Abstract
Parkinson disease (PD) and other related diseases with α-synuclein pathology are associated with a long prodromal or preclinical stage of disease. Predictive models based on diagnosis of idiopathic rapid eye movement (REM) sleep behavior disorder (iRBD) make it possible to identify people in the prodromal stage of synucleinopathy who have a high probability of future disease and provide an opportunity to implement neuroprotective therapies. However, rehabilitation providers may be unaware of iRBD and the motor abnormalities that indicate early motor system dysfunction related to α-synuclein pathology. Furthermore, there is no existing rehabilitation framework to guide early interventions for people with iRBD. The purpose of this work is to (1) review extrapyramidal signs of motor system dysfunction in people with iRBD and (2) propose a framework for early protective or preventive therapies in prodromal synucleinopathy using iRBD as a predictive marker. Longitudinal and cross-sectional studies indicate that the earliest emerging motor deficits in iRBD are bradykinesia, deficits performing activities of daily living, and abnormalities in speech, gait, and posture. These deficits may emerge up to 12 years before a diagnosis of synucleinopathy. The proposed rehabilitation framework for iRBD includes early exercise-based interventions of aerobic exercise, progressive resistance training, and multimodal exercise with rehabilitation consultations to address exercise prescription, progression, and monitoring. This rehabilitation framework may be used to implement neuroprotective, multidisciplinary, and proactive clinical care in people with a high likelihood of conversion to PD, dementia with Lewy bodies, or multiple systems atrophy.
Introduction
Traditional rehabilitation therapies for Parkinson disease (PD) are delivered when physical function is impaired and substantial neurodegeneration (approximately 50%-90% loss of nigral dopaminergic cells) has already occurred, 1 likely limiting the effectiveness of physical interventions. However, it is now well recognized that PD is associated with a long prodromal 2 or prediagnostic 3 stage of disease, lasting years to decades, that may provide a window during which neuroprotective interventions can be proactively applied. A clinical population that is known to represent the early stages of α-synuclein pathology (ie, prodromal synucleinopathy) and have a high likelihood of progression to overt neurodegenerative disease are people with idiopathic rapid eye movement (REM) sleep behavior disorder (iRBD). 4 Thus, the presentation of iRBD alone in a patient necessitates the need for the initiation of therapies that slow or prevent progression of neurodegenerative α-synucleinopathy disease. 5 Currently, there is no rehabilitation framework for how to treat people in the preclinical, prodromal, or prediagnostic stages of synucleinopathy.
Early motor abnormalities observed in iRBD offer valuable insight regarding the onset and severity of motor system degeneration and relevant early or protective therapies that may delay or mitigate future disability. The aims of this work are, first, to summarize the motor abnormalities present in people with iRBD and, second, to propose a rehabilitative framework for physical and exercise-based therapies in people with prodromal synucleinopathy. This framework may be used to design effective neuroprotective trials and proactive clinical care in iRBD. Although individuals with iRBD may progress to a variety of α-synucleinopathy phenotypes (eg, PD, dementia with Lewy bodies, or multiple systems atrophy), the focus of the present work is centered on prior research in PD and clinical applications in prodromal or early PD.
Preclinical and Prodromal Synucleinopathy
α-Synucleinopathy is a subset of neurodegenerative disorders characterized by the presence of α-synuclein insoluble aggregates in neuronal or glial cells. 6 Although PD is the most common synucleinopathy, other phenotypes of synucleinopathy include dementia with Lewy bodies and multiple systems atrophy.6,7 The time course of synucleinopathy may be separated into 3 stages of disease progression: preclinical, prodromal, and clinical (Table 1). The preclinical stage is considered the point at which the neurodegenerative process has begun, but there are no overt signs or symptoms of disease. 4 The prodromal stage is considered the time period during which nonspecific (nonmotor) and/or specific (motor) signs of synucleinopathy or parkinsonism are present, but a clinical diagnosis cannot be made. 4 Clinical synucleinopathy, for example PD, is diagnosed once the cardinal motor signs (ie, bradykinesia, rigidity, and/or resting tremor) have sufficiently manifested. Given that more than 30% to 70% of nigrostriatal dopaminergic neurons have already been lost at the time of PD diagnosis, 8 it is vital that patients in the preclinical and prodromal stages of synucleinopathy be identified as early as possible in order to initiate interventions that can slow or prevent disease progression.
Operational Definitions. a

Abbreviations: PD, Parkinson disease; REM, rapid eye movement; RSWA, REM sleep without atonia; RBD, REM sleep behavior disorder.
