Abstract
Objectives. The study aim was to develop 2 scales: predicting clinical gains and risk of acute side effects of stimulant medication in pediatric attention-deficit/hyperactivity disorder (ADHD), combining measures from EEG spectra, event-related potentials (ERPs), and a cued visual GO/NOGO task. Methods. Based on 4-week systematic medication trials, 87 ADHD patients aged 8 to 17 years were classified as responders (REs, n = 62) or non-REs (n = 25), and belonging to the side effects (SEs, n = 42) or no-SEs (n = 45) groups. Before starting the trial, a 19-channel EEG was registered twice: Test 1 (T1) without medication and T2 on a single dose of stimulant medication a few days before the trial. EEG was registered T1 and T2: 3 minutes eyes-closed, 3 minutes eyes-open, and 20 minutes cued GO/NOGO. EEG spectra, ERPs, omissions, commissions, reaction time (RT), and RT variability were computed. Groups were compared at T1 and T2 on quantitative EEG (qEEG), ERPs and behavioral parameters; effect sizes (d) were estimated. Variables with d > 0.5 were converted to quartiles, multiplied by corresponding d, and summed to obtain 2 global scales. Results. Six variables differed significantly between REs and non-REs (T1: theta/alpha ratio, P3NOGO amplitude. Differences T2-T1: Omissions, RT variability, P3NOGO, contingent negative variation [CNV]). The global scale d was 1.86. Accuracy (receiver operating characteristic) was 0.92. SEs and no-SEs differed significantly on 4 variables. (T1: RT, T2: novelty component and alpha peak frequency, and RT changes. Global scale d = 1.08 and accuracy = 0.78. Conclusion. Gains and side effects of stimulants in pediatric ADHD can be predicted with high accuracy by combining EEG spectra, ERPs, and behavior from baseline and single-dose tests. ClinicalTrials.gov identifier: NCT02695355.
Introduction
Attention-deficit/hyperactivity disorder (ADHD) is a common neuropsychiatric disorder starting in childhood, often persisting into adulthood, 1 and characterized by developmentally inappropriate symptoms of inattention and/or hyperactivity/impulsivity, resulting in impairment in multiple life domains. 2 Worldwide prevalence is estimated at 5% to 7% of the population. 3 Comorbid conditions are present in the majority of cases. 4 The most common treatment for children and adolescents is stimulant medication—methylphenidate (MPH) or dextroamphetamine (DEX)—as a part of multimodal treatment, including parent management training and adjustments in school. Therapeutic effects of stimulants (improved attention, reduced restlessness/hyperactivity, and impulsivity) are reported in approximately 70% of patients.5-9 A shift from MPH to DEX increases therapeutic effects to 80%.5,6 The frontal-basal-ganglia-thalamo-cortical networks of the brain are considered prime candidates for the source of the underlying dysfunction, including hypofunctioning dopamine and noradrenaline systems. 10 Stimulants seem to activate the frontal-striatal parts of the circuits 11 and underlie the positive clinical effects of stimulants. 12
Psychiatric diagnoses like ADHD are based on observed behavioral symptoms and developmental history. The neural mechanisms behind symptoms like inattention and hyperactivity may not always be the same, reducing the probability that the same treatment fits all cases.
Approximately 25% to 30% of pediatric ADHD patients are considered nonresponders to MPH. 6 There is no consensus, however, on what constitutes a clinically meaningful response. Rating scales from parents and teachers comparing baseline scores with scores on medication are widely used. For children older than 12, self-report scales are often included. A 25% or 50% reduction of baseline scores, or scores within 1 standard deviation (SD) above population mean,13,14 have been suggested as criteria for positive response. The critical decision in the clinic—whether to medicate—is often a global decision based on data from scales and interview feedback from parents, teachers, and patients. A separate evaluation of acute side effects (insomnia, loss of appetite, increased irritability, or anxiety) is clinically meaningful.5,15 It should be stressed that correlations between scores from parents and teachers are often low, supporting the necessity for a global clinical decision.
A significant number of nonresponders and disagreement among informants motivate the search for reliable predictors of response. Variables that have been studied include demographics, ADHD subtype, comorbidities, neuropsychological test scores, scores on rating scales, genetics, and EEG/ERP variables.16-19 The predictive power of neuropsychological tests is considered modest.20-24 Studies applying ERPs for prediction of medication effects are summarized in our 2016 publication. 25
In our previous studies on predictors of medication effects, we applied behavioral test data, EEG spectra, and ERPs from a cued visual GO/NOGO task (WinEEG). In the first study on acute side effects, we found 3 variables that contributed significantly to predictions. They were converted to quartile (Qu) scores and combined. We found 13% with side effects in Qu Group 1 of this combined scale, and 91% in Qu Group 4. 15 In another study focusing on clinical outcome, we applied a similar method, and found 36% responders in Qu Group 1 and 89% in Qu group 4. 21 In a third study, 25 we investigated the differential effects of a single dose of stimulants on responders (REs) and nonresponders (non-REs). Patients performed the cued GO/NOGO task twice, the last time an hour after receiving a trial dose. They were later classified as REs or non-REs after a systematic 4-week medication trial. In that study, we focused on changes in cognitive ERPs and behavioral parameters from Test 1 without medication, to Test 2 with medication. The main finding was that the effects of medication on ERP component P3 NOGO differed significantly between REs and non-REs, with a large effect size (d = 1.76). Results from a single-dose test improved prediction accuracy compared with predictions based on the baseline GO/NOGO task.
There is currently no reliable method for predicting response to stimulants in ADHD patients, however, without exposure to a medication trial period. 26 This method is time consuming for professionals and patients and often inconclusive because of disagreement between informants. Reliable response predictors could represent a significant improvement in clinical practice by reducing the time needed to find the best medication, if any.
