Abstract
Background and Purpose
This Point of View paper offers a commentary on challenges and opportunities discussed during the 6th International Brain Stimulation Conference held in February 2025 in Kobe, Japan, with a focus on the clinical application of repetitive transcranial magnetic stimulation (rTMS) in post-stroke rehabilitation. We argue that the major barrier lies in the field’s overreliance on standardized, one-size-fits-all protocols and its reluctance to embrace personalization in the pursuit of precision.
Results
During the conference, 2 research cultures were evident: the “Systematicists,” who rely on conventional clinical trials, and the “Personalizers,” who tailor non-invasive brain stimulation (NIBS) protocols to individual patient characteristics. This dichotomy reflects a broader challenge: how can we reconcile the need for standardization with the demand for personalization in translational research? The future of NIBS may lie in patient-specific, biomarker-driven neuromodulatory protocols that incorporate deep phenotyping and brain state-dependent stimulation, such as closed-loop TMS guided by Hebbian plasticity principles. This approach recognizes that post-stroke recovering brain is a 4-dimensional structure, shaped by space and time, which contributes to substantial intra- and inter-individual variability.
Conclusion
Understanding how NIBS interacts with each uniquely recovering brain is essential. Addressing this complexity remains a challenge for designing rigorous clinical trials and moving the field closer to effective, personalized integration in stroke rehabilitation. By delineating key components of personalization, we aim to reframe the discussion from “if” NIBS works to “for whom, for what and why, for where and when, and how” it can facilitate clinically meaningful recovery.
Keywords
Introduction
The 6th International Brain Stimulation Conference, held in Kobe, Japan from 12 to 16 February 2025, opened with a plenary lecture by Prof. Marom Bikson, who introduced a printable, disposable electrochemical technology for neuromodulation that operates without electronic components, envisioning a future where non-invasive brain stimulation (NIBS) could be dispensed in pharmacies and carried in patients’ pockets. 1 If a pill can alleviate depressive symptoms, why should not a physician prescribe a portable, non-invasive, self-discharging adhesive band as an alternative? A similar principle could apply to neurorehabilitation, potentially enabling individuals with stroke to improve motor symptoms. Yet before imagining a future where NIBS is prescribed as routinely as pharmacotherapy, it is crucial to recognize that, despite over 2 decades of research,2,3 NIBS has yet to become a standard component of patient care.
This point of view article reflects on the challenges and opportunities discussed at the conference regarding the clinical translation of NIBS for post-stroke upper limb recovery. In particular, the discussion centers on transcranial magnetic stimulation (TMS), which remains appealing due to its ability to directly modulate neuronal excitability in targeted cortical regions, 4 but has recently drawn criticism because of inconsistent findings in randomized controlled trials (RCTs). The article examines why the clinical implementation of TMS has not yet achieved widespread clinical adoption and discusses potential steps to help bridge the gap between research and routine care. We argue that this gap reflects not only methodological challenges but also a deeper conceptual issue: the historical field’s reliance on standardized, one-size-fits-all protocols that fail to account for the heterogeneity of post-stroke recovery. At the same time, advances in neuroimaging, electrophysiology, and computational modeling now provide unprecedented opportunities to move toward patient-specific, biomarker-driven neuromodulatory approaches.
During the conference, 2 distinct approaches became evident. The “
In this article, we therefore propose that meaningful clinical translation of NIBS will require creating practical models that operationalize personalization without abandoning the rigor of systematic research. We highlight emerging strategies to achieve this and most importantly, we propose that post-stroke recovering brain should be understood as 4-dimensional structure shaped by space (length, width, and depth) together with time as the fourth dimension, which gives rise to unique individual variability. By reframing the discussion in this way, we aim to shift the central question from “if” NIBS works to “for whom, for what and why, for where and when, and how.” it can facilitate clinically meaningful recovery in post-stroke rehabilitation.
