Abstract
Background
Traumatic brain injury (TBI) is a leading cause of adult morbidity and mortality. Individuals with TBI have impairments in both cognitive and motor domains. Motor improvements post-TBI are attributable to adaptive neuroplasticity and motor learning. Majority of the studies focus on remediation of balance and mobility issues. There is limited understanding on the use of interventions for upper limb (UL) motor improvements in this population.
Objective
We examined the evidence regarding the effectiveness of different interventions to augment UL motor improvement after a TBI.
Methods
We systematically examined the evidence published in English from 1990–2020. The modified Downs and Black checklist helped assess study quality (total score: 28). Studies were classified as excellent: 24–28, good: 19–23, fair: 14–18, and poor: ≤13 in quality. Effect sizes helped quantify intervention effectiveness.
Results
Twenty-three studies were retrieved. Study quality was excellent (n = 1), good (n = 5) or fair (n = 17). Interventions used included strategies to decrease muscle tone (n = 6), constraint induced movement therapy (n = 4), virtual reality gaming (n = 5), non-invasive stimulation (n = 3), arm motor ability training (n = 1), stem cell transplant (n = 1), task-oriented training (n = 2), and feedback provision (n = 1). Motor impairment outcomes included Fugl-Meyer Assessment, Modified Ashworth Scale, and kinematic outcomes (error and movement straightness). Activity limitation outcomes included Wolf Motor Function Test and Motor Activity Log (MAL). Effect sizes for majority of the interventions ranged from medium (.5-.79) to large (≥.8). Only ten studies included retention testing.
Conclusion
There is preliminary evidence that using some interventions may enhance UL motor improvement after a TBI. Answers to emergent questions can help select the most appropriate interventions in this population.
Introduction
Traumatic brain injury is a major worldwide cause of morbidity and mortality. 1 In the USA, recent reports indicate 2.87 million TBI-related visits to the emergency room, 2 with epidemiological data suggesting males being more affected than females. 3 Common causes of TBI include falls, motor vehicle accidents, and/or assaults. 4 The available total annual cost estimates for TBI range from $56–$221 billion. 5 Individuals sustaining a TBI may face cognitive, 6 behavioral, 7 and communication difficulties 8 lasting from few days post-injury to the rest of their lives. 9 Additionally, a TBI causes sensorimotor impairments to the upper (UL) and lower limbs (LL).
Motor impairments include abnormal posture, altered muscle tone, paresis, reappearance of primitive reflexes, ataxia, decreased balance, and lack of coordinated movement. 10 Individuals continue to have limited performance of activities of daily living (ADL), especially those relying on coordinated movements and UL muscle strength after a TBI. Persistent UL impairments and limitations in ADL performance impact functional independence in this population. 11
Motor improvements post-TBI are attributable in part to motor learning and adaptive neuroplasticity. 12 Provision of rehabilitation, benefits motor recovery by focusing on performing accurate repetitions of desired movement, 13 is an integral part of motor learning and promotes adaptive neuroplasticity. 14 Recent guidelines stress application of task-specific and intensive repetitive practice of functional reaching and activities including fine motor co-ordination. 15
There is a need to identify the most effective and pertinent interventions with a focus on remediation of impairments and activity limitations in this population. 16 To date, research has focused primarily on cognitive impairments and gait limitations, with less focus on UL issues.17,18 This is an important topic, given that the UL issues are more diffuse and tend to be long standing in individuals post-TBI. 19 Previous studies have identified deficits in UL functioning including impaired timing, reach accuracy and grasping ability. 20 Improving UL motor functioning helps boost the ability of individuals with TBI to perform ADL such as dress, wash clothes, cook, and groom. 21 Enhanced UL functioning also enables better community reintegration post-TBI. For example, improving the ability to drive helps commute to work and enables the ability to be competitively employed, volunteer and/or attend school.22,23
Our study objective was to systematically review the available literature focusing on rehabilitation of the UL, in individuals sustaining a TBI. Better identification of useful interventions can help select the best options to be used in the clinic and contribute to evidence-based practice. Our question in the Population, Intervention, Comparison and Outcome (PICO) 24 format was, “In individuals sustaining a TBI, does provision of rehabilitation interventions augment UL motor improvement post-intervention compared to pre-intervention?” Preliminary results have previously appeared as an abstract. 25
Methods
Systematic Literature Review
We performed a systematic search of the literature using Medline, Google Scholar, ISI Web of Science, Science Direct, and CINAHL. A Health Sciences Library Liaison helped formulate appropriate search strategies. Keywords and MeSH terms used included “traumatic brain injury,” “head injury,” “concussion,” “arm,” “upper limb,” “upper extremity,” “rehabilitation,” “intervention,” “motor recovery,” “impairment,” “activity limitation” and “motor improvement.” We used the terms “AND” and “OR” to combine keywords. Searches involved additional limits to restrict the articles to the English language literature published from January 1990 through August 2020, human species, and adult participants. Inclusion criteria were (i) exposure to or provision of rehabilitation interventions and (ii) assessment of motor impairment and/or limitations in ADL using the UL. Exclusion criteria were (i) studies focusing on effects of provision of only cognitive rehabilitation; (ii) rehabilitation focusing exclusively on LL outcomes; or (iii) review articles, single case studies, and expert opinion articles. We reviewed the reference lists of retrieved studies to identify additional relevant citations. We also checked the excluded reviews to identify any pertinent citations.
Data Abstraction and Analysis
Retrieved articles were grouped according to the intervention used. We developed a data abstraction form to extract data from the selected articles. Data were initially extracted by MKF and AFH. The first author (SKS) then verified that all relevant data were obtained from the selected articles. The extracted data included details about chronicity, type of UL intervention, outcome of intervention, and results of the study.
