Abstract
Background:
Predicting the rate of cognitive and functional decline over the course of Mild Cognitive Impairment (MCI) and Alzheimer’s disease (AD) is vital for advancing clinical care and research initiatives.
Objectives:
The current study examined if a functional upper limb motor task could predict 1-year change in cognition (Mini Mental Status Examination [MMSE]) and daily function (Quick Dementia Rating System [QDRS]) from a sample of 61 cognitively intact, 35 MCI, and 32 AD subjects.
Results:
Analyses adjusted for age, sex, and education showed that performance speed and variability on the motor task significantly predicted change in performance on the MMSE and QDRS over 1 year, and that these effects were small to medium in size (η2 = 0.04-0.05).
Conclusion:
These results suggest the potential of a simple motor task to estimate disease progression in MCI and AD, which could be used to provide additional clinical resources for and enrich clinical trials with those most likely to decline.
Keywords
Introduction
Although Alzheimer’s disease (AD) is not considered a movement disorder, dementia severity is related to apraxia and motor dysfunction can emerge in the preclinical stage and predict onset of Mild Cognitive Impairment (MCI). The majority of studies linking motor dysfunction to MCI and AD focus on gait, balance, and muscle strength, 1 with motor signs (eg, rigidity and bradykinesia) predicting disease progression. 2 This is particularly meaningful because it provides opportunities to select individuals at higher risk of progression for personalized early intervention, more appropriately managed healthcare resources, and selection into clinical trials. While a study by Scarneas 2 had a mean follow-up time of 3 years, motor tests that could predict cognitive and functional decline over a shorter period of time (eg, 1 year or less) would be more valuable. Performance on a functional upper limb motor task has been associated with amyloid deposition 3 and hippocampal volume. 4 Thus, this brief and easy-to-administer task may have clinical and research utility regarding disease progression. The current study evaluated if this motor task was able to predict change in scores on a cognitive screening tool (Mini Mental Status Examination [MMSE]) and an informant-based functional dementia staging tool (Quick Dementia Rating System 5 [QDRS]) over 1 year. It was hypothesized that baseline motor task scores would significantly predict reliable change on the MMSE and QDRS over 1 year in a combined sample of cognitively intact (CI), amnestic MCI, and AD subjects.
Methods
Data were collected from 61 CI, 35 amnestic MCI subjects, and 32 AD subjects. All subjects provided informed consent or assent while a legally authorized representative provided consent. Confirmation of group assignment was based on the AD Neuroimaging Initiative (ADNI) classification battery (see Supplemental Material for more information on recruitment and cognitive status). Of the sample, 59% self-identified as female and 99% as Caucasian. The AD group was slightly older than the CI group, while the MCI group had completed fewer years of education (see Table 1). Both MCI and AD groups had more depressive symptoms than CI subjects on the Geriatric Depression Scale. No other significant differences in these distributions were observed between the groups. Premorbid IQ measured with the Reading subtest of the Wide Range Achievement Test-4 was within the normal range for all groups.
Unadjusted Group Comparisons of Demographic Data, Predictor, and Outcome Variables.

Abbreviations: WRAT, wide range achievement test-4; GDS, 15-item Geriatric Depression Scale total score; MMSE, mini mental state examination; QDRS, quick dementia rating system; (b), baseline assessment; (1 year), 1 year follow-up assessment; (ZSRB), standardized regression-based change scores. Significant p-values (p < 0.05) are bolded.
At baseline, subjects completed 6 trials of a functional upper limb task (demonstration: https://osf.io/phs57/wiki/Functional_reaching_task). 4 In short, subjects used a plastic spoon to scoop 2 raw kidney beans at a time with their non-dominant hand from a central cup and transport them to 1 of 3 distal cups arranged at a radius of 16 cm at −40°, 0°, and 40° relative to the central cup, starting with the cup ipsilateral to their non-dominant hand, then to the middle cup, and then to the contralateral cup. They completed this sequence 5 times for a total of 15 out-and-back movements, and trial time was recorded. Six trials were completed; the mean and variability (standard deviation) were calculated for each subject. At baseline and 1-year follow-up, the MMSE was administered, and the QDRS was obtained from a collateral.
