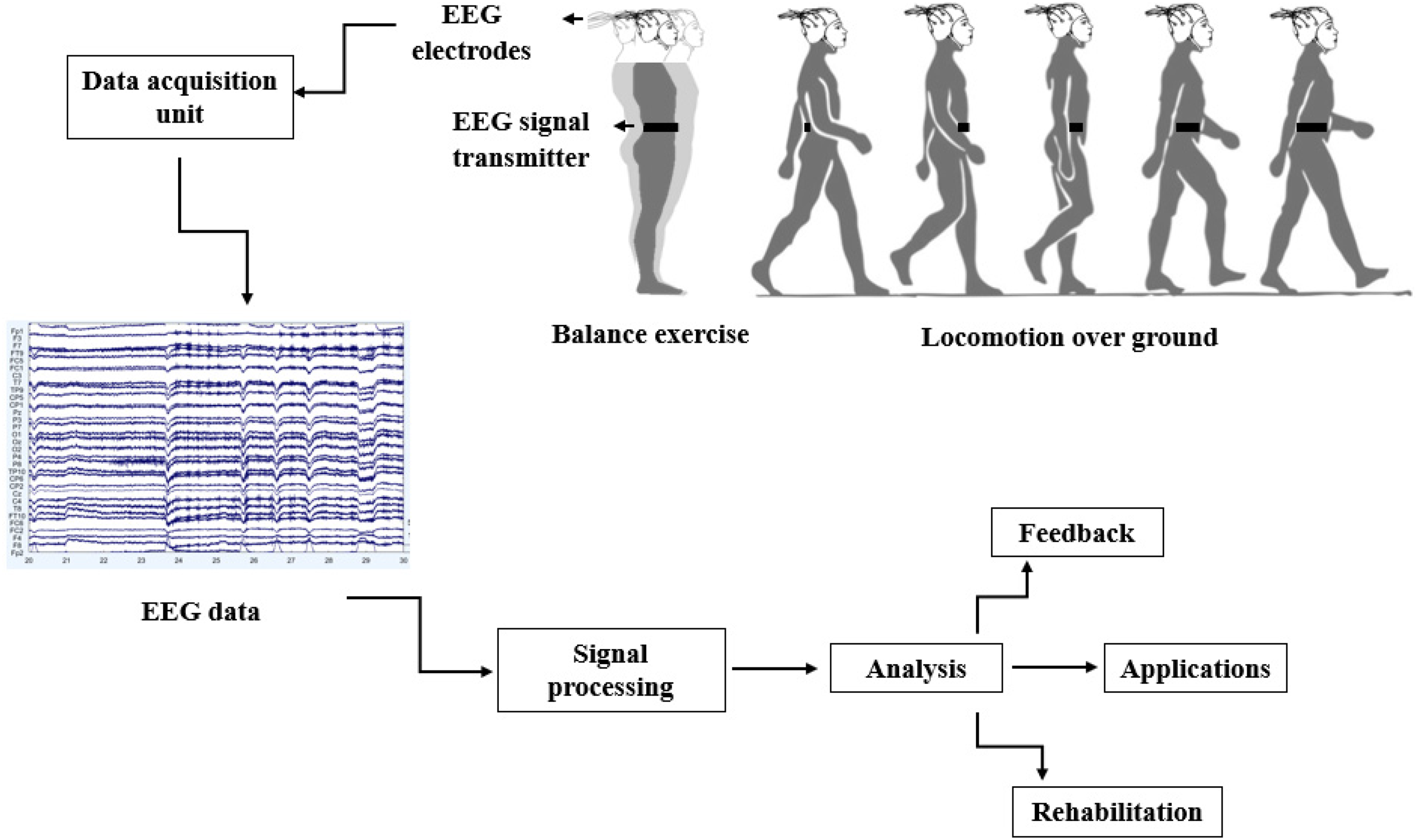
For this study, the Preferred Reporting Items for Systematic Reviews and meta-Analysis (PRISMA) method, as shown in Figure 2 is used. 25 The searches include the precise terms related to locomotion, balancing, brain activity, and EEG. The search fields are linked with “AND” to ensure that at least one term could be found in the results. The search terms for this review include EEG, standing balance, posture, treadmill walking, stepping, gait, locomotion, cortical activity, neural mechanism, and cortical dynamics. The terms in each search field are connected with “OR.” Following research equations (1-6) give a well-defined structure of the search method adopted for this review.
PRISMA flow diagram for this review.
Search Process and Inclusion-Exclusion Criteria
Various research articles included in this review are from IEEE Xplore, Ei-Compendex, PubMed, and ACM Digital Library. The inclusion and exclusion criteria for this study are mainly based on three significant objectives of the review, described in section II. The studies are only involved if they subsume the balance and locomotion challenges. Moreover, the studies that include drug trials and therapy were excluded from this review. The data selected from each article include publication year, authors, type of balance task, ie, static or dynamic, walking on over ground or treadmill, ramp or stairs, protocols used, subject attributes, and brain activity (temporal or frequency response).
Cortical Dynamics During Locomotion
For humans, locomotion is an inherent part of various life-sustaining activities. Along with the brain, different sensory inputs such as visual, vestibular, and somatosensory play integral roles in adapting the basic gait patterns over different terrains and transitions. The use of EEG systems for the investigation of brain activities is a novel research area. The research in brain activities during voluntary, rhythmic locomotor tasks in healthy subjects, the elderly population, and patients suffering from stroke, neurodegenerative diseases, amputees have begun only in the last decade. Due to locomotion's dynamic nature, brain activity investigation could lead to a better understanding of locomotion in different health conditions. In the following subsections, modulation of cortical activities during three substantial walking conditions are discussed.
Cortical Dynamics During Motor Imagery of Walking
The process of imagining movements without actually performing the physical activities of any body part is called motor imagery. The hypothesis behind performing motor imagery arises from the activation of brain areas that are comparable to that of physical motions. 26 The rehabilitation of individuals who have complications in executing physical actions can be done through motor imagery paradigms to examine the specific parts of the brain activated during imagination. The review of research studies in the area of cortical dynamics investigations during imagined walking tasks is discussed further.