Study Aims and Hypotheses
The patient group in this study is the same as in our previous “single-dose-publication.” 25 The current study has a broader scope, however, including EEG spectra from Test 1 and Test 2, behavioral data from both tests and ERP components not included in the single-dose study. In this study, we also searched for predictors of side effects in addition to medication effects on ADHD symptoms. Based on the assumption that the most robust predictors of medication response may not be the same for all patients, we hypothesized that constructing a scale combining the variables that differ significantly between compared groups (REs vs non-REs; SEs vs no-SEs) with effect sizes (d) >0.5 would improve predictions compared with our previous findings. We predicted that the 2 global scales (clinical gains scale and side effects scale) would discriminate between the compared groups with effect sizes larger than 0.8—the estimated minimum for clinical applications. 27
Methods
Participants and Diagnostic Procedures
A neuropsychiatric team comprising 2 neuropsychologists, a pediatrician, and a specialist in school psychology, all with several decades of clinical experience, diagnosed 87 patients with ADHD in accordance with DSM-IV (patient inclusion finished before DSM-5 was published). After positive screening for ADHD in the local child psychiatry outpatient clinic, patients were referred for diagnostic conclusions and extended examination. They were screened medically, developmental and medical histories were recorded, and clinical interviews, school observations, and parents’ and teachers’ rating scales (Conners’ 3, ASEBA [Achenbach System of Empirically Based Assessment], BRIEF [Behavior Rating Inventory of Executive Function], and 5-15 [FTF]28-30) were completed. Intelligence was assessed using either WISC-IV (Wechsler Intelligence Scale for Children, 4th edition) or WASI (Wechsler Abbreviated Scale for Intelligence).28-33 Patients with IQs <70 or with a diagnosed brain injury were excluded. Eleven cases were excluded because of the technical quality of the EEG recordings and 1 case for noncompliance. Patients with common comorbidities such as behavioral and emotional disorders, learning disabilities, or autism spectrum disorders were included.
The first qEEG/ERP test was part of the extended examination in the neuropsychiatric team generating hypotheses about cognitive and emotional strengths and weaknesses. Patients and parents consented to complete the second test ahead of the medication trial and were informed that we searched for EEG- based predictors of medication response.
The procedure for titration and the rules for classification as REs versus non-REs and SEs versus no-SEs are described in Ogrim et al 25 and in the supplementary material (available in the online version of the article). The Regional Committee for Medical Research Ethics approved the project.
Assessment of ERPs and EEG Spectra
EEG was recorded using a Mitsar 201 19-channel EEG system (http://www.mitsar-medical.com): 3 minutes in the eyes-closed condition, 3 minutes in the eyes-open condition, and 20 minutes of the cued GO/NOGO task. The ERP parameters measured reflect different aspects of cognitive control.34 -36 Images of animal (a) and plant (p) categories served as relevant stimuli. The trials consisted of paired stimuli with interstimulus intervals of 1000 ms and intertrial intervals of 3000 ms. Four trial categories were used: a-a, a-p, p-p, and p-h (h = human image); subjects were to respond to a-a trials. Speed and accuracy were equally emphasized. The pictures were selected from children’s textbooks; overall luminance and the image sizes of animals and plants were approximately equal. To avoid habituation, 20 different animal, plant, and human images were randomly presented in various combinations. To maintain alertness in the p-h trials, novel sounds were occasionally presented with human images. They produced an orientation reaction, confirmed by elicitation of the novelty ERP wave.
Trials were grouped into 4 blocks of 100 trials each. In each block, a unique set of 5 a, 5 p, and 5 h stimuli were selected. Each block consisted of a pseudo-random presentation (requiring equal number of trials in 4 categories) of 400 trials, with 100 trials within each trial category. Participants practiced the task before the recording started. They sat upright in a comfortable chair looking at a 17-inch CRT computer screens positioned 1.5 m in front of them; it occupied 3.8° of the visual field. Pressing the button to a-a pairs within 200 and 1000 ms after presentation of the second stimulus was considered a correct response. Failure to respond to a-a pairs within this time interval was considered an omission error. Impulsive responses to a-p pairs were considered commission errors. Subjects rested for a few minutes after 200 trials.
Input signals were referenced to earlobe electrodes, filtered between 0.5 and 50 Hz, and digitized at a sampling rate of 250 Hz, with impedance kept below 5 kohm for all 19 electrodes. An electrode cap with tin electrodes (Electro-cap International, Eaton, OH, USA) was applied, with electrodes placed in accordance with the international 10-20 system.
EEG data were re-referenced offline to the common average montage prior to data processing. Eye-blink artifacts were corrected by zeroing the activation curves of individual independent components extracted by independent component analysis (Infomax algorithm) and corresponded to eye-blink topographies.37,38 EEG epochs with excessive amplitude (100 µV) and/or excessively fast (35 µV in 20-35 Hz band) and slow (50 µV in 0-1 Hz band) frequency activities were automatically excluded from analysis.
ERPs and EEGs were registered twice—first without medication (Test 1 [T1]) and later on a single dose of MPH (n = 79) or DEX (n = 8) (Test 2 [T2]). For several practical reasons, such as parents’ need for decision-making time about medication for their children and upcoming holidays, the time between the 2 tests varied considerably, with no systematic differences between REs and non-REs or SEs and non-SEs. 23
Amplitudes of the ERP components were measured at the electrodes where the component was observed to be strongest in the grand-average ERPs of the groups (Figure 1). The sites and time intervals for ERPs after stimulus 1 were; P1 at O1 and O2 (100-220 ms), cue-P3, at Pz (270-370 ms); CNV at Cz, (1000-1100 ms). ERPs after stimulus 2: P3GO at Pz (260-400 ms); N2NOGO at Fz (220-330 ms); P3NOGO at Cz, (300-500 ms), auditory N1 at Cz (100-170 ms), and auditory P2 at Cz (170-250 ms).