The Problem
TMS is one of the most widely studied NIBS techniques. It modulates neuronal activity by delivering brief, high-intensity magnetic pulses through a coil positioned over the scalp. These pulses induce electric currents in the underlying cortical tissue, leading to depolarization of neurons and modulation of cortical excitability. Delivering repetitive pulses (rTMS) can produce longer-lasting changes in brain activity. Specifically, high-frequency rTMS (≥5 Hz) typically increases cortical excitability, whereas low-frequency rTMS (≤1 Hz) reduces it, through synaptic mechanisms resembling the long-term potentiation (LTP) and long-term depression (LTD). 3
In neuroscience research, rTMS has been extensively used in healthy adults to selectively and transiently modulate activity in specific brain regions, allowing researchers to infer causal links between neural circuits and behavior. This approach, sometimes called a “lesion” method, typically uses low-frequency rTMS to inhibit cortical regions and assess the resulting behavioral effects, much like a temporary off-switch. For instance, rTMS helps reveal 2 distinct cortical pathways for human body perception, that is, the fronto-parietal network for the upright body perception and the extrastriate body area (EBA) for the inverted body part processing. 5
In addition, TMS has been widely used to investigate the mechanisms and neural substrates underlying voluntary movement and motor learning by probing and modulating activity in specific cortical regions.6-9 These findings demonstrate that rTMS is a powerful experimental tool for investigating the functional architecture of the human brain and for inducing behavioral changes with potential therapeutic effects.
One of the most pressing challenges highlighted during the conference was the uncertain clinical impact of TMS in neurorehabilitation. Despite the 14 oral presentations and fifteen posters on NIBS and post-stroke upper limb recovery, as well as extensive ongoing research beyond the conference, 1 question remains:
Is the Evidence Strong Enough to Justify Integrating rTMS into Everyday Clinical Practice?
The application of rTMS in post-stroke motor rehabilitation has been primarily guided by the interhemispheric rivalry model, which posits that a poor process of poststroke motor recovery arises from a pathologically competitive neural circuit between the underactive ipsilesional motor cortex and the overactive contralesional motor cortex developed after unilateral brain damage. 10 Consequently, 2 possible rTMS strategies have emerged: (1) excitatory stimulation, such as high-frequency rTMS or intermittent theta burst stimulation (iTBS), targeting the ipsilesional motor cortex to enhance cortical excitability; or (2) inhibitory stimulation, such as low-frequency rTMS or continuous theta burst stimulation (cTBS), applied to the contralesional hemisphere to reduce interhemispheric imbalance. 11 While this model’s mechanistic plausibility was supported by several early proof-of-concept studies,2,7 meta-analyses of clinical RCTs consistently reveal substantial heterogeneity in recovery outcomes in patients receiving different rTMS interventions.12,13
The robust neuromodulatory effects of TMS on brain activity contrast with its variable impact on clinical outcomes in post-stroke populations, leaving its future pathway for translation and adoption unclear. While the U.S. Food and Drug Administration (FDA) has approved rTMS for specific psychiatric conditions14,15 its benefit and adoption in post-stroke motor neurorehabilitation remain limited.
While several conference sessions critically examined this translational gap, 1 particularly salient example was the navigated inhibitory rTMS to contralesional hemisphere (NICHE) multicenter RCT, which included 199 stroke participants, found no significant differences in upper limb function between the real and sham stimulation groups following therapy. 16 An extension of this study, the E-FIT multicenter RCT, replicated the NICHE stimulation protocol but used a sham coil that matched the appearance of the active coil, without delivering the weak electric field used in the NICHE. Unfortunately, the results were also replicated, showing no significant additional clinical benefit. 17 In response, the ongoing B-STARS 2 multicenter RCT was introduced as another phase III trial designed to reassess all these findings. This study employs cTBS and aims to enroll 454 participants randomized to receive either sham or active cTBS. To date, 100 participants have been recruited. 18
A key challenge underscored during the session was the high interindividual variability in response to NIBS. As Prof. Friedhelm Hummel emphasized, this variability may be influenced by multiple factors, including lesion characteristics, time post-stroke, stimulation parameters, and individual neurophysiological profiles. This heterogeneity complicates the design of standardized protocols and may contribute to the inconsistent outcomes observed across trials. Addressing this complexity is essential for optimizing treatment efficacy and advancing clinical adoption. 19
Throughout the conference, in symposiums and poster sessions on rehabilitation, 2 distinct research approaches emerged. The
Given the Variability in rTMS and Other NIBS, is the Field Ready for Large-Scale Phase III Trials That Can Truly Translate From Bench to Bedside?