Z-scores from regression-based change formulae (ZSRB 6 ) for the MMSE and QDRS total score were calculated to quantify 1-year change on these tests. This procedure 7 is a multi-step process that involves: (1) Obtaining regression coefficients for predicting 1-year follow-up scores from baseline scores, retest interval, age, sex, and education using only the CI subjects; (2) Calculating a predicted follow-up score for all subjects from all groups using the CI intercept and CI β coefficients obtained in the initial step; and (3) Generating Z-scores for all subjects from all groups, as the difference between the predicted and observed 1-year scores, divided by the residual standard deviation from the CI regression model. For these ZSRBs, values around 0 indicate stability, whereas negative values indicate decline across time relative to the development sample and positive values indicate improvement. Linear regression models were calculated to test the predictive value of baseline motor task scores on the QDRS and MMSE ZSRB scores collapsed over the 3 groups. False discovery rate (FDR) with a q-value threshold of 0.05 was used for statistical significance.
Results
Table 1 shows demographic variables and unadjusted measures of cognition and functional performance and their statistical significance between groups, as well as the ZSRB values. Average MMSE and QDRS scores at baseline and follow-up were within the expected range for each group. Regression models demonstrated mean motor task performance and performance variability at baseline significantly predicted change in total QDRS score (see Figure 1). Variability of motor task performance, but not mean performance, predicted change in MMSE performance. The effects of the statistically significant associations were small-to-medium in size (η 2 = 0.04) and survived FDR correction.
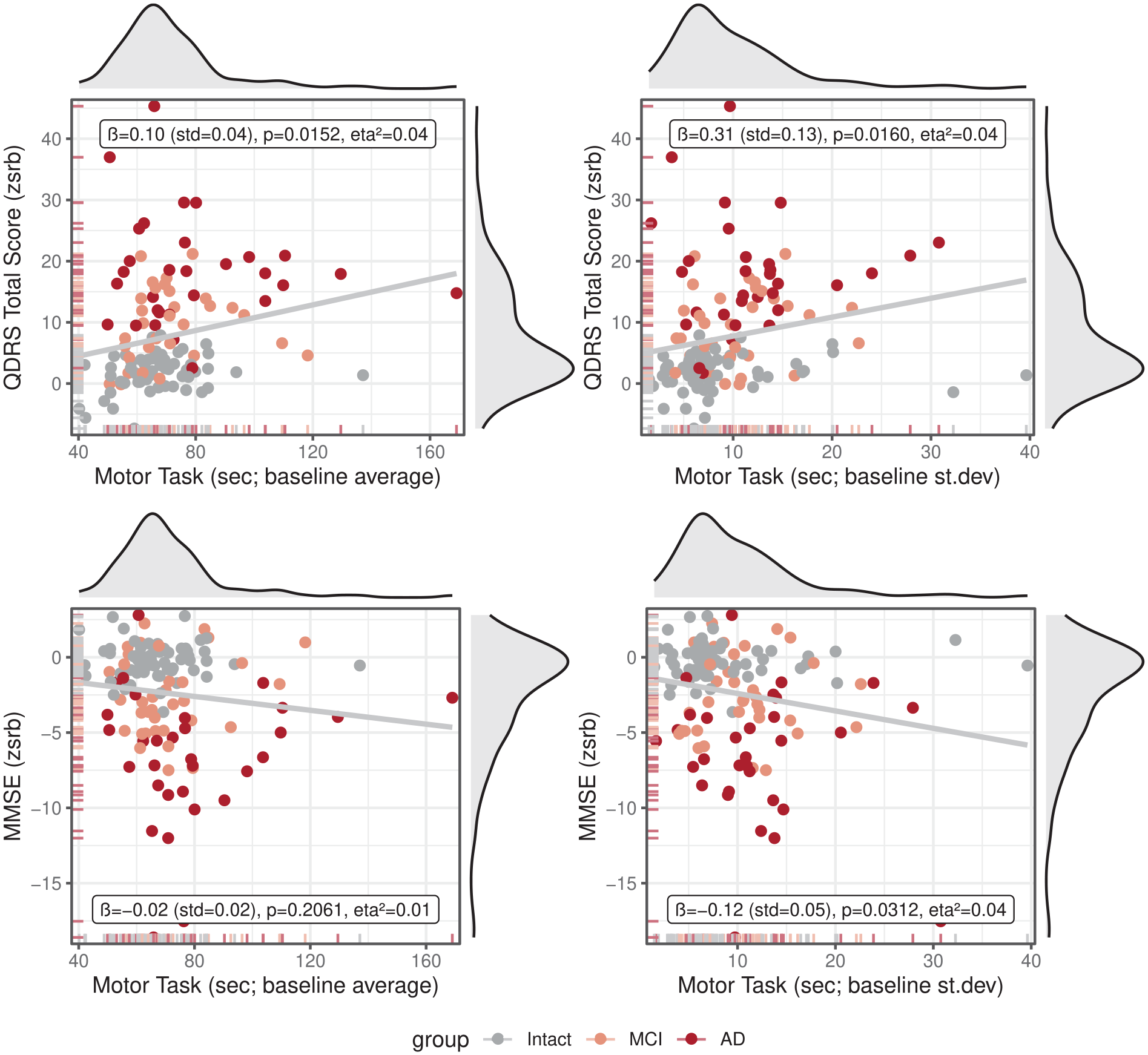
Associations between performance on the motor task (average and standard deviation over 6 sessions) and standardized regression-based change scores (ZSRB) of the Quick Dementia Rating System (QDRS) total score, as well as the Mini Mental State Examination (MMSE).
Discussion
Consistent with the hypotheses, variability in performance on a functional upper limb motor task predicted cognitive and functional change across the AD spectrum. Previous work has shown that amyloid deposition at baseline predicts QDRS decline, 8 and that multimodal data comprising cognitive performance, brain volume, and FDG-PET predicts changes on the MMSE. 9 However, such predictors are expensive, poorly scalable, and unavailable for many patients. Conversely, the current motor task is