Abstract
Cerebral palsy (CP) is a movement and posture disorder often accompanied by cognitive difficulties which can be assessed using event-related potentials (ERPs), an often-overlooked tool in this population. Here we describe our assessment protocol, examine its feasibility, and validate the use of single-subject ERP analyses in adolescents and young adults with CP, an analysis approach which recognizes the heterogeneity of the clinical population. This study involved a final sample of 9 adolescents/young adults with CP participating in the “MyStory” study (age range 16-29 years, Mage = 25.0 years; 6 female; Gross Motor Function Classification System level I [n = 4], II [n = 2], III [n = 1], IV [n = 1], and V [n = 1]). ERP components were elicited over medial prefrontal and central cortex (error- and correct-related negativities [ERN/CRN], error-positivity [Pe], N100, P200, N200, P300), as well as those generated over occipital cortex (P100, N170). Group and single-subject ERP statistics were computed for ERPs recorded over both areas. Using recently developed data analysis methods (independent components analysis and robust bootstrapped single-subject statistics), we measured the number of participants demonstrating significant condition differences at the timing of each ERP component of interest. We demonstrate good validity for ERPs recorded during 2 of our 3 tasks eliciting frontal activation (eg, 4 of 6 participants with usable data showed a significant single-subject medial frontal negativity condition difference in a context-switching task) and good validity for ERPs derived from a task engaging occipital regions (eg, 8 of 9 participants each showed a significant N170 face-object condition effect).
Keywords
Cerebral palsy (CP) is an early originating disorder involving injury to the developing brain, resulting in movement and posture difficulties 1 often accompanied by cognitive challenges. 2 Much existing neurophysiological research on CP focused on associating magnetic resonance imaging (MRI) and functional MRI measures of brain structure and function to clinical correlates of the condition 3 and treatment outcomes (eg, constraint-based, virtual-reality therapies4,5). An additional way to examine cortical function in CP is to use electroencephalography (EEG) and concomitant event-related potentials (ERPs).
ERPs are not a common tool to study cognitive processes in CP; although, a few groups have taken advantage of the superior temporal resolution of ERPs relative to MRI. Two groups examined the cognitive load associated with motor planning and execution in children with CP and comorbid developmental disregard (DD) using ERPs—with a focus on understanding the factors underlying DD for the affected upper limb.6,7 Maitre and colleagues 8 examined somatosensory processes (eg, ERP responses to air-puff stimuli), and van Elk and colleagues 9 examined ERPs associated with reaching, grasping, and rotational movements 9 and motor imagery 9 in CP populations. Byrne and colleagues 10 examined the receptive vocabulary of a single participant using ERP paradigms that do not require an overt response, and Maitre and colleagues 11 utilized this same paradigm to study receptive vocabulary following intervention. Relatively fewer groups have examined ERPs associated with higher-level cognitive processes such as attention control, error detection, and novelty detection (but see Hakkarainen et al12-14)—processes outside of the motor and somatosensory domain that rely strongly on the prefrontal cortex (PFC). Only one recently published study examined ERPs elicited over temporal-occipital regions during an emotional face processing task. 15
One reason that ERP research in this population is sparse is because CP is an impairment in movement or posture that can result in muscle activation disturbance, and the EEG signal is extremely sensitive to muscular electrical activity. Individuals with CP often demonstrate abnormalities in postural control and neck muscle activation, 16 along with more widespread muscle contractures, and both may interfere with clean EEG data collection. Consequently, researchers aware of the need for clean data may have avoided sampling CP populations, particularly those with severe motor impairments. Except for Belmonte and colleagues, 15 the above ERP studies failed to include participants with a Gross Motor Function Classification System (GMFCS) level greater than III (ie, participants who are nonambulatory and have significant difficulties controlling movement and posture), with some studies (eg, Maitre et al 8 ) only including those with a GMFCS level I (ie, participants who can walk and run but may have some difficulties in more advanced motor skills and sports).
By using bootstrapped group and single-subject statistics, we can determine how robust ERP effects are at the group level, and how consistent the results are across participants—a critical consideration given the heterogeneity of clinical samples. Perhaps only some participants demonstrate ERP effects, and modern, robust ERP analysis methods allow us to quantify this. Single-subject ERP analyses have been successfully used in clinical populations (eg, schizophrenia, 17 prosopagnosia 18 ), and researchers continue to encourage their use in other clinical and developmental populations.19,20 Single-subject statistics—in particular, single-subject bootstrapped statistics—have not been used in CP populations. The use of single-subject statistics reduces the need for a control group. In essence, each participant is their own control, and we can observe the degree that each participant shows condition effects, considering their variability in latency.
Determining the feasibility of collecting EEG across a range of CP severity and establishing optimal EEG/ERP data collection and analysis techniques in this population is critical. We evaluated the feasibility of administering a range of tasks beyond what had been previously done. Participants completed computer tasks that activate primary and secondary visual processing regions (eg, the face-house-checkerboard [FHC] task), and tasks known to recruit prefrontal regions (eg, Flanker, selective attention, and context-switching tasks). Replicating previously established ERP effects provides confidence in our EEG data analysis methods with this population.
Here, we provide proof of concept that EEG data can be collected from a range of adolescents and young adults with CP and cleaned of noncortical (eg, muscular) artifacts using independent components analysis (ICA), a statistical technique designed to separate latent variables from one another, 21 and follow this with an analysis of single-subject effects. We validated participants’ ability to perform the computer tasks while EEG data were collected by looking at accuracy data. We were less concerned about reaction time data given the motor skills impairments sometimes observed in CP. 1
Our goals were to describe the assessment protocol, examine its feasibility (ie, can the participants perform the tasks), and validate the use of our ERP methodologies in adolescents/young adults with CP. Our study advances previous research because of its (1) use of ICA and single-subject statistics, (2) inclusion of a diversity of GMFCS levels, (3) addition to existing literature on ERPs in CP across a variety of cognitive demands, and (4) inclusion of tasks measuring ERPs recorded over occipital lobes, which only Belmonte and colleagues 15 have done. We fully reported all collected data, regardless of the success of the effects.