We quantified the effectiveness of interventions using estimates of effect sizes. 26 When pre, post and retentions scores were available, effect sizes were calculated as the mean post-pre/SDpre values or mean retention - pre/SDpre values. In case only change scores were reported, we used the ratio of the mean change score to the variability in change scores. Effect sizes ranging from .2–.49, .5–.79, and ≥.8 were interpreted as small, medium, and large, respectively. 27 We assessed the quality of the selected articles using the modified version 28 of the Downs and Black 29 checklist.
The Downs and Black checklist is a reliable and valid assessment. 30 It can be used to assess the quality of both randomized and non-randomized study designs. The total scores of this assessment and PEDro scale are highly correlated in studies involving individuals with brain injuries.31,32 Scores on the modified Downs and Black checklist were rated as “excellent” (score 24–28), “good” (score 19–23), “fair” (score 14–18), or “poor” (score ≤13). 33 The quality of each study was independently evaluated by AFH and MKF, with discrepancies, if any, resolved by SKS.
Results
A total of 140 citations were identified through database searches (Figure 1). After removing duplicates, 120 citations were screened, of which 90 were excluded. Of the 30 full text articles assessed for eligibility, we excluded seven studies, as they were reviews and/or expert opinions. Twenty-three articles were included in the qualitative synthesis. The different interventions used included those to reduce muscle tone (n = 6), constraint induced therapy (n = 4), virtual reality–based gaming (n = 5), non-invasive stimulation (n = 3) [including neuromuscular electrical stimulation (n = 1) and transcranial direct current stimulation (n = 2)], arm motor ability training (n = 1), use of stem cells (n = 1), goal oriented task-specific practice (n = 1), feedback provision (n = 1), and forced use therapy (n = 1). The average (95% CI) age of participants was 36.4 (29.1 to 43.6) years. Brief highlights of the studies are presented below, with details in the accompanying tables. The scoring for the modified D&B checklist for each individual study is available in Supplementary Table 1. Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram.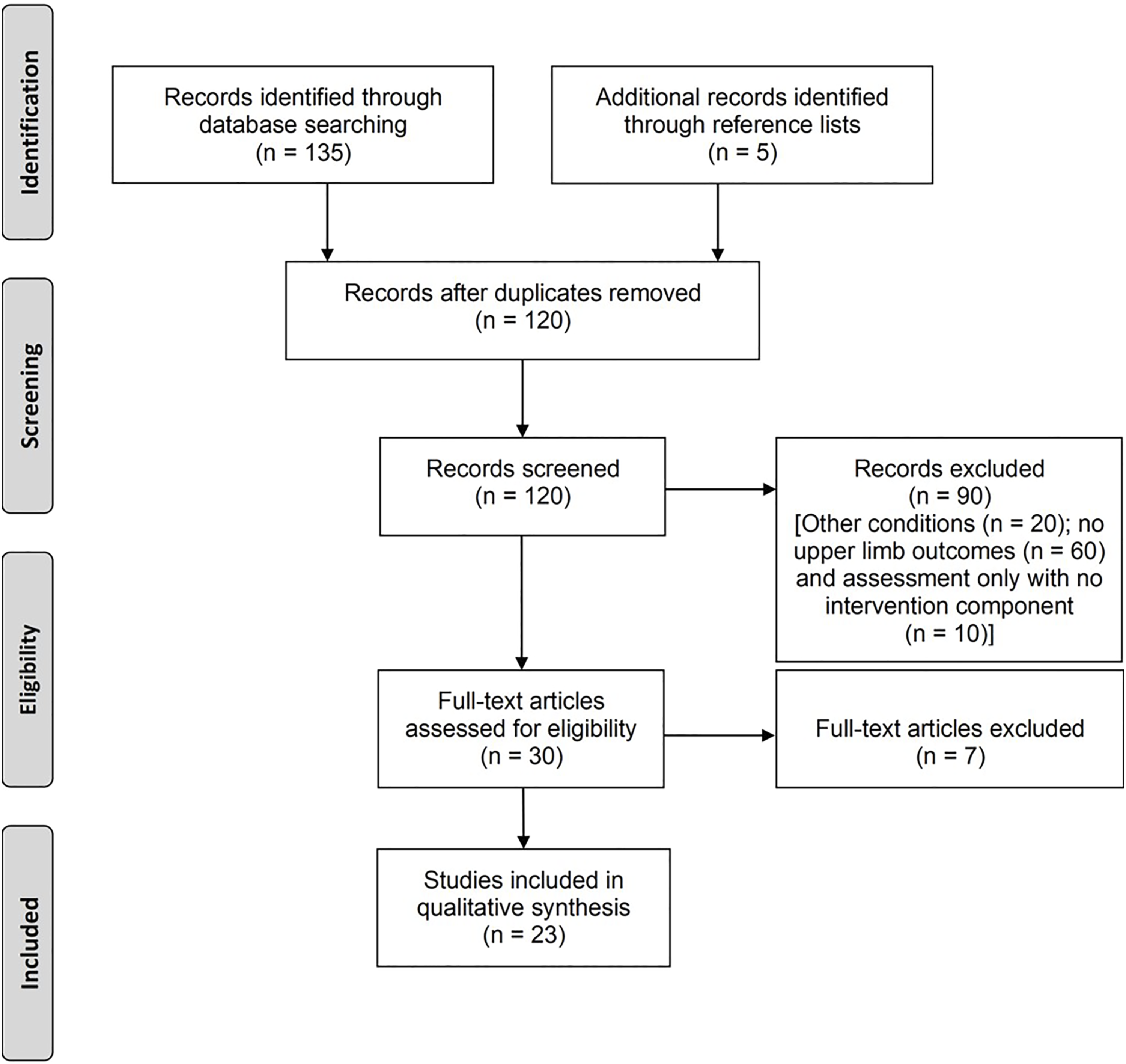
Interventions to Reduce Muscle Tone
Interventions to reduce muscle tone in the upper limbs.