The walking associated EEG features were investigated in a BCI study where the subjects performed an indicated walking task, a complex walking task in real-time. Further, the same tasks were also imagined to investigate the motor imagery activities. 27 During these tasks, the EEG data were classified into two cases, ie, actual walking and imagined walking. The classification accuracy was found to be better for actual walking than the imagined walking; therefore, it was inferred to help in BCI studies. The analyses of the EEG differences between imagining the stepping movements of the left and right leg have also been investigated, where it was shown that the frequently used mu and beta event-related desynchronization (ERD) features revealed no considerable difference between the two imaginary movements. 28 The theta band ERD significantly varied between the two imaginary actions, which can act as a basis for developing BCI systems for decoding left and right stepping imagination. 28 Another study showed that the power spectral modulations in the sensorimotor cortex depends on the walking phases (swing and stance) while walking and imagining it. 29 The phase dependent modulations were also similar to those which occur during actual walking as reported by other previous studies. The decoding accuracy of these gait phases during motor imagery was improved significantly by concurrent action observation. 30 All these findings would extend the understanding of cortical mechanisms underlying cognitive motor processes and provides clinically beneficial information.
Cortical Dynamics While Walking on a Treadmill
Several authors have worked in the area of cortical dynamics investigation while walking on a treadmill. Gwin et al presented the coupling between electrocortical activity and gait phases. 31 The electrocortical sources in the anterior cingulate, posterior parietal, and sensorimotor cortex exhibited significant intra-stride changes in spectral power. Another study required the subjects to walk on a treadmill in synchronization with a series of cue tones and instructed to quickly adapt their step rate and length according to the sudden shifts in the cue rhythm. 20 It was found that mean beta-band power was suppressed in the central midline and parietal regions and increased near the medial prefrontal and dorsolateral prefrontal cortex. This shows that the two distinct patterns of beta-band activity accompany the gait adaptations. The spatial and temporal characteristics of electrocortical activity during treadmill walking were also assessed. 21 The recorded data was related to the gait phases and based on the detection of the left heel strike; movement-related cortical potential was observed over the motor cortex's leg representation area. The inference of gait kinematics from the hip joints, knee, and ankle while walking on the treadmill has also done where it is shown that the limb kinematics both from within the body and among the subjects in human treadmill walking can be achieved which support the possibility of EEG-based BMI systems for rehabilitation. 32 The increased gamma-band power in the prefrontal and posterior parietal cortex was too found during double support and early swing phases while walking on a treadmill. 33
For controlling gait stability, EEG signals have been measured during stabilized (stabilized by elastic cords) and normal walking on a treadmill. 34 Features like the range of motion, step width, and local divergence exponent were calculated. According to the results, a substantial increase in gait stability with less local divergence exponent was found in stabilized walking. The significant difference in the cortical activity in the left premotor area has been found out with higher beta band power. The determination of electrocortical activity while walking with limited vision has also been done where the subjects were requested to walk on a treadmill with eyes closed and eyes open conditions. 35 EEG and ground reaction force signals (GRFs) were gathered, and the results indicate that theta desynchronization significantly increased in the frontal and premotor cortex while walking with closed eyes. These findings provide evidence that supports the usage of restricted vision tasks in gait rehabilitation and improvement of balance control.
Additionally, to address the brain's contribution to muscle activity during gait, coherence analysis was used to find the coupling between EMG and EEG signals during walking. 36 These findings demonstrate the contribution of the motor cortex to the muscles in steady-state treadmill walking. Another study also observed that cortical motor areas which finely control leg muscle activation during treadmill walking. 37 Effective connectivity was measured by direct transfer function between motor/non-motor cortical region of interests (ROIs) and muscle signals in the swing phase. It was found that brain-to-muscle connectivity was more substantial than the muscle to brain connectivity.
The impact of virtual environment feedback on EEG patterns during robot-assisted treadmill walking has been examined. 38 The spectral power in alpha, beta, and gamma bands was significantly decreased while walking in a virtual environment and interpreted as a correlate of an active cortical area. Another study observed significant differences in the cortical activation between active and passive robot-assisted gait training. 39 Beta and gamma rhymes were suppressed during functional gait training in central midline areas compared to inactive training. The brain dynamics in different reference frames during navigation in virtual reality were investigated. 40 A large increase in the amplitude of the alpha wave was observed in the primary visual cortex when the subjects responded to the virtual reality with an egocentric reference frame. Simultaneously, in the occipital and temporal region, the subjects responded with the allocentric reference frame.
Unique features of EEG signals were evaluated during the presence of unexpected obstacles while a subject walked on a treadmill. 41 The brain activity changes were assessed through features like average power, slope, common spatial patterns, and the coefficients of a polynomial fit. An increase in the spectral power in the SMA and premotor cortex in the delta, theta, and alpha frequency bands was also observed. 42 At the same time, the subjects performed stepping over obstacles during walking on a treadmill. It was shown that to track the obstacle location and plan the foot placement of reaching the obstacle, there was an increase in the spectral power within the posterior parietal cortex, which helps to track the obstacle while interrupting the gait cycle. Avoiding the obstacles while walking on the treadmill, the power spectral density of theta and delta wavebands was again observed to increase. 43 The obtained results from these studies can improve the user-exoskeleton interaction, improving the safety devices for current exoskeletons where there is a need to make an emergency stop command when an obstacle is detected during walking.
The differentiation between incline and level walking in humans has also been done based on electrocortical activity when the subjects walk on the treadmill with different grades. 44 Results revealed that theta power was more during walking on the inclined surface in the posterior parietal, sensorimotor and anterior cingulate clusters. Also, gamma power increases during level walking in the anterior cingulate and left sensorimotor cortex. Power fluctuation in the theta waveband over the posterior parietal, sensorimotor and anterior cingulate cortex was observed to increase with the treadmill's inclination. 44 These findings propose the changes in locomotor patterns essential to walking on an inclined surface in comparison with a level surface.