Methods
Participants
This study is part of a large, prospective cohort study: “Brain-Behaviour Correlates of Health and Well-Being in Adolescents and Young Adults with CP” (short title: MyStory Study)—part of CP-NET, an Integrated Discovery Program conducted in partnership with the Ontario Brain Institute in Ontario, Canada. Ten adolescents/young adults with CP (aged 16-29 years, Mage = 24.5 years, 6 girls/women), who were recruited for the ongoing longitudinal MyStory study, were invited as a convenience sample (Table 1). The Gross Motor Function Classification System (GMFCS), 22 Manual Ability Classification System (MACS), 23 and Communication Function Classification System (CFCS) 24 were used to objectively classify participants for descriptive purposes. Each system has 5 levels that represent meaningful differences in function, mobility, manual ability, and communication, respectively. For each system, a classification is made by determining which level best represents the person’s current function throughout the day: level I (most functional) to level V (least functional).
Baseline Characteristics and Feasibility Data (Completeness of Assessments).
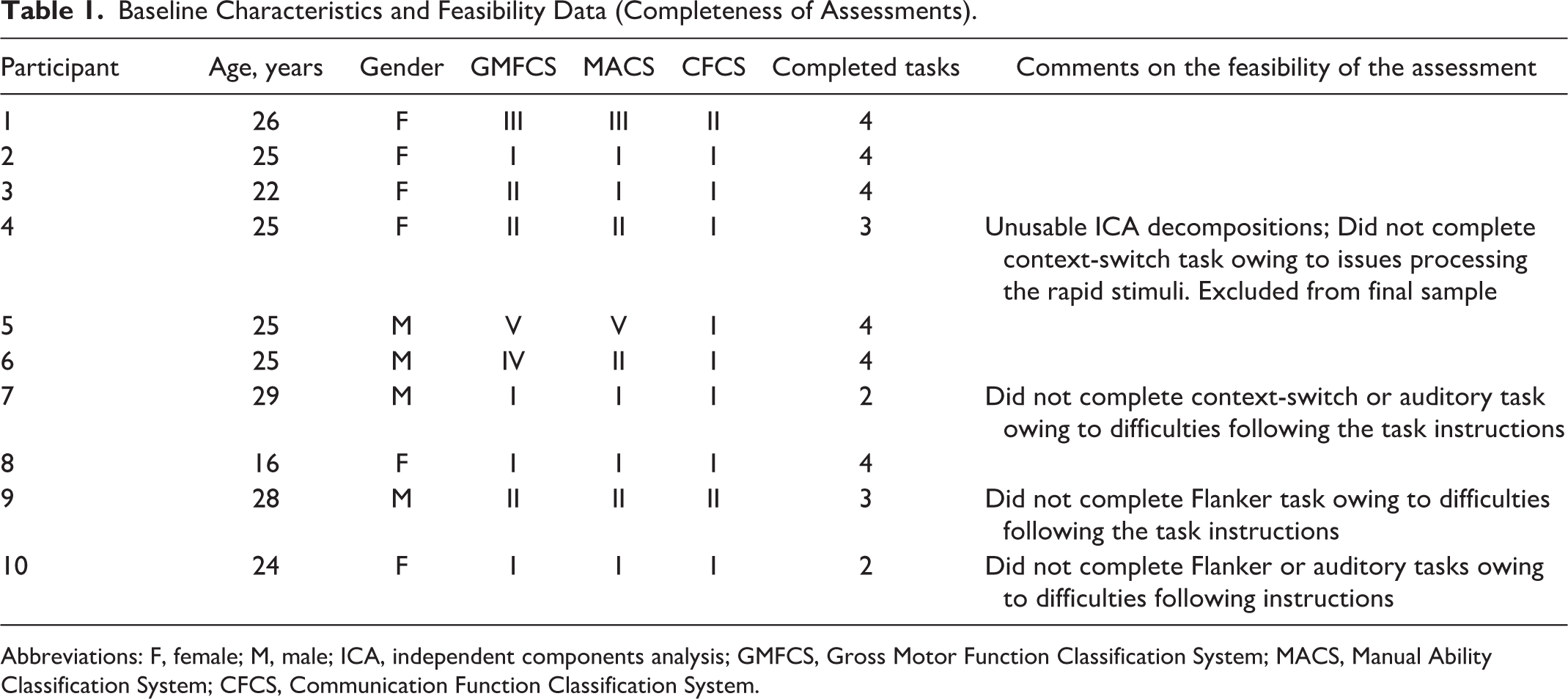
Abbreviations: F, female; M, male; ICA, independent components analysis; GMFCS, Gross Motor Function Classification System; MACS, Manual Ability Classification System; CFCS, Communication Function Classification System.
Participants were excluded if they had a low-functioning cognitive status (via self- or parental-report), could not follow simple commands, had uncontrolled seizures over the past 2 years, or had significant uncorrected visual/hearing impairments. The testing environment was described to participants and they were asked if they expected to have any difficulties performing computer tasks and responding using a modified response device. No formal assessment of cognitive or visual abilities was administered. One participant was in their final year of high school and all others had partially or fully completed postsecondary education. One participant (GMFCS level II) reported difficulties with rapidly presented visual stimuli, and therefore did not complete the context-switching task; this participant was excluded owing to poor EEG data quality, leaving a final sample of 9 participants.
Methods and Procedure
Informed consent (assent for 1 participant aged <18 years) was obtained from all participants. All participants over the age of 18 demonstrated capacity to provide informed consent (ethical clearance was obtained from the Brock University Bioscience Research Ethics Board and the Hamilton Integrated Research Ethics Board, per the Declaration of Helsinki). Participants were then fitted with a 128-channel BioSemi electrode cap along with 7 exogenous electrodes to monitor electrooculogram and facial electromyography—placed symmetrically at the zygomatic processes (2), outer canthi (2), inferior orbital bones (2), and nasion (1). Sensor coordinate maps were created for each participant (Polhemus Fastrak hardware and software). EEG was recorded at a sampling rate of 512 Hz, using a reference-free system, and electrode offsets were maintained at less than 50 µV.
