Abstract
Objective. To explore the neuropsychological effects and levels of tau protein (TAU), amyloid β 1-42 (Aβ 1-42), and lipid peroxidase after 10 sessions of anodal transcranial direct current stimulation (tDCS) in patients with mild to moderate Alzheimer disease (AD). Patients and methods. A total of 46 consecutive patients with probable AD participated in this study. They were classified randomly into 2 equal groups: active versus sham. Each patient received 10 sessions of anodal tDCS over the left and right temporoparietal region for 20 minutes for each side with the cathode on the left arm. Patients were assessed using the Modified Mini Mental State Examination (MMMSE), clock drawing test, Montreal Cognitive Scale (MoCA), and the Cornell Scale for depression. Serum TAU, Aβ 1-42, and lipid peroxidase were measured before and after the 10th session. Results. There was a significant improvement in the total score of each cognitive rating scale (MMMSE, clock drawing test, and MoCA) in the real group, whereas no such change was observed in the sham group. The Cornell depression score improved significantly in both groups. There was a significant increase in serum Aβ 1-42 (P = .02) in the real but not in the sham group, with a significant Treatment condition × Time interaction (P = .009). There was no significant effect on tau or lipid peroxidase in either group but a significant positive correlation between changes of Aβ1-42 and MMMSE (P = .005) and MoCA (P = .02). Conclusion. The observed cognitive improvements were complemented by parallel changes in serum levels of Aβ 1-42.
Keywords
Introduction
Alzheimer disease (AD) is a common form of dementia. Currently available medications have poor efficacy, are expensive, and induce adverse effects. Recent work in AD has demonstrated compelling evidence about the importance of oxidative processes (OxS) in its pathogenesis. 1 Cellular studies show that OxS is an event that precedes the appearance of the hallmark pathologies of the disease, neurofibrillary tangles, and senile plaques. 1 Indeed a greater understanding of the role of OxS mechanisms in the pathogenesis of AD may lead to novel clinical interventions. Both amyloid-β (Aβ) and tau protein (TAU) have been linked to interneuron dysfunction and have a role in mediating changes in the overall activity of neuronal circuits. 2 β-Amyloid plaques and neurofibrillary tau deposits define AD as a unique neurodegenerative disease among different disorders that can lead to dementia. 3
Because there is no satisfactory treatment for AD, searching for alternative or adjunctive approaches is of high priority. tDCS is a simple tool that can modulate brain activity using low-intensity electrical currents applied over the scalp through 2 large, saline-soaked sponge electrodes. This generates a small electrical current within the brain that can modulate ongoing levels of neuronal activity. tDCS can be applied as anodal or cathodal stimulation.4,5 In general, cathodal tDCS is thought to reduce and anodal tDCS to enhance excitability of the targeted cortical region. Interestingly, excitability enhancing tDCS (eg, by administering anodal stimulation; eg, Nitsche et al 6 ) often seems to improve cognitive functioning. In line with this, several studies have demonstrated that stimulation of the temporal cortex with transcranial direct current stimulation (tDCS) may improve some of the symptoms of AD. It can enhance name recall in healthy elderly persons 7 and improve recognition memory in patients with AD.8-10 tDCS can also increase high-frequency power in the electroencephalogram (EEG) and improve temporo-parieto-occipital coherence, both of which correlate with improvement in word recognition tasks. 11 However, in contrast with this work, other studies found no or limited effects of tDCS on cognitive rehabilitation in AD.12-14 Thus, although promising, the results of tDCS in AD to date should be considered preliminary and require further investigation.
Nguyen et al 15 suggest that one potential solution to improving the effect size of tDCS in AD is to use multisite stimulation targeting several nodes of brain networks responsible for memory and language function. In a previous open study 16 on 10 patients, they found that 5 one-hour sessions per week with 3 brain areas stimulated per session over a period of 5 to 6 weeks produced a significant improvement in cognitive scores at the end of the stimulation period.
The present study was recruiting patients before that study was published. But the aim was similar: to increase the effectiveness of tDCS in dementia by targeting more than one brain area. Our rationale was that many of the networks involved in cognition are represented bilaterally. This is not only the case for numerical cognition (number processing capabilities as deficient number comparison performance), 17 attention networks,18,19 memory, 20 or conscious processing 21 but even for functions such as language processing, which are commonly assumed to be lateralized. 22 Thus, it may well be important to deliver tDCS bilaterally.
In this exploratory study, we therefore examined whether 10 daily repeated sessions of tDCS over the temporo-parietal (TP) region bilaterally might enhance cognitive function in mild to moderate AD and produce objective changes in biomarkers of AD. The aim was to provide data to power future studies should the methodology be successful.
Patients and Methods
This trial is reported following 2010 CONSORT guidelines. In our study, the sample size was based on G power software based on the following assumptions: proportion of expected outcome in the intervention group = 30% (according to the previous study 23 ), proportion of the expected outcome in the placebo group = 1%, α level = .05, power = 0.80, with allocation ratio N2/N1 = 1, using a 1-tailed test.