Spectral and time-frequency analysis of EEG and accelerometer signals at different speeds has been done which revealed that accelerometer and EEG signals show similar time-frequency properties and also suggest the initiation of high-gamma rhythms during walking. 45 The analysis of EEG signals associated with gait speed was again done when the subjects were requested to perform speed change while walking at different rates on a treadmill. 46 Simultaneously, the speed of the treadmill belt was adjusted by the controller according to the subject’s linear speed. According to the recorded EEG signals, a classifier was also trained to distinguish between constant gait speed and gait speed changes. Results suggest that mu and beta rhythms in EEG were inhibited during gait speed changes, and there was increased activity in parietal areas, which may be due to the motor planning throughout the gait.
Treadmill walking also results in physical artifacts in EEG recordings. To quantify these artifacts, mobile brain imaging techniques were implemented. 47 Temporally independent signals were evaluated using ICA and source localization techniques, which suggests that electrocortical activity can be modeled while dynamically balancing on the treadmill. 48 All these findings are beneficial for finding cortical involvement in human gait control. These findings show that while executing locomotion on a treadmill, human cortical activity modulates accordingly.
Cortical Dynamics While Walking on the Ground
In this section, the brain activities involved in walking on the ground are discussed. The level walking to stair ascent/descent or ramp ascent/descent is generally referred to as locomotion transitions. The neural features during these transitions in locomotion are investigated using the EEG signals, which indicates that while having transitions, four clusters of the brain are involved, ie, right and left occipital lobes, parietal cortex, posterior cortex, and motor cortex .49,50 The changes in the spectral power in these regions were associated with the level of motor task demands. Also, the amplitude modulations in electro-cortical activities were varied during different walking conditions and were correlated with the levels of difficulties in locomotion tasks. At the beginning of the gait cycle, alpha and beta waves were suppressed during stair ascent and ramp accent walking compared to level walking. It is reported that there is a decrease in gamma-power and increases in theta-and beta-power in the motor area during conditions like ramp ascent and stair ascent. 51 Costa et al used the gamma-band features of EEG to decrypt the attentional demands of gait, where the participants were requested to perform four attentional tasks during gait. 52 Different features from different frequency bands are extracted, analyzed, and tested within five classifiers to distinguish between the functions. It is found that the level of attention on the environment and visible exterior information would increase as the attention on gait decreases. The variations in the spectral content of EEG were investigated to determine the cognitive workload throughout locomotion, wherein the participants were instructed to perform the cognitive task under two stages of difficulties. 53 The observations suggest that the changes in the spectral content of cortical activity index the cognitive workload with the differences in the task difficulty. Also, there is evidence for cortical information correlated with the level of attention on the gait, which can further be used as feedback with any rehabilitation strategy.
The investigations on the neuroimaging of walking were carried out to elaborate on the rhythmicity of walking. 54 These investigations were mainly based on the gamma wave's amplitude, and the results suggest that the magnitude of high gamma waves significantly increased during walking in central sensorimotor areas. The recognition of the starting and ending of gait using features from the mu-band of EEG was done to distinguish between movement onsets or offset. 55 For this, a linear classifier was used, and it is found out that the significant gait-related desynchronization occurred in the mu-band during the intended gait. This suppression in the mu-band during voluntary gait confirms the cortical activity in gait intention.
