Abstract
Purpose. To evaluate the congruence or discrepancy of the localization of magnetic resonance imaging (MRI) lesions with interictal epileptiform discharges (IEDs) or epileptic seizure patterns (ESPs) in surface EEG in lesional pediatric epilepsy patients. Methods. We retrospectively analyzed presurgical MRI and video-EEG monitoring findings of patients up to age 18 years. Localization of MRI lesions were compared with ictal and interictal noninvasive EEG findings of patients with frontal, temporal, parietal, or occipital lesions. Results. A total of 71 patients were included. Localization of ESPs showed better congruence with MRI in patients with frontal lesions (n = 21, 77.5%) than in patients with temporal lesions (n = 24; 40.7%) (P = .009). No significant IED distribution differences between MRI localizations could be found. Conclusions. MRI lesions and EEG findings are rarely fully congruent. Congruence of MRI lesions and ESPs was highest in children with frontal lesions. This is in contrast to adults, in whom temporal lesions showed the highest congruency with the EEG localization of ESP. Lesional pediatric patients should be acknowledged as surgical candidates despite incongruent findings of interictal and ictal surface EEG.
Introduction
Resective epilepsy surgery may provide seizure freedom in children with medically refractory focal epilepsy, 1 and early epilepsy surgery in pediatric patients may ameliorate detrimental effects of persistent seizures on cognition and behavior of the developing brain.2,3 A thorough presurgical evaluation aims to identify candidates for resective epilepsy surgery. 4 It needs to localize the seizure onset zone (SOZ) and delineate it from eloquent brain regions. 5 The core procedure is continuous video-EEG monitoring (VEM), where seizure semiology, ictal and interictal EEG are evaluated. Congruence of all localizing diagnostic approaches is demanded, but rarely achieved. 6 In adult patients, the localization of interictal epileptiform discharges (IEDs) and epileptic seizure patterns (ESPs) may be discrepant with the localization of the epileptogenic magnetic resonance imaging (MRI) lesions.6-9 The localization of IEDs and ESPs compared with epileptogenic MRI lesions has not been analyzed systematically in pediatric patients. The aim of the present study was to investigate that relationship.
Methods
Study Population
We analyzed all VEM data of pediatric patients with medically refractory focal epilepsies and validated MRI lesions between 1994 and 2017. All patients underwent presurgical evaluation at a Comprehensive Epilepsy Center.
Inclusion and Exclusion Criteria
Patients aged <18 years with medically refractory epilepsy who had received long-term surface-EEG and video monitoring for presurgical workup were included. Patients without EEG findings were excluded, as well as patients without MRI lesions, with more than 2 MRI lesions or MRI lesions expanding over more than 2 lobes, see Figure 1.
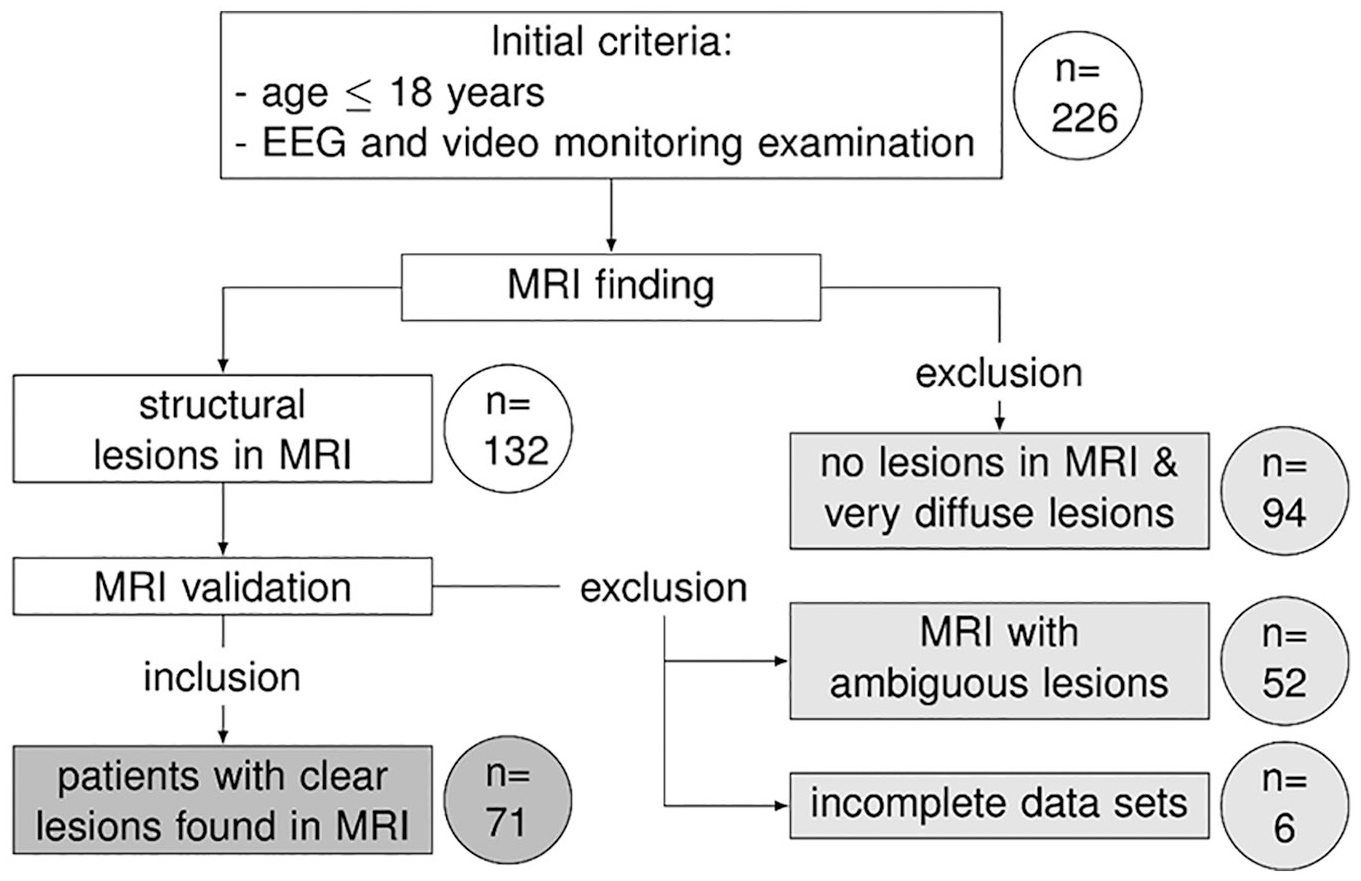
Patient disposition. The figure shows how many patients (circles) were excluded (light gray) or included (darker gray) according to the initial criteria and after the revalidation of the magnetic resonance imaging findings.
