Abstract
Fatigue is one of the most common debilitating symptoms reported by persons with multiple sclerosis (MS). It reflects feelings of tiredness, lack of energy, low motivation, and difficulty in concentrating. It can be measured at a specific instant in time as a perception that arises from interoceptive networks involved in the regulation of homeostasis. Such ratings indicate the state level of fatigue and likely reflect an inability to correct deviations from a balanced homeostatic state. In contrast, the trait level of fatigue is quantified in terms of work capacity (fatigability), which can be either estimated (perceived fatigability) or measured (objective fatigability). Clinically, fatigue is most often quantified with questionnaires that require respondents to estimate their past capacity to perform several cognitive, physical, and psychosocial tasks. These retrospective estimates provide a measure of perceived fatigability. In contrast, the change in an outcome variable during the actual performance of a task provides an objective measure of fatigability. Perceived and objective fatigability do not assess the same underlying construct. Persons with MS who report elevated trait levels of fatigue exhibit deficits in interoceptive networks (insula and dorsal anterior cingulate cortex), including increased functional connectivity during challenging tasks. The state and trait levels of fatigue reported by an individual can be modulated by reward and pain pathways. Understanding the distinction between fatigue and fatigability is critical for the development of effective strategies to reduce the burden of the symptom for individuals with MS.
Introduction
Multiple sclerosis (MS) is an inflammatory disease that targets the nervous system and progressively disables an individual.1-3 It is the most common non-traumatic cause of disability in young people and is 3 times more prevalent in women than in men.4,5 The damage caused by MS includes demyelination of axons, loss of neurons throughout the entire nervous system, and disruption of transmission along surviving pathways.4-10
The changes caused by the disease result in the emergence of several debilitating symptoms that negatively impact the ability of an individual to perform activities of daily living.11,12 Two of the most common symptoms in MS are fatigue and difficulties with walking.13-15 The prevalence of fatigue increases with disease progression and eventually becomes a primary concern for up to 80% of the people with MS, 16 irrespective of the MS phenotype.17,18 Fatigue is typically rated as one of the 2 most disabling symptoms by persons with MS,19-21 but it is difficult to manage clinically as most individuals report that its severity varies within and across days.22-25
Moreover, there is no consensus on the neurological adaptations responsible for the emergence of the symptom or its variability across time.16,26-28 One of the key reasons for this uncertainty is the diversity of definitions used for key terms and the absence of a shared conceptual framework.29-32 To address this limitation, the purpose of our point-of-view article is to distinguish between the concepts of fatigue and fatigability by focusing on the methods used to quantify each attribute. Our article is not intended to provide a systematic review on fatigue and fatigability, but rather it explains the association between fatigue and fatigability and discusses experimental strategies that can be used to identify the adaptations that influence the fatigue experienced by persons with MS.
The Symptom of Fatigue
Definitions.

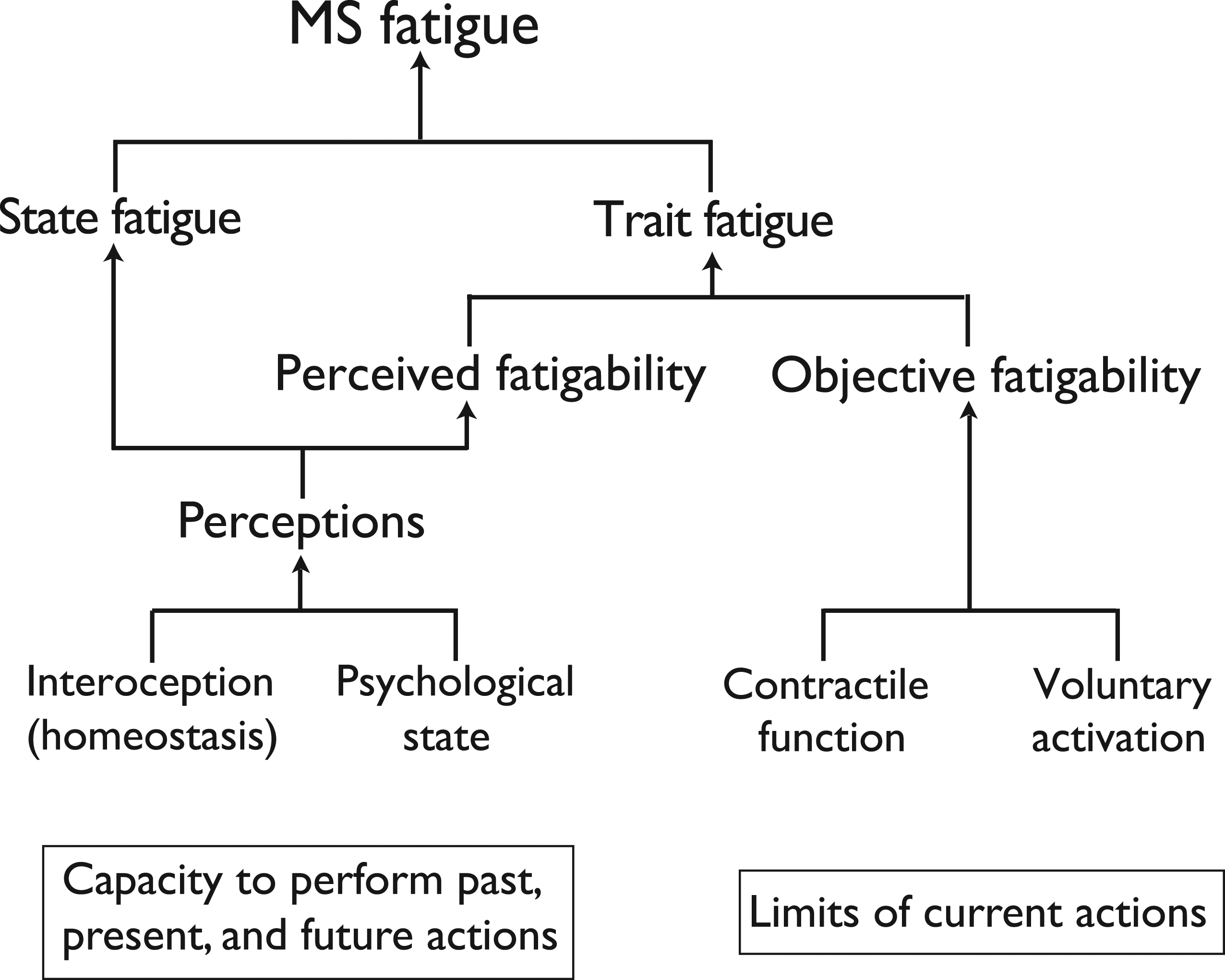
The perceptions that underlie state measures of fatigue are derived from interoceptive feedback about the homeostatic state of the body (Figure 1).18,33,39,47,48 The interoceptive signals are transmitted along ascending pathways to the insula and anterior cingulate cortex to generate emotional feelings (insula and provide the motivation (anterior cingulate cortex) for a response intended to regulate homeostasis.39,48 The signals involved in interoception also contribute to other perceptions, cognitions, and behaviors, such as predictive models, emotional impairments, and sickness behaviors.37,49 Importantly, the derivation of perceptions from the interoceptive signals can be modulated by the psychological state of the individual (Figure 1). Based on this framework, it has been proposed that the feeling of fatigue (state fatigue) arises from an inability to correct deviations from a balanced homeostatic state.
47
A scheme showing the interdependent attributes that contribute to the levels of state and trait fatigue reported by individuals with MS. State measures of fatigue indicate the level of fatigue at a specific moment in time that is derived from interoceptive signals and the psychological state of the individual. Trait measures of fatigue can be quantified from retrospective estimates of work capacity (perceived fatigability) or actual measures of physical or cognitive work capacity (objective fatigability). The level of objective fatigability derived from physical tasks depends on the contractile capabilities of the involved muscles and the level of voluntary activation provided by the nervous system. Adapted from Kluger et al.