Participants were seated 100 cm from a computer monitor. The response device was a modified keypad with two circular response buttons 6.5 cm in diameter. After adjusting the chair and other physical surroundings to participants’ comfort, participants completed 4 computerized tasks, presented using E-Prime software (Psychology Software Tools, Inc), while ERPs were collected: a face/house/checkerboard task, a context-switching task, a Flanker task, and a selective auditory attention task (see Supplementary Materials, available online). Each task took 4 to 20 minutes; most had short breaks at 5-minute intervals. Longer breaks were given between tasks. Participants indicated to the experimenter when they felt sufficiently rested to proceed. The whole appointment including informed consent, net application, testing, and net removal took approximately 2.5 hours.
Data Preprocessing
Data were re-referenced offline to the average of all sites, pruned to exclude any periods of off-task time (eg, breaks), any excessively noisy channels, and any linked channels (assessed via a nearest-neighbor correlation procedure 25 ); they were then subjected to an extended infomax ICA.21,26,27 ICA can be applied to EEG data to decompose electrical signals into various cortical and noncortical (noise) sources. Voltages caused by muscle contractions can be quantified using electromyogram (EMG) measurements. 28 In nonclinical contexts in which EEG data are not collected, ICA can cleanly separate EMG from electrocardiogram (ECG) data 29 ; thus, it may also be effective at removing EMG and other sources of noise from EEG data. ICA is effective in de-noising data from both simulated and real EEG data.27,30
Data were filtered (1-30 Hz) and dipoles fit using native EEGlab functions. 31 All independent components representing eye movements, heart rate, or other muscle activity were removed. Eye-movement components were identifiable by their extreme frontal topography and characteristic high-amplitude and rapidly resolving peaks. Components representing heart rate were identifiable by their extreme posterior topography (often left-lateralized) and characteristic ECG-like waveform. Topographies representing muscle activity were more heterogeneous in nature, but often contained high-frequency electrical activity, and had greater than 15% unexplained variance when the dipole model was fit (indicative of a noncortical source). All channels were then interpolated to a standard scalp montage by spherical spline (see Desjardins and Segalowitz 25 for further preprocessing details). Participants’ data are currently stored in the Brain-CODE Neuroinformatics Platform managed by the Ontario Brain Institute, designed to enable data sharing and to foster collaborations among a broad research community (https://www.braincode.ca/).
EEG Data Analysis
Significant condition differences were examined using robust analyses of variance (ANOVAs), calculated in STATSLAB software 32 —a free plug-in for MATLAB. STATSLAB employs bootstrap significance tests for main effects and interactions. The bootstrap technique is superior to traditional ANOVA techniques; that is, there is no need to make unverifiable and/or invalid assumptions about probability distributions prior to analysis.33-35 These techniques are useful in small sample sizes—when there are no expectations of normality (like ERPs)—and for quantifying effects across the entire time-course of ERPs. 25 Bootstrap techniques are robust—they provide control over type I error and an effective representation of the probability distribution. 36
For each participant, STATSLAB samples, with replacement, the total number of usable trials per condition. STATSLAB then sorts each of the sampled trials by amplitude independently at every time point. Twenty percent of the data were trimmed at each time point; the remaining data were averaged together to form a robust ERP. This process was repeated 1000 times, forming the bootstrap sample. Trimmed values were Winsorized to maintain qualitative robustness. 36 This bootstrap sample was used to compute the estimate of the test statistic (ie, the q statistic suggested by Wilcox 36 ), and the confidence interval (CI) about the difference at each data point. When the CI of the difference around main effects and/or interactions does not include zero, then a significant difference between groups and/or conditions exists. Thus, in the figures where the CI does not include zero—as indicated by the axis line becoming red—there is a significant difference effect (P < .05) corrected for family-wise error. Rom’s sequentially rejective method was used to help control type I error rates (see van Noordt et al 37 for further details on this robust estimation procedure). All stimulus-locked ERPs were baseline corrected from −200 to 0 ms before stimulus onset, and all response-locked ERPs were baseline corrected from −600 to −400 ms before responses.
First, we investigated 2 well-established ERP effects generated by the occipital cortex. The first occipital ERP effect relates to the sensitivity of the P100 and N170 to the presence of faces versus nonface objects (eg, houses). The N170 component is more negative in response to faces than to houses and the P100 is also more positive to faces, but less consistently. 25 The second ERP effect relates to a more subtle sensitivity of the N170 to upright versus inverted faces (the inversion effect), with larger and later N170s found for inverted compared with upright faces. 38 We measured these at P6—site of the group-maximal N170.
We also investigated 3 well-established ERP components elicited over medial PFC (mPFC), recorded at Cz and FCz. First, the error-related negativity (ERN) followed by a broad error-positivity (Pe), 39 as frequently elicited during a Flanker task, is a negative-going waveform followed by a positive ERP deflection in response to erroneous compared with correct responses. 40 Second, a set of ERP components derived from a newly developed task designed to activate mPFC—a context-switching task—that successfully elicits medial frontal negativities (MFNs) and Pe’s to responses. 37 We also looked for P300 effects, as larger P300s are observed to behaviorally relevant stimuli, such as a context change cue. Third, we looked for more negative N100 to purposefully attended versus purposefully unattended auditory tones in a selective auditory attention task.41,42
Results
Data Quality
We examined the quality of the data by looking at behavioral performance on the computer tasks. If participants could not perform the tasks as instructed, we could not ascertain EEG data quality. We additionally examined the quality of the ICA decompositions to assess the feasibility of using this technology with this population.
Behavioral Performance
FHC Task
Behavioral data were available for 4 of 9 participants; the remaining data were lost to technical difficulties. On average, these participants correctly responded to 87.7% (SD = 8.07%) of the 150 checkerboards included to maintain participant attention on the screen. Given the simplicity of the task (a left/right response and slow presentation rates), and the qualitative observation that no participants had difficulty performing the task, we analyzed all 9 participants’ EEG data.
Flanker Task
Performance on this task was bimodal. Participants either excelled or struggled greatly: mean accuracy on congruent and incongruent trials ranged from 3% to 100% and 28% to 100%, respectively. Thus, some participants may not have made enough errors for ERP analysis while others too many (eg, not understanding the task, task difficulty). We retained for EEG analysis data from 5 participants who made at least 12 errors on incongruent trials and were accurate at least 72% of the time on congruent trials.