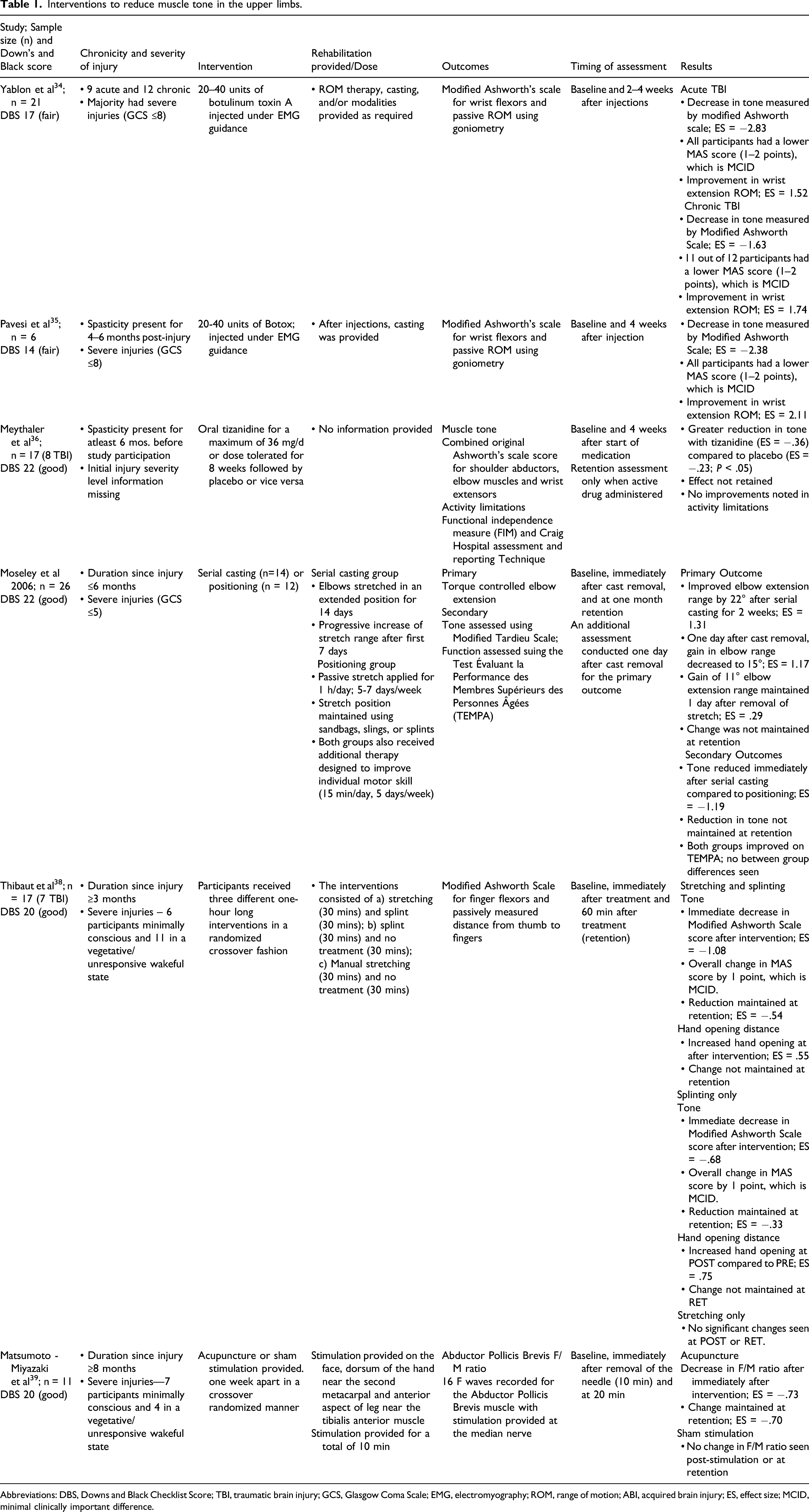
Abbreviations: DBS, Downs and Black Checklist Score; TBI, traumatic brain injury; GCS, Glasgow Coma Scale; EMG, electromyography; ROM, range of motion; ABI, acquired brain injury; ES, effect size; MCID, minimal clinically important difference.
Two studies34,35 investigated the effects of botulinum toxin A injections on wrist flexor muscle tone in 27 individuals post-TBI (18 males, 9 females) with moderate to severe muscle tone. Botulinum toxin A injections were delivered to target muscles under EMG guidance. Changes in muscle tone (quantified using Modified Ashworth’s Scale) and wrist extension range of motion (measured using goniometry) helped assess the effects of the injections. Muscle tone decreased and wrist extension range increased following botulinum toxin A injections (ES >.8).
Meythaler et al 36 assessed the effects of oral tizanidine administration on UL muscle tone in 17 individuals (14 males, 3 females) with acquired brain injuries (ABIs; TBI: n = 8, stroke: n = 9). They administered either tizanidine or placebo in a crossover fashion for 6 weeks, tapered the drug for one week and then switched over to other medication after one more week. Oral tizanidine decreased muscle tone (assessed using Original Ashworth’s scale) on the affected side immediately after treatment (ES = −.36) with no retention at 6 weeks (ES = −.1).