30
and Enoka & Duchateau.
45
Some evidence suggests that functional connectivity within interoceptive networks is disturbed in persons with MS.8,50-52 In a neuroimaging study of brain networks, for example, Chen et al. 39 found that an increase in the VAS rating of fatigue when persons with MS performed a working-memory task was associated with increased residual functional connectivity as assessed by functional MRI between both the insula and the dorsal anterior cingulate cortex with other brain areas. The connectivity patterns from selected brain regions (insula, dorsal anterior cingulate cortex, striatum, dorsolateral prefrontal cortex, and ventromedial prefrontal cortex) of the MS participants became more diffuse as the cognitively demanding task progressed, likely due to interoceptive efforts to attenuate challenges to a balanced homeostatic state. Moreover, the hyperconnectivity within the interoceptive network (insula and dorsal anterior cingulate cortex) was accompanied by a reduction in the connectivity within the reward network. Similarly, Gonzalez Campo et al. 47 found that accuracy when performing a validated interoception task (heartbeat detection) was worse for MS participants with greater levels of fatigue as assessed clinically and was associated with increased resting-state functional connectivity involving the insula and anterior cingulate cortex.
Consistent with the concept that networks involving the insula and anterior cingulate cortex contribute to the feeling of fatigue, the function of the autonomic nervous system as the neural effector in regulating homeostasis is often disturbed in individuals with MS.3,53,54 There is a strong association between clinically assessed levels of fatigue and dysfunction of the autonomic nervous system (Composite Autonomic Symptom Score 31), but the findings are mixed on the association between questionnaire-derived fatigue scores and objective measures of autonomic nervous system function (e.g., sweating response, sudomotor axon reflex, respiratory sinus arrhythmia, and Valsalva ratio). 55 When an association between fatigue scores and autonomic dysfunction has been observed in persons with MS, it has typically involved abnormal responses to tests that challenge the sympathetic nervous system. 56 Most of these studies, however, have assessed the association between measures of autonomic function and estimates of fatigue derived from questionnaires (trait fatigue).
In contrast, autonomic function may influence state measures of fatigue. This possibility was demonstrated by Sander et al. 46 in a study on the association between adjustments during an acoustic vigilance task and estimated levels of fatigue in persons with MS. Multiple regression analyses indicated that none of the outcomes could explain the variance in a questionnaire-based estimate of fatigue, whereas a moderate amount of the variance in the change in the VAS scores from before to after the vigilance test (R2 = 0.218) was explained by 2 measures of heart-rate variability. The change in one of these measures (percentage of normal interbeat intervals) suggested an increase in parasympathetic activity during the vigilance task. These results were interpreted as indicating activation of afferent fibers in the vagus nerve by proinflammatory cytokines that led to increased activity in the interoceptive brain areas and thereby increases in the feeling of fatigue. However, autonomic dysfunction only explains a moderate amount of the variance in state measures of fatigue (VAS scores). 56
The sense of fatigue that arises from interoceptive networks can be modulated by reward (motivational) pathways37,39,57-59 and pain pathways.60-62 For example, Pardini et al. 63 reported that reward responsiveness was negatively correlated with a questionnaire-derived estimate of fatigue in persons with MS who had clinically significant levels of fatigue. Moreover, the correlation explained 45% of the variance in questionnaire scores. Similarly, Dobryakova et al. 52 found that VAS scores reported by an MS group increased more from the first block of trials during a card-guessing task to the second block of trials when there was no reward compared with when there was a reward. Also, the VAS scores reported by the healthy control group only increased during the no-reward condition. These findings indicate that the presence of a reward attenuated an increase in the state level of fatigue. Presumably there were no differences in the effort required to perform the task between the reward and no-reward conditions. Critically, functional MRI measures indicated that the level of activity in a reward network (ventral striatum) changed in parallel with the VAS scores during the reward condition for the MS group. Similarly, depressive symptoms, which reflect lower levels of motivation and less engagement of reward pathways, 63 are strongly correlated with clinically assessed levels of fatigue in persons with MS.64,65 These findings demonstrate that reward-related cognition can influence both the feeling of fatigue (state fatigue) and estimates of the level of fatigue derived from clinical assessments (trait fatigue).