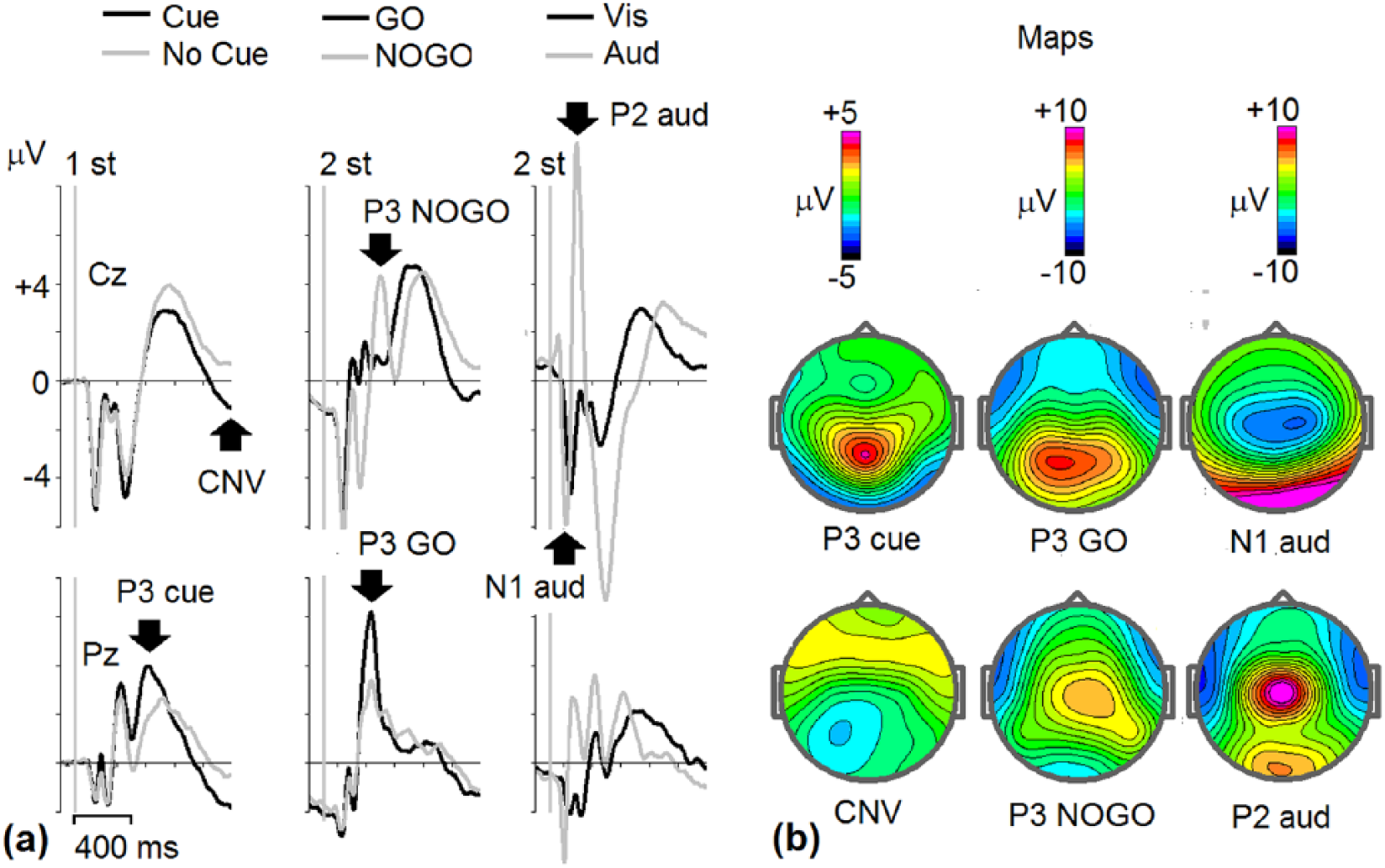
Grand-average event-related potentials (ERPs) in the cued GO/NOGO task. ERPs for all subjects and T1 and T2 are averaged (n = 176). (a) Left to right: ERPs at Cz and Pz electrodes to the first stimuli: animal-Cue (black lines) and plant-No Cue (gray lines), ERPs to the second relevant stimuli: GO (black lines) and NOGO (gray lines). ERPs to second ignored stimuli: visual alone (black lines), and visual + Novel auditory stimuli (gray lines). (b) Maps for the main components: P3 Cue, contingent negative variation (CNV) Cue for Cue stimuli, P3 GO and P3 NOGO for GO and NOGO stimuli, N1 and P2 waves to visual + Novel auditory stimuli. The arrows in (a) indicate the components.
All component amplitudes except CNV were measured manually as the local peak amplitude within a predefined time interval based on onset and offset of the component in the grand-average ERP waveform, for the whole sample at both tests. Local peak amplitude refers to the point within the defined time window for the component of interest with the largest amplitude, which is surrounded on both sides by lower voltages, thereby avoiding measuring the offset of preceding or onset of following components. 39
EEG spectra for all 19 sites in 3 conditions (eyes open, eyes closed, GO/NOGO task) were computed by the fast Fourier transformation method with epoch length of 4 seconds, 50% overlapping, and Hanning time window. For grand average spectra, posterior theta was significantly stronger in REs, and posterior alpha was significantly stronger in non-REs. These effects were best captured by computing theta/alpha ratio at site Cz (mean power in frequency band 4 to 8 Hz/mean power in frequency band 8 to 12 Hz). Comparing grand average spectra for SEs and no-SEs revealed that the alpha peak frequency in no-SEs was higher than with SEs, most clearly seen at O2 in Visual Continuous Performance Test (VCPT), Test 2. This alpha peak was scored manually for each subject.
Statistical Methods
Constructing Local Scales
Because possible confounding variables like subtypes of ADHD, comorbidities, gender, age, intelligence, and T1-T2 time interval may partially explain differences between compared groups, Chi-square tests were applied for dichotomous variables and independent samples, and t tests for continuous variables.
In our previous publication on this sample, we identified T1-T2 changes in behavioral variables and cognitive ERP components that differed significantly between REs and non-REs (omissions, reaction time [RT], RT variability, cue P3, CNV, P3 NOGO). In the present study, additional variables were checked for REs vs. non-REs and SEs vs. no-SEs. The following procedure was applied: (1) for EEG-spectra, grand average spectra (19 sites) were computed at T1 and T2. We compared REs with non-REs and SEs with no-SEs. When t tests indicated that EEG power in theta, alpha, or beta bands differed significantly between the 2 sets of compared groups, individual data from the site with the most significant difference were exported to SPSS for statistical analyses. (2) For ERPs: In our previous study, 40 individual data for CNV, P3 Cue, P3 GO, N2 NOGO, and P3 NOGO were computed. In this study, we included auditory N1 and P2 (“novelty component”), as t tests showed significant differences between SEs and non-SEs.
In summary, 3 categories of variables (behavioral, EEG spectra, and ERPs) were analyzed. Comparisons of group scores were completed at T1 and T2 separately and for the difference scores (T2 – T1). Variables that differed significantly between compared groups at P < .05, with effect size (ES, Cohen’s d) higher than 0.5 were included in the 2 global-scale computations—for predicting clinical gains and side effects. If 2 variables were correlated with coefficient higher (below) +0.7 (−0.7), the variable with the smallest d was excluded. Because the variables studied are age-dependent, we report results for the younger group (8-12 years, n = 40) and the older group (13-18 years, n = 47) separately, in addition to total-group results.