Moseley et al 37 recruited 26 individuals (23 males, 3 females) post-TBI with elbow flexion contracture, and randomized them into two groups (n = 13/group). One group received serial casting, and the other received static positioning. Serial casting increased elbow range by 22° over static positioning immediately post-intervention (ES = 1.85). One day after cast removal, elbow range gain decreased to 15° in the serial casting group (ES = 1.17), which further decreased to 11° after 2 weeks post-intervention.
Thibaut et al 38 randomized 17 participants (10 males, 7 females) with ABIs (TBI: n = 7, stroke: n = 10) to receive either soft splinting, 30 min of manual stretching, or no treatment. Provision of soft splinting resulted in increased hand opening ability (2.39 cm of major-palm distance, ES = .55). Additionally, soft splinting and manual stretching decreased finger flexor muscle tone after 30 min of treatment (ES = −.53 and −.55).
Matsumoto et al 39 assessed effects of acupuncture provision on UL muscle tone in 11 unconscious or minimally conscious males. They used a crossover study design providing acupuncture or no treatment, separated by one week. Acupuncture provision reduced the F/M ratio at the end of treatment (ES = −.73) and was retained 10 min later (ES = −.7).
Constraint Induced Movement Therapy (CIMT)
Use of Constraint Induced Movement Therapy (CIMT).
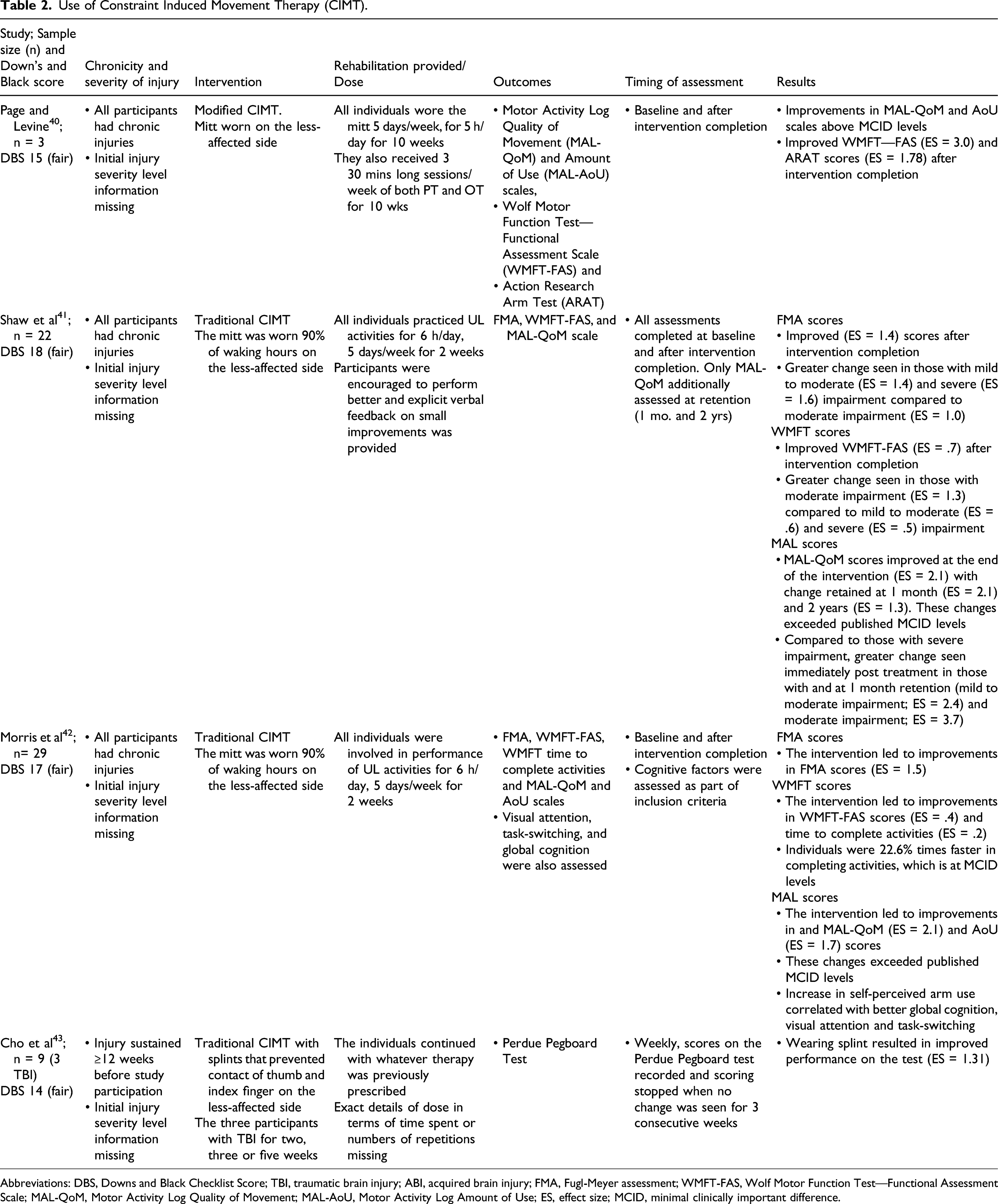
Abbreviations: DBS, Downs and Black Checklist Score; TBI, traumatic brain injury; ABI, acquired brain injury; FMA, Fugl-Meyer assessment; WMFT-FAS, Wolf Motor Function Test—Functional Assessment Scale; MAL-QoM, Motor Activity Log Quality of Movement; MAL-AoU, Motor Activity Log Amount of Use; ES, effect size; MCID, minimal clinically important difference.