Among the various motor and nonmotor biomarkers related to parkinsonism (see Heinzel et al 11 for a review), a diagnosis of iRBD is recognized as the strongest predictor of future progression to PD.4,12 iRBD is a parasomnia characterized by elevated muscle activity (REM sleep without atonia) and dream enactment behavior during REM sleep. 13 Of the estimated 1% of the population affected by RBD, 14 roughly 81% to 90% will eventually develop a neurodegenerative disorder, and therefore, this is recognized as a prodromal or early stage of α-synucleinopathy. 13 When RBD is idiopathic (iRBD), an estimated 74% of these individuals are predicted to develop a synucleinopathy disease within 12 years. 5 Thus, iRBD provides a critical opportunity to study people with early-stage synucleinopathy who have a high likelihood to progress to PD.
Furthermore, RBD or RSWA are present in roughly 40% to 55% of people with PD and are associated with a more severe, rapid-declining subtype of PD. 15 In terms of the classical phenotypes, the coexpression of PD with RBD or RSWA is less likely to be a tremor predominant subtype and more likely to present as an akinetic-rigid subtype with greater rigidity, 16 increased postural instability, gait disturbances, and increased risk of falls. 15 Clinically, it is important to identify patients with PD and RBD because they may benefit from early or intensive therapies as a result of their high risk for accelerated functional decline.
Abnormalities in iRBD
Nonmotor Abnormalities in iRBD
Nonmotor features that predict future synucleinopathy in people with iRBD include loss of olfaction, impaired color acuity, constipation, sexual and urinary dysfunction, autonomic abnormalities, and cognitive decline.7,17 Based on the presence of nonmotor impairments, longitudinal studies suggest that the prodromal phase may last 20 to 25 years.7,17 The earliest nonmotor abnormalities are reported to be loss of olfaction and orthostatic hypotension. 7 For a more in-depth discussion on nonmotor abnormalities in people with iRBD, see Högl et al. 18
Motor Abnormalities in iRBD
Roughly 34% of people with iRBD present with subtle extrapyramidal motor signs,5,19 and such signs may occur as early as 6.3 to 12.9 years before phenoconversion to a neurodegenerative synucleinopathy disease.7,20 Importantly, motor signs are among the strongest predictor of future phenoconversion. People with iRBD and motor signs are more than 3 times as likely (hazard ratio = 3.16) to phenoconvert compared to people without motor signs. 5 Motor signs identified in people with iRBD include bradykinesia, functional deficits, facial and speech abnormalities, gait and postural impairment, rigidity, and tremor. The evolution of motor signs has been shown to follow a chronological pattern starting after nonmotor signs7,20 (Figure 1).
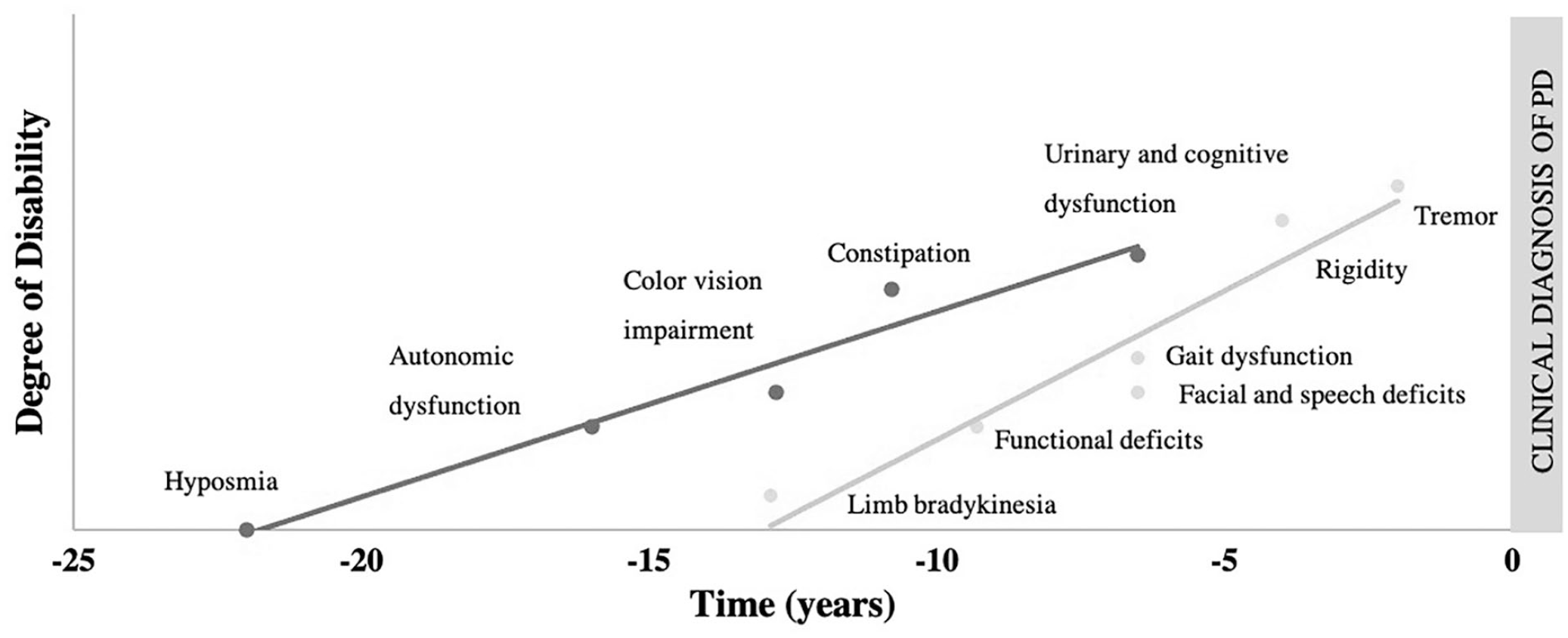
Estimated lead time of prodromal markers: Lead times for nonmotor and motor signs are approximated from data found in Postuma et al 20 and Fereshtehnejad et al. 7 Limb bradykinesia lead time is marked using quantitative data. Time in years refers to the prodromal stage with time point zero being the time of phenoconversion to an overt α-synucleinopathy such as Parkinson disease, dementia with Lewy bodies, or multiple systems atrophy.