Context-Switching Task
One participant did not understand the task, which was aborted. For the other 8 participants, mean accuracy on NoGo trials (ie, correct inhibitions) was 69% (SD = 24.3%), and mean accuracy on Go trials was 87.6% (SD = 10%). Six participants responded correctly to more than 40% of NoGo trials and more than 70% of Go trials accurately and were therefore retained for further analyses.
Auditory Selective Attention Task
Participants responded well (M = 60% on target trials; SD = 20%). Data were analyzed from 6 participants responding correctly to more than 55% of targets and with fewer than 10% false positive responses. One participant had messy EEG data and could not be included in the ERP analysis.
ICA Decompositions
Cortical ICs have a clean topography with circumscribed regions of activation, while ICs that represent noise may appear messy and have several segregated regions of activation, and generally greater than 15% of variance unexplained by the dipole model. We eliminated these from the data set. For one participant (GMFCS level II), we could not extract any clean cortical ICs (all had at least 15% residual variance); therefore, those data were eliminated from analyses. However, ICA decompositions were successful for 9 of 10 participants and successfully retained components representing cortical source data. Clean ICA decompositions were obtained from both participants with the poorest motor functioning (GMCFS levels IV and V).
Data Retention
Nine participants had usable EEG data and each participant completed four tasks; that is, 36 possible (9 × 4) EEG recordings. Twenty-six (72%) of these had acceptable behavioral performance and were analyzed (FHC, n = 9; context-switching, n = 6; Flanker task, n = 5; auditory task, n = 6). Excluding the FHC task from this calculation (owing to technical difficulties saving some behavioral data), there were 27 EEG recordings, and 17 (63%) of these were analyzed.
ERP Validation
FHC Task
At the group level, we observed significant ERP effects in the expected direction (ie, by using our techniques, we detected documented visual processing ERP effects). We found a significant group-level effects for the N170, but not the P100, for the face-house contrast (Figure 1). At the single-subject level, these results were robust. Eight of 9 participants showed a significant N170 ERP effect in the expected direction and 1 participant (a 16-year-old girl, GMFCS level I) showed an N170 effect in the opposite direction (larger N170 for houses rather than faces). See Figure 1c for all single-subject contrasts. Table 2 presents single-subject ERP effect data on a task-by-task basis.
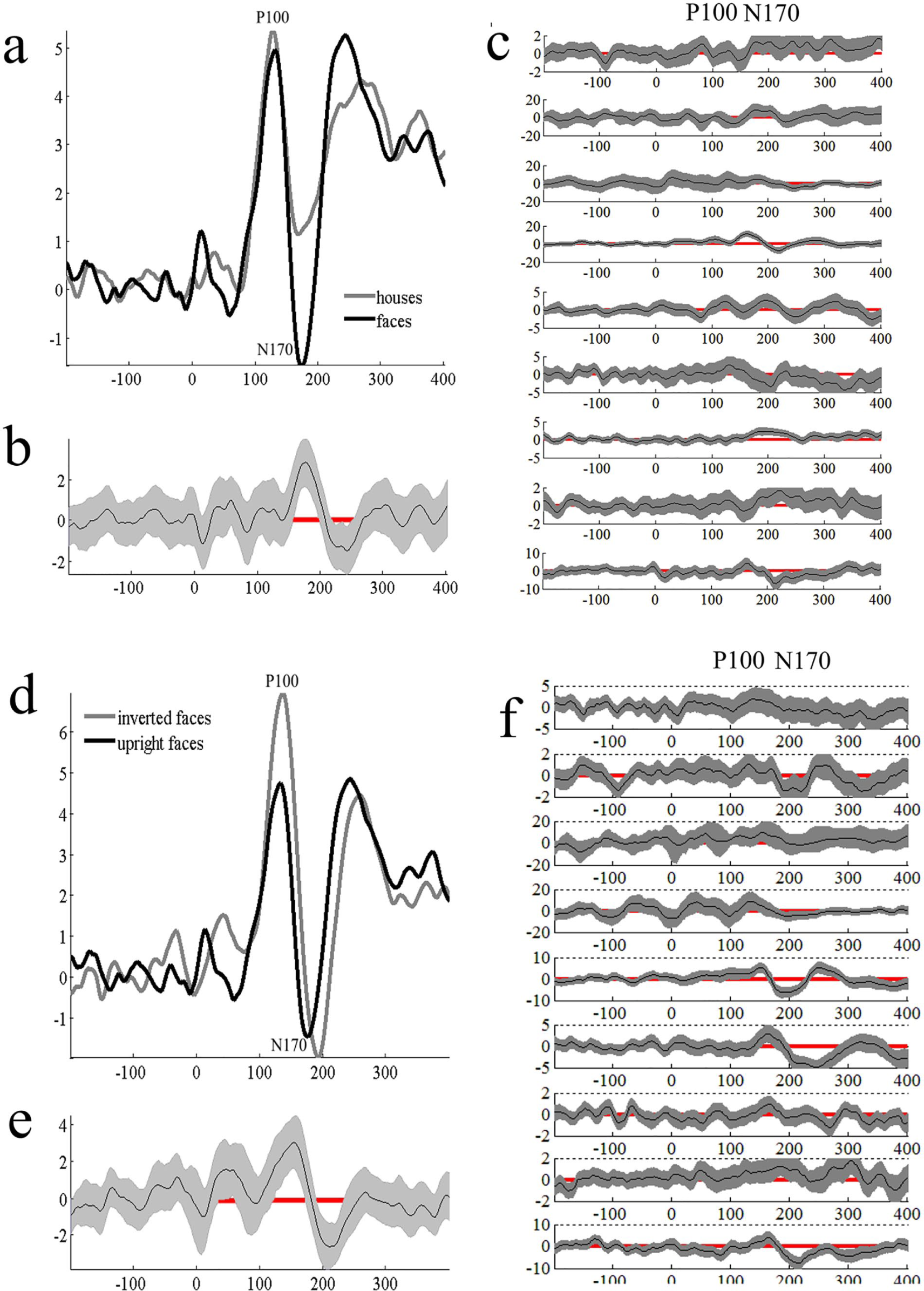
Event-related potentials (ERPs) elicited to faces versus houses at P6. Panel b shows bootstrapped significant differences for the contrast of interest. Gray envelopes represent 95% confidence intervals. When the confidence interval does not include zero (red line), significant condition differences are present. This is true for all figures in this article. Panel c shows single-subject bootstrapped condition differences to faces versus houses. Panel d shows group ERP data for upright versus inverted face contrast also at P6. Panel e shows significant bootstrapped condition differences. Panel f shows single-subject bootstrapped condition differences for upright versus inverted faces.