A total of 56 consecutive patients with a diagnosis of probable AD according to the revised criteria of the National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer’s Disease and Related Disorders Association 2011 24 were recruited from outpatient clinics and private clinics during the period from July 2016 to October 2017. These criteria have been reliable for the diagnosis of probable AD. They have been widely used in clinical trials and clinical research. We first used the outline criteria for all-cause dementia and then we used the core clinical criteria of probable AD dementia, which include the following: Meets criteria for dementia and, in addition, has the following characteristics: (1) insidious onset, symptoms have a gradual onset over months to years; (2) clear-cut history of worsening of cognition by report or observation; and (3) the initial and most prominent cognitive deficits are evident on history and examination in 1 of the following categories—(a) amnestic presentation: the deficits should include impairment in learning and recall of recently learned information; (b) nonamnestic presentations: language presentation, visuospatial presentation, and executive dysfunction. 24 In all patients, meticulous history, examinations, and computed tomography scan or magnetic resonance imaging were obtained to detect diffuse brain atrophy and to exclude other causes of dementia. Exclusion criteria were the following: substantial concomitant cerebrovascular disease, defined by a history of a stroke temporally related to the onset or worsening of cognitive impairment; the presence of multiple or extensive infarcts or severe white matter hyperintensity burden; core features of dementia with Lewy bodies other than dementia itself; prominent features of behavioral variant frontotemporal dementia; prominent features of semantic variant primary progressive aphasia or nonfluent/agrammatic variant primary progressive aphasia; evidence for another concurrent, active neurological disease, or a nonneurological medical comorbidity, or use of medication that could have a substantial effect on cognition, or epilepsy. Patients with metallic objects in the body or who had received a craniotomy in the past were also excluded.
The stage of dementia was evaluated by means of the Arabic version of the Mini Mental State Exam (MMSE), 25 with the cutoff scores being 11 and 9 out of 30 points for literate and illiterate patients, respectively. 26 In all, 10 patients were excluded because 6 of them had severe dementia, 2 patients had a history of epilepsy, and 2 others refused to participate in the study. Thus, 46 patients with mild to moderate dementia were included, but only 44 completed the study (Figure 1, see the flowchart). The mean age of the patients studied was 65.7 ± 6.4 years.
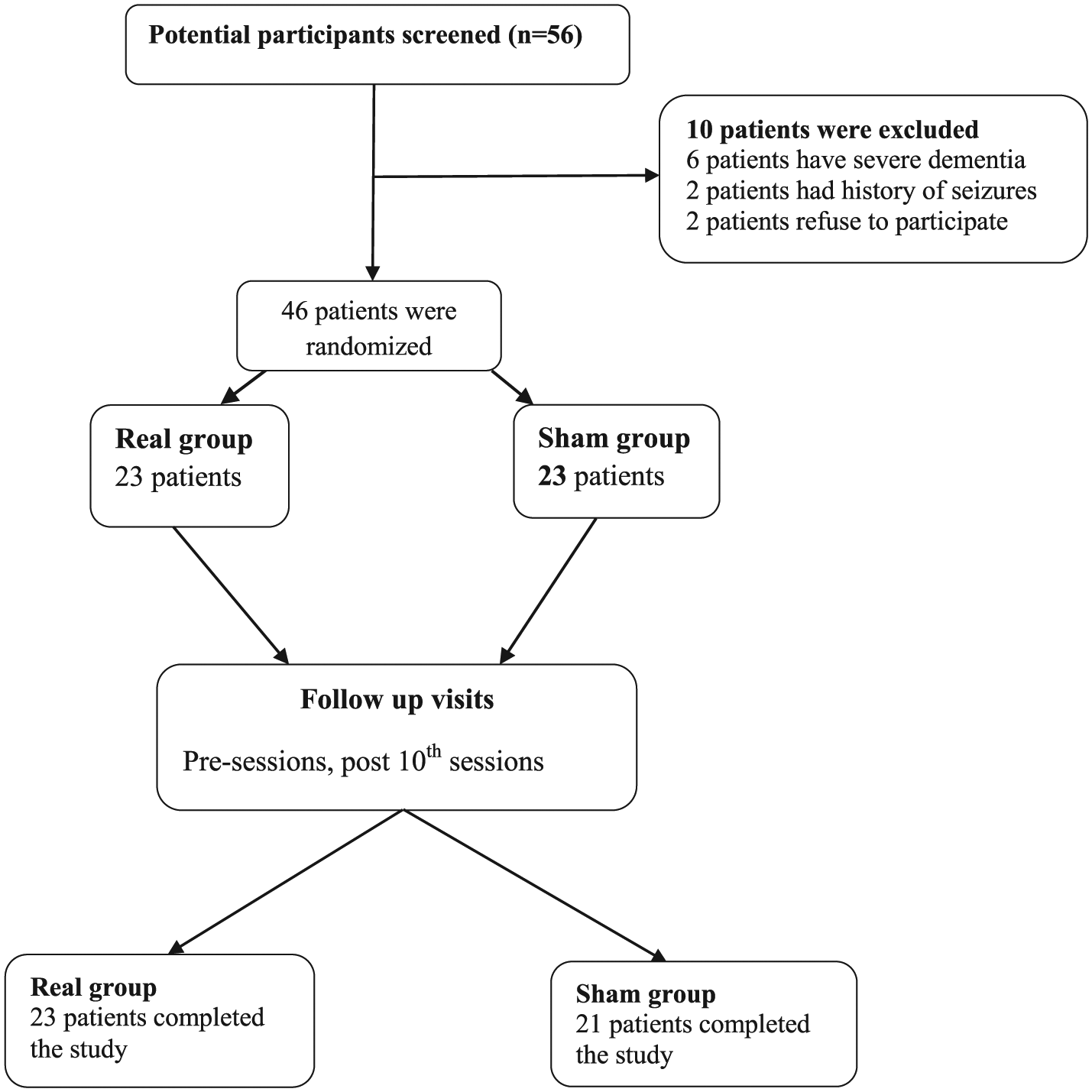
Flowchart.
All patients were given the same medical treatment for at least 2 weeks before tDCS (memantine 10 mg once daily, aspirin protect 100 mg daily, and piracetam 800 mg twice daily). This regimen was used because most recruited patients were elderly and had started this treatment before participation in the study. Other patients were treated similarly to preserve uniformity. The study design was a double-blind, sham-controlled, parallel design.