Constructing Global Scales
To combine the scores, we converted them into quartile (Qu) scores, because they were not all normally distributed. A low score on a given scale was converted to Qu score 1, a high score to Qu score 4. Qu scores were multiplied by the d of that scale, giving priority to the variables with the largest ds. Qu score 2 on a scale with d = 0.8 produced a scale score of 1.6). According to statistical adviser Rene Holst corrections for multiple comparisons are not relevant for construction of these global scales. We applied this method in 2 previous publications.19,25
Tables 1 and 2 present data for all variables that differed significantly between groups (d > 0.5): Mean, SDs, p values, and d for younger, older, and total groups. Moreover, we report accuracy, sensitivity, and specificity for the 2 global scales. We applied the receiver operating characteristic (ROC) analysis and report area under curve (AUC). A scale discriminating between groups with AUC = 0.9 to 1.0 is considered excellent, 0.8 to 0.9 is good, 0.7 to 0.8 is fair, 0.6 to 0.7 is poor, and ≤0.5 indicates failure.
Description of the Sample (N = 87).
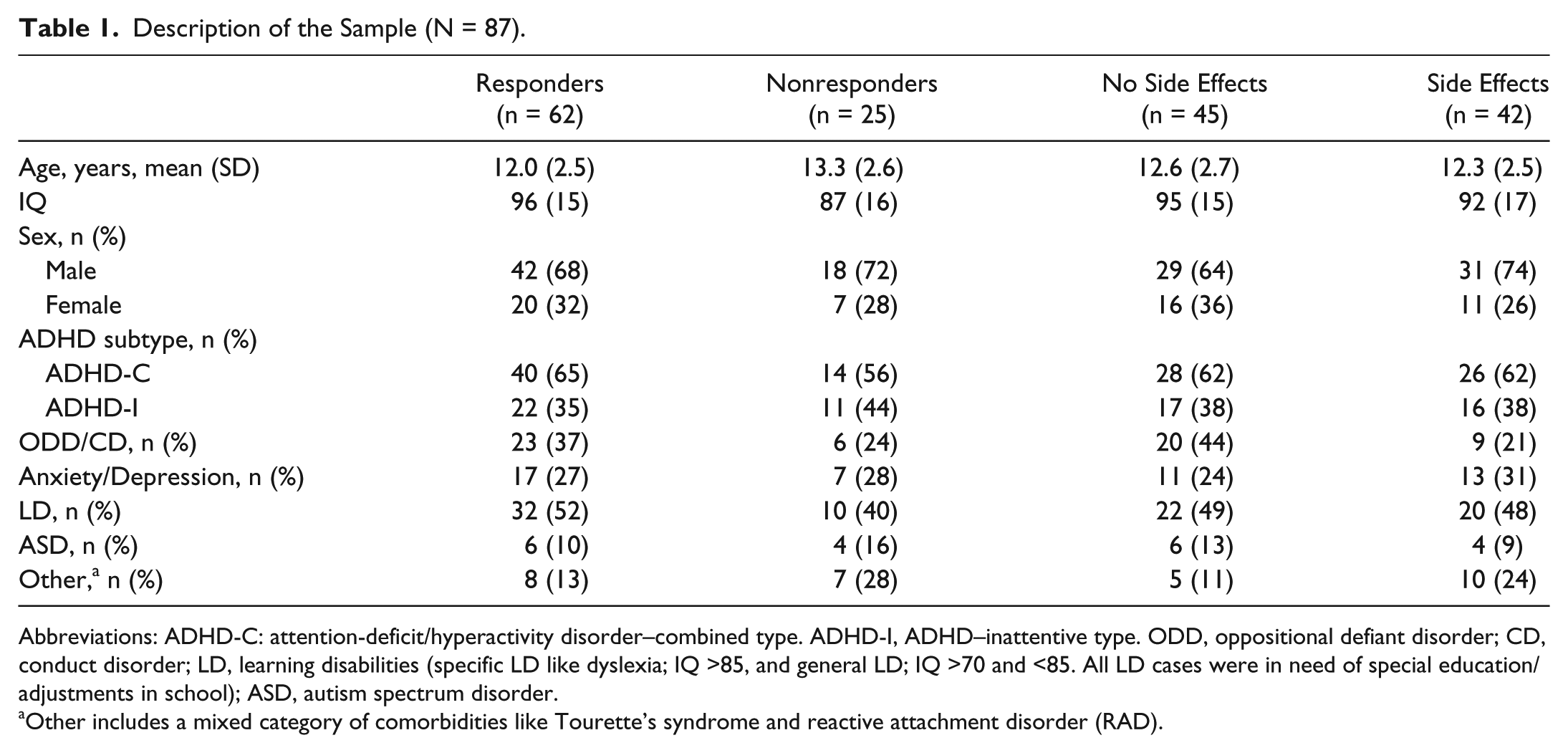
Abbreviations: ADHD-C: attention-deficit/hyperactivity disorder–combined type. ADHD-I, ADHD–inattentive type. ODD, oppositional defiant disorder; CD, conduct disorder; LD, learning disabilities (specific LD like dyslexia; IQ >85, and general LD; IQ >70 and <85. All LD cases were in need of special education/adjustments in school); ASD, autism spectrum disorder.
Other includes a mixed category of comorbidities like Tourette’s syndrome and reactive attachment disorder (RAD).