Results of Single-Subject ERP Analyses Presented on a Task-by-Task Basis.
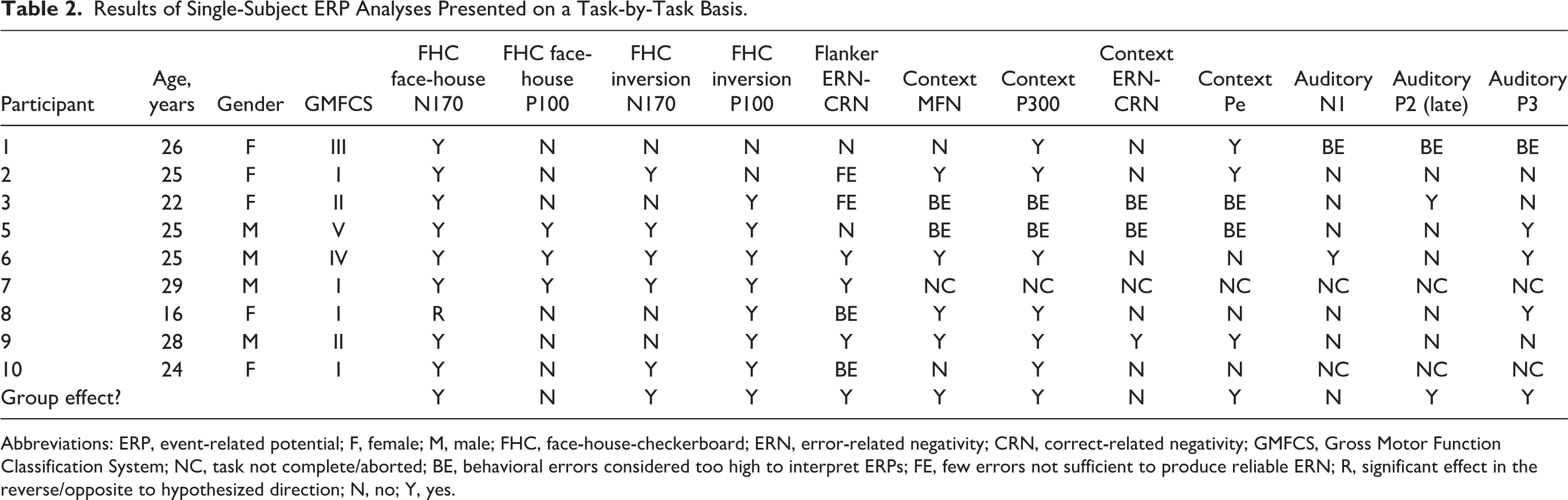
Abbreviations: ERP, event-related potential; F, female; M, male; FHC, face-house-checkerboard; ERN, error-related negativity; CRN, correct-related negativity; GMFCS, Gross Motor Function Classification System; NC, task not complete/aborted; BE, behavioral errors considered too high to interpret ERPs; FE, few errors not sufficient to produce reliable ERN; R, significant effect in the reverse/opposite to hypothesized direction; N, no; Y, yes.
We also examined the ERP data for evidence of the inversion effect. We found evidence for the inversion effect at both the P100 and N170 timing, with the N170 effect partially attributable to latency differences (Figure 1d). At the single-subject level (Figure 1f), 7 of 9 participants showed a significant P100 inversion effect, and 5 of 9 showed an inversion effect at the N170.
Context-Switching Task
Group ERP evidence for an MFN context-switching effect at the timing documented by van Noordt and colleagues 37 was found. On trials where the border color changed, we observed a more negative MFN than on Go trials without a border change. Additionally, larger (ie, more positive) P300s were observed on border change rather than Go trials (Figure 2).