Although persistent pain and elevated fatigue scores are comorbid in persons with MS,11,66-68 there appears to be little information on the impact of a reduction in the chronic level of pain on the fatigue experienced by persons with MS. 69 Taken together, these findings demonstrate that the level of fatigue experienced by an individual at a specific instant in time (state fatigue) is based on information received by interoceptive networks about the homeostatic status of body. Interpretation of the signals transmitted by these pathways, however, can be modulated by reward and pain pathways.
Clinical Assessment of Fatigue in Multiple Sclerosis
In contrast to instantaneous ratings that appear to reflect the ability to correct deviations from a balanced homeostatic state (state fatigue), clinical assessments of fatigue are based on the presumed impact of the symptom on the work capacity (fatigability) of the individual. The level of fatigability experienced by an individual can be estimated (perceived fatigability) or measured directly (objective fatigability), with the resulting values providing a measure of the trait level of fatigue (Figure 1).
Perceived Fatigability
Several questionnaires have been validated to obtain clinical assessments of the level of fatigue experienced by persons with MS.18,32,34,70-73 In this point-of-view article, we focus on the commonly used Modified Fatigue Impact Scale (MFIS) and Fatigue Severity Scale (FSS) questionnaires to illustrate the types of items included in these instruments. The MFIS is a 21-item questionnaire that assesses the impact of physical (9 items), psychosocial (2 items), and cognitive (10 items) aspects of fatigue on the daily activities of respondents over the preceding 4 weeks. Each item on the questionnaire is rated on a 5-point Likert scale ranging from never (0) to always (4). A summed MFIS score of ≥38 for the 21 items indicates a clinically significant level of fatigue and a change of 4–6 points is considered to be a minimally important difference.18,74
The FSS is a 9-item questionnaire that assesses the severity of fatigue symptoms. 38 Respondents rate each item on an 8-point scale ranging from strongly disagree (0) to strongly agree (7) based on activities performed during the preceding 1 week. The overall score is determined as the average of the scores for the 9 items and average scores ≥4 indicate a clinically significant level of fatigue severity with a change of 0.5–0.89 points denoting a minimally important difference. 18
Interpretation of the information obtained with these 2 questionnaires requires that we recognize how the items are scored. For example, consider the following 3 items included in the MFIS questionnaire: 6. I have had to pace myself in my physical activities. 10. I have trouble maintaining physical effort for long periods. 15. I have had trouble finishing tasks that require thinking.
Similar items are included in the FSS questionnaire, but they focus more exclusively on physical aspects of performance.
73
Each item on both questionnaires requires the respondent to estimate how well they were able to perform the specified activity in either the previous 4 weeks (MFIS) or the preceding week (FSS). The resulting score indicates the individual’s perception of their capacity to complete or sustain the activity indicated in each item. The assigned scores, therefore, provide an estimate of fatigability,14,32 with low scores indicating a lower level of fatigability (Table 1). As each score is based on a retrospective estimate, it represents a measure of
The impact of fatigue on physical, psychosocial, and cognitive activities as determined by the MFIS questionnaire, therefore, is based on an individual’s perceived fatigability for 21 prescribed tasks. Similarly, the FSS score for fatigue severity is derived from the average level of perceived fatigability for 9 items that focus on physical activities. Although there is a moderate correlation between the MFIS and FSS scores,18,73,79,80 the MFIS is often favored by some groups, including the National MS Society (USA), due to the multidimensional characteristics of the perceived fatigability reported by individuals with MS.11,81
Some studies have found that fatigue severity (FSS and MFIS) is greater in individuals who self-reported being more disabled (Patient Determined Disease Steps; PDDS), 19 whereas others have reported no more than a weak association between disability status (Expanded Disability Status Scale; EDDS) and the self-reported level of fatigue (MFIS) 34 or a significant association for only one MS phenotype. 18 Moreover, interventions that significantly reduce the level of fatigue often have no more than a minimal impact on disability status.82-85 One factor that confounds these findings, however, is differences in the range of disability levels included in each study.
Objective Fatigability
In addition to perceptions that underlie estimates of the capacity to perform past, present, and future actions, the self-reported level of fatigue can also be influenced by the adjustments that occur during the performance of a prescribed activity (Figure 1). The capacity to sustain a specific task is quantified as
One of the potential moderators of the association between the self-reported level of fatigue and objective fatigability considered by Loy et al.
75
was the type of task used to assess fatigability. They distinguished between tasks that comprised contractions of isolated muscle groups vs those that involved functional activities, such as tests of walking endurance. The association with self-reported fatigue (MFIS and FSS scores) for the 2 types of tasks was not statistically different (
Consistent with the conclusions of the systematic review, Steens et al. 76 found that the FSS scores reported by persons with MS were significantly associated with the decline in maximal voluntary contraction (MVC) force after a sustained maximal isometric contraction with a hand muscle for 2 min. Approximately 45% of the variance in FSS scores was explained by a combination of initial MVC force (muscle strength) and its decline (objective fatigability), despite similar reductions in MVC force for the MS and Control groups. When a self-reported measure of depression was added to the multiple-regression model, the three outcomes explained 77% of the variance in the FSS scores of the MS group. These findings indicate that there is a significant but moderate association between the self-reported level of fatigue (perceived fatigability) and an objective measure of fatigability (decline in MVC force) for a hand muscle.