All patients’ data had been thoroughly discussed in an interdisciplinary epilepsy surgery board. All data were collected in a standardized protocol, which included demographic data, such as sex, age, age at onset of epilepsy, current duration of epilepsy, and number of antiseizure drugs (ASDs) taken, as well as the results of the VEM, the MRI findings and, if available, postsurgical outcomes.
The demographic data and postsurgical results of all included patients were compared, but in case of EEG to MRI congruency a further selection had to be made to allow proper statistical comparison. Only patients with single lesions located in frontal, temporal, parietal, and occipital lobes were selected; patients with 2 lesions, large lesions or lesions classified as “other” were excluded.
Video-EEG Monitoring
The patients underwent VEM for 5 to 10 days. ASDs were discontinued for the duration of the examination, while patients were monitored continuously using a 40- to 64-channel EEG machines (Vanguard, Cleveland, OH, USA, or Xltek, Natus DBA Excel-Tech Corp., Oakville, Ontario, Canada). The EEG electrodes were set in the 10-20 system and were complimented with additional 10-10 electrodes over the presumed region of interest and sphenoidal electrodes in selected patients. A trained EEG technician was always present and marked any events, such as a clinical seizure or any possible epileptiform pattern in the EEG-recording.
Two experienced epileptologists visually reviewed the EEG data (IEDs and ESP). The time and localization of the ESP onset was defined for any seizure or subclinical seizure pattern, unless artifacts obscured. Time differences of ESP and clinical onset were noted. For IEDs, number and localization in the first 24 hours of EEG were evaluated in full. For the following recording, regular hourly 1- or 2-minute samples were evaluated for IEDs. The sampled IEDs were counted and categorized by the monopolar montage electrode with the highest amplitude.
Magnetic Resonance Imaging
High-resolution MRI (1.5-3.0 T) was performed in all patients as a part of presurgical evaluation in axial, coronal, and sagittal planes (1.3-mm slices, T1, T2, proton-density weighted techniques, and magnetization-prepared rapid acquisition gradient echo (MPRAGE). Diagnostic findings were revalidated by a neurologist with special training in brain imaging in epilepsy for any possible lesion. The revalidation was done blinded to surgical outcomes, but lesion etiology was refined when postsurgical findings were available. If no classification was possible, the lesions were categorized as “unknown.”
The MRI lesions were categorized for the purpose of this study by their localization as frontal, temporal, parietal, or occipital. When the MRI showed more than 1 lesion, the patients were classified as “2 lesions” or if the lesion covered more than 1 lobe as “large lesions.” Patients with hypothalamic or insular lesions were categorized as “other.” The EEG electrodes were also categorized as frontal (including C3, C4, and Cz), temporal, parietal, or occipital.
Postsurgical Outcomes
In case resective epilepsy surgery was performed, patient outcome was categorized by the “Engel” classification system. 10 Not-operated patients include patients who achieved seizure freedom through optimized medication, who rejected further procedures or patients with inoperable lesions.
Statistics
The level of congruence and discrepancy of IED, ESP, and MRI were noted as the fraction of all recorded IED or ESP. Patients were grouped by the localization of the MRI-documented lesions as described in the Magnetic Resonance Imaging section. The congruence between all ESPs and IEDs of the frontal, temporal, parietal, and occipital group were tested using the Kruskal-Wallis test. Demographic data between all 7 MRI lesion groups was tested using the Kruskal-Wallis test for age, age of onset of seizures, number of unsuccessful ASDs, and the duration of epilepsy and the Fisher’s exact test for differences of sex. The results were corrected for multiple testing using the Dunn-Bonferroni correction. Statistical significance was assumed at P < .05 (2-tailed).