The EEG signals have also been used to develop a system that detects the start and the stop of the gait. 56 After the preliminary analysis, the method is validated in a real-time test to enhance the brain-machine interface (BMI) training. These types of systems are beneficial to command the lower limb exoskeleton. The assessment of both the temporal and spatial gait mechanics utilizing the motion capturing machine along with the cortical dynamics was carried out for the unilateral transtibial amputees. 57 In this, the participants completed the tasks of varying difficulties while walking, which resulted in a decrease in event-related potential and an increase in the ratio of frontal theta power/parietal alpha power. The interaction between cognition and motor control for such a population has been emphasized in the conclusion of this study. The prediction of gait from pre-movement EEG signals demonstrates 76.41% accuracy for “rest versus start” classification and 74.12% accuracy for “walk versus stop” classification showing a potential for to develop real time BCI. 58 The locomotion-related potentials in EEG have been retrieved using the weighted phase lag index to quantify the variable patterns of EEG in the presence of gait-related artifacts. 59 The weighted phase lag index is the functional connectivity measure, which shows how constantly the signal from one electrode was compared while lagging in phase. It is concluded that the analysis of cortical dynamics during locomotion could be possible by using both weighted phase lag index and weighted phase lag index stability over the standard voltage-amplitude measures. The significant advantage of this method is that cognitive responses like P300 can be recovered across channels. 59
Further, Storzer et al investigated the cortical dynamics involved during walking and bicycling. It is suggested that bicycling is related to a power decrease in the high beta-band through the task's initiation and implementation. It is then followed by an increase in beta-power after the termination of the task compared to regular walking. Moreover, the characterization of walking can be done by a more potent and continuing decrease in alpha power. 60 For EEG analysis, when the subjects are required to walk and complete a motor interference task, a considerable decrease in alpha band power and an increase in beta power was observed. 61
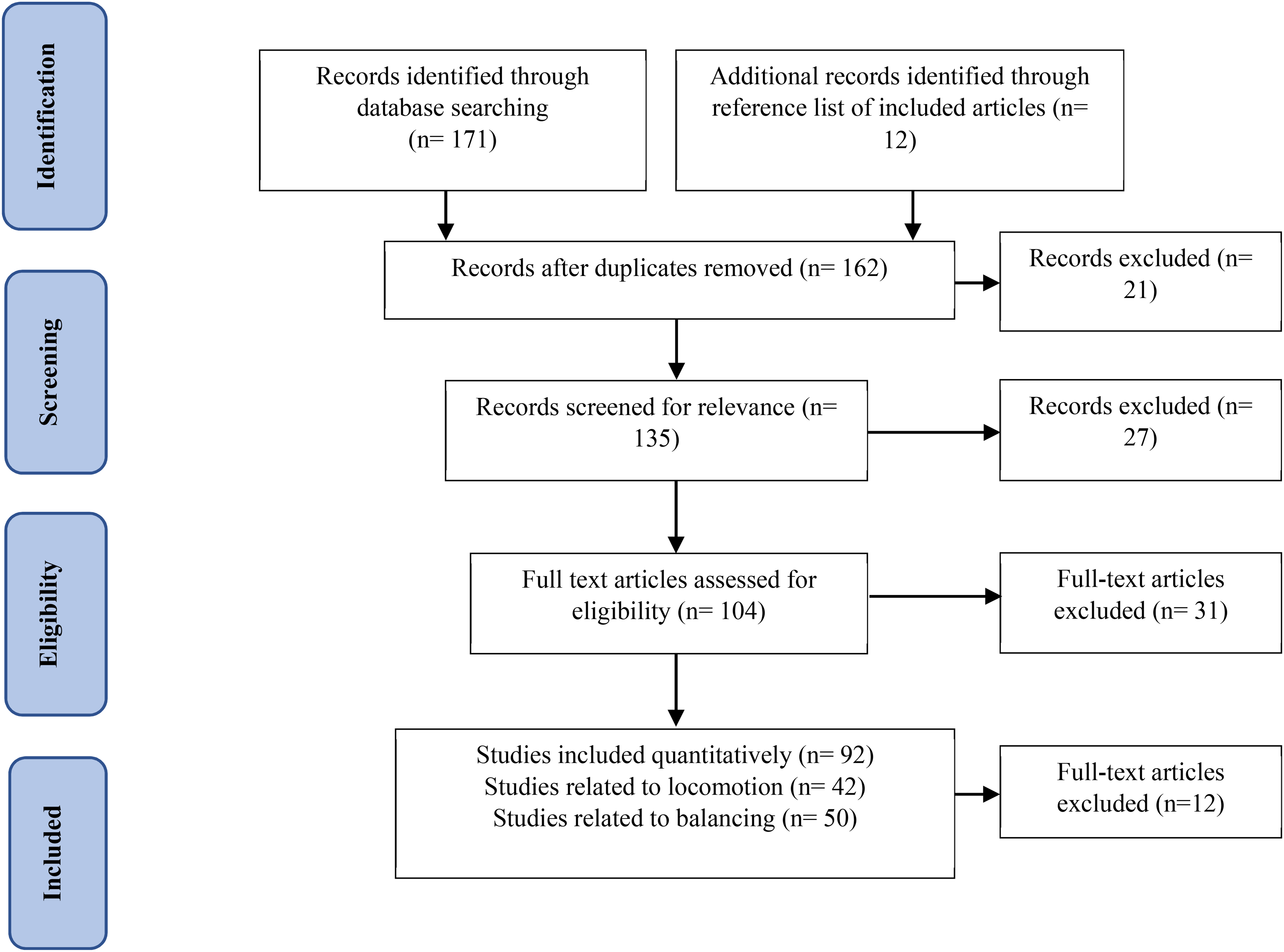
The high-density EEG recordings for different gait phases were applied with the source modeling using individual anatomy reformed from structural magnetic resonance imaging (MRI) scans. 62 The results concluded that the constant mu and beta ERD occurred during movement-related changes in cortical excitability. As low gamma frequencies have been shown to modulate the firing rate of motor neurons during a brain-machine interface task, the gait phase-related modulations in the low gamma rhythm were due to the order of motion control during gait. 63 This was suggested to represent the processes involved in motor planning. The findings presented above highlight different cortical regions which are activated while performing and envisioning various types of locomotion tasks. By extracting various EEG features from these cortical areas, an effective BCI can be formed which will be beneficial in neurorehabilitation studies. Table 1 summarizes the studies wherein the brain dynamics during locomotion have been investigated.
Cortical Activity During Locomotion.
Cortical Dynamics During Balancing
The balancing, in general, is divided into two groups: static balancing and dynamic balancing. For humans, balance refers to an individual's ability to maintain their line of gravity (LOG) within their base of support (BOS), as shown in Figure 3a. Maintaining the body in a fixed posture is referred to as Static balance. It is the postural stability with the center of mass above the base of support while the body is at rest. It has been shown in many studies that static balance involves a central coordinating mechanism in the cerebellum, which maintains the body erect. On the other hand, the ability to maintain postural stability with the center of mass above the base of support while the body is in motion is referred to as dynamic balance, as shown in Figure 3b. The central coordinating mechanism involved in dynamic balancing includes the cerebellum and basal ganglia, which helps maintain stability. The vestibular and visual systems convey natural impulses during dynamic balancing, which relate to the movement and environment. In this Section, the EEG-based findings during the balancing tasks are discussed in detail. It is mainly divided into two sub-sections: (A). Cortical activity during static balancing and (B). Cortical activity during dynamic balancing. The studies related to the variations in cortical dynamics during balancing are discussed as per the flowchart shown in Figure 4.
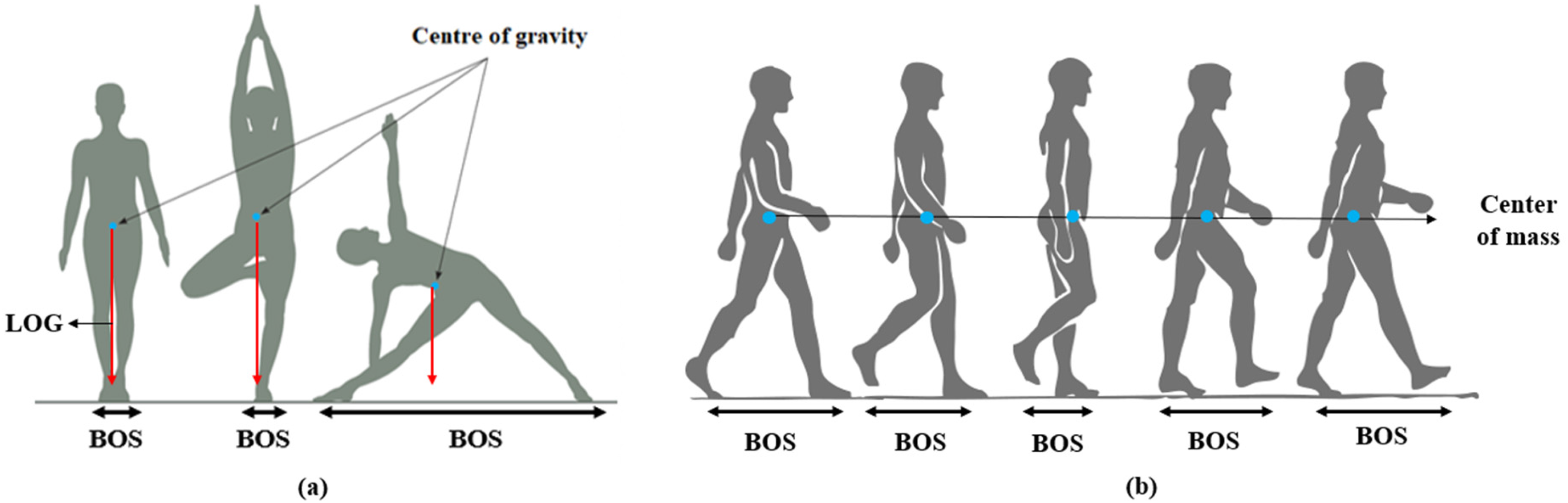
(a) static balance (b) dynamic balance (locomotion).