In contrast, we found that the self-reported level of fatigue was not associated with a measure of objective fatigability (decline in MVC torque) derived from a series of intermittent, fatiguing contractions with the knee extensor muscles.
86
In our study, the MFIS scores were significantly greater (43 ± 14; n = 12) for an MS group than for a Control group (11 ± 8; n = 12) (Figure 2(A)), yet there was no difference between groups in the decline in MVC torque after 60 isometric contractions (10-s on, 5-s off) to a target of 25% MVC torque (Figure 2(B)). These results indicated that a difference in MFIS scores between the 2 groups of participants was not associated with the values achieved on an objective measure of fatigability for this particular protocol. However, Taul-Madsen et al.
87
did find a statistically significant association (r2 = 0.20, Association between fatigue and fatigability in persons with MS. (A): The self-reported level of fatigue (MFIS) at baseline was significantly (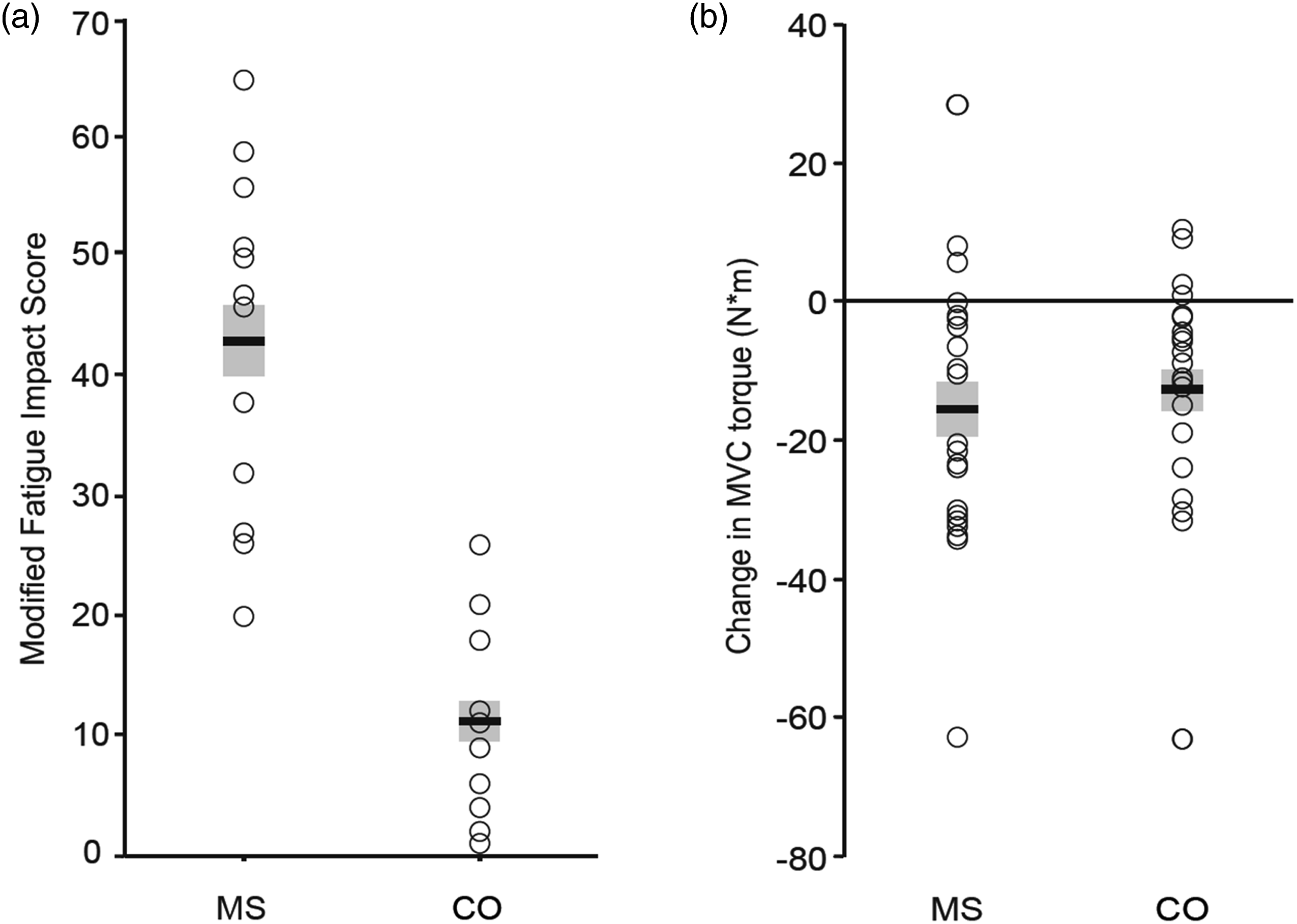
The other approach to examining the association between self-reported fatigue and fatigability is to derive measures of objective fatigability from the performance of functional tasks, such tests of walking endurance. One such metric is the distance or average speed during the last minute of a 6 min test relative to the first minute.88-92 With this approach, Ramari et al. 92 found that the distance (%) walked during the last minute was less for a group of mildly disabled MS participants (EDSS scores 1-3) than a Control group; that is, they exhibited greater objective fatigability during this test. Ramari et al. 92 also found that 39% of the variance in this index of objective fatigability for the MS group was explained by the strength of the knee flexor muscles and measures of postural sway when standing on a foam surface with eyes closed. Unfortunately, they did not measure the self-reported level of fatigue. Although the 6 min test is a common measure of disability status in persons with MS, 93 no studies have examined the association between the variance in the self-reported level of fatigue (MFIS or FSS scores) and measures of objective fatigability derived from tests of walking endurance.