Each patient was evaluated with the following:
Cognitive evaluation using the Modified Mini Mental State Examination (MMMSE) 27 ,clock drawing test 28 and Montreal Cognitive Scale (MoCA; Arabic version) 29 : The clock-drawing test is a tool used for detecting cognitive impairment and is also used to assess visuoconstructive ability. MoCA is a 30-point screening test administered in 10 minutes to identify elderly people with MCI. The total possible score is 30: a score of 26 or more is considered normal, and a score <26 with no functional impairment indicates MCI. Additionally, a score <26 with functional impairment indicates early dementia, and an extra point is added if the individual has 12 years or fewer of formal education. To screen for depressive symptoms, we used the Cornell Scale for Depression in Dementia (Arabic version), 30 which is a questionnaire completed by an informant (ie, a relative). Assessment was performed just before the first session and immediately after the last session. Psychometric evaluation was performed in our psychometric laboratory by a psychologist who did not know which type of stimulation the patients had received.
Assessment of neurodegenerative serum biomarkers in the Medical Research Center-Faculty of Medicine, Assiut University: Because it is difficult to measure cerebrospinal fluid (CSF) biomarkers in AD, in the present study, we measured biomarkers in serum as outlined by Hanon et al. 31 In that study, plasma Aβ was associated with cognitive status and CSF biomarkers, suggesting that plasma amyloid biomarkers could play a role in the diagnosis of AD. For this, 3 mL of blood was drawn, then centrifuged and serum separated and kept at −80°C. Both human TAU and human Aβ peptide 1-42 (Aβ 1-42) were measured by ELISA kits (Catalog no. SG-10797; no. SG-10347, respectively, China). The detection range for TAU was from 7 to 200 ng/L and for Aβ from 20 to 580 pg/mL. Lipid peroxide was measured as thiobarbituric acid that reacts with malondialdehyde in acidic medium at 95°C for 30 minutes to form thiobarbituric acid reactive product measured at 534 nm. All patients were tested at baseline (before the starting of sessions) and at the end of the treatment.
Group Allocation
Anodal or sham group allocation indications, in a 1:1 ratio, were placed in serially numbered, opaque, closed envelopes. Each patient was given a serial number from a computer-generated randomization table and was placed in the appropriate group after opening the corresponding sealed envelope, with a total number of 23 patients in each group.
Transcranial Direct Current Stimulation
tDCS was delivered (neuroConn, Ilmenau, Germany) using a current intensity of 2 mA (because 2 mA anodal tDCS produces a slightly longer lasting facilitation of excitability than 1 mA 14 ), and the stimulation duration was 20 minutes for each side. Each patient received 5 sessions/wk for 2 consecutive weeks. A pair of 35-cm2 rubber electrodes were inserted into sponge pads soaked with 10 mL of sterile water as recommended in other studies.32-34 We stimulated the left TP lobe for 20 minutes then (5 minutes in between) the right TP lobe for 20 minutes, with the anode (positive electrode) placed at the T3-P3 orT4-P4, respectively, according to the 10-20 EEG system. We used bilateral bicephalic tDCS with 2 active anodal electrodes in order that homologous structures in each hemisphere are facilitated because both hemispheres are thought to contribute to performance during cognitive testing.35-39 An extracephalic (over the deltoid muscle of the left arm) reference was used to avoid stimulation of structures at the vertex as previously mentioned.8,9,40
For sham tDCS, the placement of the electrodes, current intensity, and ramp time was identical to those of the real tDCS group; however, the stimulation lasted only for 30 s. The investigator responsible for delivering tDCS had no contact with the patients. The investigator administering the cognitive tests and the patients did not know which type of stimulation the patients received.
Follow-up
At the end of the therapy, patients were asked whether they thought they had anodal tDCS or sham. We followed up the patients clinically after the end of the 10th session using the same scales for assessment of cognitive function as the primary outcome and changes in serum level of brain damage biomarkers and lipid peroxidase as the secondary outcome. We also asked patients specifically whether they experienced any of the common side effects of tDCS, such as irritation under the electrodes, headache, and dizziness. Two patients in the sham group refused to complete the sessions: one developed gastroenteritis, and the other left the study because there was no improvement after the sixth session.
Ethics
The examiner was blind to the degree of dementia and the experimental condition. All participants or their caregivers gave informed consent before participation in the investigation and after full explanation of the study protocol, which was approved by the Local Ethical Committee of Assiut University Hospital.
Data Analysis
All data of the 44 patients who completed the study were analyzed with the aid of SPSS version 16. The results were expressed as means ± SDs. Because the distribution of the data did not differ statistically from normality (Shapiro-Wilk test, P > .05), statistical analysis of the scores in each test was performed with repeated-measures analysis of variance (2-way ANOVA) with time, as the within-subject factor, and treatment condition (active and sham tDCS) as the between-subject measure. The data from the groups had similar variances and hence satisfied the usual ANOVA requirement of homoscedasticity. Post hoc comparisons of means following significant interaction terms are uncorrected for multiple comparisons because they are only exploratory analyses. Greenhouse-Geisser degree of freedom corrections were applied to correct for the nonsphericity of the data. P <.05 was considered significant for all statistical analyses. Data represented in the figures with bars were used (mean ± SD). The Fisher exact test was used to test differences in the side effects of tDCS and sex distribution between the groups.
Results
All the patients tolerated tDCS well without major adverse effects. Minor effects were observed in 2 patients in the active tDCS group who recorded itching, headache, and dizziness that disappeared after a few hours after the first session, but not in subsequent sessions. This observed side effect had no significant effect between study groups (P = .48). The mean age (SD) of each group was 64.22 (3.64) and 65.23 (4.52) years for the active and sham groups, respectively (P = .41), with male to female ratios of 13:10 and 13:8 for anodal and sham groups, respectively. There was no significant difference in the duration of dementia between groups (P = .99), with mean durations (SDs) of 14.08 (5.79) and 14.09 (4.68). There were no significant differences between the groups in the different clinical rating scales at baseline assessment (Table 1).
