Abstract
This study evaluates the clinical efficacy and safety of intravenous allogeneic mesenchymal stem cell (MSC) therapy as an adjunct to standard immunosuppressive treatment in dogs diagnosed with primary immune-mediated hemolytic anemia (IMHA), particularly in cases with inadequate response to conventional immunosuppressive protocols. A total of 157 client-owned dogs with IMHA received MSC therapy at Safari Veterinary Care Center or collaborating clinics. A core cohort of 43 dogs met strict inclusion criteria: confirmed primary IMHA, no significant comorbidities, and receipt of at least two MSC treatments. A retrospective analysis was performed on medical records of treated dogs. Packed cell volume (PCV) trends were used to assess treatment response, with success defined as sustained improvement to ≥30% PCV following MSC therapy. Safety was assessed by reviewing for adverse reactions postinfusion. In the core cohort, 76% (33/43) of dogs achieved successful hematological recovery. Across the broader cohort, clinical improvement was also observed, particularly in dogs with poor initial response to standard therapy. No serious adverse events were recorded following MSC infusion. Allogeneic MSC therapy appears to be a well-tolerated and potentially effective adjunctive treatment for canine IMHA, particularly in refractory cases. These findings support further investigation in controlled clinical trials.
Introduction
Background and clinical challenge
Immune-mediated hemolytic anemia (IMHA) is a life-threatening autoimmune disorder, which destroys red blood cells (RBCs) through aberrant immune responses. In affected dogs, the immune system targets RBC surface antigens, resulting in premature hemolysis that occurs either within the bloodstream through complement activation (intravascular hemolysis) or in organs such as the spleen and liver via phagocytosis by macrophages (extravascular hemolysis). 1 The resulting anemia is often severe and rapidly progressive, with mortality rates reaching approximately 25% in extravascular cases and up to 75% in intravascular IMHA cases. 2
Traditional therapies for IMHA focus on immunosuppressive medications, including corticosteroids, cyclosporine, azathioprine, or mycophenolate mofetil. While these treatments can be life-saving for many patients, they often fail to correct the underlying immune dysfunction leading to frequent relapses, infectious complications, and chronic morbidity. For many dog owners, this leaves few options beyond prolonged illness, drug-related side effects, or euthanasia. 2
Mechanisms of IMHA
In healthy dogs, RBCs have a lifespan of ∼100 days, with approximately 600 billion cells produced daily. IMHA disrupts this equilibrium when autoantibodies (typically IgG or IgM) bind to RBC antigens causing rapid hemolysis. In intravascular hemolysis, complement-mediated lysis causes destruction of RBCs within blood vessels. In extravascular IMHA, antibody-tagged RBCs are phagocytosed by splenic or hepatic macrophages, resulting in anemia and the appearance of spherocytes on blood smears. 1,2 Chronic red cell destruction exacerbates systemic inflammation, thromboembolism, and multiorgan dysfunction—especially involving the lungs, liver, and kidneys.
IMHA may be classified as
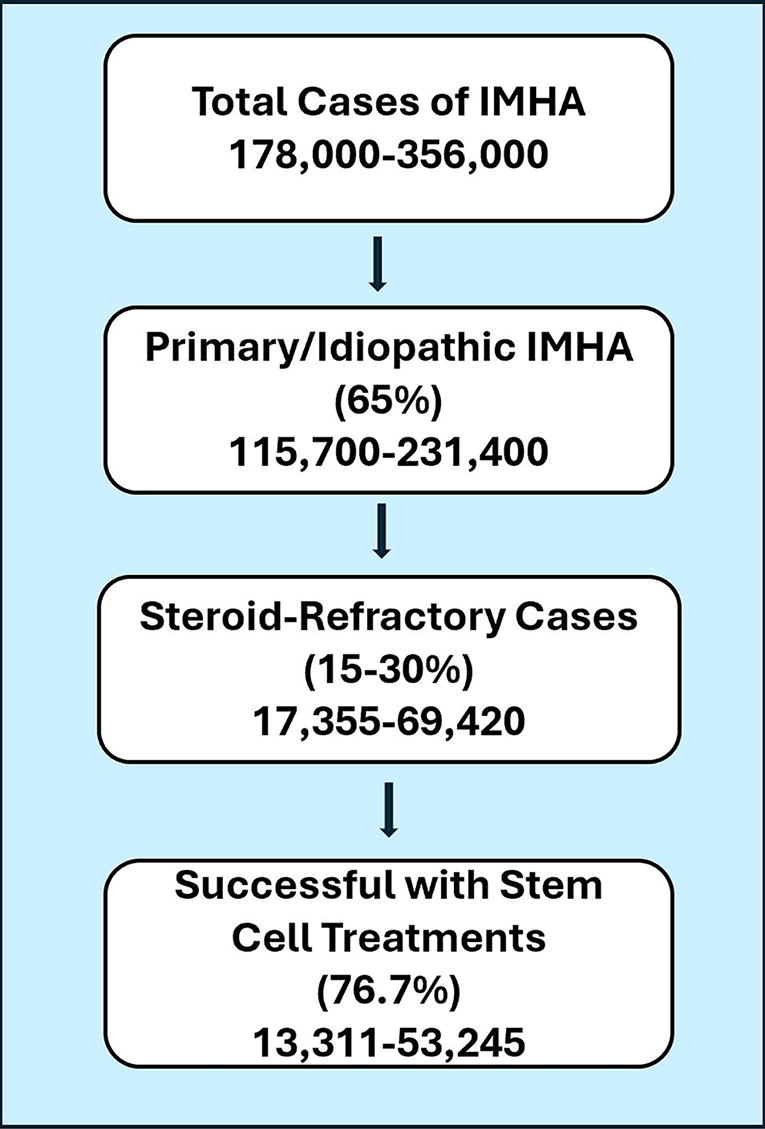
Flow chart representing the subsets of IMHA incidences in the United States per year. IMHA, immune-mediated hemolytic anemia.