There are key conceptual limitations in the prevailing clinical frameworks guiding rTMS. Although class A evidence supports the therapeutic efficacy of low-frequency (1 Hz) rTMS over the contralesional motor cortex in post-acute motor stroke, effect sizes remain moderate. These findings stem from small studies (n < 20) with high intersubject variability and limited clinical impact. A major concern in the field relates to the theoretical mechanism of rTMS interventions, particularly the persistent reliance on a “one-concept” approach, the assumption that simply increasing ipsilesional M1 excitability or suppressing contralesional M1 excitability, combined with rehabilitative therapy, will universally improve motor outcomes in stroke. 22 This oversimplification overlooks the complex and diverse nature of post-stroke recovery processes.
The Solution
In response to the challenges outlined above, ranging from variable clinical outcomes and patient heterogeneity to shortsighted theoretical frameworks, medicine is increasingly leaning toward precision approaches across evaluation, diagnosis, intervention, and monitoring. NIBS aspires to make a similar shift within neurorehabilitation. Precision rehabilitation in stroke has been defined as “applying the optimal type and dose of therapy at the ideal time to maximize return of function for individual patients.”
23
A growing body of research is examining this principle across the entire stroke continuum, from prediction of stroke events
24
to the characterization of individual profiles (commonly referred as
To support this shift, translational research should adopt a patient-specific, parameter-oriented approach, using personalized protocols based on deep phenotyping that integrates clinical, neuroimaging, and neurophysiological data.19,25 Crucially, this level of customization must account for the multiple variables that shape recovery within the personalized space. These include lesion location and extent, corticospinal tract integrity, stroke onset and stage of recovery, comorbidities, medications, concurrent non-pharmacological interventions, and stimulation parameters, as well as variability both between patients (inter-individual) and within the same patient over time (intra-individual). Recognizing these variables as central inputs, rather than confounding factors, is essential for developing clinically meaningful personalization neuromodulatory strategies. However, this level of individualization is complex to implement within standardized research designs, which require uniform interventions to ensure comparability and replicability.
Because all of these variables cannot realistically be addressed at once, beyond both practicality and the scope of most RCTs, emergent approaches are being proposed prioritizing specific factors as stepping stones toward personalization. Several projects presented at the conference illustrated this approach. For example, a promising avenue for refining TMS application is targeting specific brain states, such as oscillation phases. For instance, the negative peak (trough) of sensorimotor mu oscillation is associated with LTP-like plasticity, while the positive peak is linked to LTD-like plasticity. 27 Repetitive state-dependent stimulation over a specific brain state would lead to a robust LTP/LTD plasticity. 28 An ongoing multicenter trial is evaluating personalized brain state-dependent rTMS in subacute motor stroke, comparing it to sham stimulation and conventional, non-brain state-dependent rTMS. EEG-TMS-based brain state-dependent stimulation could revolutionize NIBS by maximizing therapeutic plasticity effects. 22
Similarly, Prof. Sarah Hussain presented a real-time, machine learning-driven EEG-TMS system that modulates brain states with 86–95% accuracy to enhance residual corticospinal tract function. 29 Closed-loop stimulation represents a promising path for individualized therapy and may lay the groundwork for future trials. Along these lines, Dr. Caroline Tscherpel emphasized concurrent TMS-EEG as a tool for personalized assessment of network pathology, aiming to optimize rehabilitation through biomarker-driven neuromodulatory interventions. 30
Particular attention should also be given to motor evoked potentials (MEPs), which have been shown to be predictive of early upper limb recovery. On top of the conventional MEP, a novel MEP-derived threshold matrix framework has been proposed to examine peri-threshold corticomotor activation. 31
Overall, the field presents a mix of promising and inconsistent findings. The challenge remains in translating neuromodulation into consistent and clinically meaningful outcomes, and in identifying the therapeutic components required across the post-stroke recovery spectrum. The key may not lie in universal application, but in tailoring NIBS to individual brain states while monitoring recovery with reliable biomarkers. Yet brain state is only 1 aspect of the broader personalized space of post-stroke recovery previously outlined earlier, a crucial variable, but not the only one. To operationalize personalization, we propose framing the clinical translation of NIBS around 4 guiding model questions: for whom, for what and why, for where and when, and how.