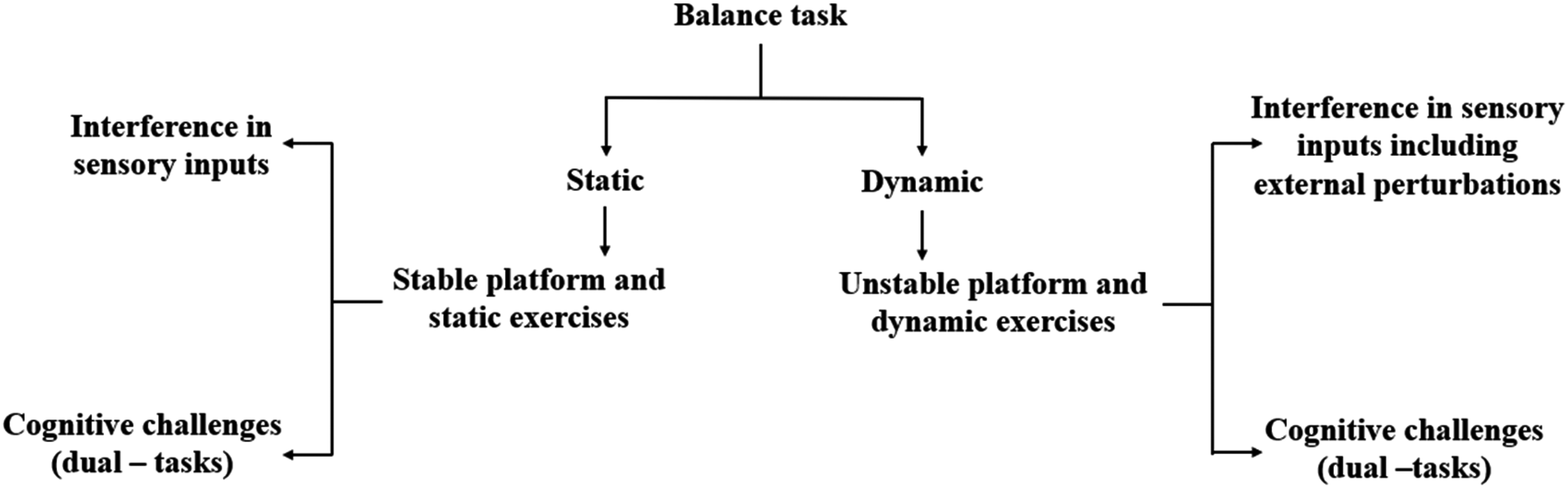
Flowchart for the presentation of review studies related to cortical activities during the balance control asks.
Cortical Correlates of Static Balance Tasks
Several existing studies have examined the EEG variations related to static balancing. The power spectral density of different EEG wavebands was investigated over various balancing tasks. 64 The variations in the power density of different EEG wavebands have also been found in response to balance task challenges.65,66 For example, the gamma-wave activation has been observed, to increase as the balance challenges increases. 65 Several authors have also investigated the event-related potential and its analysis during different balancing tasks.16,17 The detailed review of the studies on the cortical activity variations with balancing tasks are discussed in the following subsections. Moreover, the balance performance when subjects encounter dual-task sensory inputs and cognitive challenges are also discussed.
Interventions in Sensory Input While Balancing
Sensory inputs (visual, vestibular, and somatosensory) are essential for maintaining the balance of the human body. To challenge the balance control, the cortical dynamics were examined while intervening in these sensory inputs. The event-related potentials that occurred due to the sensory inputs intervention during balancing have been observed in the brain's fronto-central area.16,17 It is validated that there is an increase in event-related potential amplitude with increasing difficulties in postural challenges. 18
Ishigaki et al investigated the effect of sensory feedback on balancing. 67 It was shown that due to light touch, the postural sway reduces independent of the mechanical support. Analysis of different EEG wavebands was done to examine the brain activity related to the light touch effect during standing with closed eyes condition, which results in decreased alpha power in the left posterior parietal cortex and primary sensorimotor area. 67 While performing different standing tasks on the compliant foam surface, T3-Fz EEG coherence did not differ significantly. This proved that young adults do not pose higher real-time conscious postural control when task difficulty increases. 68 Unipedal Stance on stable and unstable surfaces revealed the significant interaction effects in left and right motor clusters while balancing. 69 Here, the leg dependent changes in alpha-2 power indicate lateralized patterns of cortical processing in motor areas during single-leg stance. Challenging the postural control system by disrupting the upright stance with mechanical skeletal muscle vibrations applied to the calve muscles, the predominance of frontal lobe connections and the strengthening of temporal-parietal network connections were found in the alpha band. 70
Moreover, the auditory biofeedback was applied to the subjects during quiet standing, both with closed and open eyes, suggesting that the postural sway reduces and postural stability increases with auditory biofeedback. 71 On the other hand, the continuous balance tasks of increasing difficulties show that the spectral power of alpha-band reduces as the task difficulty is increased. 72 It was also revealed that the coherence in the alpha-band increases with the balance task difficulty. This study demonstrates the functional importance of bilateral central and parietal cortices in continuous balance control. 72
Cognitive Challenges (Dual-Tasks) While Balancing
The dual-task refers to the allocation of the attentional resources to perform both cognition and balancing. The postural task was paired with a cognitive job to investigate cortical activity while balancing. 73 It has been shown that with the increase in the center of pressure trajectory, there is a reduction in the event-related potential in the right sensory and left cortical motor areas. The presence of visual feedback for maintaining balance on a tilt platform results in the decrease of the amplitude of event-related potential over the motor cortex and sensorimotor areas. 16 The emotional response of fear and anxiety has been studied by placing the subjects at a prescribed height in which N100 event-related potential was observed before an unpredictable perturbation, and the amplitude of N100 was found to be increased with increasing height. 74 The effective connectivity during standing or walking with or without a visual oddball discrimination response task was compared, which shows that the connectivity was weaker during standing when performing a cognitive task in the prefrontal cortex, posterior parietal cortex, and anterior cingulate cortex. However, more vital connectivity was found in standing conditions than walking, proposing that more cognitive resources may be required while standing than walking. 75 These indications directly prove the role of the central processing system between the two processes in humans.