Variables Differing Significantly Between Responders (REs) and Nonresponders (Non-REs). a
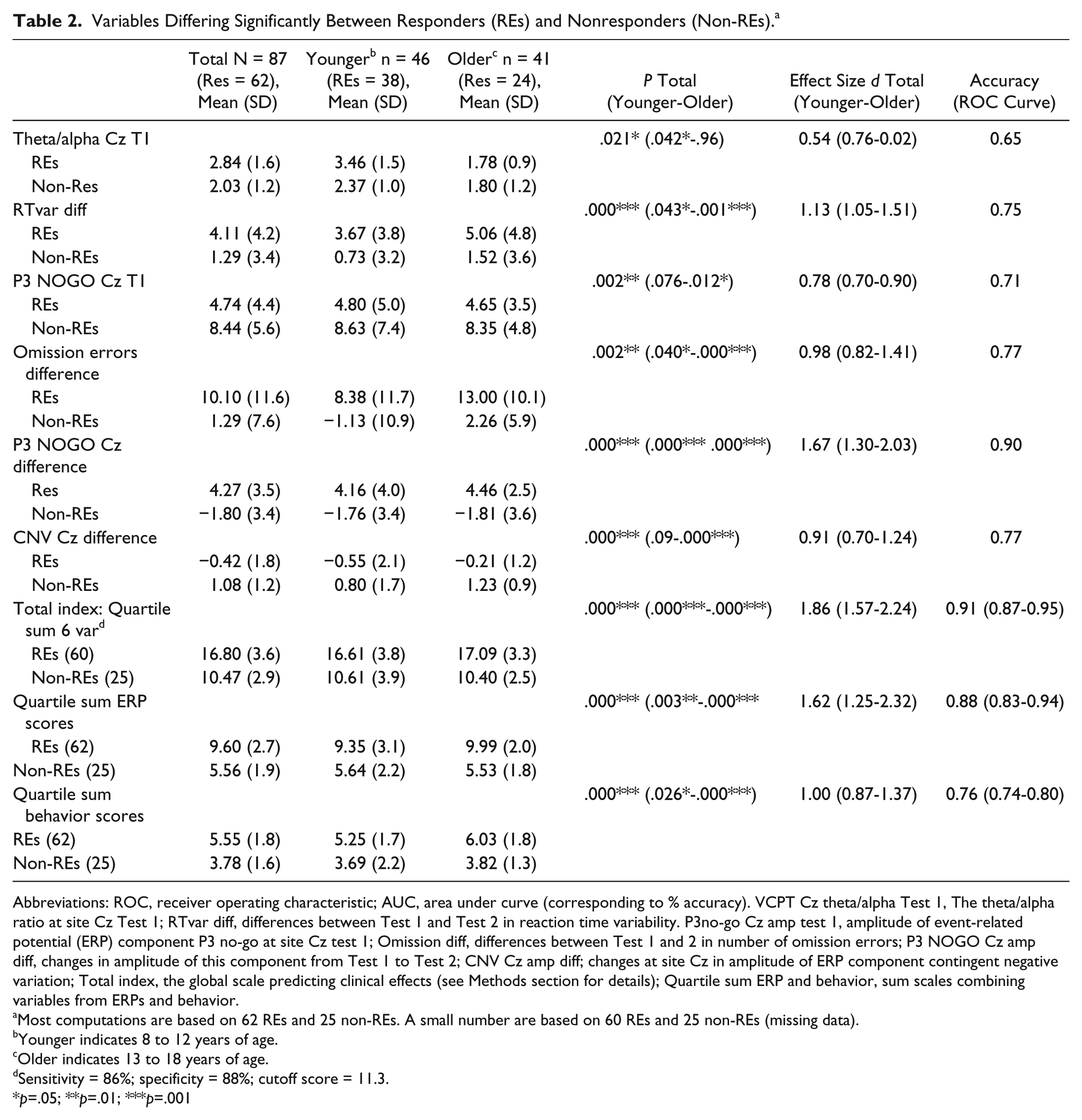
Abbreviations: ROC, receiver operating characteristic; AUC, area under curve (corresponding to % accuracy). VCPT Cz theta/alpha Test 1, The theta/alpha ratio at site Cz Test 1; RTvar diff, differences between Test 1 and Test 2 in reaction time variability. P3no-go Cz amp test 1, amplitude of event-related potential (ERP) component P3 no-go at site Cz test 1; Omission diff, differences between Test 1 and 2 in number of omission errors; P3 NOGO Cz amp diff, changes in amplitude of this component from Test 1 to Test 2; CNV Cz amp diff; changes at site Cz in amplitude of ERP component contingent negative variation; Total index, the global scale predicting clinical effects (see Methods section for details); Quartile sum ERP and behavior, sum scales combining variables from ERPs and behavior.
Most computations are based on 62 REs and 25 non-REs. A small number are based on 60 REs and 25 non-REs (missing data).
Younger indicates 8 to 12 years of age.
Older indicates 13 to 18 years of age.
Sensitivity = 86%; specificity = 88%; cutoff score = 11.3.
p=.05; **p=.01; ***p=.001
Statistical analyses were performed using SPSS Version 18 (http://www.spss.com), with significance = 5%. Cohen’s d effect sizes were calculated with correction for different sample sizes.
Results
As in our previous study, 40 neither time interval between tests nor type of medication (MPH vs DEX) differed significantly between REs and non-REs. No significant differences between the selected groups were found in terms of ADHD subtype, comorbid disorders, or gender. REs’ mean age was 1.2 years younger than non-REs (P = .04), and the non-RE group scored 9 IQ points lower than REs (P = .05). IQ data were unavailable for 13 patients.
We found no significant differences between SEs and non-SEs for age; IQ, gender, ADHD subtype, MPH versus DEX, or comorbid emotional, learning, or autism spectrum disorders.
Responsers Versus Nonresponders
Table 2 shows 6 variables to be significantly different between REs and non-REs, with ES > 0.5. ERPs and behavioral parameters captured the differences better than EEG spectra. Figure 2 illustrates lower amplitude of P3 NOGO wave in REs compared with non-REs at T1 and increase of this wave at T2 for REs but not for non-REs.