“For whom” addresses the first axis of stratification: patient-specific biological and clinical characteristics. These include lesion location, corticospinal tract integrity, comorbidities, medications, and concurrent rehabilitation programs, to identify subgroups most likely to benefit.
“For what and why” refers to the selection of neuromodulation modality, whether rTMS, transcranial direct current stimulation (tDCS), transcranial alternating current stimulation (tACS), or other techniques, recognizing that each interacts differently with the injured brain. The “why” addresses the intended therapeutic mechanism of the stimulation, for example, whether to excite, inhibit, or synchronize specific networks.
“For where and when” highlight the importance of unique neuroanatomical and functional brain structures that are not identical across individuals, owing to genetic factors and lifelong exposure to environmental influences. These differences also manifest within the same individual over time, particularly across the dynamic course of post-stroke recovery. The dimension of when also encompasses both the recovery phase (acute, subacute, or chronic) and temporal fluctuations in brain states. These time-sensitive windows, along with inter- and intra-individual anatomical and functional variability, represent critical opportunities for targeted neuromodulation to maximize neuroplasticity, as guided by EEG or neuroimaging derived biomarkers.
Finally, “how” concerns the optimization of stimulation parameters, such as frequency, for instance, how many sessions are sufficient, whether tolerance develops over time, and whether increasing intensity, such as the number of pulses or current (mA), can offset lower frequency to accelerate results. It also includes considerations of how sessions should be sustained over time to ensure clinically practical outcomes, the use of standardized electrode placements versus neuronavigation with or without neuroimaging, coil orientation, electrode montage, priming strategies, or even combinations of stimulation modalities, all aimed at fine-tuning therapeutic effects.
Together, whether used in full or in part, the guiding questions of “for whom, for what and why, for where and when, and how” offer a practical model for developing structured approaches to post-stroke precision rehabilitation in NIBS.
Recommendations
While research continues to refine its understanding of NIBS and its most effective implementation, clinical application has already begun. For instance, TMS is being used in some rehabilitation clinics for post-stroke upper limb recovery, despite ongoing uncertainties in outcomes and limitations in methodology. Although clinical guidelines have been established and emerging data suggest its potential for therapeutic integration,11,32,33 delivering TMS without a clear understanding of when, how, and for whom it works risks navigating uncertain ground.