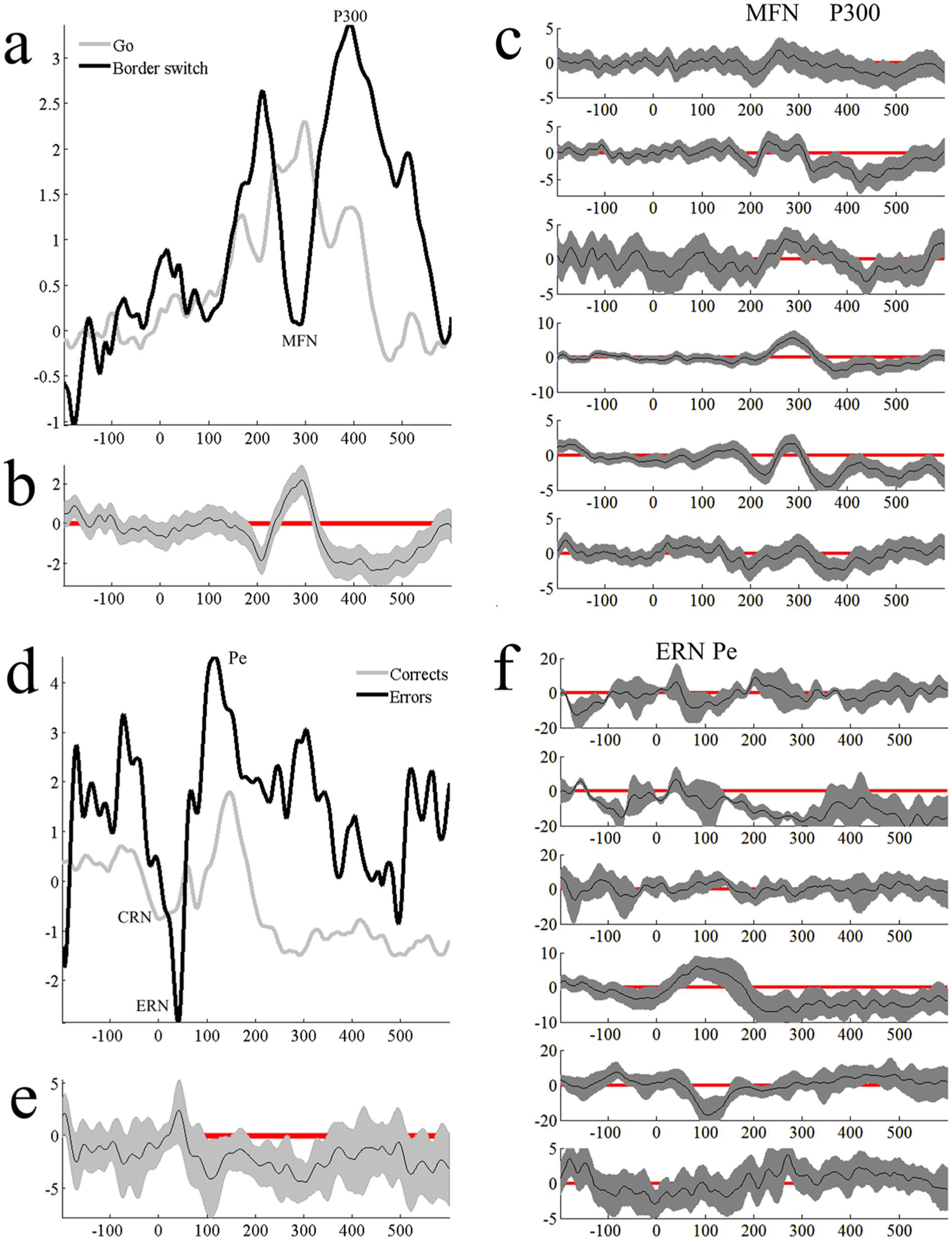
Panel a shows group-level stimulus-locked context-switching effects at FCz. Panel b shows single-subject context-switching effects. Panels d, e, and f are response-locked ERN-CRN differences for the group ERP, group condition differences, and single-subject ERN-CRN differences, respectively. ERN, error-related negativity; CRN, correct-related negativity; ERP, event-related potential.
At the single-subject level, four participants demonstrated a statistically significantly more negative MFN to border change trials than Go trials, and all six participants demonstrated more positive P300s to border change compared to Go trials (Figure 2c).
We then compared response-locked error trials to correct trials to examine the possibility of replicating ERN and CRN differences. At the group level, no significant ERN-CRN differences were observed; although, the depicted differences were in the expected direction. The ERN was followed by a series of Pes that were significantly larger for errors than corrects (Figure 2d and e).
At the single-subject level, the ERN-CRN difference was not robust, with just 1 participant showing significantly more negative responses to errors versus corrects. The Pe differences were observed among several participants. Several participants showed baseline condition differences (Figure 2f); although, this could represent variation in motor preparation and execution (ie, awareness of the error and thus the ERN occurred prior to the motor response).
Flanker Task
Five participants provided usable Flanker EEG data. A significant error-correct difference was found at the group level (Figure 3). At the single-subject level (Figure 3c), 3 of 5 participants showed the expected ERN effect; that is, more negative ERPs to errors rather than corrects, with this effect being driven mostly by 1 participant.
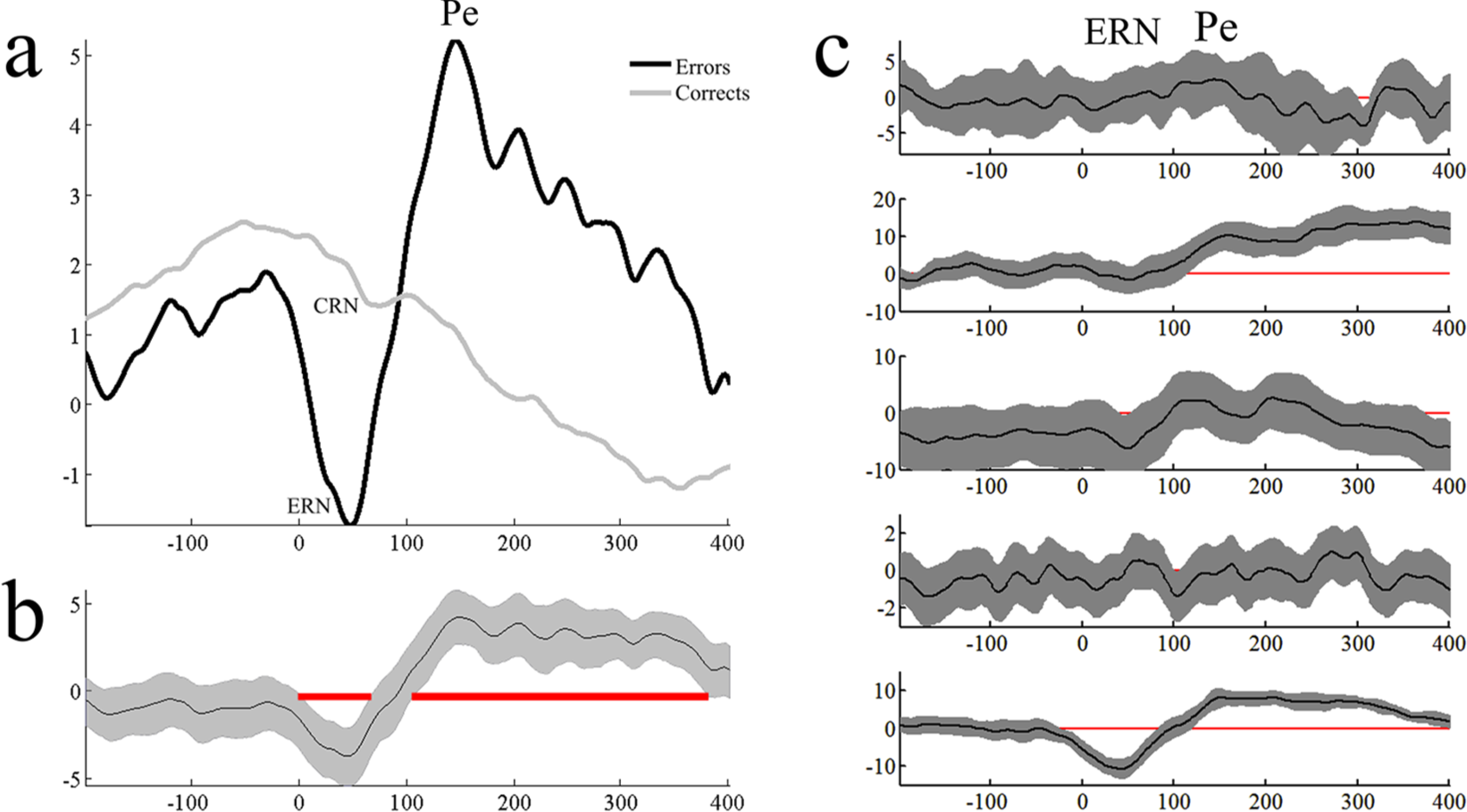
Panel a shows group-level error – correct ERPs at Cz recorded during the Fish Flanker task. Panel b shows the confidence interval around the difference and significant differences are highlighted by the presence of the red line. Panel c shows single-subject error – correct differences. The final 3 participants shown have ERN differences in the expected direction, with the effect largely driven by the final participant. ERP, event-related potential; ERN, error-related negativity.