Limb Bradykinesia
Limb bradykinesia is the earliest motor feature to manifest in people with iRBD who are likely to convert to a neurodegenerative α-synucleinopathy.7,20 Clinical assessment of bradykinesia severity in people with iRBD is conducted using the Unified Parkinson Disease Rating Scale part III (UPDRS-III) using repetitive alternating movements of the fingers, hand, forearm, and legs. Abnormal bradykinesia scores on the UPDRS-III manifest roughly 5 years prior to diagnosis.7,20 However, more sensitive quantitative measures of upper-limb movement speed and accuracy, such as the alternate-tap test, 21 may show deviation from age-normal scores 6 to 12 years before conversion. 7
Parkinsonian bradykinetic behaviors have also been detected in iRBD using high-precision, quantitative methods. Instrumented analyses of index and thumb tapping have revealed decreased movement speed 19 and a sequence effect 22 in people with a short duration of iRBD symptoms (~5 years), suggesting early parkinsonian bradykinesia in people with iRBD. However, there are no longitudinal studies utilizing instrumented measures of bradykinesia in iRBD. It remains unclear how parkinsonian bradykinesia manifests initially and if characteristics of bradykinesia, hypokinesia, or akinesia can differentiate between populations with healthy aging, dementia, or parkinsonism.
Motor Experiences of Daily Living and Speech
Physical and functional deficits in people with iRBD have been identified using the UPDRS-II and III, with a deviation from normal scores beginning up to 9.8 years before phenoconversion. 7 Basic activities of daily living demonstrate a deviation from normal, but near the time of phenoconversion (approximately 1-3 years). 7 Specific deficits related to activities of daily living include self-reported changes in handwriting, turning in bed, walking speed, and speech.
Items from the UPDRS-II and III that demonstrate predictive value in iRBD include corticobulbar-mediated functions of speech and facial muscle tone. The lead time of facial and speech abnormalities has been estimated to range from 6.5 to 9.8 years.7,20 Although facial and speech abnormalities are noted perceptually in only 2% of people with RBD, quantitative speech abnormalities are estimated to be present in up to 88%. 23 Specific vocal characteristics related to monopitch tone, inappropriate silences, and rate of follow-up speech segments have been shown to best discriminate between people with PD and controls, whereas people with iRBD demonstrate large individual variability. 24 Speech abnormalities associated with iRBD are hypothesized to reflect bradykinesia and impaired muscle initiation (ie, akinesia) in vocal musculature. 24 When comparing instrumented voice assessments to other motor features such as tremor, hand tapping, and posture, vocal quality was the best group discriminator between those with iRBD, PD, and controls. 25 These findings may indicate that instrumented evaluations of speech may yield a highly sensitive marker of future phenoconversion in iRBD. There are no published reports of quantitative speech analysis in longitudinal studies of iRBD.
Gait and Postural Abnormalities
In people with iRBD, reduced gait speed and postural abnormalities are present and resemble gait dysfunction in early PD. Reduced gait speed as measured by the Timed-up and Go outcome have been shown to first occur approximately 6 years before phenoconversion in people with iRBD.5,7,20,26 Although instrumented studies of gait speed indicate mixed results,26-28 gait variability appears to be dysregulated in iRBD. Step time variability has been shown to be increased in iRBD cohorts, similar to age-matched individuals with PD. 27 Yet the variability of gait characteristics has also been found to be reduced when at-home, long-duration gait bouts are assessed in iRBD compared to controls. 26 These seemingly conflicting reports may reflect a lack of automaticity and flexibility of gait regulation in people with iRBD or result secondary to poor postural control.