State and Trait Fatigue
In a study that involved a more comprehensive assessment of the association between self-reported fatigue and objective fatigability, Aldumghmi et al.
44
compared state (VAS scores) and trait (questionnaire) measures of fatigue with objective measures of fatigability on three tasks. The trait level of fatigue was measured with the Neurological Fatigue Index (NFI) questionnaire, which estimates perceived fatigability by requiring respondents to answer 23 questions about physical capacity, cognitive capacity, and sleep quality during the preceding 2 weeks. Objective fatigability was assessed with the 6 min walk test, intermittent maximal contractions with a handgrip dynamometer, and an attention-demanding cognitive test. In addition, VAS scores were obtained before and after each of the three tests of objective fatigability. Although the VAS scores increased significantly after each fatigability test, the NFI scores (perceived fatigability) were not significantly associated with the level of objective fatigability observed after either the 6 min walk or the handgrip tests. However, the scores for the physical subdomain of the NFI questionnaire were significantly correlated with the decline in performance (response variability) on the cognitive test (r = 0.33,
Nonetheless, some evidence suggests there is a significant association between VAS scores (state fatigue) and questionnaire-derived estimates of perceived fatigability.
40
For example, Krupp et al.
38
found a significant correlation (r = 0.68,
Adaptations That Influence the Level of Fatigue
Although the level of fatigue experienced by persons with MS can be reduced by such interventions as physical exercise,95-98 cognitive behavioral therapy,95,96 and self-management of fatigue,99-101 the critical adaptations that need to be achieved to lessen the burden of fatigue remain uncertain.16,26-28 One approach to identifying the adaptations responsible for the progressive worsening in the level of fatigue experienced by persons with MS is to examine the associations that emerge during intervention studies. This can be accomplished by comparing changes in state and trait measures of fatigue with changes in key outcome variables, such as the capacity of the nervous system to generate muscle-activation signals (voluntary activation), functional connectivity within interoceptive networks, measures of cognitive function, and quality-of-life metrics.
Perceived and Objective Fatigability
Most studies that have examined the association between the self-reported level of fatigue and objective fatigability have used a measure of motor function as the outcome variable to quantify fatigability. The outcomes have included such measures as MVC force,71,76,77,86,102 muscle endurance,103-105 contractile function of leg muscles,103,106 walking performance,44,82,107-110 and metabolic cost of walking.111-114 Also, a few studies have examined the association between self-reported levels of fatigue and objective measures of cognitive function (attention, episodic memory, executive function, processing speed, and working memory).52,94,115,116
Despite the moderate association between self-reported levels of fatigue (MFIS, FSS) and objective measures of fatigability, multiple-regression models that include several other outcome variables can often explain greater amounts of the variance in the self-reported scores for fatigue. For example, Coates et al.
71
found that much of the variance in FSS scores for persons with MS was explained by three variables (R2 = 0.739,
Although these studies have been able to characterize differences in motor function between people with MS and healthy controls, they typically fail to capture the substantial influence of the extra demands on the nervous system of persons with MS when performing prescribed tasks.33,117-127 Critically, the reduced capacity of the central nervous system in persons with MS to generate an adequate level of voluntary activation is significantly correlated with the self-reported level of fatigue.71,121,128,129
One example of this effect is shown in Figure 3, which compares the increase in rating of perceived exertion (RPE) for 2 groups of participants during the intermittent fatiguing contractions shown in Figure 2. Despite similar declines in MVC force immediately after the 60 intermittent contractions (MS: −16 ± 19 N•m; Control: −13 ± 16 N•m), we found that RPE scores increased more rapidly during the task for MS participants.
86
A similar finding was reported by Thickbroom et al.
126
during 120 intermittent (7-s on, 3-s off) contractions (40% MVC force) with a hand muscle. They observed similar declines in MVC force after the fatiguing contraction for the 2 groups of participants (MS: −29 ± 3%; Control: −24 ± 5%), but greater final RPE scores at the end of the task for the MS group (MS: 17 ± 0.34; Control: 15 ± 0.7). However, Thickbroom et al.
