Abstract
Stimulus induced repetitive periodic or ictal discharges (SIRPIDs) are a commonly observed EEG pattern in critically ill patients. However, the epileptic significance of SIRPIDs remain unclear. We identified and reviewed 55 cases with SIRPIDs according to the ACNS criteria. SIRPIDs occurred after standardized painful stimuli during a standard 20-minute EEG. These cases were investigated regarding their relation to non-convulsive status epilepticus (NCSE) according to Salzburg Consensus Criteria and in-hospital mortality. In 37/55 patients (67.3%), SIRPIDs were associated with NCSE. In most patients (26/37 cases, 70.3%) with concurrent status epilepticus, SIRPIDs occurred after status epilepticus (on average 4.8 days later), but in 3/37 patients (8.1%) they were observed before a later status epilepticus. In four cases (4/37 cases, 10.8%), SIRPIDs appeared both before and after an episode of NCSE and in other four cases the two patterns coexisted in the same EEG. In 50% of the patients, status epilepticus was refractory, super-refractory or the patient died before its resolution. The overall mortality in the cohort was high at 58.2%. These findings corroborate the hypothesis that SIRPIDs might represent a state with increased epileptogenic potential, commonly co-occurring with NCSE. Furthermore, SIRPIDs are associated with therapy-refractory course of status epilepticus and high mortality.
Introduction
Stimulus induced rhythmic, periodic, or ictal discharges (SIRPIDs) are a frequently unrecognized electroencephalographic finding in critically ill patients.1–4 SIRPIDs are defined by the American Clinical Neurophysiology Society as periodic or rhythmic electroencephalographic discharges “reproducibly brought up by an alerting stimulus”. The stimuli can be induced by acoustic, tactile and any other noxious or non-noxious stimulation and SIRDIPs can appear independently from clinical arousal. 5 The EEG shows typically high amplitude self-limiting discharges that can resemble the electrographic pattern of a non-convulsive epileptic status.
The neuronal networks involved in the pathogenesis of SIRPIDs are unknown, but an underlying pathological cortico-subcortical dysregulation, associated with diffuse cortical hyper-excitability, has been suggested.6,7 Conversely, the influence of vigilance on epileptic activity in EEG recordings is a well-known phenomenon.8,9 In particular, phases of transition between different states of consciousness are susceptible to an enhancement of epileptic ictal or inter-ictal phenomena. For example, epileptic spikes are often associated with sleep wake transitional states (sleep onset and awakening). A similar mechanism might play a role in the emergence of SIRPIDs, considering its association to stimulation and change of vigilance states.
Since their first description in 2004, the significance of SIRPIDs and their epileptogenic potential has been debated.
10
Many experts relate the pattern to the “ictal-interictal continuum”—
In summary, these studies suggest that SIRPIDs are a pathological reaction to alerting stimuli, which appear in critically ill patients who frequently experience clinical or electroencephalographic epileptic activity over the course of the acute disease. However, the association of SIRPIDs with status epilepticus and its prognostic value in patients treated in an intensive care unit (ICU) remains unclear. Based on the clinical observation that many ICU patients with SIRPIDs also show EEG patterns of status epilepticus with similar frequency and morphology, we hypothesize that SIRPIDs might represent a state with increased epileptogenic potential. The primary aim of our retrospective, case controlled analysis is to assess the association between SIRPIDs and NCSE in our cohort. As a second endpoint, we assessed in-hospital mortality in the cohort showing SIRPIDs.