Abnormal postural responses are also similar in people with iRBD and early PD. Gait tasks in combination with a cognitive task (ie, dual-task gait) have been shown to unveil deficient postural compensation strategies in people with iRBD. During dual-task gait, people with iRBD increase step asymmetry and step width variability, 28 which is similar to changes seen in early PD. Additionally, during gait initiation, people with iRBD have been shown to demonstrate PD-like deficits, with reduced anticipatory postural adjustments and fractionated muscle burst patterns. 29 Considering that iRBD is associated with α-synuclein–driven brainstem dysfunction, 14 loss of postural control may be the earliest manifesting gait impairment because the pedunculopontine nucleus and other brainstem structures are integral to regulate gait and postural control. 29 There are no longitudinal studies in iRBD using quantitative gait and postural response analyses.
Rigidity and Tremor
Clinically, rigidity and tremor are quantified using the UPDRS-III and are estimated to manifest 3.5 to 4.4 years and 1.3 to 1.5 years, respectively, before phenoconversion in people with iRBD.7,20 The relatively late manifestation of rigidity or tremor, when combined with bradykinesia, typically fulfills the diagnostic criteria for clinical PD and, therefore, offers little additional predictive value. There are no longitudinal studies utilizing instrumented measures of tremor or rigidity in people with iRBD.
Framework for Prodromal Exercise and Rehabilitation in IRBD
In an effort to reduce the severity and slow the progression of neurodegenerative disease, it is imperative to identify persons with a high risk of future synucleinopathy or early signs of motor system dysfunction to implement neuroprotective interventions. Currently, there are no known pharmacological or nonpharmacological interventions that are known to delay or prevent PD onset. 30 However, there is compelling evidence from experiments in animal models of parkinsonism 31 and trials in humans 32 that exercise and physical medicine interventions may affect the expression and progression of disease, with little risk of adverse effects. Thus, it is ideal to start exercise and physical medicine interventions during the prodromal stage or in people with iRBD.33,34 To date, a framework for exercise application or clinical management in iRBD has not been established. Here, we propose a framework for implementing exercise and rehabilitative services in individuals with iRBD. This clinical framework includes the primary role of sleep clinicians and movement disorders neurologists to initiate consultative services from traditional rehabilitation providers. Rehabilitation providers can then initiate an intervention centered on exercise prescription, progression, and monitoring or address physical impairments as they manifest. The rationale for this framework comes from existing evidence in animal models, epidemiological studies, and exercise interventions applied in early or mild PD cohorts.
Rationale for Neuroprotective Effects of Exercise in Prodromal Synucleinopathy
There is a surmounting body of evidence that indicates physical activity and exercise have neuroprotective effects that may reduce or mitigate the severity of neurodegenerative disease. 35 The biological mechanisms recognized to underlie exercise as a neuroprotective therapy include enhanced neurogenesis, elevated neurotrophic factors, reduced neuroinflammation, and decreased oxidative stress. 31
Exercise interventions in animal models of α-synuclein driven PD indicate that there is a beneficial effect of exercise on motor and nonmotor symptoms while also mitigating degeneration of nigral systems. Two animal models of PD that are relevant to understanding how exercise therapy may be beneficial in iRBD are the rotenone and the adeno-associated virus α-synuclein model. Both the rotenone and the adeno-associated virus α-synuclein model lead to the development of α-synuclein in nigral neurons and the production of parkinsonian motor symptoms. Using the rotenone model, it has been shown that early aerobic exercise for 2 or 6 weeks was effective to prevent elevated α-synuclein levels while increasing tyrosine hydroxylase levels in the substantia nigra.36,37 In addition, aerobic exercise for 2 weeks following rotenone exposure was found to reduce the loss of serotonergic markers in the dorsal raphe and mitigate depressive behaviors. 38 Rats that receive aerobic exercise following rotenone exposure also demonstrate improved motor performance in the rotarod test compared to sedentary rats, 37 suggesting preservation of motor function in exercising animals. A study of voluntary aerobic exercise using the adeno-associated virus α-synuclein model has also indicated that exercise reduces neuronal loss in the substantia nigra. 39 Although this study failed to demonstrate a beneficial effect of voluntary running on motor function, it did indicate beneficial effects of exercise on hippocampal-associated memory. While the mechanism of neuroprotection from exercise is unclear, these findings support the idea that aerobic exercise can delay the neurodegenerative process related to α-synucleinopathy in the substantia nigra and dorsal raphe while mitigating losses in motor and cognitive domains. For a full review of exercise-induced effects in animal models of PD, please see Crowley et al. 31
Epidemiological studies also indicate that physical activity and exercise may play a protective role in reducing the risk of PD (for a review, see Fang et al 40 ). A relationship between increased physical activity and decreased prevalence of prodromal nonmotor symptoms in aging adults has been recently reported. 41 A large longitudinal trial has provided further evidence that even a medium level of physical activity lowers the risk of developing PD. 42 Following PD diagnosis, regular physical activity has been associated with slower declines in functional mobility and health-related quality of life, 43 indicating the value of early exercise and physical activity intervention.