Rationale for stem cell therapy
MSCs have emerged as a promising treatment for autoimmune diseases capable of modulating immune responses without broad immunosuppression. These cells exert therapeutic effects through modulating immune cell activity (paracrine signaling), regulatory T-cell activation, inhibition of proinflammatory cytokines, and restored immune tolerance. 4 Unlike conventional drugs, MSCs adaptively respond to the local immune environment, reducing the risk of overtreatment or systemic toxicity.
In the context of IMHA, MSC therapy offers a 2-fold benefit: (1) modulation of autoreactive immune responses to halt antibody production by suppression of the immune system’s attack on RBCs and (2) protection of antibody-tagged RBCs from accelerated destruction. 5 These mechanisms are especially valuable for patients with poor or partial response to immunosuppressive drugs.
Immune tolerance failure
The immune system is a dynamic network of specialized cells designed to protect the body from infections, abnormal cells, and other threats. Among its key players are lymphocytes, which are responsible for recognizing and responding to specific foreign antigens. Their development, selection, activation, and regulation are tightly controlled processes that, when disrupted, can lead to autoimmune diseases such as IMHA.
Immune tolerance re-education
The breakdown of immune tolerance is central to the pathogenesis of IMHA. These mechanistic insights establish a biologically grounded basis for using MSCs in the treatment of IMHA and inform the dosing rationale.
MSCs are responsive cells that exert potent immunomodulatory effects. They secrete transforming growth factor beta (TGF-β) and complement regulatory proteins such as Factor H. TGF-β plays a central role in promoting the development, differentiation, and expansion of regulatory T-cells (Tregs), thereby restoring immune tolerance and preventing inappropriate immune activation that occurs in autoimmune diseases. 5 Concurrently, MSCs produce Factor H, which inhibits complement activation on RBCs. This helps prevent complement-mediated lysis and protects erythrocytes from destruction. 6,7
Recent studies have further clarified MSC mechanisms, showing that MSCs and the newly minted Tregs they produce act by:
The therapeutic strategy in this retrospective study mirrors a “vaccination” model for immune re-education. The initial MSC dose was intended to rapidly suppress active RBC destruction through anti-inflammatory and complement-inhibitory actions, while simultaneously initiating Treg recruitment. Subsequent booster doses were designed to reinforce and expand memory T-cell populations, fostering long-term immune tolerance rather than transient suppression.
MSC dosing rationale
A dosing regimen of 2 million cells/kg body weight was employed. This protocol was based on pilot data demonstrating superior clinical efficacy compared with the more traditional 1 million cells/kg standard used in human protocols. The rationale for higher cell dosages included
Emerging evidence suggests a dose-dependent relationship in MSC therapies, where larger cell numbers promote more robust Treg expansion and longer-lasting immune regulation, particularly in refractory autoimmune diseases. 10,11 Thus, the selection of a 2 million cells/kg dosing protocol was both biologically rational and clinically validated for achieving durable therapeutic success in canine IMHA.