Auditory Task
Six participants completed this task accurately and had clean EEG data. We did not observe expected N100 differences at the group level; however, late-P200 and P300 effects were observed (Figure 4). At the single-subject level, 1 participant showed the P200 effect and 3 showed P300 ERP effects (Figure 4c).
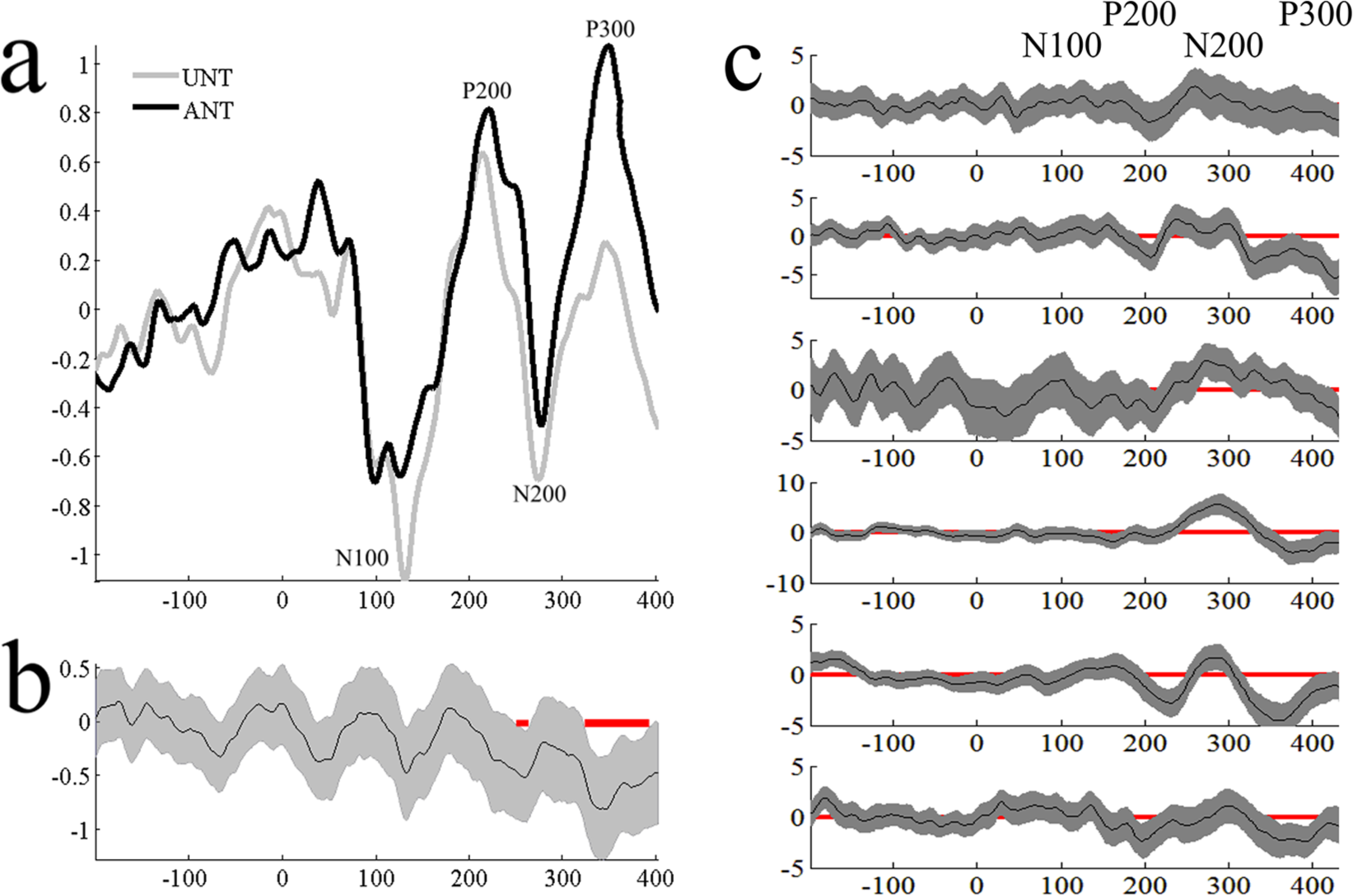
Panel a shows group auditory ERPs at FCz to ANT and UNT tones, while panel b shows times of significant differentiation. Panel c shows single-subject ANT-UNT bootstrapped confidence intervals. ERP, event-related potential; ANT, attended nontarget; UNT, unattended nontarget.