Cortical Correlates of Dynamic Balance Tasks
In this Section, the neural correlates of the dynamic balancing tasks are discussed in detail. Several existing studies have examined the EEG variations related to dynamic balance control. The movement-related potential during the postural adjustments and adjacent weight shifting exercises was explored. 76 The ERD in EEG wavebands was found preceding postural adjustments in the fronto-central cortical areas of the brain. 76 The movement's classification intent for the transition from sitting to a standing posture has been decoded from EEG before movement execution. 65 It was observed that while shifting from a stable to an unstable stage (single leg standing), there was a burst in theta waveband, which results in the reduction in theta power before an actual fall. 77 This section thoroughly explains the cortical correlations of dynamic balance paradigms, including intervention in sensory inputs and dual cognitive tasks.
Interventions in Sensory Inputs Including External Perturbations While Balancing
This section presents the review of studies on interventions in sensory inputs with external perturbations for the balancing act. By manipulating the sensory inputs (visual, vestibular, proprioception) experimentally, balance control can be confronted, and simultaneously, cortical activity can be recorded. Understanding the cortical correlations while performing dynamic balancing and the intervention in these sensory inputs has been done in many previous studies. The differential modulation of EEG wavebands was found out as a postural sway function using Morlet wavelet techniques. 78 When the subjects were instructed to produce self-paced voluntary postural sways in the anterior-posterior and medial-lateral directions, it was found that the amplitude of movement-related cortical potentials was significantly higher before medial-lateral sways. Therefore, it was concluded that medial-lateral sway appears to be more difficult and energy-demanding tasks than the anterior-posterior sway. The investigation of cortical processes related to the balancing tasks of varying difficulty on the different bases of support has been done. 19 As the balance tasks become more challenging, the theta band power increases in the parietal, central, and frontal regions of the brain. The spectral characteristics of cortical activity in balance control were found out by the individual alpha peak frequency of the EEG signals. 79 The participants completed different balancing tasks varying in the stability of the surface and base of support, resulting in an increase in alpha activity in the cortex's frontocentral region. The alpha band was also suggested to support the visual information essential for stabilizing the body while balancing. In this network, the occipital electrodes act as sources, and the interactions propagate from occipital to parietal areas. 80 Simultaneously, the theta band was proposed to support the somatosensory information required for motor responses for the subjects performing a balancing act on stable and unstable surfaces. 80
Different balance responses with varying demands in posture have been compared. 81 The subjects reacted to the rearward action of the support surface with one step forward or keeping their feet in place. Observations revealed a more substantial suppression of alpha and low gamma rhythms in the SMA with higher postural demands. This study inferred that the SMA is involved in the preparatory balance control for reactive balance responses with higher postural needs. 81 Postural kinematics” interpretation by cortical activity was also determined on a Biodex balance system. 82 The correlation coefficient between the modeled and measured anterior-posterior and medial-lateral velocities was estimated, which proved that postural balance performance could be predicted from EEG signals. The alpha-band power spectrum from frontal and central channels was considered to check the reliability of EEG measurements from a wireless device using dry electrodes during different balancing tasks. 83
The postural control during balancing was influenced by presenting expected or unexpected perturbations and their effects on the cortical activity was analyzed. 84 Observations show that negative event-related potential occurred when a sudden perturbation was given, succeeding in a sequence of expectable perturbation, which may be associated with the brain's error recognition capability. The relation between the characteristics of perturbation and the neurophysiology of reactive balance control was shown per the latency of functional postural response after perturbation evoked potential (N1), which was found faster for forward than for backward perturbation at a constant speed and was not affected by the rate of perturbations. 85 While standing and walking in response to physical and visible perturbations, identifying cortical activity was done. 86 The participants were pulled mediolaterally at the waist or presented with a virtual reality environment. The findings reveal the time-frequency variations in theta and beta wavebands were the strongest in the occipital-parietal region during visual perturbations and strongest in sensorimotor areas during pull perturbations. A cable held the subjects while weight shifting in forwarding position, and perturbations were induced by releasing the cable at regular time intervals. 87 The outcomes suggest that the amplitude of cortical activity was increased when the upcoming perturbation was unknown. In the course of recurring balance perturbations, while balancing on the leading leg, positive and negative event-related potentials were detected in the parietal and midline frontocentral cortex, respectively. 88 The self-initiated and unexpected perturbations were also introduced by liberating the load attached with a cable and, the participants stayed on a force platform. 89 The negative event-related potential was observed after the perturbation was presented for the expected trials. The analysis of cortical activity before the externally activated perturbations was carried out, suggesting the involvement of cortical activity in postural adjustments. 90 The spatiotemporal characteristics of evoked potentials in cortical activity have been explored. 91 It was demonstrated that due to instability, both upper and lower extremity balance improving responses were related to the evoked cortical potentials. The support-surface perturbations of unpredictable timing and amplitude were presented and cortical N1 response amplitude was correlated with the balance ability of the subjects. 92 A significant interaction was found between balance performance and N1 amplitude which reflect greater cortical involvement in balance recovery when challenged.