Materials and Methods
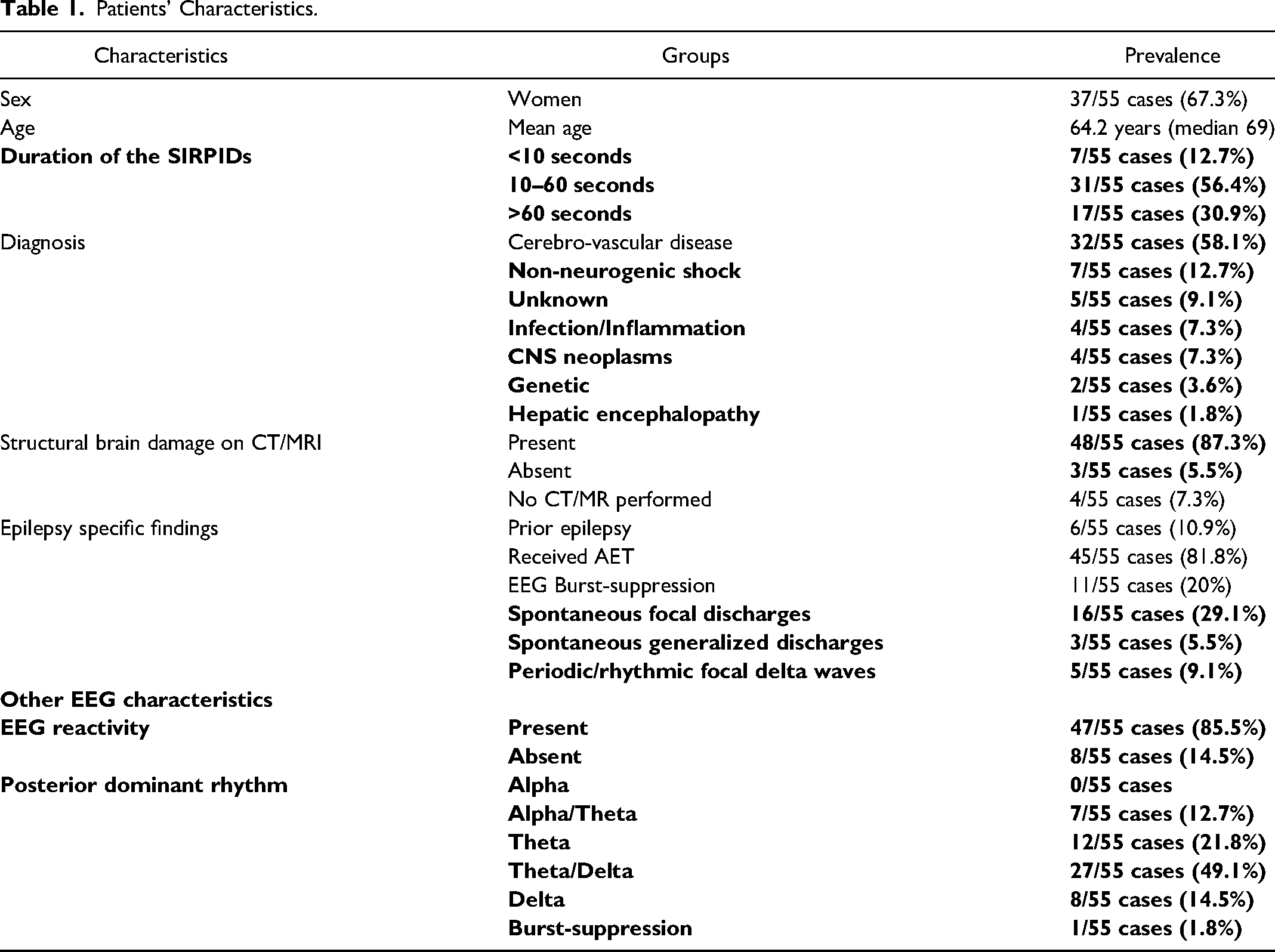
We screened our clinical database in the period from 1.1.2007 to 31.12.2020 for ICU patients who received at least two 20-min standard EEGs in our tertiary care center in a state of reduced vigilance. In total, we identified 67 patients with reproducible stimulus related EEG effects. These cases were then visually reviewed for electroencephalographic diagnosis of SIRPIDs according to criteria of the American Clinical Neurophysiological Society 5 (Table 1: Patients’ Characteristics). A typical EEG with SIRPIDs is shown in Figure 1. Blinded review of these EEGs by two epileptologists (FC, LI) showed agreement in 55 patients (37 women, 67.3%, mean age 64.2 years). We excluded the 12 cases without agreement from further analysis. In the excluded EEGs, the time-locked appearance of the discharges after application of the stimulus could not be confirmed or a precise analysis of the EEG examination was not possible due to artifacts. Furthermore, we assessed the duration and the frequency of the SIRPIDs, as well as the electroencephalographic reactivity (changes of the EEG background from baseline after stimulation other than SIRPIDs), the dominant EEG background rhythm and the presence of other pathological discharges for all patients.