Two studies examined the effects of CIMT on participants with chronic TBI. In both studies, participants wore the mitt on the less-affected limb for 90% of waking hours. All participants (n = 22; 14 males, 8 females) in the first study by Shaw et al 41 decreased UL motor impairment measured using the Fugl-Meyer Assessment (FMA; ES = 1.4) and improved in performance of ADL (measured using WMFT; ES = .7) immediately after treatment. Participants also reported an increase in self-perceived quality of movement immediately after the intervention (ES = 2.1), which was retained at one month (ES = 2.1) and at two years post-intervention (ES = 1.3).
Participants in the other study by Morris et al 42 (n = 29; 19 males, 10 females) similarly had better scores on the FMA (ES = 1.5) and WMFT (ES = .4). Participants reported an increase in the amount (ES = 1.7) and quality (ES = 2.1) of self-perceived UL use after the intervention. Participants reporting better use of the more affected UL had better global cognition (assessed using the Mini-Mental Scale) and visual attention and task-switching (measuring using the Trail Making Tests A and B).
Cho et al 43 examined the effects of CIMT on fine motor function of the hand in 9 participants (8 males, 1 female) with ABIs (TBI: n = 3, stroke: n = 6). The less-affected side was partially constrained with an opposition restriction splint that blocked use of the thumb and index finger. All participants were evaluated weekly using the Perdue Pegboard test, until no change was seen in three consecutive assessments. Constraining the less-affected side resulted in improved performance on the pegboard test (ES = 1.31).
Virtual Reality Gaming
Use of virtual reality gaming.

Abbreviations: DBS, Downs and Black Checklist Score; TBI, traumatic brain injury; PTA, post traumatic amnesia; UL, upper limb; FMA, Fugl-Meyer Assessment; BBT, Box and Blocks Test; ES, effect size.
In two studies, Ustinova and colleagues examined the effects of task-practice of reaching movements from a standing position. In the first study, 44 13 participants post-TBI (6 males, 7 females) practiced 10 trials of reaching movements. Movements were recorded using a motion analysis system. At the end of 10 trials, participants reached faster to the targets (ES=.54), and further improved (ES = .74) at retention testing (30 min post-intervention). The participants also had straighter reaching movements (ES = −1.07), which were retained (ES = −.97).
The second study 45 examined the effects of multiple sessions of playing games on balance, reaching and co-ordination. Participants (n = 15; 10 males, 5 females) with chronic TBI played games for 15 sessions (thrice weekly). All participants were assessed at baseline, after practice, and one-month post-practice. Dynamic balance (ES = 1.33) and reaching movement straightness (ES = −1.16) improved after practice and at one month, these changes were retained.
Mumford et al 46 examined the effects of repetitive practice of unimanual and bimanual UL movements in nine individuals (5 males, 4 females) with severe chronic TBI. Assessments included kinematic measures of reaching as well as the number of blocks transferred on the Box and Blocks Test (BBT). After training, all participants had more accurate movements (ES = .62) and transferred more blocks (ES = .42).
Syed and Kamal 47 assessed the effects of VR-based gaming on 34 individuals with a variety of neurological disorders (26 males, 8 females) including TBIs (n = 9). Participants received 12 sessions of either VR-based training (n = 17) or conventional training (n = 17). Both groups improved after training with greater changes noted with VR-based (P < .001) compared to conventional training (P < .05) for both balance (assessed using Berg Balance Scale, BBS) and self-reported UL ADL performance [assessed using Disabilities of the Arm, Shoulder and Hand (DASH) questionnaire]. When results were compared only for the participants post-TBI, greater within group changes were noted after VR-based training compared to before for BBS (ES = 5.73) and DASH (ES = −2.35).
In another study, Buccellato et al 48 examined the effects of VR-based gaming on a group of 21 participants (15 males, 6 females) with ABIs (TBI: n = 13, stroke: n = 4, a combination of stroke + TBI: n = 4). Participants were randomized to an early treatment group (n = 11) or a delayed treatment group (began training 3 weeks after study initiation; n = 10). The effects of this system on UL function, dexterity, and activity performance were assessed using the FMA, BBT, and Jebsen Taylor Hand Function test, respectively. Early or delayed training did not result in improved function or dexterity. However, activity performance was improved (ES = .52).
Non-Invasive Stimulation
Use of non-invasive stimulation and arm ability training.

Abbreviations: DBS, Downs and Black Checklist Score; TBI, traumatic brain injury; ABI, acquired brain injury; ROM, Range of motion; MMSE, Mini-Mental Scale Examination; FMA, Fugl-Meyer Assessment; BBT, Box and Blocks Test; ES, effect size; MCID, minimal clinically important difference.
Alon et al 49 assessed the effects of provision of NMES enabling reciprocal finger flexion and extension along with grasp and release in 20 individuals (14 males, 6 females) with chronic ABIs (TBI: n = 7, stroke: n = 13). All participants received an average of 3.5 h stimulation daily over the course of the intervention, which lasted for almost four months. All participants had a more extended posture at the elbow (ES = 4.09) and wrist (ES = 3.71) at rest at the end of the intervention. At the wrist, participants improved their range of passive extension (ES = 2.69) as well as active flexion and extension (ES = 2.73). At the elbow, active ROM of elbow movement increased (ES = 6.91).
Kang and colleagues 50 assessed the effects of 2 mA anodal tDCS to the left dorsolateral prefrontal cortex on reaction time to an attention task. Nine individuals (8 males, 1 female) with chronic TBI participated and were randomized to receive active tDCS for 20 min or sham stimulation after one week in a crossover fashion. Reaction time on a computerized timed task decreased after application of real tDCS vs sham stimulation at the end of the intervention (ES = −.89). However, this change was not maintained at the two retention assessments (3 h and 24 h after the end of stimulation).