Results
Patients
A total of 226 patients aged 18 years or younger were evaluated with presurgical VEM. Structural MRI lesions were found in 132 patients. After MRI revalidation 71 patients (42.3% female) met the criteria described above, see Figure 1. The MRI lesions were mostly frontal (n = 21; 29.6%) and temporal (n = 24; 33.8%), and more rarely occipital (n = 4; 5.6%) and parietal (n = 5; 7.0%). Also less frequent were patients with two lesions (n = 7; 9.9%), large lesions involving more than 1 lobe (n = 6; 8.5%) and “other” lesions (n = 4; 5.6%). There were no significant differences regarding sex (42.3% female, P = .255) and age (median 14 years, interquartile range [IQR] 7.5-16, P = .094). There was a global statistical significance between age of onset (median 8 years; IQR 2.5-12, P = .013), duration of epilepsy (median 3 years; IQR 1-1.5, P = .019), and number of unsuccessful ASDs (median 2; IQR 1-4, P = .019) prior to VEM evaluation; however, pairwise comparisons showed no significant results.
Interictal Epileptiform Discharges
The localization of IEDs with regard to the MRI lesion rarely achieved full congruence. In the group of patients with frontal MRI lesions, 70.2% of IEDs were recorded over the frontal region. For temporal lesions, parietal, and occipital lesions IEDs were also most frequently located over the corresponding lobe at 49.3%, 42.6%, and 38.0%, respectively. The group results were not statistically significantly different (P = .455), see Figure 2.
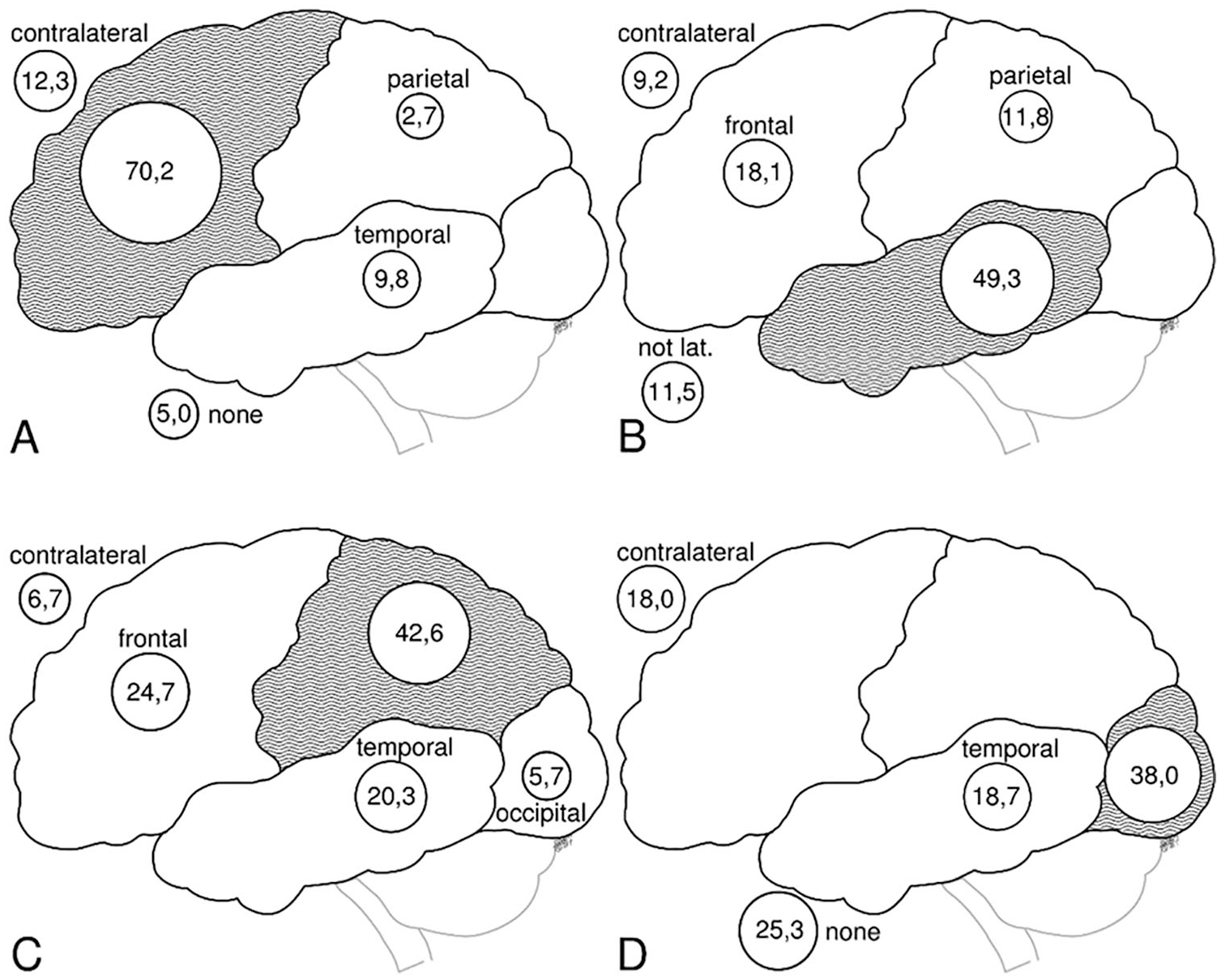
Interictal epileptiform discharge (IED) by magnetic resonance imaging (MRI) lesion. IED distribution over 4 different lesions (A = frontal, B = temporal, C = parietal, and D = occipital) is shown. Each subpicture shows the percentage of IEDs over the own lesion (gray area), the other cortex areas, as well as the contralateral lobe of the lesion. Given are the percentage values of all IEDs in the respective MRI group.