A more nuanced view of brain recovery goes beyond 3-dimensional spatial structure (length, width, and depth). The fourth dimension, the passage of time, shapes plasticity through experience (Figure 1). Interindividual variability, then, can be viewed as an emergent property of this temporal dimension, reflecting the unique ways each brain responds and adapts.
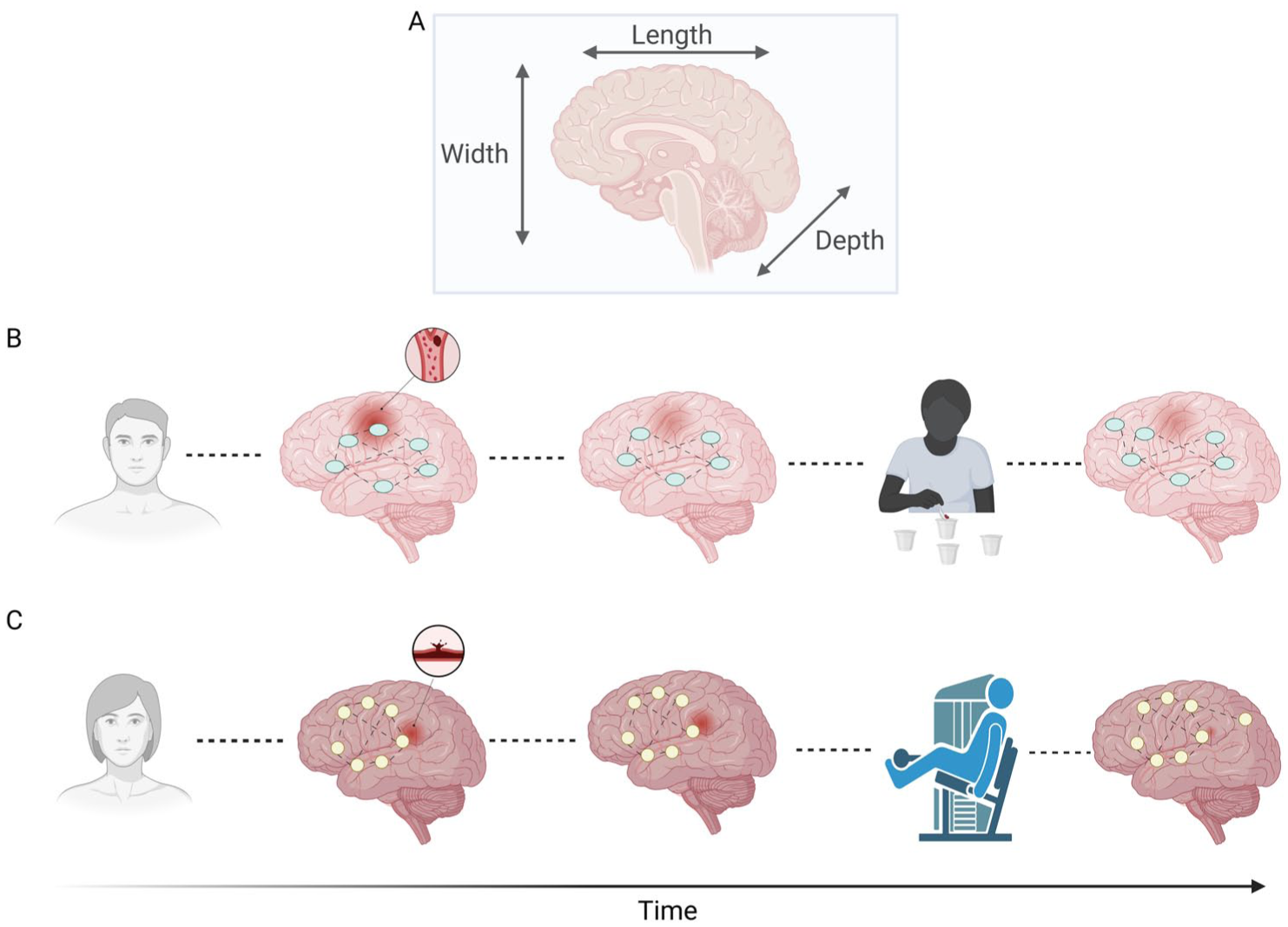
The 4-dimensional view of brain recovery. (A) Spatial dimensions of the brain (length, width, and depth). (B–C) Two individuals (different gender and age) with distinct brains who experience a stroke, each with different lesion locations and network changes. These panels illustrate how different post-stroke exposures, such as rehabilitation tailored to current impairments, interact with evolving brain connectivity and lesion characteristics over time. This progression highlights both interindividual variability in recovery trajectories and intraindividual variability when accounting for the passage of time. Both structural and functional changes can occur in the recovering brain, and each lesion in each brain is unique, as is its aftermath. In the stroke spectrum, if the central component is the person, then interventions should be designed to be central to the individual rather than to the stroke alone. Created with BioRender.
Variability is not merely a source of noise but may serve as a meaningful biomarker and a pillar for personalization models: greater variability could reflect an unstable system, while reduced variance could be associated with consistent motor performance or reduced abnormal neural signals, which may indicate reacquired control. In neuromodulatory terms, the potential effects of NIBS on variability warrant further exploration, particularly since it remains uncertain whether variability may increase following stimulation.
Building on this, the recovery journey of each stroke survivor is inherently individual. Given this multidimensional complexity, it is unlikely that a one-size-fits-all NIBS protocol will yield optimal results across patients. Each unique brain interacts differently with the type of NIBS technique and its stimulation parameters, such as duration, intensity, and electric field distribution. As such, a systematic application of NIBS in neurorehabilitation may or may not lead to therapeutic benefits. The true potential of NIBS likely resides in how it engages with this 4-dimensional model of brain recovery. Understanding these dynamic interactions is essential before NIBS can realistically become available in pharmacies, ideally in the not-so-distant future 1 though perhaps only in the more distant one if key uncertainties remain unresolved.