Middleton et al 51 examined the effects of bi-hemispheric stimulation followed by robotic training on five participants with ABIs (TBI; n = 1, stroke: n = 3, stroke + TBI: n = 1). All participants performed strengthening and functional activities for a total of 40 min. Each participant received a 1.5 mA intensity concurrent stimulation for the first 15 min. Results for participants post-TBI (2 males) revealed improvements only in FMA scores (ES = −.47), which were retained (ES = −.42). Participants post-TBI also reached the targets faster at the end of the intervention (ES = .37; assessed by the KINARM© robotic device) and continued to improve 6 months later (ES = .7).
Arm Ability Training (AAT)
We found one study (excellent quality; Table 4C) that assessed the effects of AAT on motor performance outcomes and hand function. In this study by Platz and colleagues, 52 60 participants (36 males, 24 females) with ABI (TBI: n = 15, stroke: n = 45) were randomized into three groups: a control group, a group receiving AAT and a group receiving AAT + knowledge of results (KR) feedback (n = 20 each). Activity performance was assessed using the time to complete the TEMPA (Test Evaluant les Membres Superieurs des Personnes Agees). Kinematic assessment of an aiming movement on a stylus between two targets was also conducted. Provision of AAT resulted in faster performance on the TEMPA (ES = −.95), which was retained one year later (ES = −.75). Participants receiving AAT also had faster aiming movements (ES = .67). Providing KR feedback did not enhance task performance.
Stem Cell Transplantation
Use of stem cells and other interventions.
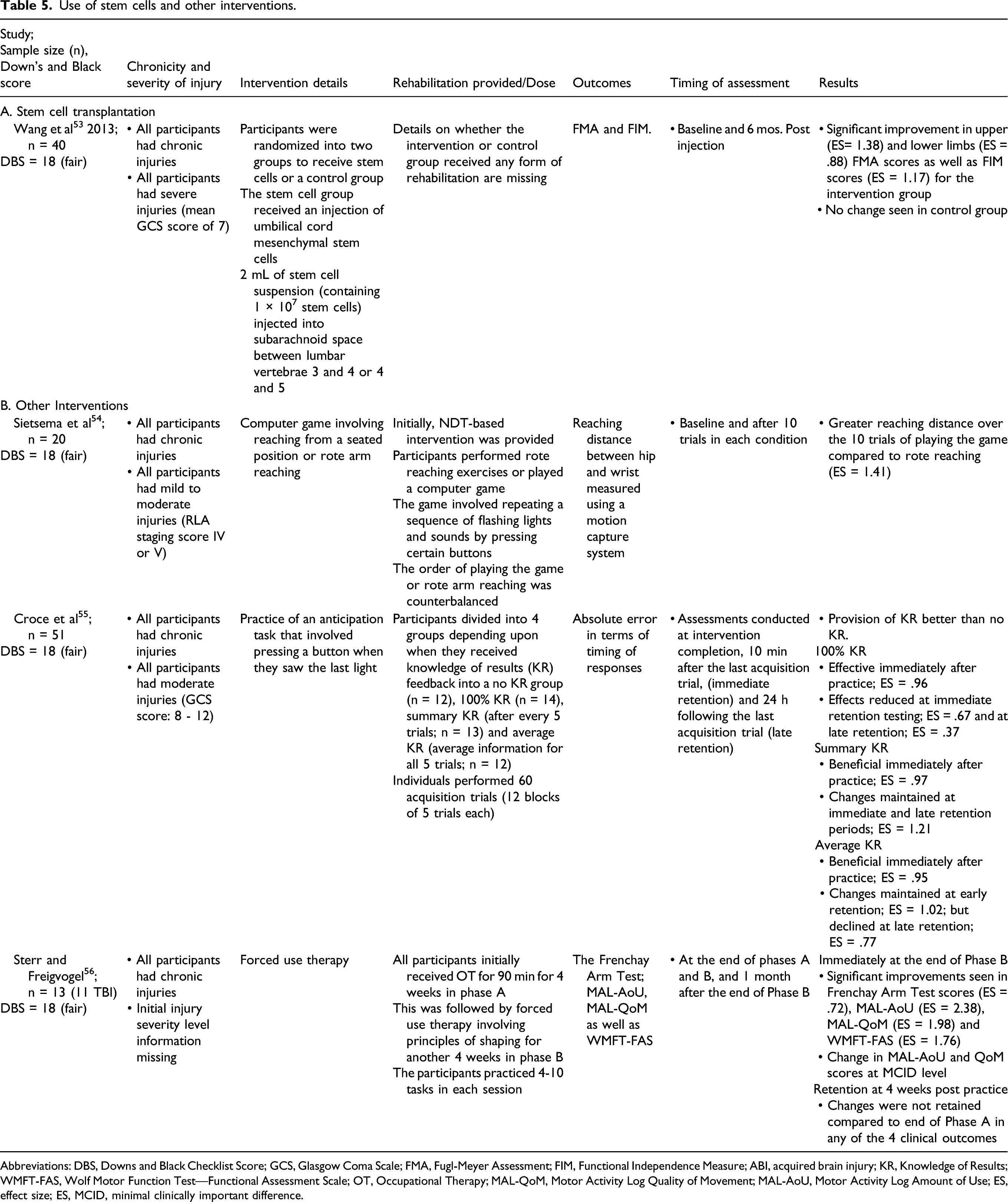
Abbreviations: DBS, Downs and Black Checklist Score; GCS, Glasgow Coma Scale; FMA, Fugl-Meyer Assessment; FIM, Functional Independence Measure; ABI, acquired brain injury; KR, Knowledge of Results; WMFT-FAS, Wolf Motor Function Test—Functional Assessment Scale; OT, Occupational Therapy; MAL-QoM, Motor Activity Log Quality of Movement; MAL-AoU, Motor Activity Log Amount of Use; ES, effect size; ES, MCID, minimal clinically important difference.