The reduction of cortical activity in experts (athletes) was evident by comparing the EEG data of athletes and non-athletes while standing on a stabilogram. 93 Observations proposed that high-frequency alpha amplitude was lower in athletes at right and middle parietal and left and right central areas than non-athletes. Similarly, the correlation between body sway and cortical activity of athletes and non-athletes during standing was analyzed, which shows that the ERD of the alpha band was more in amplitude with the reduction of body sway in athletes than in non-athletes. 94
The effects of the magnitude of perturbations on postural responses and the EEG measures were examined in Parkinson’s disease subjects. 95 An increase in the ERD in beta waveband was found to reduce the adaptability of postural responses, which suggests that cortical activity directly influences postural response scaling for people with Parkinson’s. The reorganizations of the functional brain network following amputation have been investigated using phase synchronizations, which show the functional connectivity between alpha and beta wavebands. 96 The above findings demonstrate the contribution of the cerebral cortex in retaining erect posture during dynamic balance tasks.
Cognitive Challenges (Dual-Tasks) While Balancing
The effects of a cognitive challenge on cortical activity for the balancing act with the moving platform's unexpected translations have been observed. A considerable decrease in the extent of cortical activity (N1 response, the first negative peak after perturbation onset) was found in comparison to the control state (stable platform). 97 The event-related potentials were found to be maximum over the frontal-central regions when the balance stand is disturbed by perturbation along with a cognitive task. 98 The examination of the effects of two and three-dimensional virtual reality environments on brain functions during balancing has also been carried out. 99 The frontal midline EEG power increase in the three-dimensional virtual reality in comparison to two-dimensional virtual reality was found. When postural and cognitive tasks were combined, higher delta rhythm was found over the anterior cortex in elderly population. 100 Addition of visual oddball displayed an increase in theta activation over the sensorimotor and occipital cortices.
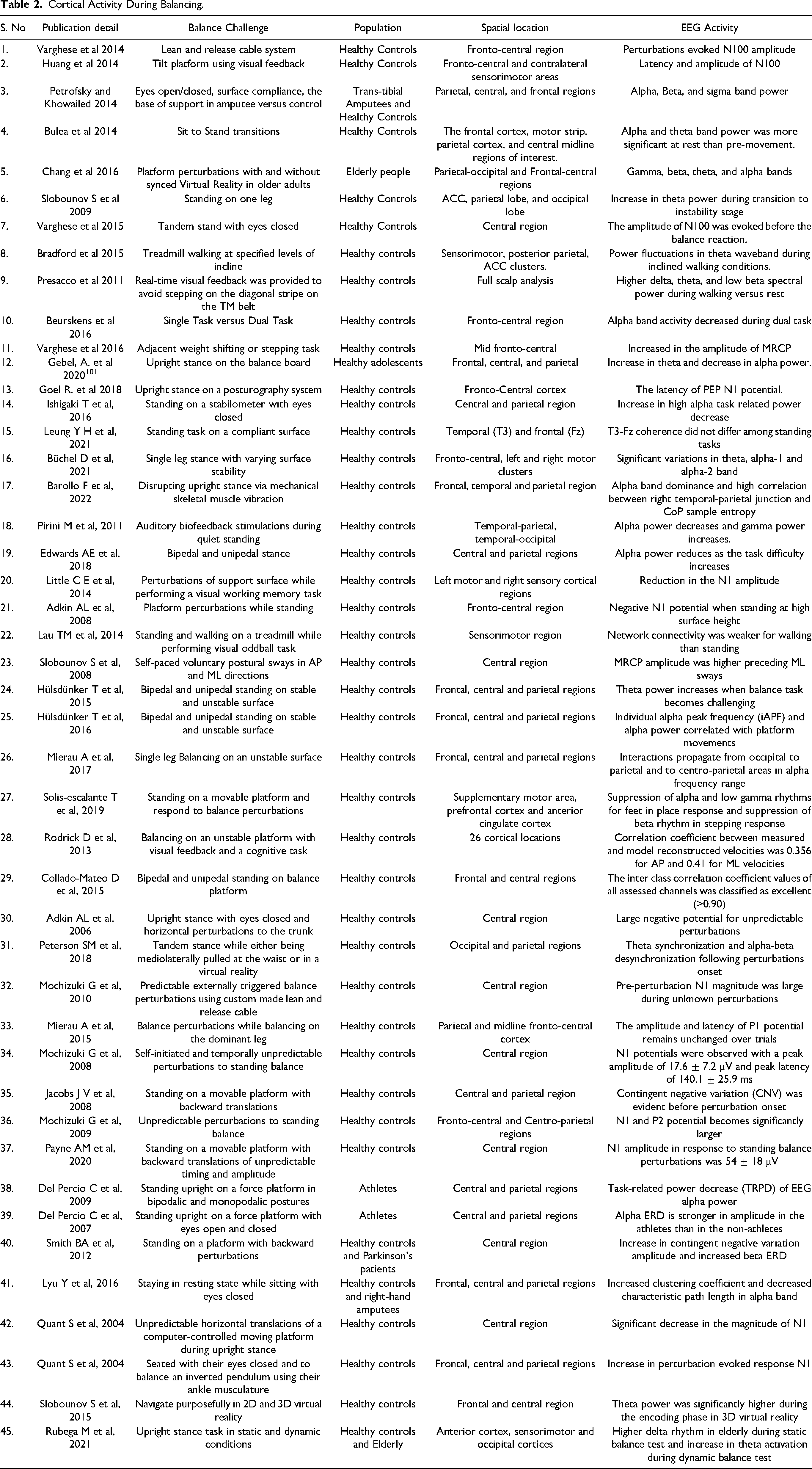
The above literature highlights the cortical regions involved while performing different balance tasks. The inputs from these activated areas can be considered of high importance in developing neuroprostheses and performing effective rehabilitation for amputees and subjects suffering from neurodegenerative disorders. Some neuro-feedback strategies can also be developed which aim at improving the postural performance of patients (Amputees, Parkinson’s and stroke) by enhancing the flexibility of highlighted cortical areas via targeted cognitive training or local stimulation techniques. This may also contribute to the reduction of training time for the efficient rehabilitation of patients. One of the most crucial requirements of restoration is to regain the balance of the patient's body. Variables like power spectral density, the frequency of various EEG bands, phase synchronization, event-related potentials, etc, have been figured out in the above discussed studies to investigate the cortical correlations during balancing and summarized in Table 2.