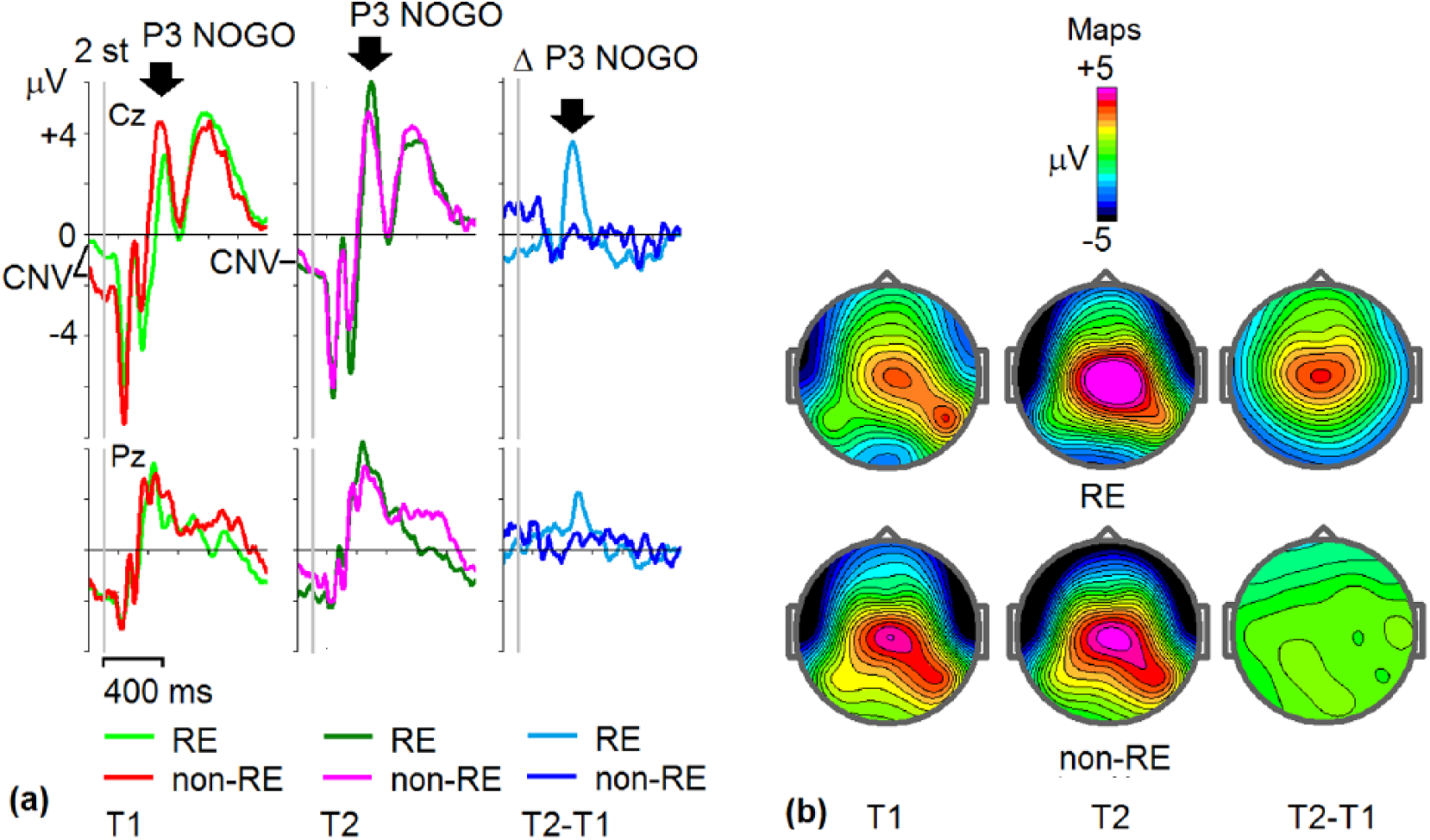
Grand-average event-related potentials (ERPs) for responders (REs) and non-REs. (a) Grand-average NOGO ERPs at Cz and Pz electrodes for REs and non-REs at T1 (light green = RE, red = non-RE), T2 (dark green = RE, pink = non-RE), and T2-T1 differences (light blue = RE, dark blue = non-RE). Vertical gray line = onset of stimulus presentation. Contingent negative variation (CNV) indicates prestimulus baseline. (b) Maps of ERPs for REs and non-REs taken at maximums of P3 NOGO waves indicated by arrows at (a).
We computed 3 global scales; quartile sum scores on 6 variables (Total index), quartile sum ERP scores (ERP index), and quartile sum behavior scores (Behavior index). The procedure is described in the Methods section. The main findings are related to Total index, which combines the 6 variables of interest. The ES of this scale was d = 1.86 (younger group = 1.57; older group = 2.24). The accuracy of the scale was 91% (younger group = 87%; older group = 95%). We also computed sensitivity and specificity for the Total index: sensitivity = 86%, specificity = 88%.
Side Effects Versus No Side Effects
The results are presented in Table 3. SEs and no-SEs differed significantly for four variables. The SEs had a faster reaction time at T1 but did not increase their speed on medication. The no-SEs were 34 ms faster at T2 than at T1, but 20 ms slower than SEs. The frequency of the alpha peak at T2 (site O2, VCPT, task condition) was highest in the no-SE group. This difference was larger in the older group.
Variables Significantly Differing Between Side Effects Group (SE) and No-SE Group With Effect Size >0.5.
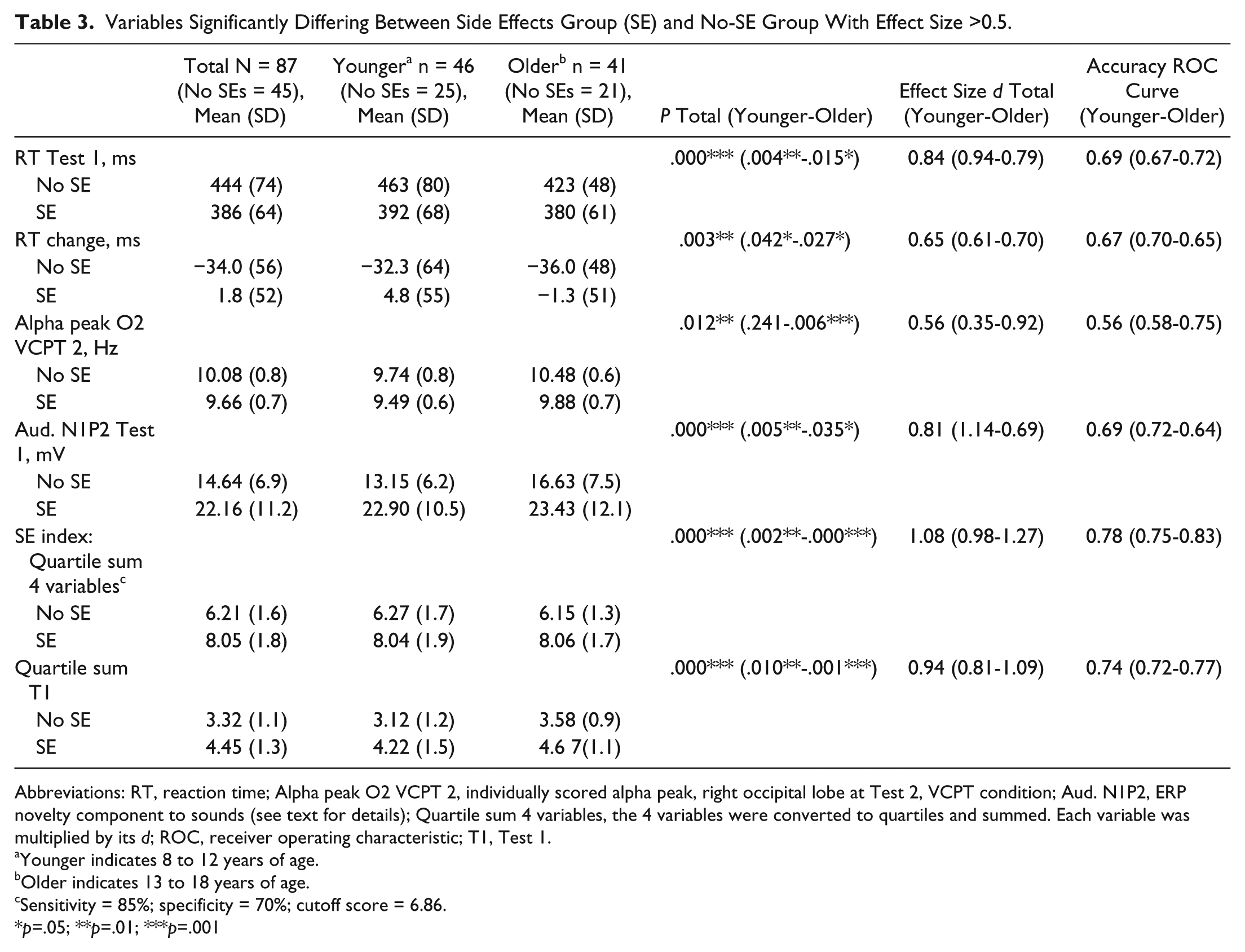
Abbreviations: RT, reaction time; Alpha peak O2 VCPT 2, individually scored alpha peak, right occipital lobe at Test 2, VCPT condition; Aud. N1P2, ERP novelty component to sounds (see text for details); Quartile sum 4 variables, the 4 variables were converted to quartiles and summed. Each variable was multiplied by its d; ROC, receiver operating characteristic; T1, Test 1.