This point of view article addresses these questions previously raised by proposing a 4-dimensional model of post-stroke recovery of the brain for researchers to carefully consider when formulating hypotheses, and for clinicians when deciding whether to implement an rTMS or other NIBS-type protocol, or whether such an intervention is necessary at all, based on the current evidence and individual patient needs. In addition we propose a paradigm shift from protocol-centric to patient-centric neuromodulation through a guiding model structured around 5 core questions: “for whom,” “for what and why,” “for where and when,” and “how.”
We suggest that personalization in NIBS should be operationalized through measurable, stratifiable elements, which can be prioritized flexibly depending on the research or clinical context. As a first step, patient stratification (“for whom”) should focus on lesion type, stroke phase of recovery and demographics variables like age, gender, ongoing treatments, pre-existing medical conditions. This includes factors such as post-stroke depression, which is often excluded in research criteria but highly prevalent, as well as other psychiatric conditions, 34 leading to underrepresentation in study findings. 35 Thus, it is important to measure physiological indicators, such as MEPs, and structural measures, such as diffusion tensor imaging (DTI), to gain a clear picture of corticospinal tract integrity. Even more importantly, these metrics should ideally be collected longitudinally, before, during, and after the intervention, rather than cross-sectionally.
Second, defining “what and why” requires selecting NIBS modalities. This includes a rationale for choosing a specific form of TMS, or alternatives within transcranial electrical stimulation (tES), based on feasibility and mechanistic alignment with the intended outcomes. This process should be guided by the therapeutic aim, such as promoting functional recovery in targeted anatomical regions or inhibiting maladaptive compensation patterns.
Third, “where and when” can be guided by individualized neuroimaging or EEG biomarkers to determine accurate target site and timing. With access to a neurophysiological atlas that integrates structural, functional, and temporal information, more precise decisions can be made in both space and time to capitalize on neuroplastic windows.
Finally, “how” involves adapting stimulation parameters and delivery methods in response to individual biomarkers and feedback loops, in a continuously responsive and flexible manner based on the patient’s fluctuating neurophysiological profile.
Acknowledging that RCTs are the gold standard and one of the highest levels in the evidence pyramid, researchers should consider allowing more flexibility in trials using neuromodulation, given the high variability in post-stroke recovery. For that reason, future trials could adopt adaptive designs such as variable stimulation trials that allow parameter adjustments during the intervention, N-of-1 studies that capture individual responses over time, or stratified randomized controlled trials that isolate and evaluate specific components, or even case series o single-subject designs that provide greater methodological elasticity before progressing to full RCTs.
Once these personalized elements are systematically accounted for, we advocate for the creation and use of shared data banks that allow for inter-study comparisons across stroke types and conditions. This approach moves from individual stratification to population-level interpretation, enabling both the construction of personalized profiles and their alignment with broader patterns in post-stroke recovery. Such efforts could facilitate the development of a core set of personalization metrics, such as MEPs or mu rhythm phase in EEG.
This point of view article was originally inspired by findings presented in the proceedings and by debates emerging during the panel sessions at the conference on stroke rehabilitation. As such, some relevant information may not be included in this manuscript, but these proceedings fueled our reflection on the challenges that remain in translating non-invasive brain stimulation into clinical practice. We subsequently searched for additional published evidence to complement and expand upon the ideas developed in this article. Although the primary focus is on TMS, given its status as the most established NIBS technique and the substantial resources allocated to high-quality trials, other non-invasive brain stimulation methods were also presented during the conference (Table 1), such as tDCS, transcutaneous auricular vagus nerve stimulation (taVNS), and low-intensity focused ultrasound (LIFU).
Proceedings Presented at the 6th International Brain Stimulation Conference Related to Non-Invasive Brain Stimulation (NIBS) for Post-stroke Upper Limb Motor Recovery.