Feedback and Other Interventions
We found 3 studies (fair quality; Table 5B) that assessed the effects of different interventions on UL motor impairment and activity levels in individuals post-TBI. Sietsema and colleagues 54 assessed the effects of playing a game within an occupational context compared to rote exercises on UL movement patterns. Twenty individuals (17 males, 3 females) with mild to moderate TBI participated in the study. Participants practiced 10 trials in both conditions. The total forward reaching distance from the hip to the wrist was measured using motion analysis. Game playing resulted in greater reaching distance (13 cm, ES = .63) than rote arm reaching exercises.
Croce and colleagues 55 evaluated the effectiveness of provision of KR feedback at different schedules in subjects with severe TBI (n = 51; 42 males, 9 females). All participants practiced 60 trials (5 trials/block, 12 blocks) of an anticipation task. Participants received KR feedback on timing errors after each trial at different schedules—no KR (n = 12), 100% KR (n = 14), summary KR (n = 13) and average KR (n = 12). They were then tested for immediate (after 10 minutes) and delayed (after 1 h) retention. All the three KR groups were more accurate in the last block compared to the first block of trials (ES = .96). At early retention testing, this effect was decreased in the 100% KR group. However, the summary KR (ES = 1.21) and average KR (ES = 1.02) groups continued to improve accuracy. At the late retention testing, the effects were further reduced in the 100% KR group (ES = .37) and average KR group (ES = .77) but was retained only in the summary KR group at the same level (ES = 1.21).
Sterr and Freivogel 56 examined the effects of shaping principles on UL activity performance in 13 individuals (9 males, 4 females) with ABIs (TBI: n = 11, stroke: n = 2). All participants were evaluated using the MAL, WMFT, and Frenchay Arm Test. Compared to provision of occupational therapy, task-practice using shaping principles resulted in greater motor improvement on all outcomes (MAL; AoU: ES = 2.23, QoM: ES = 1.98, WMFT; ES = 1.76 and the Frenchay Arm Test; ES = .72).
Discussion
We examined the effectiveness of different interventions to augment UL motor improvement in individuals post-TBI. Majority of the studies reported moderate to large effect sizes for intervention effectiveness. In terms of quality assessment, one study was excellent, five good, and the rest were fair.
Outcomes Used to Assess Motor Improvement
A variety of outcomes were used to assess motor improvements at different levels of the International Classification of Functioning (ICF). 57 At the motor impairment level, the FMA was commonly used.41,42,48,51,53 Goniometry34,35,49 and torque-controlled passive extension 37 helped assess changes in wrist and elbow ranges of motion. In addition, kinematic motor performance outcomes including speed, reaching path straightness, and accuracy helped quantify motor impairment.44-46 These kinematic measures were obtained using motion capture equipment, robotic manipulandum, or using instrumented tablets. All the above-mentioned measures have well established psychometric properties.58,59
Muscle tone was most commonly quantified using the Ashworth’s scale or the MAS.34,35,38 Other measures used included the Modified Tardieu Scale 37 or neurophysiological (H-Reflex) measures. 39 The MAS has been recommended as a measure of choice in published guidelines. 58 However, both the MAS and Modified Tardieu Scale have poor inter-rater reliability in individuals post-TBI. 60 Use of the MAS alone does not distinguish between the tonic and phasic components of spasticity. 61 Changes noted in H-reflex based parameters do not automatically translate to better functional performance after rehabilitation. 62 The utility of other neurophysiological measures (e.g., based on spatial threshold control of muscle activation) 61 alone or in conjunction with existing clinical measures to assess muscle tone remains to be estimated.
Similar to motor impairment, a variety of assessments were used to measure activity limitations. The WMFT was commonly used40-42,56 across the different studies. Dexterity was measured by using the BBT,46,48,51 Purdue Pegboard Test, 43 TEMPA,37,52 and Jebsen Taylor Hand Function test 48 in different studies. Limitations in ADL performance were also quantified using the FIM,36,53 the CHART, 36 Frenchay Arm Test, 56 and the ARAT. 40 In addition, studies also used the DASH 47 MAL amount and quality scores,40-42,56 and the Stroke Impact Scale 51 which report participant self-perceived levels of UL use. All the measures have excellent psychometric properties, 59 and the FIM and ARAT are part of the published guidelines. 58 Inclusion of the DASH, MAL, and Stroke Impact Scale across studies is encouraging, given the suggestion to use patient reported measures as outcomes in intervention studies. 63
Follow-Up Assessments
It has been suggested that motor improvement after TBI is attributable in part to motor learning. 12 Retention of improvements in performance noted at the end of the intervention denote motor learning. However, only 1036-39,41,44,51,52,55,56 studies included any form of retention testing. Amongst these studies, the timing of testing varied widely. Retention was tested at the following periods post-intervention: 10 min, 55 20 min, 39 30 min, 44 1 h, 38 3 h, 50 24 h,37,50,55 4 weeks,37,41,56 6 weeks, 36 6 months, 51 1 year, 52 and 2 years post-intervention. 41
Not all studies found that changes were retained. While hypertonia was reduced in the short-term (≤24 h) using casting 37 and acupuncture, 39 long-term retention (>24 h) was absent with oral tizanidine. 36 Only short-term retention was assessed with VR 44 and feedback provision. 55 Use of shaping principles with 41 and without 56 constraints as well as Arm Motor Ability Training 52 resulted in long-term retention. Both short 50 and long-term 51 retentions were seen with the use of tDCS. It remains to be seen if use of VR technology and use of different interventions including acupuncture and botulinum toxin A result in long-term retention in individuals post-TBI.