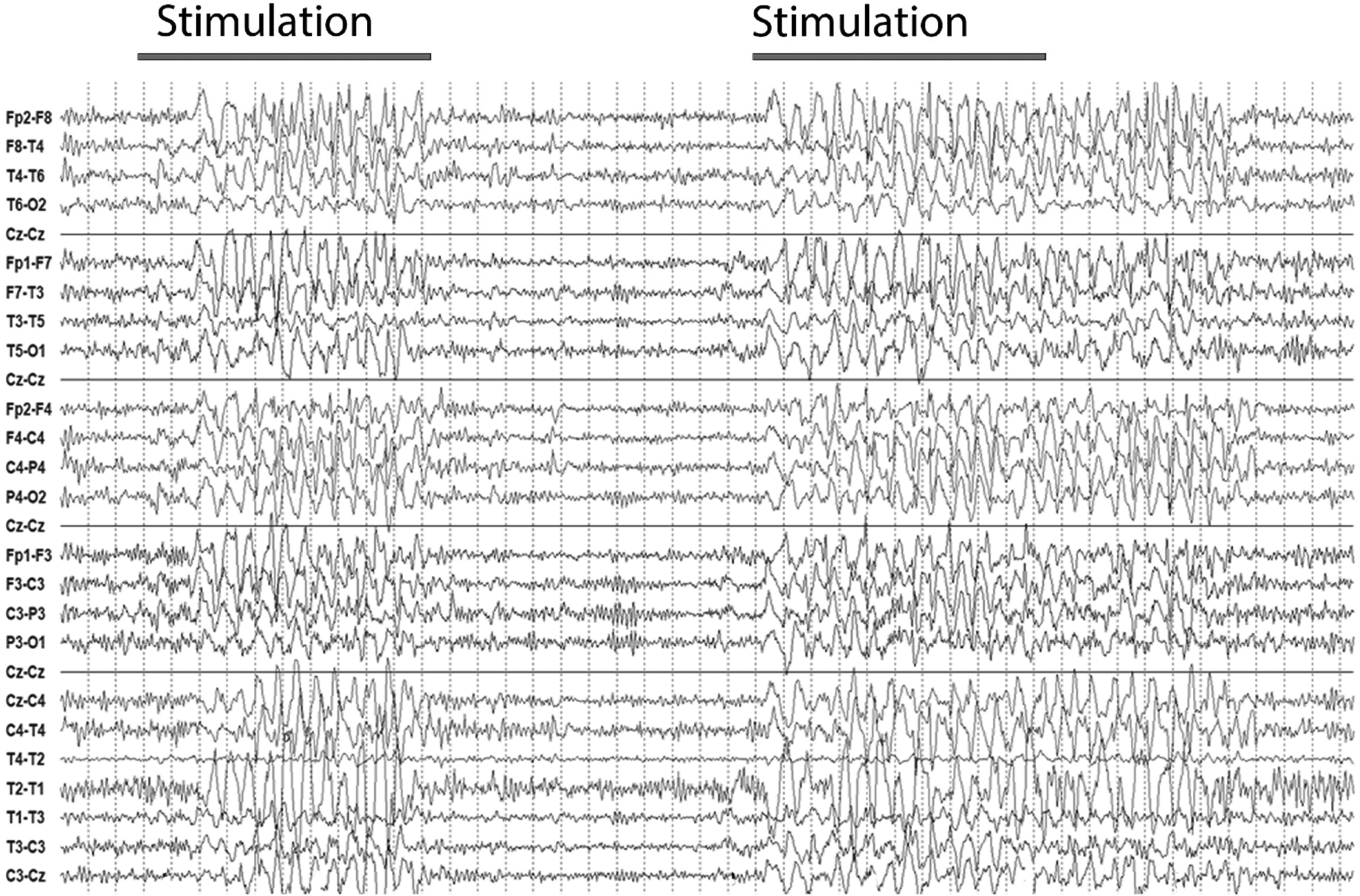
EEG over a time span of 50 seconds. Two episodes of SIRPIDs after stimulation on the right and left side. In particular, rhythmic and incompletely generalized sharp-and-slow- waves with a bifrontal maximum can be observed for respectively
Patients’ Characteristics.