Demographic and Clinical Baseline Data of the Studied Groups.
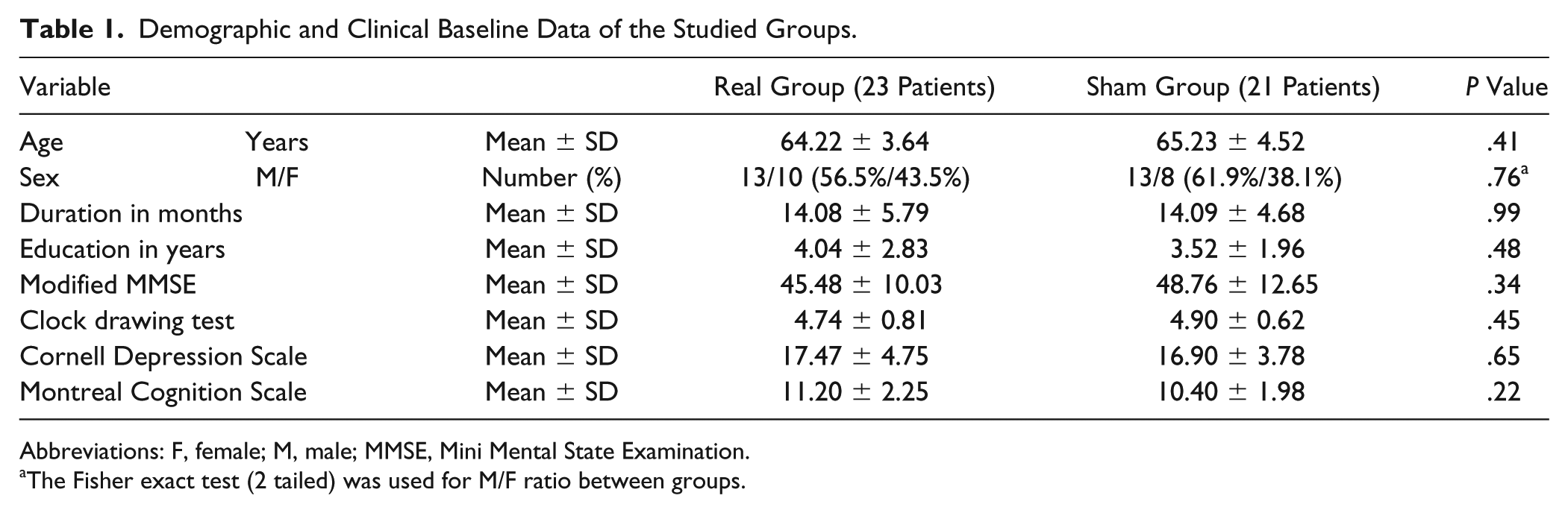
Abbreviations: F, female; M, male; MMSE, Mini Mental State Examination.
The Fisher exact test (2 tailed) was used for M/F ratio between groups.
Effect of tDCS on Cognitive Assessments
Two-way repeated-measures ANOVAs on the scores for each rating scale, with treatment condition (anodal and sham tDCS) and Time (baseline, postsessions) as main factors, revealed significant interactions for each rating scale [MMMSE: df = 1(42), f = 32.97, P = .0001; clock drawing test: df = 1(42), f = 22.82, P = .0001; MoCA: df = 1(42), f = 46.76, P = .0001; and Cornell Depression Scale: df = 1(42), f = 13.47, P = .001]. Post hoc analyses showed that there was a significant improvement in the total score of each cognitive rating scale (MMMSE, clock drawing test, MoCA) in the active group (P = .001, .0001, and .01, respectively), whereas no significant changes were observed in the sham group (Table 2 and Figure 2). Both groups improved significantly in the Cornell Depression Scale, although the significant interaction term in the ANOVA indicates that the effect was greater after active tDCS than sham. The subitems of MoCA (visuospatial, executive function, attention, reading digits, language, recent memory, delayed recall, orientation) all significantly improved in the active group compared with the sham group, except the 7 serial subtraction item. In the Cornell Scale subitems, only physical and ideational scores significantly improved in the active group compared with the sham group, with P =.0001 and .03, respectively.
Effects of tDCS on MMMSE, Clock Drawing Test, Montreal Cognition Scale, and Cornell Depression Scale in the Group Studied.