Looking at individual patients, only 25.0% of patients with temporal lesion had 100% of their IEDs over the temporal region, and 41.7% had no IEDs over the temporal lobe at all. In frontal MRI lesions, only 19.0% of patients had IEDs exclusively over the frontal leads and 20.0% of parietal and 25.0% of occipital lesional patients, see Table 1.
Congruency of Interictal Epileptiform Discharges (IEDs) Over the Lesion Grouped by Magnetic Resonance Imaging (MRI) Lesion. a .
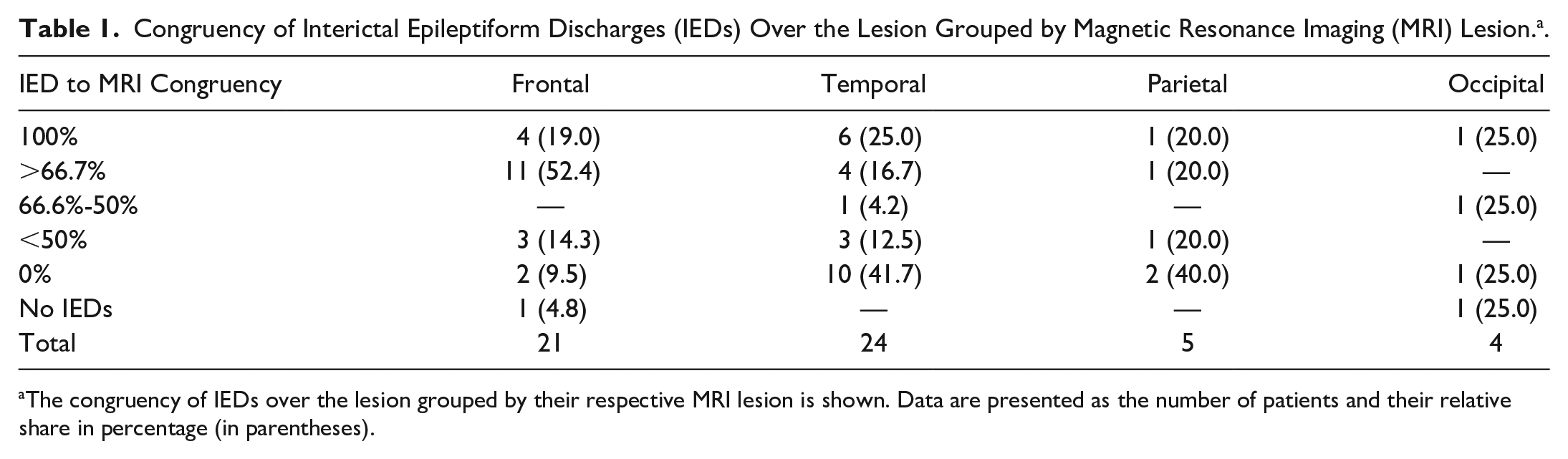
The congruency of IEDs over the lesion grouped by their respective MRI lesion is shown. Data are presented as the number of patients and their relative share in percentage (in parentheses).
Epileptic Seizure Patterns
In patients with frontal and temporal lesions, onset of ESP was congruent (ie, EEG seizure pattern onset of frontal lesions was recorded in frontal electrodes) with the corresponding EEG electrodes over these lobes in 77.5% and 40.7% of all recorded seizures, respectively. In patients with parietal and occipital lesions, congruence was at 74.0% and 58.3%, respectively. The distribution difference of ESP onset between patients with temporal and frontal lesions was statistically significant (P = .009; see Figure 3). The distribution difference of ESPs between all other lesions was not statistically significant (P = 1). Frontal ESPs onset was the second most frequent localization in temporal, parietal, and occipital lesions (30.8%, 24.0%, and 35.4%, respectively, see Figure 3).