Study objective
This study retrospectively evaluates the clinical efficacy and safety of intravenous allogeneic MSC therapy in dogs diagnosed with IMHA, most of whom exhibited inadequate response to conventional immunosuppressive treatments. A defined core cohort meeting strict inclusion criteria was analyzed to assess MSC dose frequency, hematological recovery, and treatment-related adverse events.
Materials and Methods
Stem cell preparation and quality control
Adipose-derived MSCs were harvested from purpose-screened donor dogs following informed owner consent. Donors were healthy adults screened via complete blood count, serum biochemistry, and infectious disease testing.
MSCs were isolated by enzymatic digestion followed by adherence selection and then expanded to passages 3–6 before cryopreservation. The isolated MSCs were cultured in Dulbecco's modified Eagle medium: Nutrient Mixture F-12 media supplemented with 5% fetal bovine serum and 1% penicillin-streptomycin. Cells were maintained at 37°C, 5% CO2, and 95% humidity.
Identity was confirmed by flow cytometry (CD73+, CD90+; CD34−, CD45−). Cells were also characterized through induced differentiation with Thermo Fisher’s StemPro differentiation kits. Sterility testing, mycoplasma testing, and viability assays (>85% postthaw) were performed (see supplementary data for further results).
Study design and patient selection
This study was designed as a retrospective, multicenter cohort analysis of 157 client-owned dogs diagnosed with IMHA and treated with allogeneic MSC therapy. Medical records were collected from both general practice and specialty veterinary hospitals between 2019 and 2024. The study sought to evaluate clinical outcomes and treatment safety in actual veterinary settings.
Dogs were eligible for inclusion if they had a confirmed diagnosis of IMHA based on clinical findings, hematological parameters (e.g., regenerative anemia, spherocytosis, autoagglutination, or positive Coombs’s test), and no evidence of secondary causes. All patients received traditional immunosuppressive therapy—corticosteroids alone or in combination with agents such as cyclosporine or mycophenolate mofetil—and many also underwent blood transfusions prior to or during MSC administration. A substantial proportion of patients were refractory to these conventional treatments at the time stem cell therapy was initiated.
For focused outcome analysis, a core cohort of 43 dogs was selected based on: Confirmed Receipt of at least Availability of complete hematologic follow-up, including serial packed cell volume (PCV) data.
The ages ranged from 10 months to 12 years and 2 months (Table 1). Out of the 43 patients, 61.8% were treated externally at referring hospitals and 38.2% were treated at Safari Veterinary Care Centers.
Accepted Patient Information
mo, month; yr, year.
MSC therapy protocol
Each dog received MSCs intravenously at a standardized dose of
Ethical approval and informed consent
This study was conducted at Safari Veterinary Care Centers, a private veterinary hospital. As the study involved clinical treatment of client-owned dogs with allogenic MSCs, formal Institutional Animal Care and Use Committee approval was not applicable. All treatments were performed with the informed, written consent of each pet owner, following a detailed discussion of the procedure, potential risks, and benefits. All procedures adhered to the ethical standards of the American Veterinary Medical Association and applicable veterinary regulations.
Results
Outcome measures
Medical records were reviewed for baseline PCV, immunosuppressive treatment history, MSC administration dates and doses, and follow-up PCV outcomes. The
In the broader cohort of 157 dogs treated with MSCs, 35 were excluded due to insufficient hematological data (e.g., missing PCV values or platelet-only records). Among the remaining 122 dogs, 27 died or were euthanized before the evaluation of treatment response could be conducted, leaving 95 evaluable cases. Of these, 68 dogs achieved a PCV ≥30%, yielding a therapeutic success rate of 72% in the more inclusive population, which encompassed dogs with comorbidities and those who received only a single MSC dose. This highlights the potential benefits of MSC therapy even outside the core analysis cohort.