Younger indicates 8 to 12 years of age.
Older indicates 13 to 18 years of age.
Sensitivity = 85%; specificity = 70%; cutoff score = 6.86.
*p=.05; **p=.01; ***p=.001
The automatic reaction to novel sounds in p-h + novel sound pairs is reflected in 2 ERP components: auditory N1 (100-170 ms after stimulus presentation) and auditory P2 (170-250 ms after presentation). These components were significantly stronger in SEs than in no-SEs (Figure 3). The amplitudes of these 2 components were individually scored and summed (auditory N1 was multiplied by −1).
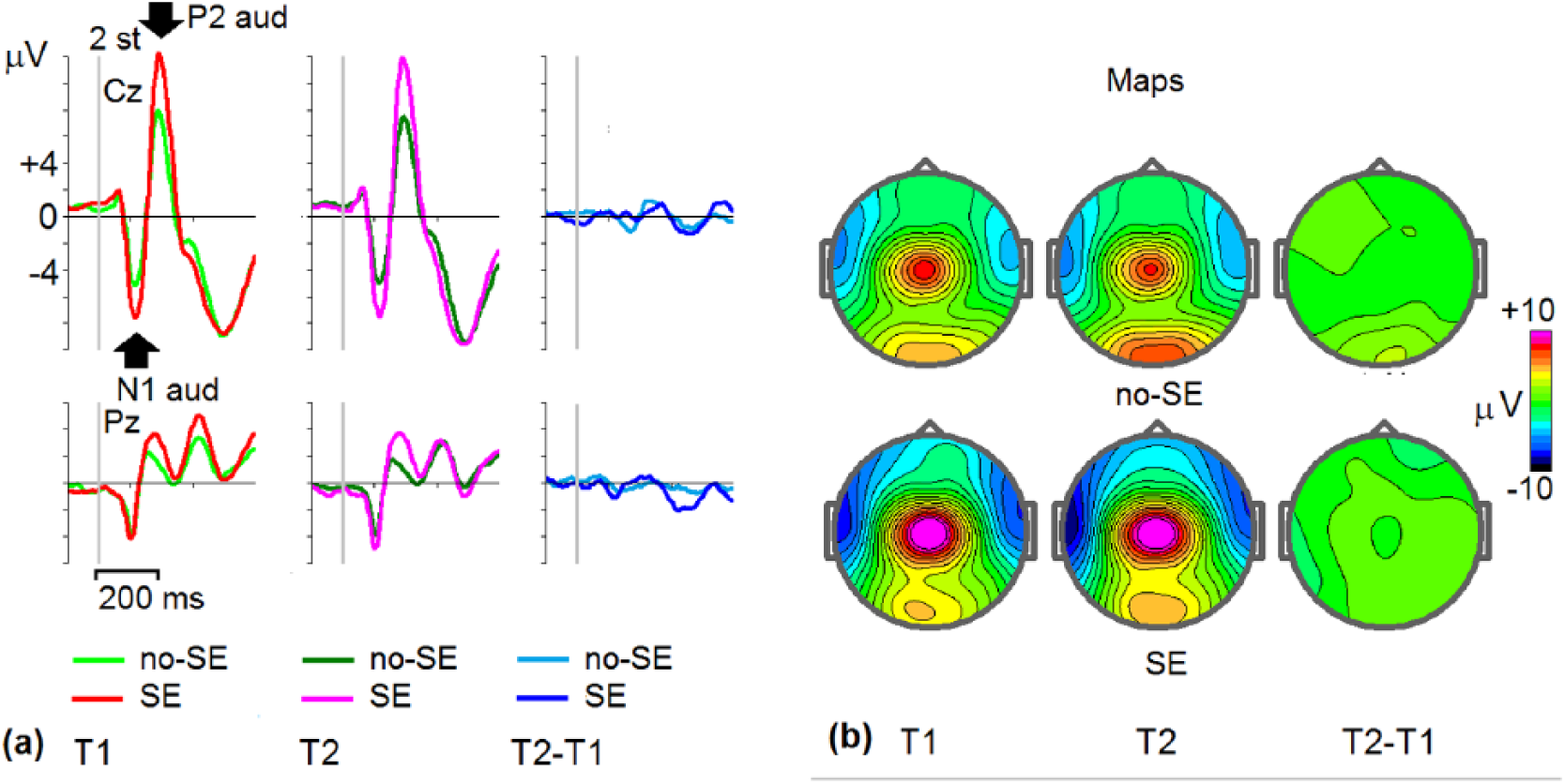
Grand-average event-related potentials (ERPs) in the group of patients with side effects (SE) and no-side effects (no-SE). (a) Grand-average ERPs to novelty at Cz and Pz electrodes for SEs and no-SEs taken at T1 (light green, no-SE; red, SE), T2 (dark green, no-SE; pink, SE), and T2 − T1 differences (light blue, no-SE; dark blue, SE). Maps of ERPs for SEs and non-SEs taken at maximums of P3 NOGO waves indicated by arrows at (a).