126
found that the increase in the RPE scores (state fatigue) reported by the MS participants was not statistically associated with their MFIS scores (trait fatigue). Increase in rating of perceived exertion (RPE) as measured on a Borg scale during 60 intermittent contractions performed with the knee extensor muscles. The isometric contractions were performed to a target force of 25% of maximum. The increase in RPE score was significantly (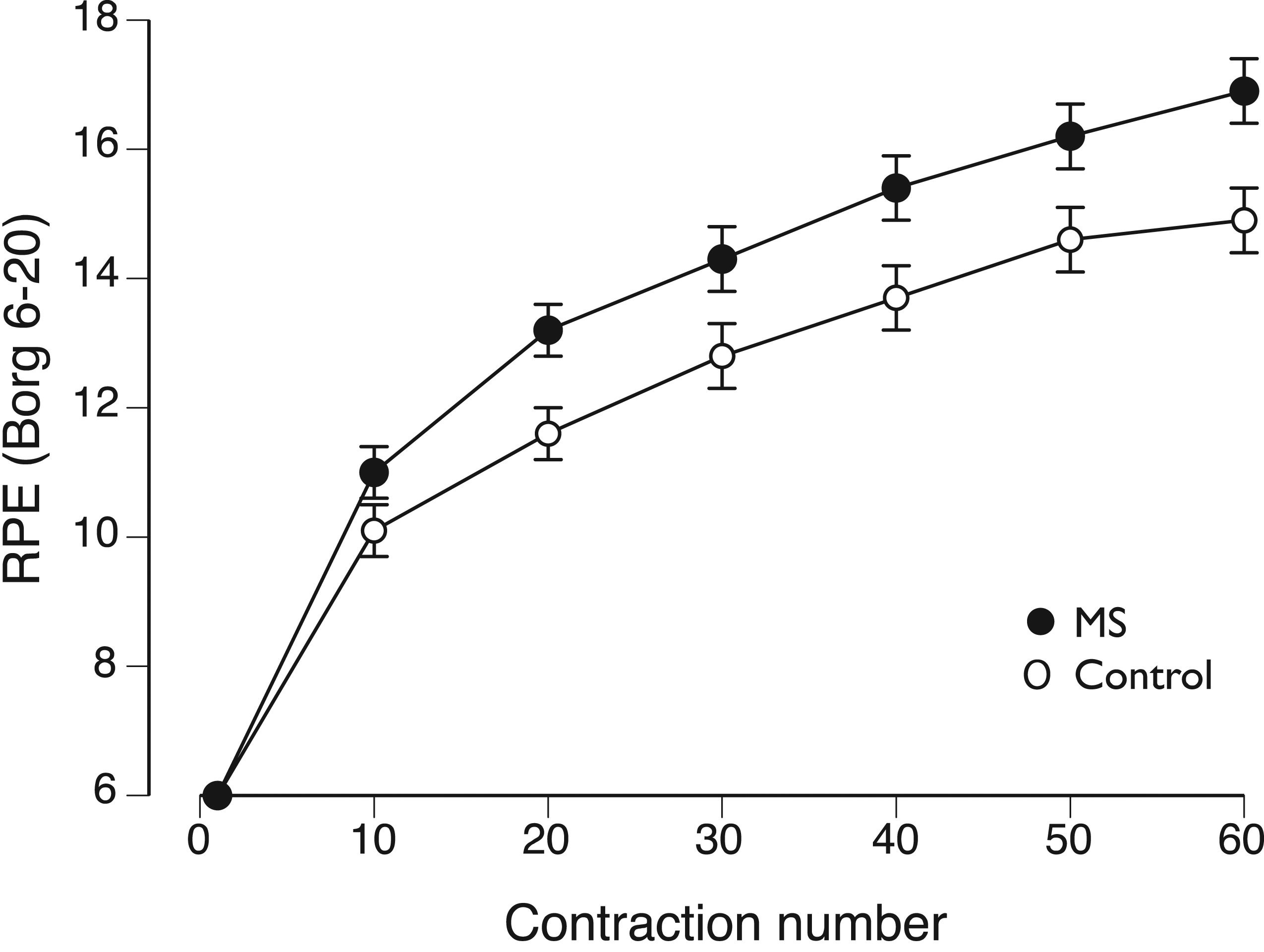
Thickbroom et al.
126
suggested that the more rapid increases in RPE scores for persons with MS was attributable to their reduced capacity to generate adequate levels of voluntary activation during the fatiguing contraction. Consistent with this suggestion, Steens et al.
77
found that despite a similar decline in MVC force for a hand muscle after a maximal contraction that was sustained for 2 min (MS: −67 ± 9%; Control: −65 ± 8%), the decrease in MVC force was associated with a different outcome measure for each group: MS group – the decline in voluntary activation as assessed with the twitch-interpolation technique (R2 = 0.51, Decrease in voluntary activation during an MVC sustained for 2 min with a hand muscle. Voluntary activation was measured by pairs of electrical stimuli applied to the ulnar nerve once every 18 s during the sustained contraction. The decrease in the level of voluntary activation was significantly (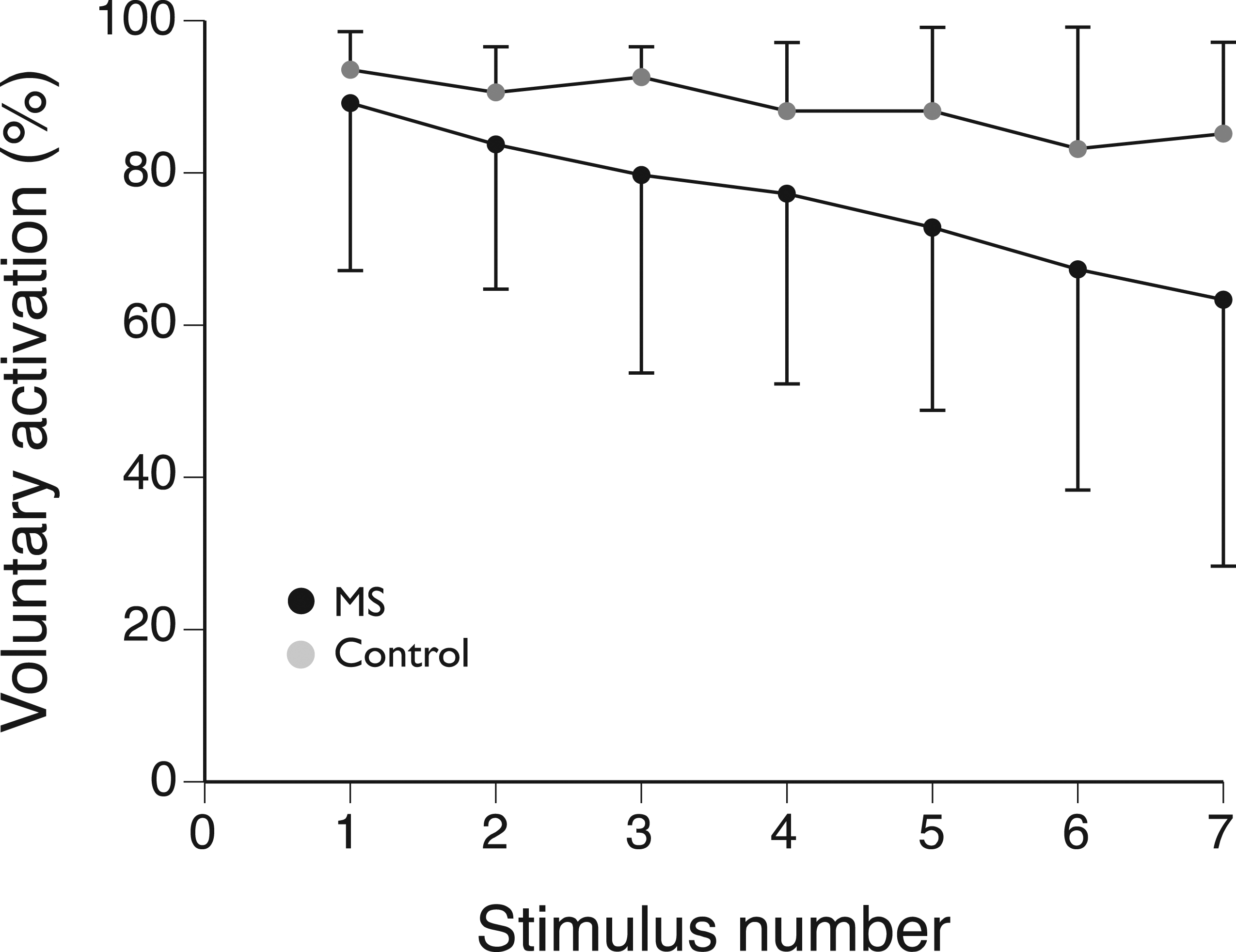
Although these studies demonstrate that the state level of fatigue (RPE scores) increases more rapidly for persons with MS during fatiguing contractions, the adjustments are largely unrelated to baseline measures of self-reported fatigue (MFIS and FSS scores). One approach to identifying the adaptations underlying self-reported levels of fatigue is to examine the associations observed after exercise interventions.96,130-133 Despite the many exercise interventions that have been examined, including studies of endurance training, muscle power training, strength training, task-oriented training, combined types of training, and training with less-conventional protocols (eg, yoga), relatively few have examined the mechanisms of action underlying exercise-induced decreases in the severity of fatigue. One exception was a study that examined the influence of 12 wks of strength training on muscle strength (knee extensors and flexors), walking performance, stair-climbing ability, self-reported fatigue, mood, and quality of life. 131 They found that the intervention reduced the self-reported level of fatigue (FSS scores), but that the decrease in fatigue was not correlated with changes in muscle strength, walking performance, or stair-climbing ability. However, they suggested that the improvement in fatigue (lower FSS scores) was likely related to an increase in the capacity of the nervous system to activate muscles (voluntary activation), which they had previously found to be deficient in persons with MS. 128
Self-Reported Fatigue and Voluntary Activation
Measures of Walking Performance and Disability Status for Individuals with MS Before and After a 6-week NMES Intervention. 82
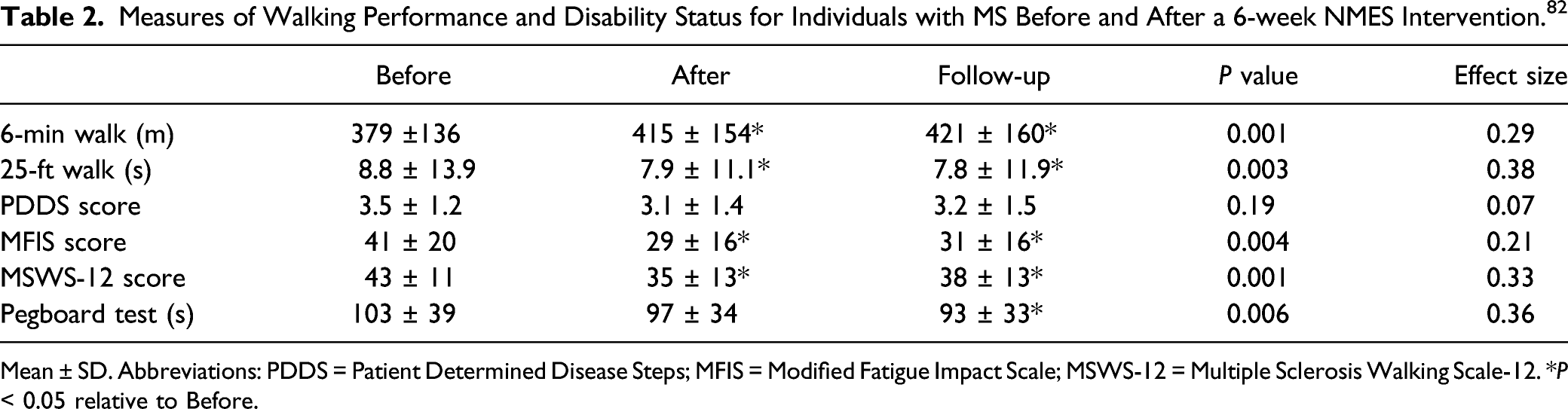
Mean ± SD. Abbreviations: PDDS = Patient Determined Disease Steps; MFIS = Modified Fatigue Impact Scale; MSWS-12 = Multiple Sclerosis Walking Scale-12. *
In a follow-up study, Alenazy et al. 138 examined the influence of 9 treatment sessions (3x/week for 3 weeks) with transcutaneous electrical nerve stimulation (TENS) on motor function and disability status in individuals with MS. TENS involves the application of a lesser current (< 20 mA) than NMES (∼50 mA) and targets the activation of sensory fibers. 139 TENS was delivered through conductive pads placed over rectus femoris, tibialis anterior, the median nerve, and the thenar eminence. Participants sat in a relaxed position while TENS was delivered to each location in one limb at a time. As indicated in Table 3, the MS group experienced improvements in walking endurance (2 min test), maximal walking speed (25 ft walk), the number of repetitions performed in the chair-rise test, and time to complete the grooved pegboard test with the right hand. The R2 values for the MS group indicated moderate effect sizes (Table 3).