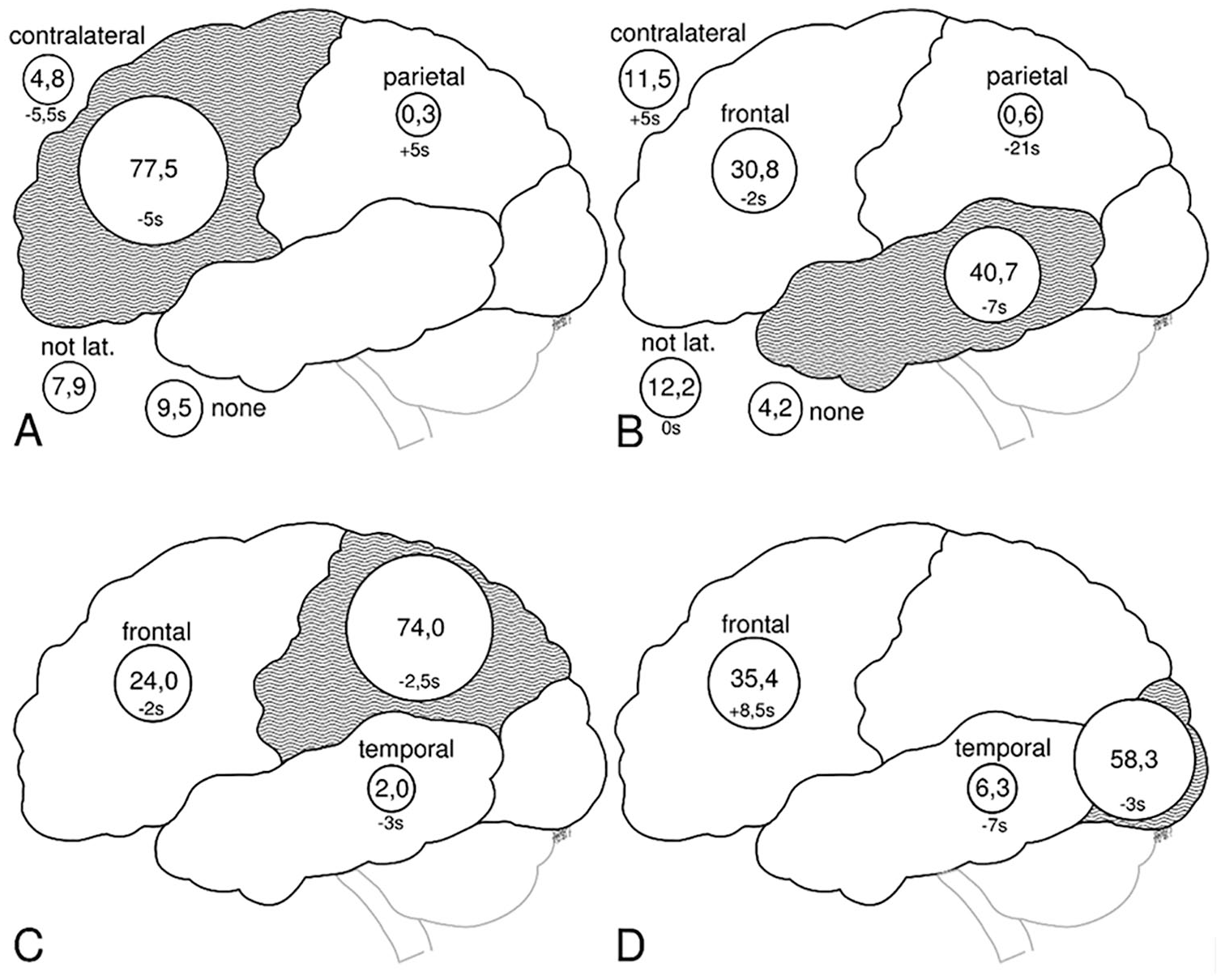
Epileptic seizure pattern (ESP) by magnetic resonance imaging (MRI) lesion. Seizure propagation over 4 different lesions (A = frontal, B = temporal, C = parietal and D = occipital) is shown. Each subpicture shows the percentage of ESPs over the own lesion (gray area), the other cortex areas, as well as the contralateral lobe of the lesion. Given are the percentage values of all ESPs in the respective MRI group. The median of the time difference (in seconds) of ESP onset to clinical seizure onset for the different ESP localizations is also shown.
We also examined the proportion of patients in whom ESPs were exclusively localized over the lesional lobe. In 71.4% of patients with frontal lesions the congruent ESPs were exclusively detected over the frontal lobe, see Table 2. In patients with temporal lesions, only 29.2% had all their ESP in the temporal EEG-leads, while 45.8% of the patients had no ESPs over the lesion at all.
Congruency of Epileptic Seizure Patterns (ESPs) Over the Lesion Grouped by Magnetic Resonance Imaging (MRI) Lesion. a
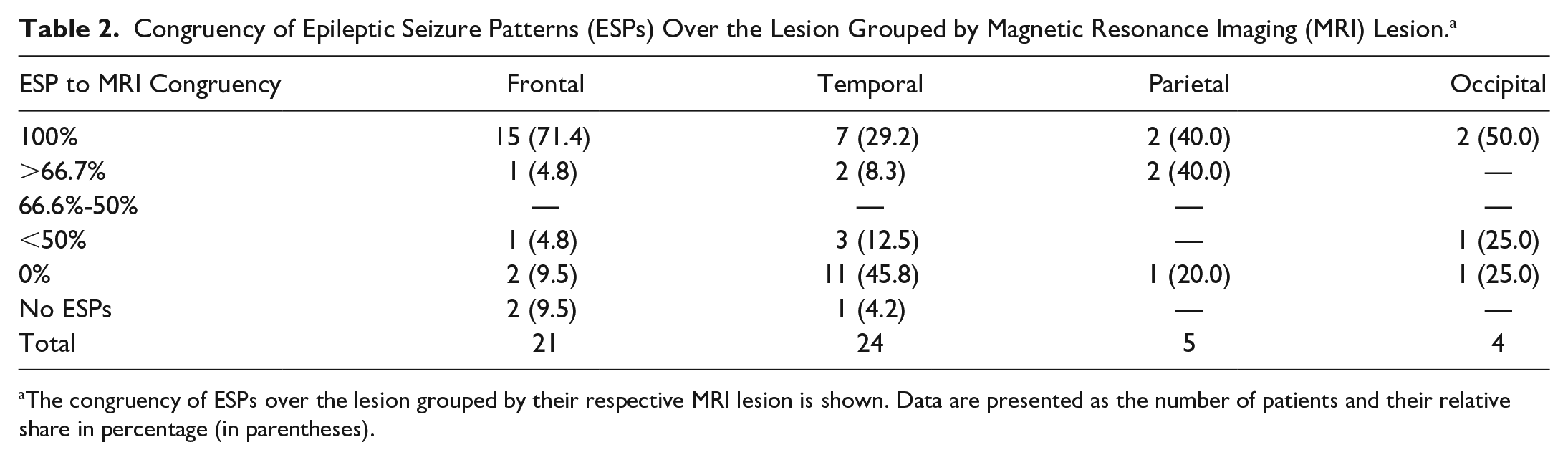
The congruency of ESPs over the lesion grouped by their respective MRI lesion is shown. Data are presented as the number of patients and their relative share in percentage (in parentheses).