brief (~5-7 minutes), easy to administer, and does not require highly trained personnel or expensive equipment, 10 which makes it an ideal tool to use in clinical practice and clinical trials to identify patients most likely to decline across time. Additionally, this task could be employed in primary care clinics and community settings (eg, health centers and churches). The lack of an association between mean performance over the 6 trials of the upper limb motor task with behavioral decline in contrast to the significant association between variability in performance over the 6 trials of the upper limb motor task with behavioral decline may be related to fluctuations in motor abilities, but also attention or motivation, all which are affected in AD. This could indicate that, while AD pathology may not affect how well one is able to perform the upper limb motor task, it does limit the ability to perform the upper limb motor task consistently well. A strength of this work is that we used ZSRB scores to account for baseline performance and test–retest effects on the MMSE and QDRS which more sensitively tracks change across time. A limitation of the current work is the modest sample size. Future studies should include larger and more diverse samples to obtain more reliable estimates. These results are promising as they indicate that a brief motor task could be used in clinical practice and research to help predict disease progression over a 1-year interval, enabling more flexible treatment planning and lifestyle modification.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683241309591 – Supplemental material for Using a Functional Upper Limb Motor Task to Predict Cognitive and Functional Decline in Mild Cognitive Impairment and Alzheimer’s Disease
Supplemental material, sj-pdf-1-nnr-10.1177_15459683241309591 for Using a Functional Upper Limb Motor Task to Predict Cognitive and Functional Decline in Mild Cognitive Impairment and Alzheimer’s Disease by Vincent Koppelmans, Kevin Duff and Sydney Y. Schaefer in Neurorehabilitation and Neural Repair
Footnotes
Data Availability Statement
The data supporting the findings of this study are available on request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sydney Schaefer is co-founder and managing member of Neurosessments LLC; Kevin Duff is a member of Neurosessments LLC. Vincent Koppelmans does not have a conflict of interest to report.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH/NIA grants R01AG055428 awarded to Kevin Duff, K01AG073578 awarded to Vincent Koppelmans, and K01AG047926 awarded to Sydney Schaefer.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