The SE index scale (sum of the weighted quartile scores of 4 variables) differed significantly between SEs and no-SEs, P < .001***, with large ES (d = 1.08; younger group, d = 0.98; older group, d = 1.27). The accuracy of the scale was 0.78, in the “fair” category (0.70-0.80); younger group = 0.75, older group = 0.83; sensitivity = 85%, specificity = 70%.
We also computed a scale based on the 2 variables at T1: P < .001***; younger, P = .01**; older, P = .001***; ES total, d = 0.94; younger group, d = 0.81; older group, d = 1.09. Overall accuracy was 0.74; younger group, 0.72; older group, 0.77.
Discussion
The aim of this study was to develop 2 composite scales: one for predicting clinical gains from stimulant medication in pediatric ADHD and another for predicting risk of acute side effects. The scales combined measures from EEG spectra, ERPs and behavioral data from a cued visual GO/NOGO task designed specifically for studying components of cognitive control and performed by ADHD patients twice: at T1 without medication and at T2 on a single dose of stimulant medication given a few days before the 4-week trial. Combining EEG spectra, ERPs and behavioral variables into a global composite score makes prediction of clinical gains with effect size d = 1.86 (younger group = 1.57; older group = 2.24). Because many studies, including our own, show that the number of nonresponders increases with age, we believe that the effect size seen in the older group is particularly interesting. As a general clinical principle, we prefer to base decisions on several sources of information.
The predictive power of the global SE variable was not as strong as for the global RE variable, but d =1.08 (younger 0.98, older 1.27) is larger than d = 0.80, which is suggested as a necessary criterion for clinical applications. 27
Relationship to Theory and Previous Research
In our previous study, 21 predictors of clinical gains were based on T1. These findings indicate that the stimulant responders had dysfunctions related to the frontal executive system, whereas the dysfunctions of the non-REs primarily seemed to be associated with posterior areas of the brain, not relying on dopamine as strongly as the frontal areas. Applying independent component analysis (ICA) to the cue P3 component, we found close-to-normal values in REs, but a significantly smaller component in non-REs. In the current study and in the “single-dose-study,” 25 we confirm the findings that reduced function of the frontal executive system, as reflected in reduced amplitudes of the P3 NOGO and CNV components, predicts a positive medication response.
In the current study, REs and non-REs differ significantly in the younger group in posterior theta/alpha ratio, in accordance with studies finding excess theta in ADHD to be related to a positive medication response. 41 The increased level of posterior alpha in non-REs may reflect underactivation of brain areas involved in processing of visual stimuli. 27 Excess posterior alpha is characterized as one of several qEEG-based subtypes of ADHD, and may represent posterior hypoarousal.27(p308) We speculate that patients in this subgroup may not be the best candidates for stimulant medication.
The theory of anterior and posterior attention systems is widely accepted 42 and corresponds to Posner’s theory of 3 attention networks43,44 responsible for alerting, orienting, and executive control. It has been shown that the alerting and executive networks can be influenced by stimulants. The orienting network is associated with superior parietal lobe activation. This network may be the site of primary deficits in non-REs.45,46 According to Arnsten, 47 posterior brain areas assess the salience of stimuli. The prefrontal cortex (PFC) regulates attention based on relevance/meaning, screening distractions, sustaining, shifting, and dividing attention. The PFC is sensitive to changes in dopamine and norepinephrine, requiring optimal levels for optimal executive function and supporting the view that ADHD patients with deficits related to the anterior attention system are good candidates for stimulant medication. Posterior attention deficits may require different treatments.
In our previous study on side effects, 15 we compared SEs, no-SEs, and healthy controls. We argued that all 3 variables contributing to prediction were related to cortical activation; the SE group was not underactivated. SEs had a near-normal response preparation (ICA CNV late), their emotional activation was stronger than normal (increased ICA ERP visual early component, generated in posterior cingulate cortex), and their reaction time was faster than that of the no-SE group.
In the current study, we also found that the SE group was faster at T1, and their novelty component was stronger—perhaps an indication of an alerted, activated state.
Limitations of the Study
Predictors based on a specific group of patients should be tested on an independent sample. This has not been done so far. The majority of cases referred to our clinic are complicated cases, probably with more comorbidity than usually seen, underscoring the need of replication studies. The data are generated in a clinical setting, making proper blinding difficult. External experts should ideally complete the evaluation of clinical gains and side effects in a future study. Inclusion of 8 patients on DEX can be criticized, but they were not significantly different from the rest regarding the distribution of REs versus non-REs and SEs versus no-SEs. Drug studies usually include placebo to control for expectation effects. In this study conclusions are based on reports from parents, children and teachers, and it seems unlikely that expectations differ significantly among the groups.
Clinical Implications
ADHD is a heterogeneous disorder, and the neural mechanisms behind the symptoms are not always the same across patients. This is probably a key reason that 25% to 30% are considered nonresponders to stimulants. The trial-and-error method based on questionnaires presently used to find the right medication is time consuming and often surrounded by uncertainty, because informants may disagree. Evidence-based predictors of medication response represent a useful supplement. Evaluation of ongoing medication could also be improved by applying the single-dose method described in this article. Predictors should also be in accordance with accepted theory and previous research. The variables that we combine into global indices in this study are in accordance with accepted theory and published research.
Supplemental Material
Global_study-supplementary_file-050518 – Supplemental material for Predicting Clinical Gains and Side Effects of Stimulant Medication in Pediatric Attention-Deficit/Hyperactivity Disorder by Combining Measures From qEEG and ERPs in a Cued GO/NOGO Task
Supplemental material, Global_study-supplementary_file-050518 for Predicting Clinical Gains and Side Effects of Stimulant Medication in Pediatric Attention-Deficit/Hyperactivity Disorder by Combining Measures From qEEG and ERPs in a Cued GO/NOGO Task by Geir Ogrim and Juri D. Kropotov in Clinical EEG and Neuroscience
Footnotes
Acknowledgements
Coauthors of a previous publication, Jan F. Brunner and Ida E. Aasen, Department of Neuropsychology, Helgeland Hospital, 8656 Mosjøen, Norway, contributed significantly to ERP calculations.
Author Contributions
Dr. Ogrim tested the patients, completed the statistical analyses supported by a statistical adviser, and wrote most of the paper. Professor Kropotov created the figures, wrote the paragraphs related to ERPs and reviewed the paper, suggesting changes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Østfold Hospital Trust, Fredrikstad, Norway.
Supplemental material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