Exercise as an Intervention for Prodromal Synucleinopathy
Several promising exercise interventions have been studied in PD that may translate to iRBD. The following interventional recommendations have been drawn from randomized controlled trials in samples of people with early PD (disease duration of less than 5 years) and intervention of at least 12 weeks. Because of a paucity of exercise research in early PD, studies of cohorts with an average disease duration of 5 to 7 years are also considered. There is an immediate need for research to be conducted in individuals with prodromal synucleinopathy or early PD to better understand the impacts of different exercise types on improving or slowing the progression of parkinsonism and synucleinopathy.
Aerobic Training
Aerobic exercise is recommended as a prodromal intervention for iRBD to promote neuroprotection and possibly mitigate or delay the onset of synucleinopathy disease. Aerobic exercise in early PD has been shown to increase aerobic capacity and endurance and slow the progression of motor symptoms.44-46 A study comparing treadmill training in de novo PD patients (ie, before medications are initiated) at high or moderate intensities (80% or 60% maximum heart rate, respectively) for 6 months showed that only high-intensity aerobic exercise was superior to usual care with respect to the UPDRS-III measures of progression. 44 Home-based stationary cycling in early PD for 6 months has also been shown to significantly reduce the rate of progression of motor symptoms and improve cardiovascular fitness compared to stretching and relaxation. 45 Recently, cycling for 3 months has been shown to generate increased cortico-striatal neuroplasticity and dopamine release compared to stretching exercises. 46 Furthermore, evidence for aerobic exercise to mitigate the loss of motor function was found in mild PD cohorts who complete 3 to 6 months of Nordic walking exercise.47,48 Improvement in alternating movements following 6 months of moderate-intensity aerobic exercise has been noted in mild PD, 48 suggesting a possible therapy for early-onset bradykinesia in iRBD. In summary, evidence from aerobic exercise trials in early PD, as well as in parkinsonian animal models, suggests that aerobic exercise may impart beneficial effects on motor function by preserving dopaminergic and corticostriatal systems in the presence of disease.36,37,46 For a review of aerobic exercise in PD, please see Schootemeijer et al. 32
Prescription of aerobic exercise in iRBD should include factors of intensity, duration, and frequency. Moderate- to high-intensity exercise with a duration of 30 to 45 minutes a minimum of 3 times per week is recommended.44,45 If safely tolerated, high-intensity (>70% maximal heart rate) aerobic exercise should be recommended over moderate-intensity aerobic exercise. 44 Common modes of aerobic exercise include cycling, treadmill exercise, and walking programs; however, patient preference should be prioritized if intensity and duration goals can be met.
Progressive Resistance Training
Progressive resistance training (ie, resistance exercise that increases the resistance load or intensity over time) is recommended in iRBD because it may reduce the progression of both motor and cognitive symptoms. Recent reviews in PD indicate that progressive resistance exercise has a potent effect on muscle strength, physical function, and parkinsonian symptoms in mild to moderate PD.49,50 To our knowledge, there are no published resistance exercise trials in early PD cohorts with an average disease duration of less than 5 years. However, long-term resistance exercise studies in mild to moderate PD, with an average disease duration of 5 to 7 years, have shown that there are beneficial effects on strength, 51 physical function,52,53 and Parkinsonian motor symptoms.52,54-56 The longest resistive exercise trial to date in PD was 2 years in duration and revealed that progressive resistance exercise, but not nonprogressive general exercise that included some strengthening, significantly reduced clinical measures of motor severity, improved physical function, reduced bradykinesia, improved cognitive function, and increased strength.54,55 Resistance exercise has also been found to improve standardized ratings of limb bradykinesia, 56 peak velocity of ballistic arm movements, 55 and functional gait speed.52,57 Relevant to the motor abnormalities observed in iRBD, resistance exercise may be effective to address early-onset limb bradykinesia and gait hypokinesia. Yet it is unclear if progressive resistance exercise has disease-modifying effects despite the consistent reports of mitigating parkinsonian symptoms. 50