Presence of Cognitive and Mood Impairments
Dysfunction in different cognitive domains influences generalized motor improvement in individuals post-TBI. 6 Only two42,50 of the selected studies, examined the association between UL motor improvement and cognitive impairment. Few other studies provided information on baseline levels of cognitive functioning,40,48,54,55 but did not examine the effects of baseline cognitive dysfunction with motor improvement. Only one study assessed the levels of baseline depressive symptomatology, 48 which can predict motor improvement and satisfaction with life after discharge from rehabilitation in this population. 64 The presence of cognitive impairments 65 and depressive symptoms 66 influence motor improvement after a stroke. Future studies will need to focus on the relationship between cognitive dysfunction, mood disorders, and motor recovery in individuals post-TBI to better understand their association with motor improvement.
Level of Injury Severity
Out of the 23 included studies, only few studies reported initial injury severity levels. The Glasgow Coma Scale,34,35,37-39,53,55 duration of post-traumatic amnesia, 45 or Rancho Los Amigos original scale 54 helped quantify initial injury severity levels. This information is an important prognostic indicator for changes in overall motor improvement and levels of activity performance assessed using the Barthel Index 67 as well as a composite score of ADL and social participation (assessed using the Glasgow Outcome Scale Extended measure). 68
The other studies did not specify the injury severity levels, but some provided FMA scores.41,42,44,45,47,51 FMA scores ≥50/66 and ≤49/66 represent as mild and moderate to severe levels of post-stroke UL motor impairment. 69 The FMA scores in the acute post-stroke stage can predict subsequent UL recovery potential. 70 Whether UL FMA scores can be used to make similar predictions in individuals post-TBI remains to be estimated.
Sex and Gender Considerations
As stated previously, a greater proportion of males (1.2–4.4) sustain TBIs compared to females.3,71 The greater proportion of males amongst the included participants across the different studies are indicative of the above findings. Only two studies had an almost equal distribution of sexes, 46 or included more females. 44 Despite consistent calls for considerations of sex and gender on functional outcomes,72,73 only one study 47 assessed the effects of sex on outcomes. Future studies must strive to include more female participants and consider the effects of sex and gender on functional outcomes.
Effect of Chronicity
Studies on interventions including acupuncture, CIMT, VR-based games, NMES, stem cells, game playing, feedback, and forced-use therapy exclusively included participants with chronic injuries. While studies using Botulinum toxin A included acute, sub-acute and chronic participants, separate analyses were conducted by chronicity. 34 Use of serial casting 37 included only participants in the sub-acute stage. Other studies including participants across all stages did not conduct separate analyses based upon chronicity.38,50,52 Future studies must strive to include participants across all stages or conduct sub-analyses based on time since injury.
Limitations
Heterogeneity amongst the interventions used prevented performance of an overarching statistical synthesis like a meta-analysis. Among the 23 studies included in this review, only 9 studies were designed as RCTs. Although the wide variability in presenting symptoms and underlying injury severity present serious challenges in designing RCTs involving individuals post-TBI, 74 encouraging efforts are underway. 75 Only three studies included in this review had sample size calculations37,45,52 and one study 48 provided an estimate for numbers of participants needed for future trials. Nine of the 23 studies included participants with stroke and TBI. Thus, generalization of the findings is limited to a certain extent, except for two studies,47,51 which conducted separate analyses for individuals post-TBI. Future studies should include only individuals post-TBI or conduct separate sub-analyses for this population. Limits placed on age (adults), language (English only) and non-inclusion of terms like shoulder, elbow, wrist, and hand. may have led to exclusion of some studies.
Conclusion
Preliminary evidence suggests that different rehabilitation interventions may facilitate UL motor improvement in individuals post-TBI. This review has identified several new questions in individuals post-TBI such as when compared to baseline, does provision of: (i) Botulinum toxin A followed by intensive rehabilitation result in maintained reduction of muscle tone at 1 and 3-month retention assessments; (ii) CIMT results in better motor improvement compared to conventional rehabilitation following the intervention; (iii) a combination of interventions such as VR-based gaming and tDCS is more beneficial than provision of one single intervention following the intervention and at retention; and (iv) provision of knowledge of performance feedback is useful and results in similar or better improvements than KR feedback at the end of the intervention and at retention assessments. We hope that these questions will help guide and foster further research to evaluate the efficacy of the most suitable interventions to reduce impairment and improve activity performance post-TBI.
Supplemental Material
sj-xlsx-1-nnr-10.1177_15459683211056662 – Supplemental Material for Upper Limb Motor Improvement after Traumatic Brain Injury: Systematic Review of Interventions
Supplemental Material, sj-xlsx-1-nnr-10.1177_15459683211056662 for Upper Limb Motor Improvement after Traumatic Brain Injury: Systematic Review of Interventions by Sandeep K. Subramanian, Melinda K. Fountain, Ashley F. Hood and Monica Verduzco-Gutierrez in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgments
The authors would like to acknowledge Dr Kate Aultman for her support and encouragement in this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SKS was funded by a pilot grant awarded by the School of Health Profession, UT Health San Antonio.
Supplement material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
