Abstract
Background. Effects of Tai Chi (TC) on specific cognitive function and mechanisms by which TC may improve cognition in older adults with amnestic mild cognitive impairment (a-MCI) remain unknown. Objective. To examine the effects of TC on cognitive functions and plasma biomarkers (brain-derived neurotrophic factor [BDNF], tumor necrosis factor-α [TNF-α], and interleukin-10 [IL-10]) in a-MCI. Methods. A total of 66 older adults with a-MCI (mean age = 67.9 years) were randomized to either a TC (n = 33) or a control group (n = 33). Participants in the TC group learned TC with a certified instructor and then practiced at home for 50 min/session, 3 times/wk for 6 months. The control group received educational material that covered information related to cognition. The primary outcome was cognitive performance, including Logical Memory (LM) delayed recall, Block Design, Digit Span, and Trail Making Test B minus A (TMT B-A). The secondary outcomes were plasma biomarkers, including BDNF, TNF-α, and IL-10. Results. At the end of the trial, performance on the LM and TMT B-A was significantly better in the TC group compared with the control group after adjusting for age, gender, and education (P < .05). Plasma BDNF level was significantly increased for the TC group, whereas the other outcome measures were similar between the 2 groups after adjusting for age and gender (P < .05). Conclusions. TC training significantly improved memory and the mental switching component of executive function in older adults with a-MCI, possibly via an upregulation of BDNF.
Introduction
It is estimated that the number of people with dementia worldwide will reach 75 million by the year 2030. 1 Accumulating evidence suggests that the beneficial effects of interventions for Alzheimer’s disease (AD), the most common form of dementia, are achieved in the preclinical stage. 2 Thus, individuals who are in an early stage of cognitive decline beyond that associated with aging, known as mild cognitive impairment (MCI) or mild neurocognitive disorder, 3 have become a target group for early intervention. Amnestic MCI (a-MCI) is particularly of interest because this subtype is common and a prodromal symptom of AD. 4
Exercise is a promising nonpharmacological intervention to improve cognition. 5 Tai Chi Chuan (Tai Chi [TC]), a form of mind-body exercise, may be particularly beneficial to older adults with a-MCI because it involves movement recall, task switching, and visuospatial processing simultaneously with physical movements. 6 Meta-analyses have reported clinically relevant improvements in global cognitive function after 12 weeks to 1 year of TC training in cognitively impaired individuals. 7
Physiological factors mediating exercise-induced cognitive improvement remain unclear. A recent systematic review suggested that exercise may attenuate the progression of neurodegenerative processes, facilitate the production of neurotrophic factors, or reduce vascular risk factors, such as inflammatory markers, consequently resulting in cognitive improvement. 8 Brain-derived neurotrophic factor (BDNF) is a protein involved in neuron survival and plasticity critical for learning and memory. BDNF has been linked to the protection of brain structures against neuronal damage,9,10 and the benefits of exercise in improving peripheral BDNF levels have been demonstrated in previous studies.8,10,11 Low-grade systemic inflammation has also been linked to neurodegenerative process such as MCI, 12 with studies reporting that levels of tumor necrosis factor (TNF)-α, C-reactive protein, interleukin (IL)-6, and IL-10 are associated with cognitive impairment.13,14 Furthermore, evidence from previous studies suggests that regular exercise has anti-inflammatory effects in older adults both with and without chronic inflammatory diseases.15,16 Taken together, it is reasonable to propose that these biochemical markers are the modulating factors linking exercise training and cognitive improvement.
To date, few studies have examined the effects of TC on specific cognitive domains. Moreover, studies investigating the mechanisms by which TC may improve cognition are scarce. To address these knowledge gaps, we conducted a pilot randomized controlled trial to explore whether TC training could improve functioning across specific cognitive domains, peripheral BDNF, and reduce inflammatory markers in older adults with a-MCI. This research builds on our interim 15th-week analysis that showed that a combined center- and home-based TC training was feasible for older adults with multiple-domain a-MCI and that TC training significantly improved cognitive function (ie, memory, visuospatial ability, and executive function) and physical performance associated with falls in this group. 17 This report focuses on outcomes at the completion of a 6-month trial that included physiological biomarkers posited to be responsible for the beneficial effects of TC on cognitive function.