Patients with “2 lesions” had 66.7% of their ESPs over one of the lesions, while patients with “large lesions” had either generalized ESPs (33.3%) or almost no congruence between the localization onset of ESP and the MRI lesion. “Other” patients with hypothalamic and insular lesions show mainly frontal ESP onset.
Lesion Etiology
No differences of EEG findings (both IED and ESP) in relation to etiology of the lesion were observed. For lesion classification by MRI localization, see Table 3.
Lesion Classification by Magnetic Resonance Imaging Lesion. a
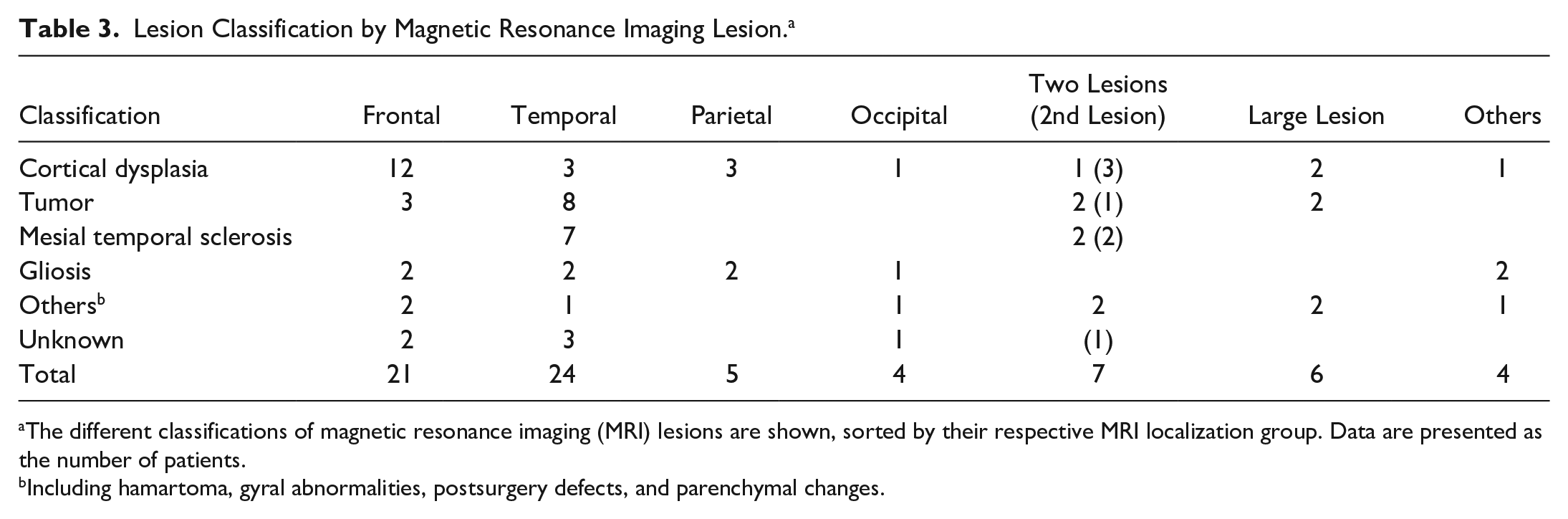
The different classifications of magnetic resonance imaging (MRI) lesions are shown, sorted by their respective MRI localization group. Data are presented as the number of patients.
Including hamartoma, gyral abnormalities, postsurgery defects, and parenchymal changes.
Patient Outcome
A total of 28 patients (39.4%) underwent resective epilepsy surgery. 82.1% of these were free of disabling seizures (Engel class I 10 ) after surgery at a mean follow up of 1 year. Of the 29 patients (40.9%) that were not operated upon, 11 achieved seizure freedom for a 2-year follow-up without surgery, while 6 had inoperable lesions, due to bihemispheric lesions, lesions next to eloquent cortex or inconclusive invasive studies. A total of 75.0% of patients in the groups “2 lesions,” “large lesions,” and “other” did not undergo surgery, making up 12 of the 29 patients (41.4%) in the “no surgery” group. Fourteen patients were lost to follow-up (19.7%), see Table 4.