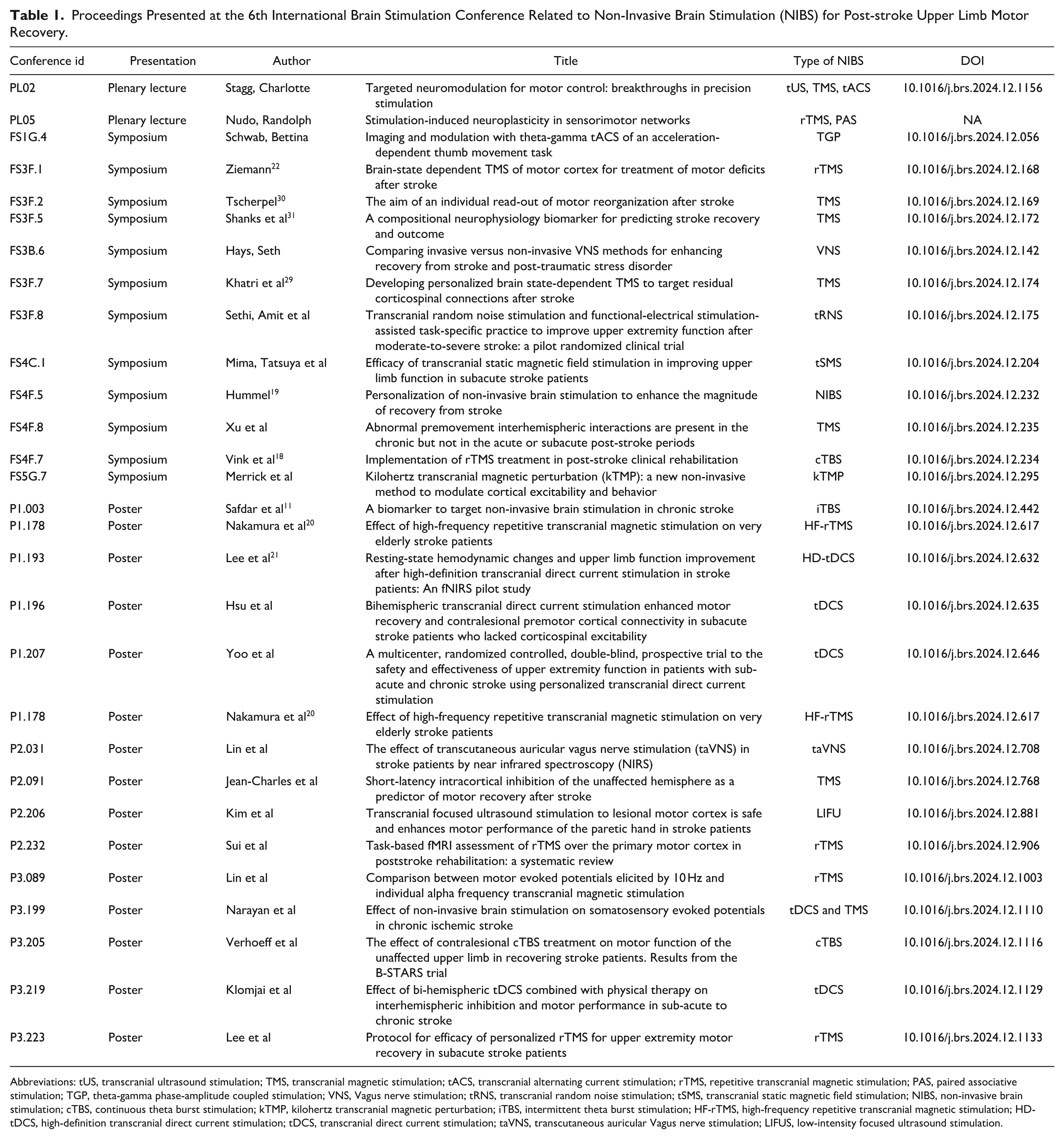
Abbreviations: tUS, transcranial ultrasound stimulation; TMS, transcranial magnetic stimulation; tACS, transcranial alternating current stimulation; rTMS, repetitive transcranial magnetic stimulation; PAS, paired associative stimulation; TGP, theta-gamma phase-amplitude coupled stimulation; VNS, Vagus nerve stimulation; tRNS, transcranial random noise stimulation; tSMS, transcranial static magnetic field stimulation; NIBS, non-invasive brain stimulation; cTBS, continuous theta burst stimulation; kTMP, kilohertz transcranial magnetic perturbation; iTBS, intermittent theta burst stimulation; HF-rTMS, high-frequency repetitive transcranial magnetic stimulation; HD-tDCS, high-definition transcranial direct current stimulation; tDCS, transcranial direct current stimulation; taVNS, transcutaneous auricular Vagus nerve stimulation; LIFUS, low-intensity focused ultrasound stimulation.