All EEG recordings except one were acquired in an ICU setting according to the 10/20 placement system (23 electrodes, recording time at least 20 minutes, sampling rate 200 Hz, off sedatives). One EEG showing SIRPIDs was recorded in the general neurological ward while the patient was still in a reduced vigilance state. As an alerting stimulus, we applied standardized painful stimulation of the fingernail as part of the standard EEG examination for all patients with reduced quantitative level of consciousness in our center. The application of the painful stimulus occurred after registration of at least ten minutes of EEG without stimulation. The stimulus was applied bilaterally with an interval of ca. 30 seconds between the two sides.
We investigated this cohort of
As a control group, we selected 40 consecutive patients from a neurosurgical ICU between 2018 and 2020 who met following criteria: (1) Soporous or comatose state (2) ICU stay >3 days (3) at least two standard EEG performed during the hospital stay 4. Known cerebral pathology. The only exclusion criterion was the presence of SIRPIDs. In this group (22 women, 55%, mean age 63.7 years), we assessed the prevalence of status epilepticus and the in–hospital mortality, which we compared with those in the
The local ethics committee approved the study, all patients or legal representatives gave informed consent. However, this consent did not include a provision stating that individual data can be made freely accessible.
Results
We found episodes of NCSE in the majority of patients (37/55 cases, 67.3%) preceding or following the EEG diagnosis of SIRPIDs (Figure 1(A)).Four more patients who did not experience NCSE showed clinical seizures or electroencephalographic epileptic seizure patterns during their hospital stay (4/55 cases, 7.3%). In the control group, the prevalence of NCSE was 22.5% (9/40 cases) and the difference was statistically significant (odds ratio (OR) 7.08 [2.7889–17.9750], p < .0001).
Next, we analyzed the temporal relationship between the occurrence of NCSE and SIRPIDs. In most
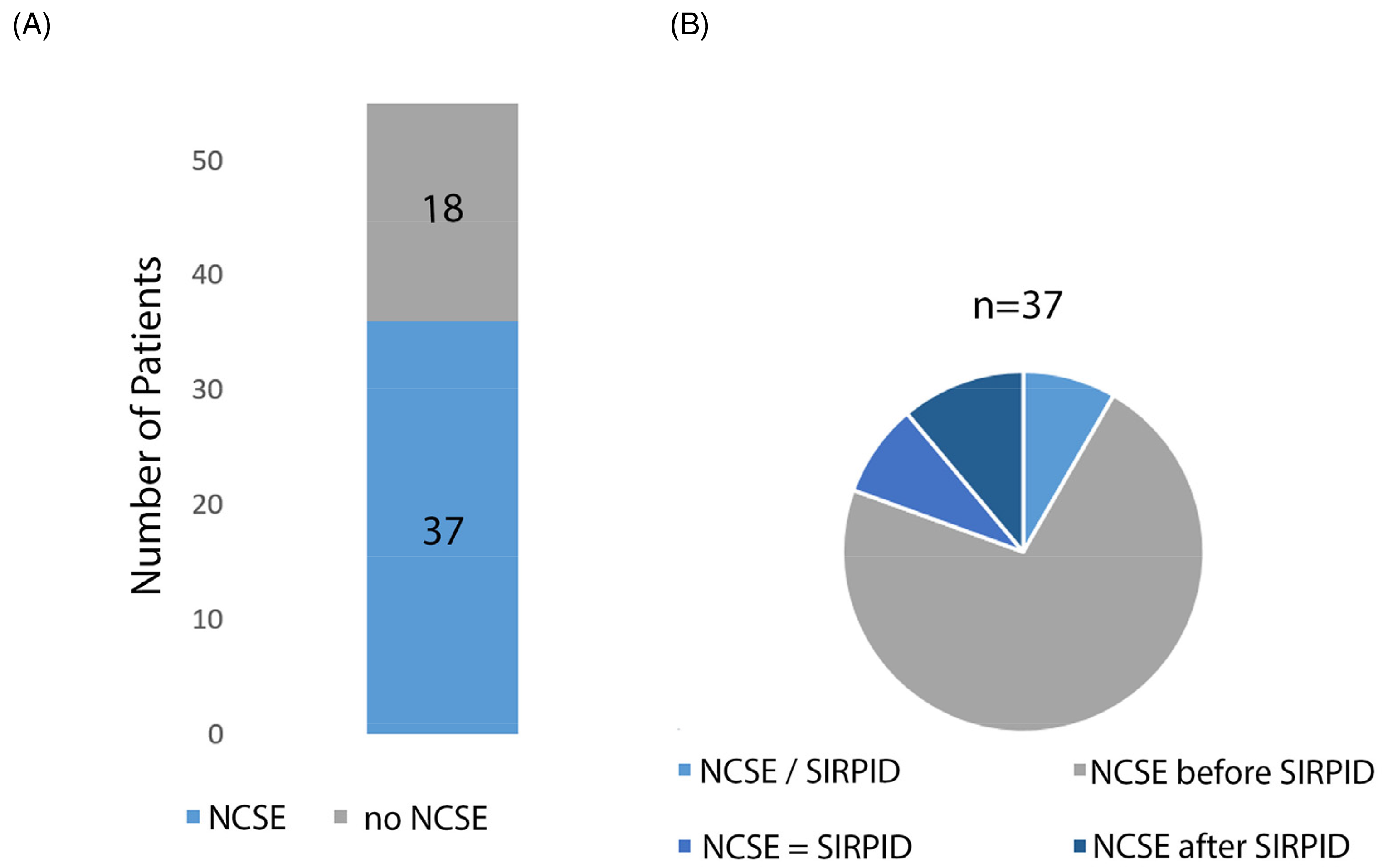
(A) prevalence of NCSE in our cohort of patients with SIRPIDs is represented (dark blue). (B) Patients are divided in four subgroups: NCSE occurring before SIRPIDs (
In the analysis of seizure responsivity based on the clinical medical records, we found that NCSE was