Patient Outcome by Magnetic Resonance Imaging (MRI) to Epileptic Seizure Pattern (ESP) Congruency. a
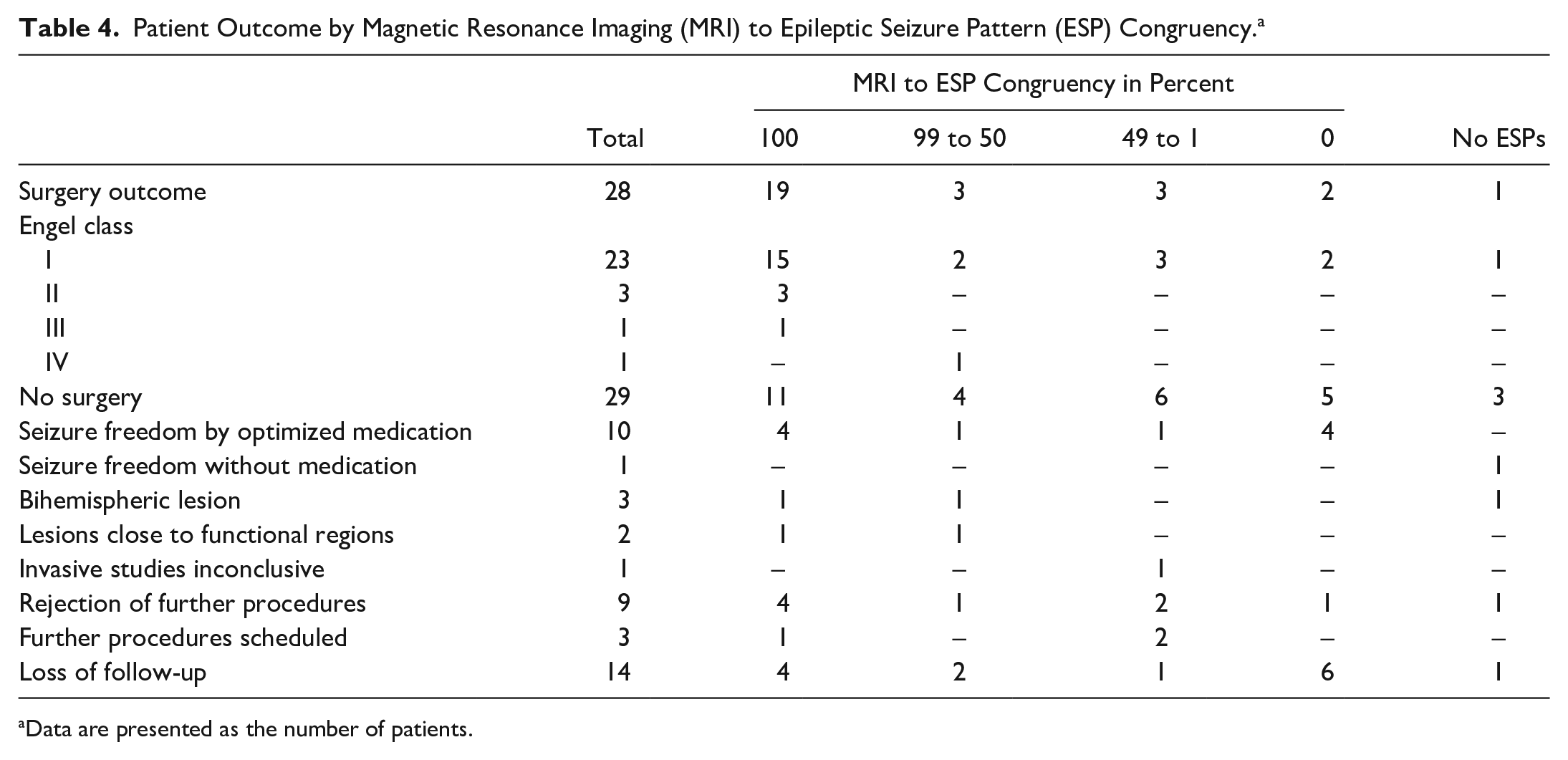
Data are presented as the number of patients.
A 100% congruency between MRI lesion and ESP onset zone was seen in 19 (70.4%) of the 27 operated patients and 11 (42.3%) out of 26 not operated patients (patients with no ESP excluded). Vice versa, 8 (29.6%) of the 27 operated patients and 15 (57.7) of 26 not-operated patients showed incongruent results (99%-0% congruency), see Table 4. Statistically, patients with congruent results of MRI lesion and ESP onset zone were not more likely referred for epilepsy surgery than patients with incongruent results (P = .078).
Discussion
Our study demonstrates that complete congruence of interictal and ictal EEG findings with the MRI lesion is rare in pediatric epilepsy patients as shown previously for adult epilepsy patients.6,11 We detected significant differences in ESP distribution between patients with frontal and temporal MRI lesions.
The most prominent localization of IEDs in our study was over the lesion, but except for patients with frontal lesions the congruence was typically below 50%. In addition, failure to record IEDs due to the limitations of surface EEG recording must be considered. 6 The IED localization is known to differ from the MRI lesion, particularly in extratemporal epilepsies.7-9,11
There are sparse data on the relation of ESP to a given MRI lesion in children and adolescents with focal epilepsy. In a pediatric study with a comparable number of patients and demographics, only 22 of 39 patients with frontal lobe epilepsy (FLE) had ESP localizing to the frontal lobe, whereas almost all patients with temporal lobe epilepsy (TLE) had ESP over the temporal lobe (16 of 17). 12 The discrepancy of these finding to our results might be explained by the high number of MRI negative patients within the FLE group and by the high incidence of hippocampal sclerosis within the TLE group in the cited study compared with a broader spectrum of etiologies in our sample. As in our study the age distribution was a median of 14 years, it represents mainly older children. A cohort with an even younger age may also represent different results.