Progressive resistance training protocols for iRBD should be prescribed 2 to 3 times/wk, for 20 to 60 minutes or more.54,57 Specifically, progressive resistance exercise that systematically increases the intensity of muscular demand over time is recommended to maximize neuromuscular gains. 54 Considerations should be given to extensor muscle–focused exercises because extensor muscle weakness tends to be greater than flexor weakness in people with PD 58 and may contribute to postural abnormalities, fall risk, and loss of physical function. Additionally, resistance exercises may be most beneficial when progressive resistance and instability are combined to maximize effects on physical function and neuromuscular adaptation. 59
Multimodal Exercise
Multimodal exercise programs performed individually or in a group recreational exercise setting are recommended for people with iRBD because they may improve physical function and quality of life and reduce sedentary habits. In early PD, multimodal exercise programs have been found to be effective to mitigate progressive mobility loss. Particularly dance and yoga appear to be effective at improving parkinsonian motor symptoms, physical function, and gait speed when delivered at a dose of 60 minutes, twice per week.60-62 Sardinian dance training compared to usual care in early PD has been shown to improve motor symptoms, functional mobility, balance, and fatigue. 61 When expanding the list of studies to those including people with PD for 5 to 7 years, we found further support for Tango dance 63 and yoga 64 as well as for Tai Chi/Qigong.65,66 Tango dance lessons given twice weekly in mild-moderate PD have been shown to have a variety of beneficial effects such as improved balance, functional mobility, and walking speed and slowing the progression of parkinsonian motor symptoms. 63 Tai Chi/Qigong are associated with mixed effects on motor symptoms but may be effective to improve balance and physical function.65,66 Other multimodal exercise programs that may be applicable in prodromal synucleinopathy, but are not discussed because of variations in study design or disease duration, include group activities such as boxing, 67 sensorimotor agility programs, 68 individualized home exercise programs, 69 and physical therapy (PT). 70
For patients with iRBD who show no overt motor abnormalities, multimodal exercise may be especially effective to promote sustained independent community exercise. To promote adherence to long-term exercise participation, recommendations should be made for a mode of exercise that is appealing and likely to be completed by the individual. Therefore, factors that ensure consistent exercise participation (3 d/wk at a minimum) should be prioritized and recommendations for mode of exercise centered on patient preference. Clinically, it may be especially important for a multimodal exercise prescription in iRBD to include remote or in-person consultative services with a rehabilitation expert.
Framework for Integrating Rehabilitation Into IRBD Care