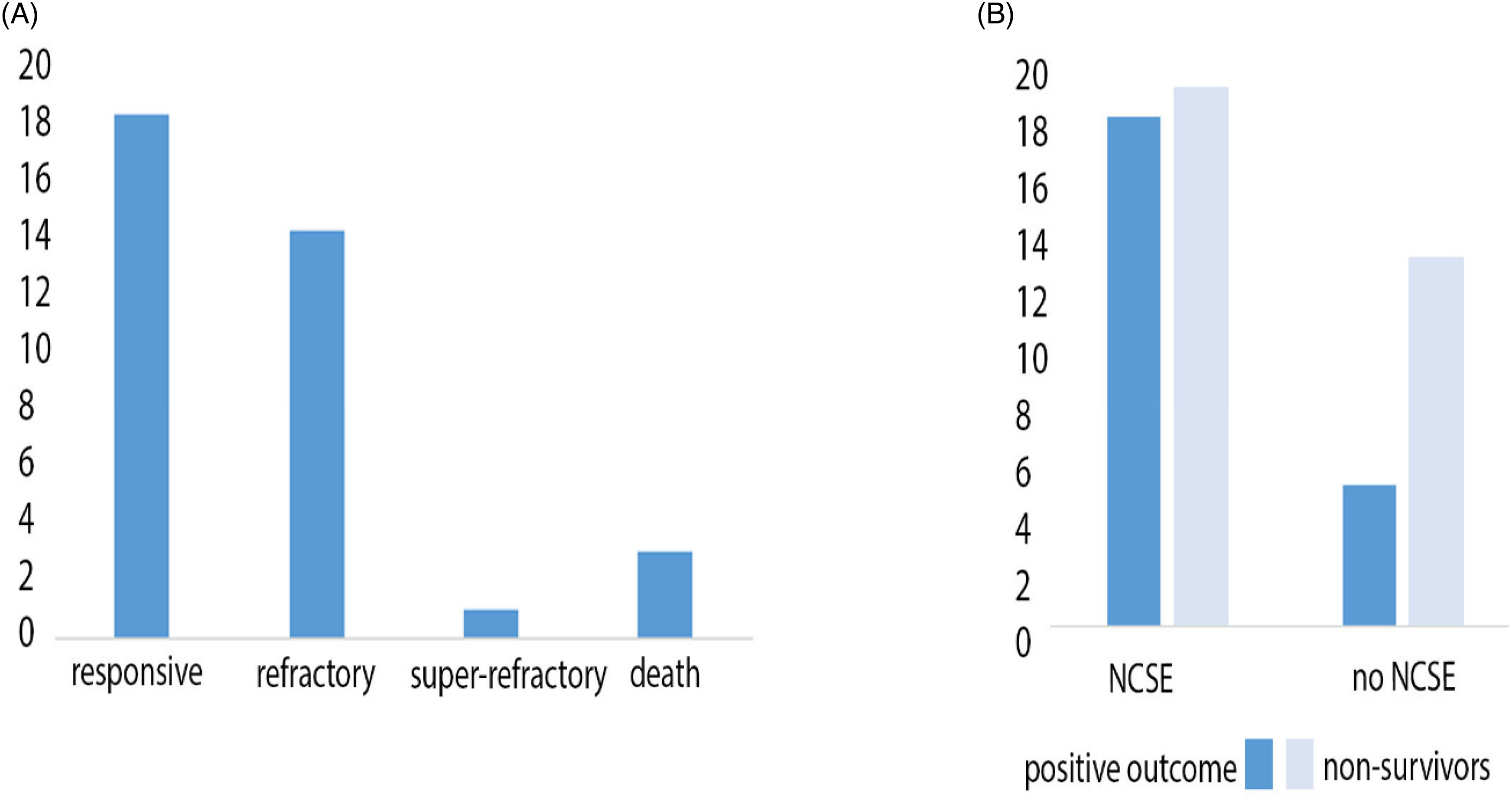
Treatment response and mortality in patients with and without NCSE. (A)
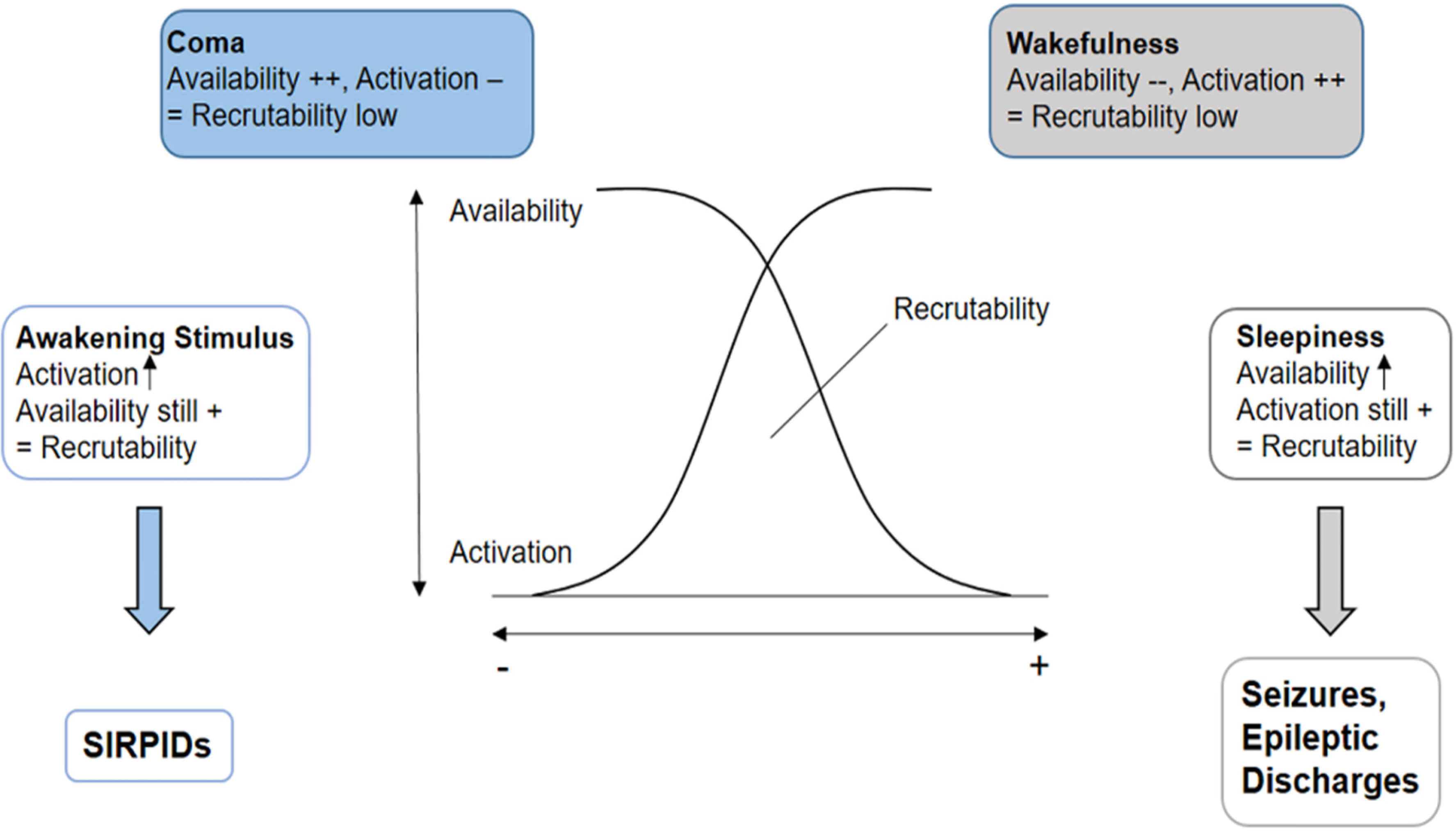
Modified graphical representation of the “recruitability theory” by Speckmann 22 to explain the raised epileptogenicity of changes of vigilance states. On the right side of the graphic, neuronal recruitability increases (gray arrow) during the transition from wakefulness to light sleep due to the raise of the neuronal availability and persistence of the neuronal activation typical of wakefulness. In contrast, on the left side of the graphic, a raise of the recruitability can be observed (blue arrow) if neuronal activation increases, as deep coma is characterized by a very high availability of neuronal cells. We suggest that the activation caused by the awakening stimulus could lead to a transient elevation of the recruitability and, consequently, to SIRPIDs.
IV midazolam was administered in sixteen cases during the EEG examination. All except one patient showed electroencephalographic responsivity of the SIRPIDs in terms of suppression of the rhythmic activity (15/16 cases, 93.8%), while no clinical response was observed.
Review of the available imaging data showed structural brain damage in all but four patients who received brain imaging (48/51 cases, 94.1%). In four cases, no neuroimaging was performed. Cerebro-vascular pathology was the most common cause of hospitalization in both
The mean duration of SIRPIDs was 85 seconds ± 118 seconds, with great variability from a minimum of five seconds to a maximum of ten minutes. In only seven patients, SIRPIDs lasted less than 10 seconds (7/55 cases, 12.7%). In most cases, the duration was between ten and sixty seconds (31/55 cases, 56.4%), while in seventeen cases SIRPIDs lasted more than one minute (17/55 cases, 30.9%). Moreover, we compared the mean duration of SIRPIDs in patients with or without status epilepticus (83.5 ± 126.9 seconds vs 88.2 ± 103.1 seconds, t = 0-137, p = .89) and between non-survivors and survivors (92.4 ± 112.6 seconds vs 74.9 ± 124.6 seconds, t = −0.535, p = .59). Direct pairwise statistical comparison did not reveal any significant difference between the subgroups. The frequency of rhythmic discharges in SIRPIDs was between 1/s and 2/s in all but one case, where we observed particularly slow periodic discharges with a frequency of 0.5/s.