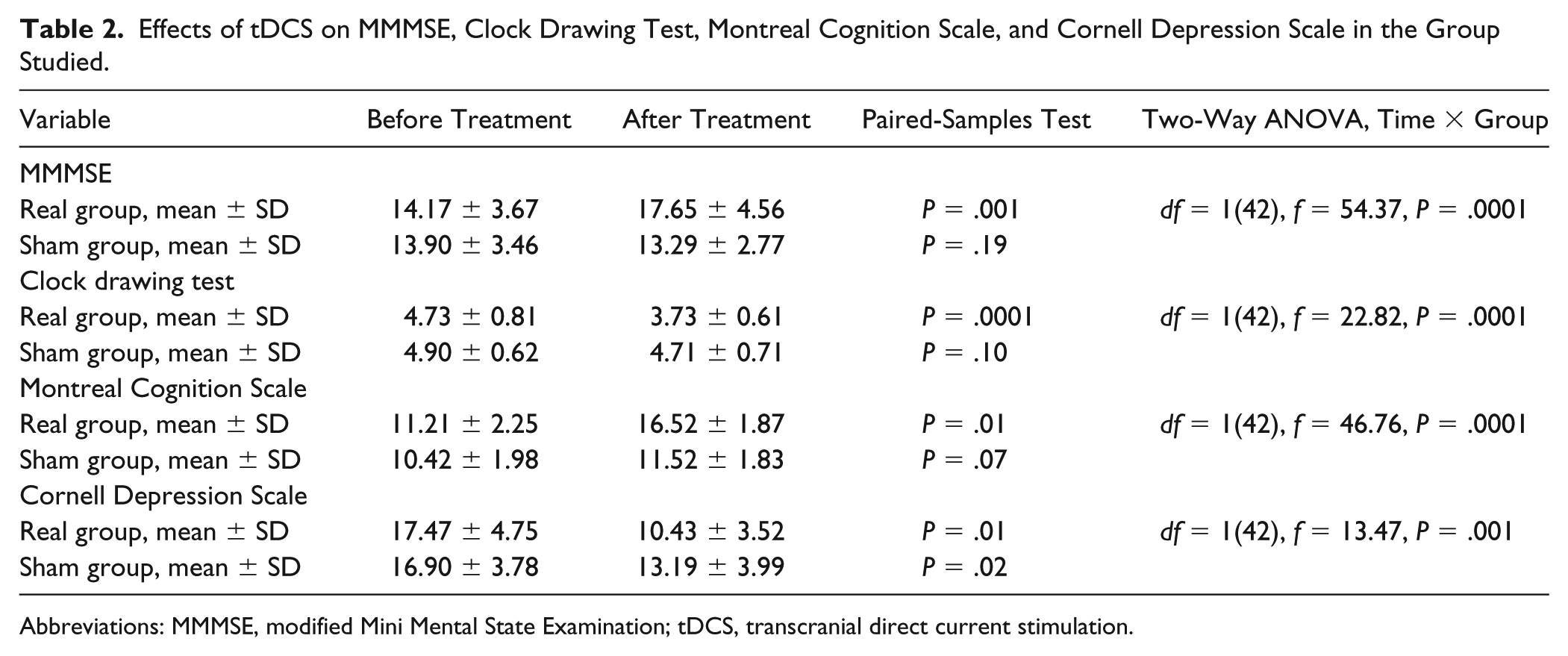
Abbreviations: MMMSE, modified Mini Mental State Examination; tDCS, transcranial direct current stimulation.
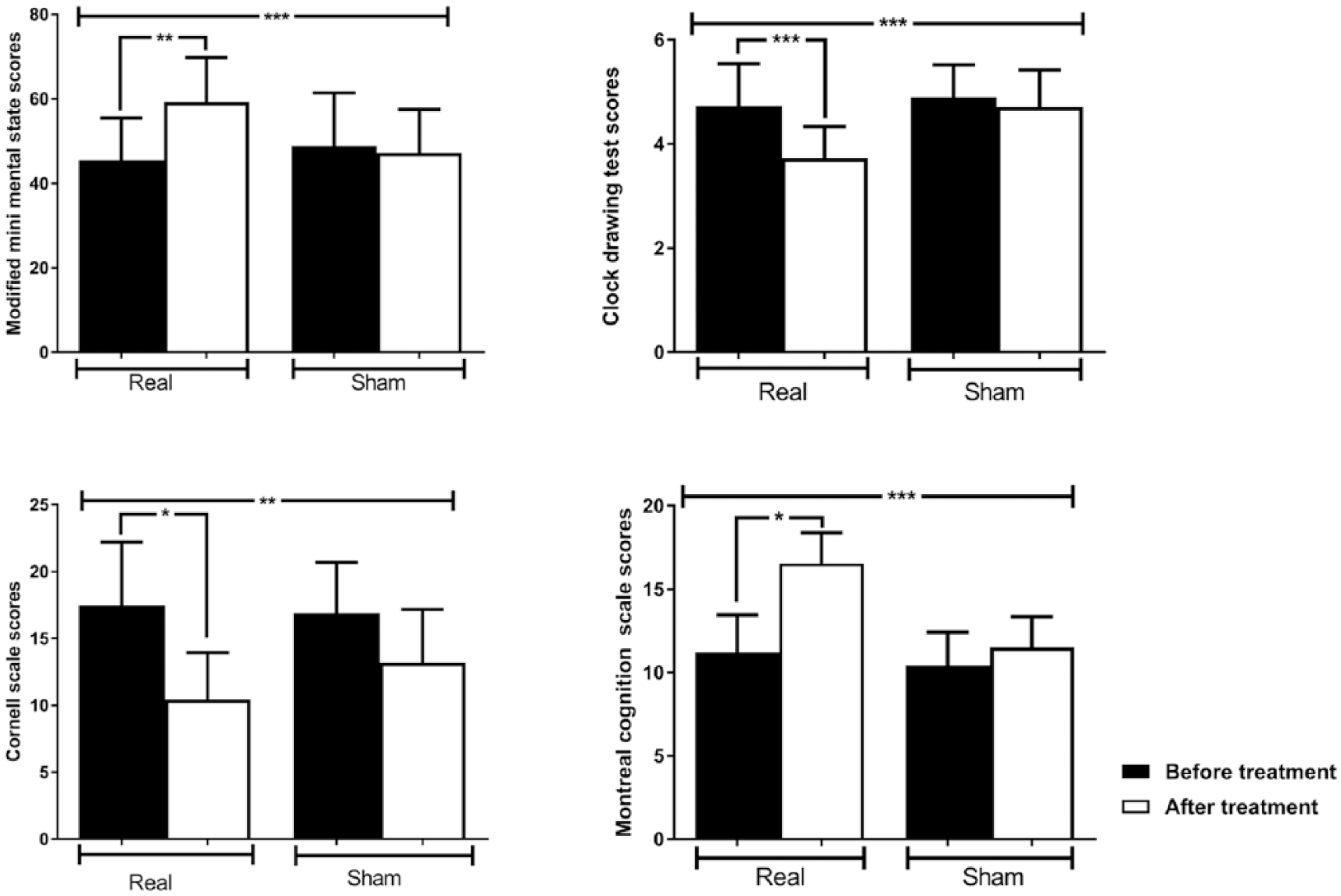
The effects of transcranial direct current stimulation on the modified Mini Mental State Examination (upper left trace), clock drawing test (upper right trace), Cornell Depression Scale (lower left trace), and Montreal Cognition Scale (lower right trace) in studied groups. There was a significant improvement in the total score of each cognitive rating scale in the real group, whereas no such change was observed in the sham group; for the Cornell Depression Scale, both groups were significantly improved. A significant treatment condition and time interaction was seen for each rating scale. Data are mean ± SD
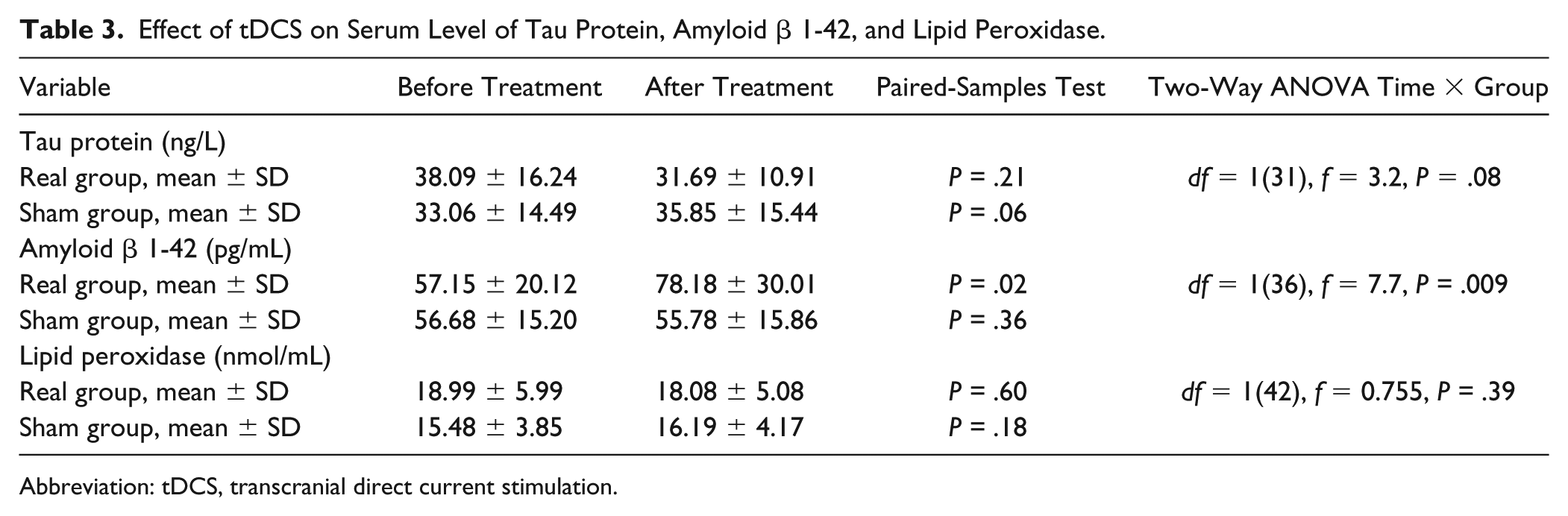
Effect of tDCS on TAU, Aβ 1-42, and Lipid Peroxidase in the Studied Group
There were no significant changes in TAU in each group separately, and 2-way ANOVAs revealed no significant treatment condition (active and sham tDCS) and time (baseline and postsessions) interactions [df = 1(31); f = 3.2; P = .42], with a tendency for levels to fall after active (P = .06) and rise after sham tDCS (P = .21).
There was a significant increase in Aβ 1-42 levels (P = .02) in the active group but no significant effect after sham. A 2-way ANOVA on the serum Aβ 1-42 level, with treatment condition (active and sham tDCS) and time (baseline and postsessions) as main factors revealed a significant interaction [df = 1(36); f = 7.7; P = .009].There was no significant change in lipid peroxidase in either active or sham groups [Table 3A and Figure 3A; df = 1(42), f = 0.755, P = .39].
Effect of tDCS on Serum Level of Tau Protein, Amyloid β 1-42, and Lipid Peroxidase.
Abbreviation: tDCS, transcranial direct current stimulation.