Measures of Motor Function and Disability Status for Individuals with MS Before and After a 3-Week TENS Intervention. 138
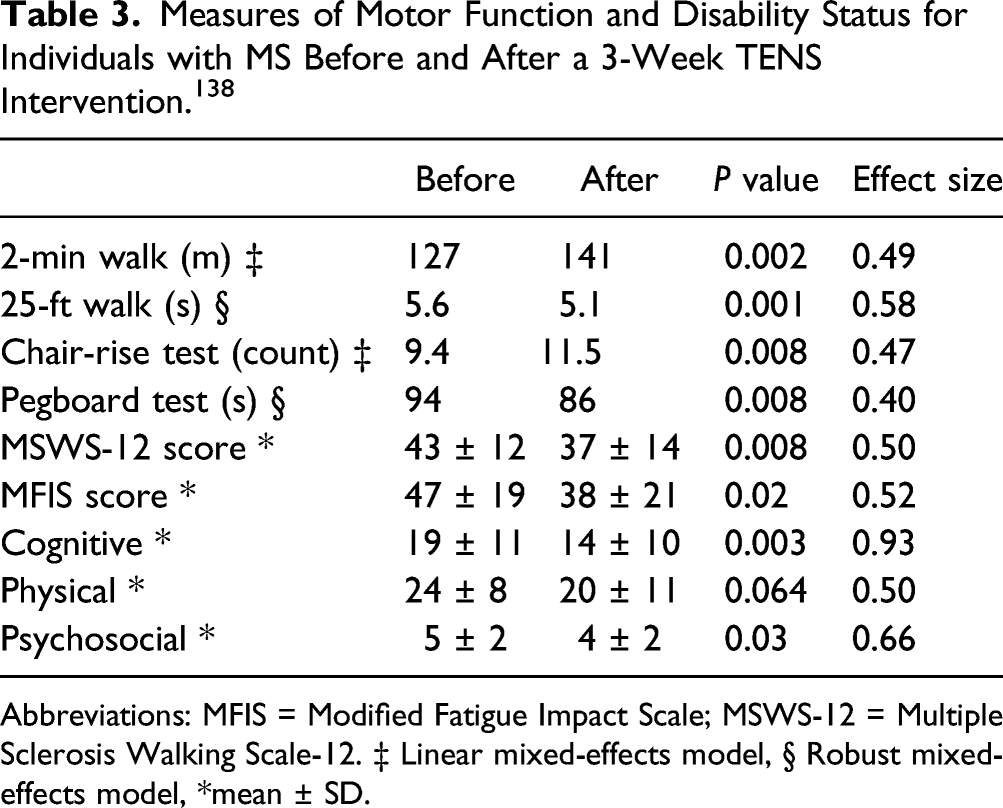
Abbreviations: MFIS = Modified Fatigue Impact Scale; MSWS-12 = Multiple Sclerosis Walking Scale-12. ‡ Linear mixed-effects model, § Robust mixed-effects model, *mean ± SD.
These findings indicate that several treatment sessions in which electrical stimulation is applied to the limbs of persons with MS can reduce the self-reported level of fatigue.82,135,138 This outcome has been found in studies with protocols that differ in the intensity and distribution of the applied current, the target muscles, the concurrent actions of the participants, and a range of disability levels. However, the findings on the association between the decrease in the self-reported level of fatigue and the improvements in motor function are mixed. Moreover, little is known about either the adaptations responsible for the decrease in fatigue severity or the optimal strategies required to achieve this benefit. Nonetheless, our point-of-view paper suggests that the framework needed to address these issues has been clearly established (Figure 1).
Summary
The level of fatigue reported by an individual at a specific instant in time (state fatigue) is derived from interoceptive pathways that provide information about the regulation of homeostasis. A reduced capacity to accommodate a challenge to homeostasis, such as due to the presence of MS, appears to be responsible for elevated state levels of fatigue. In contrast, the typical approach used clinically to assess fatigue is to quantify its impact on the work capacity (fatigability) of the individual. Clinical measures of fatigue can be obtained from questionnaires (MFIS, FSS, and NFI), which are based on retrospective estimates of work capacity (perceived fatigability), or from the change in an outcome measure during the performance of a prescribed task (objective fatigability). Changes in the state level of fatigue (VAS scores) are no more than weakly correlated with questionnaire scores (perceived fatigability), which indicates that the 2 measures assess different aspects of the fatigue experienced by persons with MS. Significantly, objective fatigability (measured work capacity) has no more than a moderate association with the self-reported level of fatigue. However, the levels of fatigue and fatigability can be modulated by reward and pain pathways and the self-reported level of fatigue can be reduced by various interventions that involve either or both voluntary and electrically evoked contractions. To advance knowledge in this field and to enable its translation to the effective management of the fatigue reported by persons with MS, it is critical that we understand the distinction between fatigue and fatigability and appreciate that the 2 attributes emerge from different sets of adaptations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship or publication of this article.