Cortical Activity During Balancing.
Identification and Removal of Movement Artifacts
In this section, the method for the identification and removal of movement artifacts is discussed. The EEG modality is sensitive to head and body movements, introducing movement artifacts into the recorded brain signals. Although it is expected to have sudden movements during walking and maintaining balance to introduce movement artifacts into the acquired EEG signals, to identify the exact cortical dynamics involved in locomotion and balance control, movement artifacts must be appropriately detected and removed. Therefore, it is essential to locate the artifact removal techniques and methods used in locomotion and balance control studies.
Several authors have been working in the area of advanced signal processing techniques for biomedical applications in recent times. Using the template regression artifact removal process, the event-related potential was recognizable during running on a treadmill. 101 However, it was found nearly identical during walking before and after movement artifact removal. Weighted phase lag index methodology has been used across all EEG channels and recovered P300 response to a stimulus while the subjects walked on a treadmill. 59 A low correlation was found between EEG and accelerometer signals while walking on the treadmill, and it was suggested not to use the uniform filtering method across all scalp electrodes because the signal correlation varied for each of the three-electrode locations (Cz, Oz, T8). 45
Furthermore, there was variation in movement artifact at different electrode locations, walking speeds, and across various subjects. In contrast, no large spectral amplitude spikes were found corresponding to stepping frequency obtained from accelerometer signals. 47 A strong coherence was also observed using the wavelet coherence analysis between delta band EEG signals and accelerometer signals for higher walking speeds suggesting the occurrence of movement artifacts. Here, the artifacts subspace reconstruction method has been used to remove the artifacts which appear at higher walking speeds. 47 A mathematical tool called ICA is used to separate a signal into its statistically independent components. 48 This method was used along with dipole fitting technique, which accurately localizes 99% of the independent components originating outside the brain to localize the movement artifacts. The residual 1% of independent components remained in the signal, giving more opportunities to remove movement artifacts during walking. The conventional ICA or adaptive mixture ICA (AMICA) is used to identify and remove movement artifacts from EEG signals collected during walking or balancing. The muscular and ocular artifacts have been identified and removed based on frequency spectrum and time course components using ICA.19,79 A source localization algorithm for dipole fitting (DIPFIT), has also been used by several studies after performing ICA to refine artifact identification and removal. Each independent component has been evaluated for location within the head model to estimate the residual variance between the scalp projections. 102 Movement artifacts were identified in each independent component using a performance-based method that utilizes the participant’s stepping frequency, derived from a forehead-mounted accelerometer. 103 A template correlation rejection method was also used to identify and reject EEG channels and independent components carrying motion-related artifacts. 104 The template of amplitude patterns was created, and the fraction of total epochs presenting relevant correlation to the template were determined. A novel artifact removal method named constrained ICA with online learning (cIOL) was presented. 105 It was shown that cIOL has the highest accuracy in removing movement artifacts and provides highest signal to noise ratio among the state of art methods. During a cognitive balance task in virtual reality, a movement artifact suppression procedure involving wrap correlation filter effectively minimizes correlation with the movement data for all channels and improves the overall classification performance. 106 An adaptive signal denoising method was shown recently to supress the motion artifacts from EEG signals. 107 A hybrid signal denoising framework which comprises modified empirical mode decomposition and an optimized Laplacian of Gaussian filter was proposed. The results revealed that the proposed algorithm outperforms the existing EEG signal denoising algorithms for the suppression of motion artifacts. Many authors have also used the principal component analysis to identify and remove the movement artifacts.16,47,65 Lastly, a recent study provided recommendations for the use of various artifact removal methods depending on the intensity of the movement during the locomotion tasks. 108 Table 3 summarizes the artifact identification and removal methods used by various EEG studies.
Artifact Removal Methods.

Conclusion
In this review, the role of the EEG system in the study of brain dynamics especially during balancing and locomotion activities is discussed. It is inferred that the amount of cortical activity during balancing and locomotion in different brain regions depends entirely on the complexity of the task, intervention in sensory inputs, and cognitive dual-tasks. Such assessments could have a wide range of neurological gait rehabilitation functions and can help in developing the brain-computer interface to control lower limb neuroprostheses for the patients. From this review, it is clear that the use of EEG in the human balance and locomotion control field is still in its initial stages. There are still many chances to learn more about the brain networks that control human balance and locomotion, as well as to apply these systems to problems that are actually mobile and encountered in everyday life. Several researchers investigating the neuroimaging modalities using EEG have an apparent possibility to couple this modality with sensory, mechanical, or cognitive difficulties to further investigate the cortical dynamics. In future, using multiple neuroimaging modalities, a deeper understanding of the temporal and spatial dynamics of brain activity in balance and locomotion control can be developed. For example, pairing fMRI and EEG modalities, could take advantage of each modality's spatial and temporal resolution and offer new insights into the brain activity. Engineers should focus on the development of mobile neuroimaging hardware including design elements that will dampen the noise introduced in the signal by the user movement. Furthermore, design engineers should be conscious of human factors elements, such as hardware usability and user satisfaction for all user groups, throughout the design process. Lastly, mobile neuroimaging acquisition systems need to be benchmarked, particularly in terms of repeatability, signal quality, and user satisfaction of the various hardware and software systems.
Footnotes
Abbrevations
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Engineering Research Board, (grant number ECR/2016/001282).