Clinical practice guidelines in PD suggest that PT, occupational therapy (OT), and speech language pathology (SLP) should be considered early after diagnosis.71-73 This antiquated approach fails to recognize the clear evidence that PD is a characterized by years to decades of progressive neurodegeneration and loss of motor function. Furthermore, considering the growing evidence that aerobic exercise may be neuroprotective, in addition to the known cardiovascular and mental health benefits of physical activity, rehabilitation services should be integrated into care for people with prodromal synucleinopathy. The evidence is clear that functional deficits, bradykinesia, gait impairment, and voice deficits are commonly present in iRBD indicating impending phenoconversion to a neurodegenerative synucleinopathy such as PD, dementia with Lewy bodies, or multiple systems atrophy. Therefore, rehabilitation services should be started before diagnosis of a neurodegenerative α-synucleinopathy disease in people with iRBD. Staging of services should be based on symptom progression because of the predictive value these signs have in identifying those at risk of future neurodegenerative synucleinopathy (Table 2).
Timeline of Services in a Rehabilitation Framework.
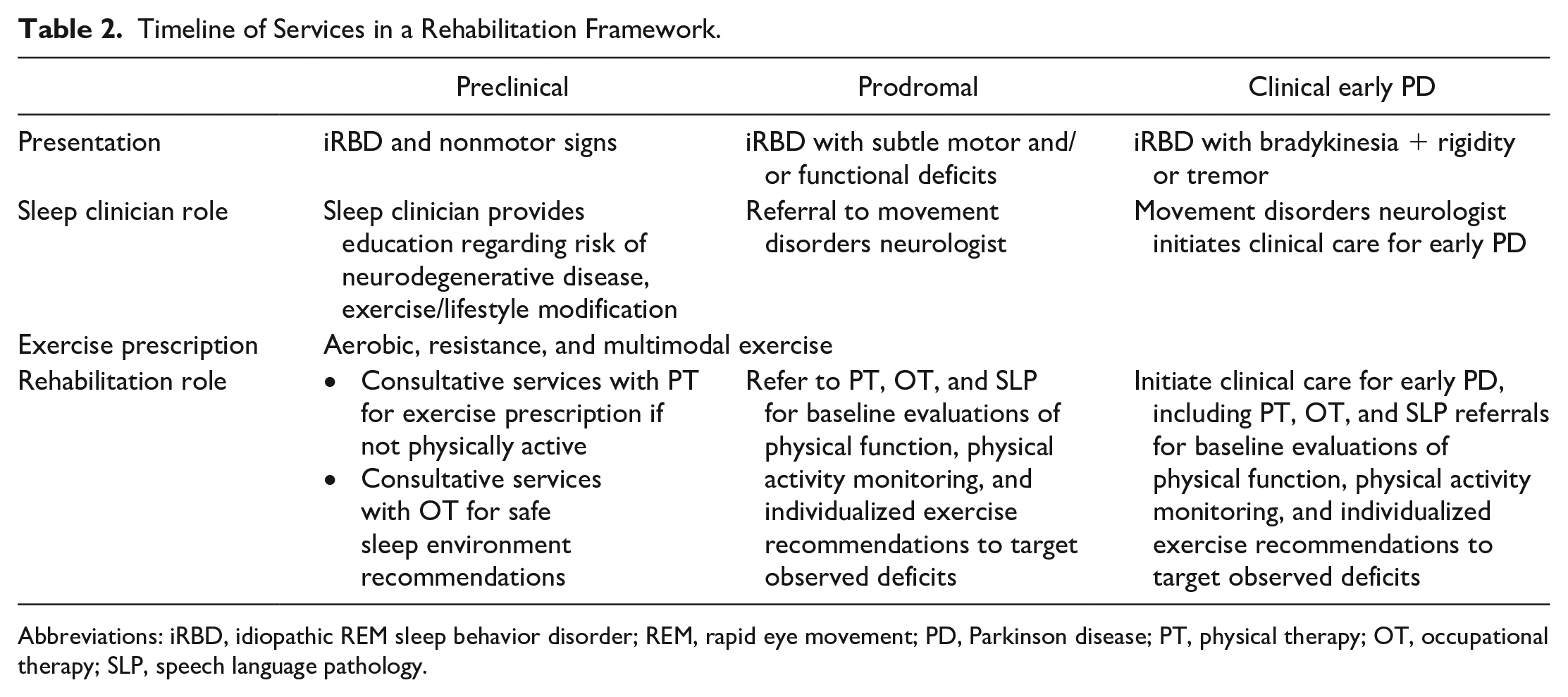
Abbreviations: iRBD, idiopathic REM sleep behavior disorder; REM, rapid eye movement; PD, Parkinson disease; PT, physical therapy; OT, occupational therapy; SLP, speech language pathology.
Early Prodromal Synucleinopathy
People with iRBD in the earliest stages of prodromal synucleinopathy disease may present only with RSWA and/or dream enactment and no other predictive signs. These people are primarily managed by their sleep clinician. In addition to the standard care established for iRBD, we recommend that sleep clinicians educate the patient on the risk of future neurodegenerative disease and the evidence regarding the benefits of exercise. 74 In individuals who are sedentary or would like assistance designing an exercise plan, consultative visits with a physical therapist may increase their exercise participation.69,70 Therefore, we recommend consultative services with PT for a subset of iRBD patients who need an exercise prescription or have comorbidities that require activity adaptation. Strategies to promote increased exercise may include motivational interviewing, general exercise education, behavioral change elements, and mobile technology/app-based telemedicine in sedentary or inactive individuals. 69
Late-Stage Prodromal Synucleinopathy
Patients with iRBD who exhibit motor and nonmotor signs that strongly predict future phenoconversion to a neurodegenerative synucleinopathy should be evaluated for subtle parkinsonism. When mild bradykinesia is observed, we recommend that the sleep clinician consider referral to a movement disorders specialist and a knowledgeable physical therapist. Early referral to PT can promote increased participation in physical activity and exercise and possibly mitigate the progression of motor signs such as bradykinesia.48,55,56
In later stages of prodromal synucleinopathy, when functional deficits are reported by the patient, the sleep clinician or movement disorders specialist should consider involving the full interdisciplinary physical medicine and rehabilitation team, including PT, OT, and SLP. 33 The interdisciplinary team may then address and monitor the progression of functional deficits (eg, handwriting changes, walking changes, speech changes). Because consultation with all 3 disciplines may overwhelm an individual with iRBD, we recommend taking an interdisciplinary or transdisciplinary approach. 75 For example, if an individual with iRBD and motor symptoms is referred to PT for exercise promotion, the physical therapist should screen for deficits in the domains of motor experiences of daily living and speech. If there are deficits, the physical therapist can provide education and referrals to OT and SLP as appropriate.
We propose that physical therapists, occupational therapists, and speech-language pathologists utilize a similar approach in people with iRBD manifesting mild motor deficits as they do in early clinical PD.71-73 Rehabilitation should include a comprehensive discipline-specific evaluation of physical function with attention to establishing a baseline performance assessment using standardized outcome measures and identifying the presence of any deficits. Treatments should include addressing any observed functional deficits, education on the role of exercise and multidisciplinary therapy, and prescribing/monitoring exercise programs.