Ongoing research beyond the conference continues to explore the combined use of multiple NIBS approaches for post-stroke motor rehabilitation, such as pairing high-definition tDCS (HD-tDCS) with electrical theta burst stimulation (eTBS). This strategy aims to integrate the therapeutic advantages of both modalities, enhancing stimulation focality and practicality and has shown encouraging results in sham-controlled study. 36
One notable recent contribution is the TRANSPORT2 study, a phase II multicentre randomized controlled trial that investigated the effects of tDCS combined with one of the most evidence-based interventions for motor recovery: modified constraint-induced movement therapy (mCIMT). The trial enrolled 129 stroke survivors, between 1 and 6 months post-stroke, who were randomized to receive sham stimulation, 2 mA, or 4 mA of bi-hemispheric tDCS during the first 30 minutes of therapy, followed by 120 minutes of active mCIMT per session, delivered over ten sessions within a 2-week period. The results showed no added benefit from tDCS. 37 Unfortunately, this well-designed study reflects the findings of the multicenter TMS trials discussed earlier, raising further concern within the scientific community about whether NIBS can ultimately provide a meaningful translational clinical benefit in stroke rehabilitation.
It is also worth noting that further preclinical studies using animal models focused on the customization of NIBS for motor restoration may help address key uncertainties and support the design of future phase III clinical trials in human stroke survivors. This is especially important given the considerable difficulty, and, at times, the unrealistic nature of conducting high-quality, evidence-based trials involving NIBS and other non-pharmacological therapies in a robust manner, particularly in diverse stroke survivor populations.
As the field advances, careful clinical translation grounded in individualized science will be key to ensuring that NIBS delivers functional motor restoration in post-stroke upper limb rehabilitation. A major challenge ahead is designing protocols and clinical trials that can rigorously evaluate complex, personalized interventions, without narrowly restricting inclusion criteria. Stroke survivors often present with diverse comorbidities and varied recovery trajectories, which must be reflected in study populations to ensure clinical relevance. Embracing this heterogeneity aligns with the broader, emerging shift toward personalized, patient-centered medicine. Doing so will support a more comprehensive understanding of how NIBS interacts with real-world post-stroke recovery.
Footnotes
Author Note
During the preparation of this manuscript, the authors used ChatGPT-4 for language editing to enhance readability and clarity. During the peer-review process, the authors utilized ChatGPT-5 to address reviewers’ comments and to draft and integrate responses into the revised manuscript. After employing these tools, the authors thoroughly reviewed and edited the content as necessary and take full responsibility for the final version of the published article. Plagiarism was subsequently checked using iThenticate for each submitted and re-submitted version of the manuscript. The authors also used BioRender.com to create ![]() , and the publication license for this figure is available upon request.
, and the publication license for this figure is available upon request.
The authors would also like to thank all the presenters of the 6th International Brain Stimulation Conference, held in Kobe, Japan, from 12 to 16 February 2025, particularly those presenting during Symposium 4F: Novel Brain Stimulation Therapies to Promote Recovery After Stroke and Symposium 3F: Mapping and Facilitating Descending Motor Pathways After Stroke: Implications for Recovery, whose work served as an inspiration for writing this piece. Beyond the scope of this conference, the authors also wish to acknowledge Dr. Jesús Martín-Fernández, whose talks on neurosurgery and brain individual variability further stimulated the application of these concepts within neurorehabilitation.
Author Contributions
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by the Rehabilitation Research Institute of Singapore (RRIS). Dr. Pablo Cruz Gonzalez and Dr. Ananda Sidarta are supported by the funding from the Agency for Science, Technology and Research (A*STAR), Nanyang Technological University (NTU), and the National Healthcare Group (NHG). Additional support was provided by the National Natural Science Foundation of China (NSFC) through the Young Scientists Fund (Ref. No. 82402987) and the RGC Germany/Hong Kong Joint Research Scheme 2024/2025 (Ref. No. G-PolyU508/24) awarded to Dr. Jack Jiaqi Zhang.
Ethical considerations
Ethical approval was not required.
Consent to participate
Not applicable.