Most patients showed a preserved EEG background reactivity to painful stimuli other than SIRPIDs (47/55 cases, 85.5%). There was no significant difference between patients with (32/37 cases, 86.5%) or without NCSE (15/18 cases, 83.3%) or patients who died (26/32 cases, 81.2%) or not (21/23 cases, 91.3%).
We classified the posterior dominant rhythm in alpha, alpha/theta, theta, theta/delta, delta or burst-suppression. In most cases we observed a mixed theta/delta activity (27/55 cases, 49.1%), corresponding to a moderate slowing of the background activity. In about one third of the patients, we found mild background slowing with isolated theta (12/55 cases, 21.8%) or admixture of alpha/theta activity (7/55 cases, 12.7%). A severe, delta slowing was present in eight patients (8/55 cases, 14.5%) and the last patient had a burst-suppression pattern. We found no significant differences between the subgroups. Finally, we looked for the presence of spontaneous focal or generalized, ictal or interictal discharges or periodic/rhythmic patterns. Focal discharges were the most frequent finding (16/55 cases, 29.1%), while we found generalized triphasic waves only in three patients (3/55 cases, 5.5%). In five more patients we observed spontaneous focal periodic or rhythmic delta waves, mostly bifrontal (5/55 cases, 9.1%). Again, there was no significant difference regarding our primary and secondary outcome variables among the subgroups.
Discussion
In this study, we examined the prevalence of NCSE in an ICU cohort of
Up to
From a neurophysiological point of view, we support the interpretation that SIRPIDs might be an equivalent of NCSE in deep unconscious patients due to brainstem and thalamic dysfunction or pharmacological intervention, as proposed by Hirsch et al.
5
A possible explanation for this is based on the increased epileptogenicity during changes of vigilance states, according to the “recruitability model” proposed by Speckmann in 1986.
22
In this model, neuronal recruitability (= the probability of a neuronal cell or a neuronal circuit to be recruited by an epileptic focus) depends on the state of activation of cortical neurons and on their availability, which reflects the number of neuronal cells which could potentially be recruited by an epileptic focus at a certain time. On the one side of the spectrum, active wakefulness is characterized by high activation of cortical neuronal cells, but low availability, as most neurons are already processing the constantly incoming afferent information. The opposite happens in coma, where cortical activation is at its lowest, which leads to low recruitability despite the high number of available neuronal cells. Thus, both conditions are not favorable for the
This mechanism could also explain the higher mortality in cases who did not experience status epilepticus. In particular, the deeper level of unconsciousness of these patients, which does not allow the
We recognize that this study has several limitations, such as retrospective, non-standardized patients’ selection based on EEG pattern or different underlying brain pathologies, which probably had an influence on the mortality data in our cohort. Thus, the retrospective nature of the study does not allow to establish a causal relation between SIRPIDs and NCSE. Furthermore, the lack of a standardized treatment protocol in subjects with or without NCSE does not allow us to draw conclusions on the optimal therapeutic approach in patients with SIRPIDs. It remains an open question, whether the pragmatic initiation of an
Footnotes
Author Contributions
Francesco Capecchi collected, analyzed and interpreted the data and wrote the manuscript. Andrea di Giacopo partecipated to the collection of the data, especially those contained in Table 1. Emanuela Keller was involved in the interpretation of the data and revised the manuscript for content and form. Ian Mothershill was involved in the interpretation of the data and revised the manuscript for content and language. Lukas L. Imbach analyzed and interpreted the data and revised the manuscript for content and form.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