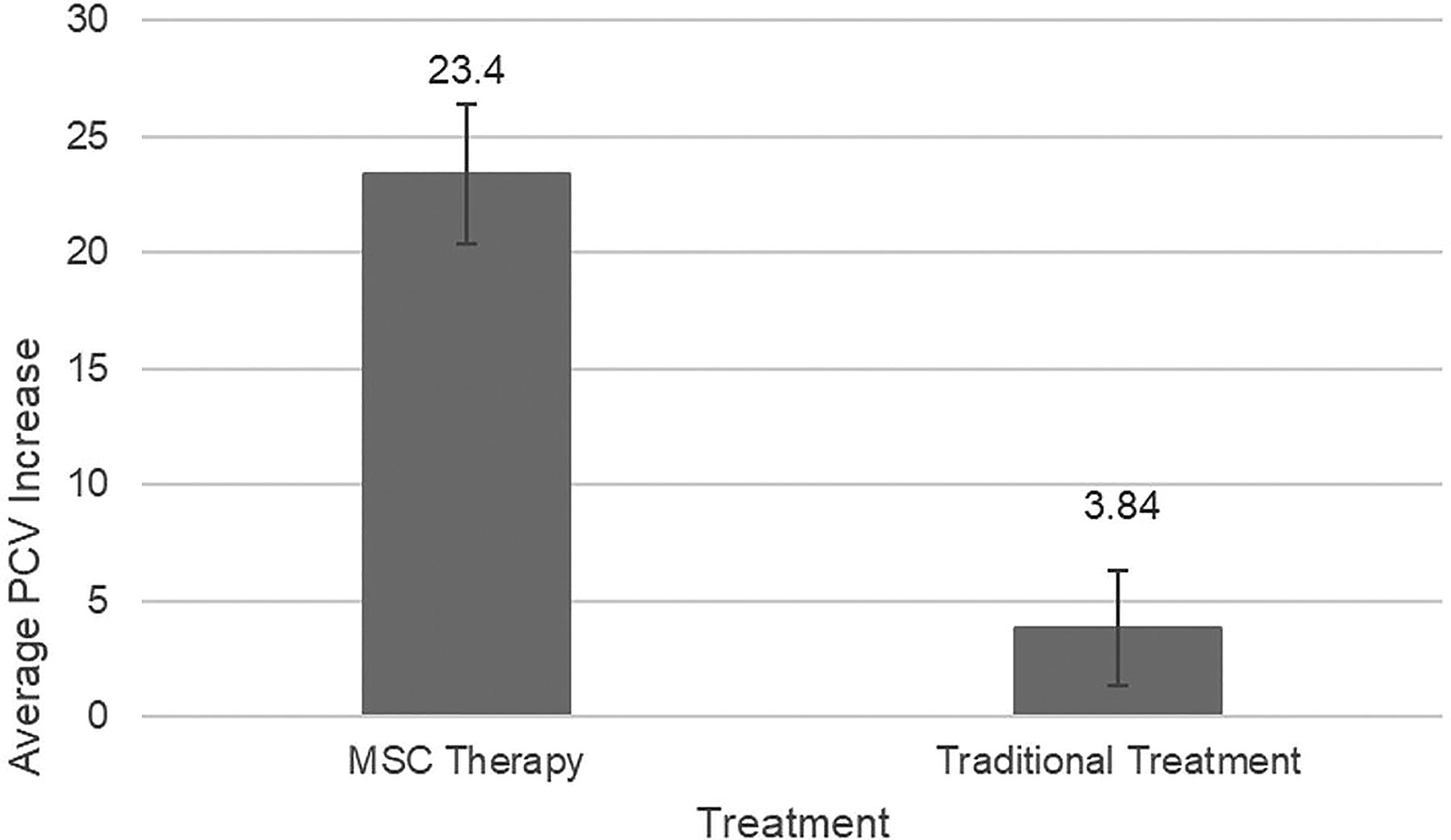
Increase in PCV after MSC therapy versus traditional treatments. MSC, mesenchymal stem cell; PCV, packed cell volume.
Number of MSC rounds
Most dogs in the study received two rounds of MSC therapy, with 68.9% (20/29) achieving a post-treatment PCV ≥30% and an average PCV increase of 23.5 points, the highest observed among all dosing groups. Dogs receiving three rounds also responded well, with a 77.8% success rate (7/9) and a mean PCV increase of 23.2 points. A small number of outliers received five or more rounds, but due to limited sample size, these were excluded from comparative analysis (Table 2).
Number of IV MSC Rounds and Their Success Rates
MSC, mesenchymal stem cell; PCV, packed cell volume.
Due to only one patient having either five or eight rounds, they were not considered statistically significant.
Safety assessment
Adverse reactions were identified through retrospective review of clinical notes. Among the 157 dogs treated with stem cell therapy for IMHA, 9 dogs (5.7%) experienced one or more adverse events. Eighteen adverse event instances were documented across these nine dogs, including symptoms such as fever, collapse, diarrhea, and transient seizure activity. No serious adverse events or deaths were observed or attributed to MSC therapy. All adverse events were mild to moderate in severity and resolved with supportive care. Deaths attributable to underlying IMHA severity were not classified as treatment-related adverse events.
Discussion
Administration of MSCs was associated with meaningful clinical improvement and a favorable safety. MSC treatment resulted in substantially greater increases in PCV and higher rates of therapeutic success. The consistency of outcomes across multiple hospital settings—both internal and external—supports the potential utility of MSC therapy as an effective adjunct in the management of canine IMHA.