Potential rehabilitative treatment strategies for iRBD have been described 33 and include services from PT, OT, and SLP. PT treatments should address exercise prescription to improve or maintain aerobic function, strength, balance, agility, mobility, and posture. In the early stages of the disease, treatments should focus more on restoration of function, but may also include compensatory strategies. OT treatments should focus on maintaining or improving all domains of occupational performance and quality of life, including employment, and instrumental and basic activities of daily living, particularly related to fine motor control. OT treatments may include restorative exercises, environmental adaptations, and behavioral compensatory strategies. In addition, fall prevention and safety should be addressed by PT and OT. SLP may address domains of speech and voice while also monitoring cognition and swallowing because early impairments in these areas should be considered indicative of possible dementia with Lewy bodies or multiple system atrophy and immediately communicated to the treating neurologist.
Transitioning to Care for Clinical PD
Once early clinical PD has been diagnosed, we recommend interdisciplinary rehabilitation treatment following treatment guidelines for PT, OT, and SLP as stated in previously published clinical practice guidelines.71-73 We refer readers to these guidelines and the several published real-world examples of how interdisciplinary rehabilitation programs can be implemented.70,76
Future Directions and Considerations
The existing literature in iRBD indicates that motor abnormalities and subtle parkinsonism can be observed for greater than 10 years prior to phenoconversion from iRBD to a neurodegenerative α-synucleinopathy; therefore, protective or early therapies in iRBD should be tailored to promote neuroprotection and address the manifesting motor features. Several considerations and limitations are relevant to the implementation of early or protective therapies in iRBD.
One important consideration of the proposed rehabilitation framework is that it applies to patients with iRBD who represent prodromal synucleinopathy and may convert to not only PD, but also to dementia with Lewy bodies and multiple system atrophy. Interestingly, the available predictive models do not indicate a substantial difference in prodromal motor signs between those who convert to PD, dementia with Lewy bodies, or multiple systems atrophy.5,7 This observation may suggest that the proposed framework for rehabilitative services in iRBD may be more widely applicable in a variety of α-synucleinopathies.
Future rehabilitative and exercise-based studies in iRBD are needed. Clinical trials to examine the efficacy of neuroprotective interventions in iRBD have been designed 77 and sample sizes estimated. 5 Whereas the time to phenoconversion is the ideal primary outcome in therapeutic trials, 77 exercise-based trials should also include outcomes sensitive to the progression of parkinsonian motor signs evident in iRBD (ie, MDS-UPDRS) and instrumented measures of bradykinesia, rigidity, tremor, and voice. These outcomes should be assessed in the absence of cholinergic, dopaminergic, or noradrenergic medication that may mask exercise-induced benefits on the motor system 54 and compared to cohort data in people with PD and coexisting RBD to control for differences in PD phenotype.
Finally, it remains unknown if exercise-based interventions can halt or mitigate the neurodegenerative process associated with α-synucleinopathy. Although it is clear from previous trials that exercise can slow motor symptom progression in people with PD,44,54,63 exercise may mask motor symptoms without interfering with age- and time-dependent neurodegenerative processes. Therefore, it is imperative that future studies include biomarkers of neurodegeneration 18 to assess the effectiveness of interventions to prevent or slow progression of disease.
Summary and Conclusion
Extrapyramidal or subtle parkinsonian signs manifest in people with iRBD years to decades before phenoconversion to a degenerative α-synucleinopathy disease.7,20 A survey of the literature indicates that specific motor signs evident in iRBD include bradykinesia, impaired motor experiences of daily living, speech abnormalities, and gait/posture deficits. The proposed rehabilitation framework for iRBD includes early exercise-based interventions of aerobic exercise, progressive resistance training, and multimodal exercise with consultative services provided by rehabilitation experts to address exercise prescription, progression, and monitoring.
Finally, there is an immediate need for exercise and rehabilitation research to be conducted in individuals with prodromal synucleinopathy to better understand the role of exercise, physical activity, and early rehabilitation services on α-synuclein–driven neurodegeneration. Using the clinical population of iRBD, it is now feasible to evaluate neuroprotective interventions; however, development of effective therapies will require increased awareness of iRBD by clinicians and require collaboration between specialists in the fields of neurology, sleep medicine, and physical medicine. The proposed framework is recommended for clinicians who wish to integrate rehabilitative services into the care plan for patients who show signs of prodromal or subclinical synucleinopathy.
Footnotes
Authors’ Note
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health’s National Center for Advancing Translational Sciences or National Insitute of Neurological Disorders and Stroke.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Institutes of Health’s National Center for Advancing Translational Sciences, Grants TL1R002493 and UL1TR002494, and the National Institutes of Health’s National Insitute of Neurological Disorders and Stroke Grant RO1 NS088679.