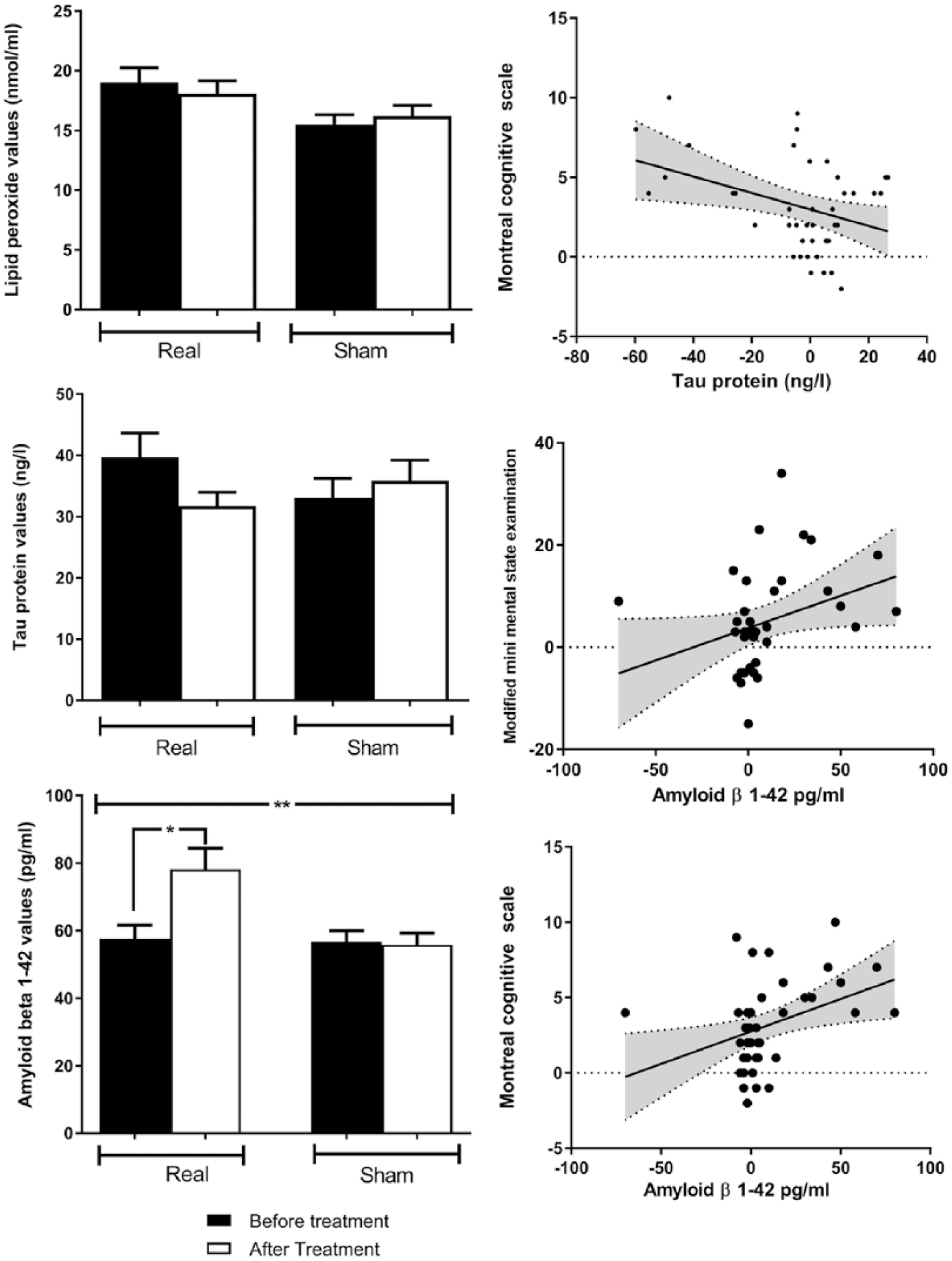
A. The effects of transcranial direct current stimulation on tau protein (top panel) and on amyloid β 1-42 (bottom panel) in real and sham groups. There were no significant changes in tau protein in either group or no interaction between them. There was a significant interaction between group and time for amyloid β 1-42 protein (P = .009), with an increase in amyloid β 1-42 level in the real group (P = .02) but no change in the sham group. Data are mean ± SD (*P < .05, **P < .01). B. Correlation between the changes in tau protein level and changes in Montreal cognitive score (left). Remaining panels are correlations between changes in amyloid β 1-42 protein and changes in Modified Mini Mental State Examination (MMMSE; middle) and Montreal cognition score (right). There is a significant negative correlation between changes in tau protein and Montreal cognitive score (r = −0.37; P = .014). In addition, there are significant positive correlations between the changes in amyloid β 1-42 protein and MMMSE and Montreal cognition score (r = 0.45, P = .005; r = 0.37, P = .02, respectively). The gray zone represents the 95% CI of the correlation line. All data points were included in the calculation.
There was a significant negative correlation between the changes in TAU and changes in MoCA (r = −0.37; P = .014). Significant positive correlations were seen between the changes in Aβ 1-42 protein and changes in MMMSE and MoCA, with r = 0.45 and P = .005, and r = 0.37 and P = .02, respectively; there were no other significant correlations between lipid peroxidase and changes in different cognitive scales (Figure 3B).
Discussion
The currently available medications for dementia have poor efficacy, are expensive, and induce adverse effects. Here, we explored the possibility of using new nonpharmacological therapeutic approaches using tDCS as a tool for noninvasive modulation of spontaneous neuronal activity. Although there have been some previous encouraging positive studies using tDCS in AD,8-10,23,41 the variability in the stimulation parameters used, the target site of tDCS stimulation, and the intensity of the stimulation make it difficult to draw firm conclusions about efficacy. The aim of our study was to add more data to these studies and to expand current knowledge by evaluating markers of brain damage (ie, tau and β-amyloid level in serum) and oxidative stress (ie, lipid peroxidase in serum).
The present study shows that active tDCS produces significant improvement in the cognitive rating scores, whereas there was no effect of sham. However, effects on the Cornell depression score were similar in both groups, suggesting that this is a nonspecific effect of the procedure. Because there was a significant effect on depression in the sham group who had no improvement in cognition, we think that it is unlikely that effects on depression are responsible for the changes in cognition. In fact, effects of most medications on depression are also slow and take many weeks to achieve maximum effect.