Discussion
CP can affect neurocognitive processes and ERP research can shed light on the precise nature of these cognitive impacts on adolescents and adults with CP. Relatively few research groups examined ERPs associated with higher-level cognitive processes such as attentional control, error detection, and novelty detection (but see Hakkarainen et al12-14), processes outside of the motor and somatosensory domain. As a first step in our MyStory research program, we needed to demonstrate the feasibility of recording ERPs during tasks across a variety of cognitive demands. Our results show initial promise for recording EEG and analyzing ERPs from individuals with CP across a range of gross motor functioning. ICA allowed us to clean the data of a substantial portion of muscle activity and thereby examine ERPs measured at the scalp but derived from cortical sources. The novel use of single-subject statistics allowed us to recognize the heterogeneity of this clinical population and to document individual differences in the robustness of an ERP effect within a single participant with CP (ie, to not average ERPs across groups of patients vs controls).
We investigated two ERP effects generated by the occipital cortex—the P100 and N170—to the presence of faces versus nonface objects (i.e., houses) and the N170 face-inversion effect. We investigated 3 ERP complexes measured at frontocentral sites: the ERN and Pe elicited during a Flanker task, the N100 to attended versus unattended auditory tones in a selective auditory attention task, and MFNs such as the ERN/Pe and NoGo N200. Additionally, we examined the P300 elicited during a context-switching task. We demonstrated proof of concept in 9 participants with CP by replicating some well-established ERP effects.
Regarding feasibility, clean ICA decompositions resulting identifying cortical components were achieved for 9 of 10 participants. Participants performed reasonably well on the computer tasks—most accurately on the FHC task and with modest success in tasks designed to activate mPFC. Between 63% and 72% of the EEG recordings were retained for analysis. Given the novelty of our study, we lack comparable results; however, it is not uncommon in clinical or developmental samples to exclude a modest proportion of participants for behavioral or electrophysiological reasons 43 and existing ERP studies in CP have equally small samples.8,9 Nonetheless, we are confident that, with some modifications, we may increase participant retention in future investigations.
Our main focus was to replicate well-documented ERP effects to ensure their validity in this population. We successfully replicated ERP effects for basic visual perceptual processes eliciting the P100 and N170. Eight of nine participants showed a statistically significant N170 face-effect, and we found evidence for the face-inversion effect at both the P100 and N170 timing from 7 and 5 participants, respectively. Additionally, we had some success with more prefrontally mediated ERP components. In the context-switching task, the MFN and P300 were sensitive to context changes, with 4 of 6 participants demonstrating a significantly more negative MFN to border switch trials than Go trials, and all 6 demonstrating more positive P300s to border switch rather than Go trials. In the Flanker task, a group-level error-correct difference was found for the 5 participants with usable data. At the single-subject level, 3 of 5 participants showed the expected and statistically significant ERN effect. Given the length of the testing session and that the Flanker task was the final task, we recognize that fatigue may have reduced the strength of these effects.
Six participants’ data were retained for the auditory task. We did not observe expected N100 differences at the group level, although late-P200 and P300 effects were observed. At the single-subject level, 1 participant showed the P200 effect and three participants showed P300 effects. Thus, when participants successfully completed the task, we replicated common ERP effects, except for the auditory task.
We understand these tasks may only be used on a circumscribed population of individuals with CP. We excluded participants without the ability to perform motor movements with one of their hands/wrists (ie, an inability to use our response device; note that this did not preclude the inclusion of those with GMFCS levels IV and V), those with uncontrolled epilepsy, and those who could not follow basic task instructions. Nonetheless, we could use these tasks with a relatively heterogeneous population as demonstrated by the range of GMFCS, MACS, and CFCS levels. We hope this report encourages others to collect EEG data in this population and will help improve the quality of data collected as other researchers will not have to reduplicate the efforts detailed here. Using our assessment protocol and analysis methods, EEG data can be collected from individuals with CP, including data recorded from mPFC.
In future data collection, we will allow for a greater dynamic interstimulus interval adjustment and shorter testing sessions to accommodate the heterogeneous nature of the population. We plan to change the Flanker task from one involving directionality to one not based on stimulus orientation as some individuals with CP have difficulties with directionality. 44 We will examine whether ERP effects are a function of CP severity, underlying brain etiology, comorbidities, personal stress experienced during development, and degree of executive function challenge in larger samples.
Supplemental Material
sj-pdf-1-eeg-10.1177_1550059420977318 – Supplemental material for Cognitive Event-Related Potentials in Young Adults With Cerebral Palsy: A Proof-of-Concept Study
Supplemental material, sj-pdf-1-eeg-10.1177_1550059420977318 for Cognitive Event-Related Potentials in Young Adults With Cerebral Palsy: A Proof-of-Concept Study by Christine L. Lackner, Jan Willem Gorter and Sidney J. Segalowitz in Clinical EEG and Neuroscience
Footnotes
Acknowledgements
We would like to acknowledge the participation of the adolescents and young adults in the MyStory project for their considerable time and energy put toward this research. We would like to acknowledge the research assistance from members of the MyStory Research Group, including staff, postdocs and clinicians: Sarah Hopmans, Julie Wilson, Helena Viveiros, John Secen, Paul Stacey, Caitlin Cassidy, Diana Parvinchi, and Sophia Roth. The MyStory Research team includes Darcy Fehlings, Mark Ferro, Andrea Gonzalez, Geoffrey Hall, Anna McCormick, Robert Palisano, Peter Rosenbaum, and the authors of this article. Thanks also to CPNet for their continual support of this project. We would like to acknowledge Jessica Callegari and Cassandra Jones for their help with EEG data collection and Stefon van Noordt and James Desjardins for their work programming and designing original versions of the computer tasks used here.
Author Contributions
CLL contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. JWG contributed to conception and design; contributed to interpretation; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. SJS contributed to conception and design; contributed to interpretation; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. MSG contributed to conception; critically revised manuscript; and gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was conducted with the support of the Ontario Brain Institute, an independent nonprofit corporation, funded partially by the Ontario government, with a partner contribution by the Scotiabank Chair in Child Health Research held by Dr Jan Willem Gorter.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