Similar to adults, children with mesial temporal sclerosis (MTS) reveal a distinct neurophysiological pattern with ESP most likely seen over the temporal leads: while children with MTS had both ESPs and IEDs over the lesion, children with temporal tumors revealed discrepant ESP onset as well as extratemporal IEDs. 13 This should be considered, as etiologies other than MTS are more commonly investigated for resective epilepsy surgery. 14
Data on correlations of lesion localization and EEG seizure onset have been more intensively investigated in adult patients with epilepsy than in children and contrast our findings. In a study on 390 lesional adult epileptic patients, only in 37.7% of patients with frontal lesions were the ESPs exclusively located over their respective lesion. 6 In contrast, 63.5% of patients with temporal lesions had ESPs exclusively in the respective region in that survey. This higher concordance of EEG and MRI in temporal compared with extratemporal lesions was also significant in a meta-analysis evaluating the diagnostic accuracy of VEM. 11 Furthermore, in case the temporal lesion was an MTS, ESP congruence has been shown to be up to 100%. 15 The differences of ESP and MRI lesion congruence between adults and children might be due to different distribution of etiologies and their distinct neurophysiological phenotypes with more MTS in adults than in children15,16 and the different anatomical representation of the temporal lobe on surface EEG.
Other lesion locations are often known to provide poor SOZ scalp localization, such as insular, deep, or midline lesions, which might explain the lack of congruency in some cases of our study. It was shown, that 10 to 20 cm2 of cortex activated through a subcortical lesion are required to localize an interictal or ictal EEG pattern. 17
Considering the time of occurrence, the congruent ESPs have been discussed to occur earlier than discordant ESPs. 18 However, there were no significant differences between the time of ESP onset over the actual MRI lesion and ESP onset over other regions in our study. Invasive studies could reveal more exact time resolution differences with regard to localization of the ictal onset zone, which may not be seen in surface EEG recordings. But as surface EEG is always the first step in presurgical evaluation, the relationship of surface EEG and MRI and its significance is relevant for the presurgical workup.
The success of epilepsy surgery with regard to seizure freedom and absence of functional impairment is dependent on the ability to localize the SOZ. When patients are free of disabling seizures after surgery, a resected MRI lesion can be considered as the actual epileptogenic focus. 82.1% of our operated patients achieved seizure freedom (Engel class I 10 ). In the present study, patients with incongruent results were as likely referred to surgery as patients with congruent result and seizure outcome was equally distributed. Although discrepancy of MRI lesion and ESP is a common occurrence in our study, it only demonstrates the poor localizing power of surface EEG and should not lead to an automatic deferral of these patients from surgery. The usual approach to those patients is to proceed to invasive EEG studies, 4 as they may need a more comprehensive coverage with intracranial electrodes. 19 All pediatric patients should be considered for epilepsy surgery, as they might benefit greatly from the surgical treatment option.2,3
The limitations of our study are a smaller sample size than in adult patients, 6 but our patient number is higher than in previous pediatric studies. The histological type of the lesion might also influence anatomo-electroclinical correlations. However, the present sample size was too small to reach enough power to find conclusive statements of this correlation. The interpretation of our postsurgery outcome is also restricted. Overall, 9 of all 71 patients (12.7%) or their legal guardians rejected further procedures or surgery and 11 of 71 (15.5%) achieved seizure freedom through optimized medication. Also, as only 28 of our 71 patients (39.4%) underwent surgery, the analysis of congruence between MRI lesion (as the suggested seizure onset zone) and EEG is only descriptive for patients without surgery.
Conclusions
As the localizing yield of surface EEG is low in children, we conclude that the IED distribution is not a reliable source for definite localization of the epileptogenic zone.6,7 The ESP distribution may be reliable in children with frontal lesions, but not in temporal lesions, while in patients with parietal and occipital lesions there is not enough data to form a conclusive argument. Surface EEG should be used as confirmative and not as excluding information and epilepsy surgery should still be pursued when discrepant surface EEG results are present.
Footnotes
Acknowledgements
We thank R. Picinotti, R. Tschakert, and O. Klein for technical assistance.
Author Contributions
JR contributed to conception and design, contributed to acquisition and interpretation, critically revised manuscript, and gave final approval. SS contributed to design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, and gave final approval. MT contributed to acquisition, drafted manuscript, critically revised manuscript, and gave final approval. PP contributed to analysis, critically revised manuscript, and gave final approval. LG contributed to acquisition and gave final approval. AP contributed to acquisition and gave final approval. MK contributed to acquisition and gave final approval. CV contributed to acquisition and gave final approval. SN contributed to acquisition and interpretation, critically revised manuscript, and gave final approval. IB contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The Ethical Review Board of the Medical Faculty of the Ludwig-Maximilians-University of Munich declared no relevant ethical conflict for this anonymized retrospective analysis (Project-No. 17-326).