The data, thus, support the work of Boggio et al, 9 who demonstrated that 5 repeated sessions of tDCS improved cognitive performance that persisted for 4 weeks after neurostimulation therapy ceased, and Penolazzi et al, 13 who reported that after 10 repeated sessions, tDCS stabilized patients’ global cognitive functioning for approximately 3 months. In another study performed by Ferrucci et al, 8 10 sessions of anodal, cathodal, and sham tDCS were applied over the TP cortex in 3 separate sessions (15 minutes at 1.5 mA, at least 1 week apart). Anodal tDCS significantly improved recognition memory; cathodal tDCS significantly decreased accuracy in the word recognition task; and sham tDCS did not change it. Moreover, no effects were observed in a visual attention task, suggesting that the effects of tDCS were likely specific for recognition memory.
In the present study, we use MMSE as a screening test to diagnose dementia and its severity; however, we applied the MMMSE to determine changes in cognitive function because this is a more comprehensive examination that has more refined scaling and has greater usefulness in longitudinal studies. The modified items in MMMSE are drawn from common human experiences (eg, date and place of birth, body parts, laughing/crying, eating/sleeping), so they will be equally applicable to individuals from different cultural and regional backgrounds.
The mechanism of the effect is still unclear. Although stimulation was applied over TP lobes, it is difficult to predict the distribution of current that reaches the cortex. 42 The large area of our bilateral protocol may have produced widespread activation in brain structures that may increase the effectiveness of the cognitive reserve pool that is still operational in mild and moderate AD. Indeed, widespread tDCS-induced changes in cortical activity have been demonstrated in previous neuroimaging studies. 43 Such activity could also have led to a greater reduction in depression scores in the tDCS group compared with sham and indirectly contributed to the improvement in cognition.
General cognitive processes such as selective attention, alertness, and executive functioning may be slowed down in our patients but not completely blocked. 37 Many of these processes are influenced by bilateral cortical structures. By using bilateral, bicephalic tDCS with 2 active electrodes of the same polarity, we hoped to achieve maximum effect on processing. As a result, the neuronal network may become more reactive and enhance cognitive ability.
Emerging evidence also suggests that weakened integrity of the corpus callosum directly contributes to a decline in cognitive function in aging adults,44,45 whereas increased callosal thickness in typical childhood development correlates with intelligence,46-48 processing speed, 49 and problem-solving abilities. 50 Several structural and functional neuroimaging studies have revealed changes in the corpus callosum in the early stages of AD and even in mild cognitive impairment, supporting its role in cognitive decline.51-53 Moreover, disease severity seems to correlate with the degree of callosal atrophy. 54 We postulate that bilateral, bicephalic tDCS may enhance interhemispheric functional connectivity and thereby improve cognitive function. 55
Plasma Aβ 1-42 is lower in patients with AD than in normal individuals. 31 It decreases over time in patients with newly acquired AD and may be associated with mortality in AD patients. 56 Plasma Aβ 1-42 levels were also reduced in a 4 year longitudinal study, and high baseline Aβ 1-42 levels and a decrease during follow-up were associated with decline in MMSE scores. 57 With the initiation of Aβ deposition, plasma Aβ levels decline, and once the disease is established, the lowest Aβ levels seem to predict more rapid progression. 58 Thus, in the present study, it was interesting to find that real (but not sham) tDCS produced a significant increase in Aβ 1-42 (P = .02) and that this may relate to the changes in cognitive scores because there was a significant positive correlation between the changes in Aβ 1-42 and the changes in MMMSE and MoCA. It may, therefore, be that use of tDCS could act as a neuroprotective tool in AD patients and prevent Aβ 1-42 intracellular deposition.
In the present study, there was a tendency for TAU to decrease after tDCS sessions, but this was not significant. Nevertheless, there was a negative correlation between the change in the TAU level and changes in MoCA because TAU levels in plasma were significantly higher in AD patients compared with both controls and MCI patients. 59 The observed cognitive improvements were complemented by parallel changes in the AD biomarkers levels—TAU and Aβ 1-42 levels.
In a previous study, 60 we found a significant correlation between the change in serum endorphin levels and the changes in ratings of pain, depression, and anxiety following tDCS in Fibromyalgia (FM), suggesting that the increase in β endorphin after tDCS stimulation is associated with pain relief. Altar et al 61 also reported that blood biomarker tests may be used in clinical trials of treatment for AD. In the present study, the parallel changes in AD biomarkers and TAU and Aβ 1-42 serum levels support the possibility that serum biomarker changes are consistent with the concept that applications of blood tests in AD go beyond screening and diagnostic testing; other potential uses are risk assessment, prognostication, and evaluation of treatment target engagement as well as outcome as stated in the Galasko and Golde review. 62
This study was a relatively small exploratory study to examine the possible effects of our bilateral tDCS protocol in the absence of previous data using a similar montage to treat AD. However, this does mean that it is difficult to compare our present results with previous work using different tDCS methodology. Another limitation was the use of MMMSE and MoCA for follow-up of the patients; in retrospect, a specific cognitive task for executive function, working memory, and visuospatial function would also have been useful. Another limitation was the difficulty in obtaining CSF samples to assess the biomarker of neurodegeneration because most of our patients were unwilling to give a CSF sample. One final limitation is that when we explored significant 2-way interaction terms, we did not correct for multiple comparisons in the post hoc t-tests. The results are only descriptive but do not affect the overall conclusions about difference in treatment effects.
Conclusion
A total of 10 sessions of bilateral tDCS applied over the TP region can improve cognitive function in AD and may reduce the human and social burden of dementia as well as the social costs resulting from chronic cognitive disability. The observed cognitive improvements parallel the changes in level of the AD biomarkers, TAU, and Aβ 1-42.
Recommendation
Long-term follow-up is recommended because the study did not measure whether the effects of the study were long lasting or not.
Footnotes
Acknowledgements
The authors would like to thank Prof John Rothwell for useful discussion during the preparation of this article.
Authors’ Note
ClinicalTrials.gov Identifier: NCT03313518.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received financial support from Assiut University research center.