The primary aim of this case series was to investigate the effectiveness of MSC therapy in the treatment of IMHA in dogs, with a specific focus on achieving immune modulation and restoring immune tolerance. 15 The results indicate a notable rate of successful outcomes among the treated dogs. This outcome is particularly encouraging, suggesting that MSC therapy can serve as a valuable intervention in managing IMHA, especially when conventional treatments have proven inadequate. 16 This study demonstrates that intravenous administration of allogeneic MSCs is a clinically effective and safe strategy for canine IMHA management.
Durable immune modulation
MSC therapy offers a
Dose response and clinical implications
The observed correlation between the number of MSC therapy rounds and the improvement in PCV levels highlights the importance of treatment consistency. Specifically, two to three rounds of MSC therapy were associated with the most significant enhancements in PCV, suggesting that repeated exposure to MSCs may be necessary to achieve a sustained therapeutic effect. This repeated exposure may foster long-term remission by promoting regulatory and memory T-cell populations and more permanent immune tolerance mechanisms. 21
The administration of two to three rounds further confirmed that the “vaccination” model of administration is effective in this case. It is hypothesized that the first round of stem cells given is to mitigate the acute destruction of RBCs through anti-inflammatory paracrine factors. 22 The destruction of RBCs may also be mitigated through the inhibition of the complement system against RBCs. Stem cells have been shown to secrete Factor H, which acts as a protective barrier against complement activation. 23 The second to third “booster” rounds of MSC therapy are then able to restore immune tolerance. Further exploration into the specific mechanisms driving this response is warranted, potentially uncovering insights into ideal treatment intervals and dosing strategies.
Treatment scalability and safety profile
Importantly, shipment of cells resulted in MSC therapy that was consistent across clinical settings, reinforcing the broader scalability and accessibility of this approach—a critical consideration for broader clinical adoption.
The study protocol provided a standardized framework for dosing (2 million cells/kg) and administration, minimizing variability. The low incidence of adverse events, predominantly mild infusion reactions, and no treatment-related deaths suggests that MSC therapy can be safely integrated into IMHA treatment protocols. Overall, the safety profile of MSC therapy, combined with its potential for immune modulation and restoration of immune tolerance, positions it as a promising treatment modality for IMHA in dogs.
Future directions
Several future directions are essential to advancing the clinical adoption of MSC therapy for IMHA. First, randomized controlled trials are needed to validate these outcomes in a statistically rigorous framework and account for confounding variables. Comparative investigations should also evaluate MSC therapy as an adjunct to conventional immunosuppressants, potentially identifying synergistic regimens that reduce the need for chronic pharmaceutical intervention.
Beyond IMHA, there is strong rationale to expand the application of MSC therapy to other autoimmune conditions including immune-mediated thrombocytopenia, Evan’s syndrome, and inflammatory bowel disease. Such studies will help define disease-specific protocols and broaden the therapeutic scope of MSCs in veterinary medicine.
Equally important is the standardization of cell manufacturing practices. As regulatory scrutiny increases, harmonizing MSC production under current good manufacturing practice standards will be critical for FDA Center for Veterinary Medicine recognition and eventual product licensing. Safari stem cell’s current protocols, including rigorous donor screening, validated cryopreservation, and consistent cold-chain logistics, lay the groundwork for this transition. The end goal is to bring these technologies to the private practitioner.
Conclusion
Intravenous allogeneic MSC therapy restores immune tolerance, improves hematological outcomes in canine IMHA, and has a strong safety profile. By promoting the recruitment and expansion of regulatory T-cells, inhibiting complement activation, and reducing the inflammatory destruction of RBCs, MSCs target the immunological cause of IMHA. This represents a meaningful shift from traditional generalized immune suppression toward targeted immune recalibration. Moreover, the therapy’s ability to be shipped without loss of efficacy supports clinical feasibility of this approach, marking an advancement in the accessibility of regenerative treatments. These findings support MSC therapy as an adjunctive treatment for canine IMHA and lay the foundation for adoption in veterinary regenerative medicine.
Footnotes
Acknowledgments
The following individuals associated with Safari Stem Cell and Safari Veterinary Care Centers were contributors to this project: Seymone Batiste, Melinda Wharton, Michael McCord, Susan Mooney, and Lori McCord.
Author Disclosure Statement
The authors were both affiliated with Safari Veterinary Care Centers at the time of the study.
Funding Information
No funding was received for this article.
Supplementary Material
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