Methods
Study Design and Participants
This study was an assessor-blinded, prospective interventional, randomized controlled trial. Participants were recruited from the local community. They were eligible to participate if they met Petersen’s criteria for diagnosing amnestic multiple-domain MCI (a-MCI), 4 had a score of ≥24 on the Mini-Mental State Examination and <26 on the Montreal Cognitive Assessment, 18 had adequate memory with cues, and comprehended instructions. Participants were excluded if they took medications for their cognitive impairment, had neurological conditions (eg, Parkinson’s disease, stroke, multiple sclerosis), had depressive symptoms, had vision and/or hearing problems that were uncorrected, had acute or chronic conditions that would preclude exercise, and exercised regularly (≥30 min/d, ≥3 d/wk).
The sample size for the study was determined for the 2 main outcomes—memory and digit span—based on the effect sizes obtained from previous findings. 19 With 80% power and 5% type I error, the estimated sample sizes of 34 and 58 were required to detect change on the memory and digit span tests, respectively. To accommodate 15% dropout, 66 participants with a-MCI (33 participants per group) were required.
The study protocol was approved by the Human Ethical Review Board of the Faculty of Medicine, Chiang Mai University, Thailand (NONE-2556-01912), and the trial was registered on the US Clinical Trials Registry (NCT02552329). All participants signed a written informed consent form prior to participation. Participants were randomized to either the control or TC group. A research person not involved in the study performed permuted block randomization with a fixed block size of 6 and 1:1 allocation using a computer-generated random allocation with sequentially numbered, opaque sealed envelopes.
Intervention
Before starting the 6-month home practice, participants in the TC group learned TC principles and the 10-form TC for 9 sessions (3 times per week for 3 weeks). The 10-form TC, similar to that used by Wolf et al, 20 was chosen for feasible application to older adults with a-MCI. TC classes with the group size of 6 to 7 participants per class were taught at the Department of Physical Therapy by one certified TC instructor. After completion of the 9 learning sessions, participants then practiced TC at home 3 times per week for 6 months (72 sessions). A 50-minute video was provided to assist participants to undertake their TC home exercises. Each 50-minute session included a 10-minute warm-up (range of motion, muscle stretching), 30 minutes of TC exercise, and a 10-minute cool down (muscle stretching and breathing exercise). Participants were instructed to fill in a logbook immediately after exercise to maximize the accuracy of their records. In addition, several strategies were implemented to assist with exercise adherence, including asking family members to remind participants, posting a note about their exercise schedule in the house as a visible reminder, and administering a weekly reminder telephone call. Participants in the educational control group received information related to cognitive impairment and fall prevention. They attended a 1-hour presentation on cognitive impairment (eg, signs, symptoms, tips to manage cognitive decline and improve cognition) and falls (eg, risk factors, consequences, tips to prevent falls), followed by a question/discussion section. The booklet, which contained information on cognitive impairment and fall prevention, was given to participants to read at home. They were encouraged to ask questions or discuss its content with research staff during a weekly phone call.
Participants in both groups were asked to maintain their routine lifestyle throughout the study period and to inform research staff if they changed their routines or health events occurred. In addition, all participants received a phone call from a research staff member once a week to monitor health and routine changes (eg, attending a recreation club, supplement and medication use, illnesses and hospital admissions). Intervention group members were also asked about the frequency and duration of their TC exercise and any adverse events in these calls.
Outcome Measures
All outcomes were assessed at baseline and at the completion of the trial by trained assessors blinded to participant group allocation. The primary outcome measure was cognitive performance, including memory, visuospatial ability, and executive function. Memory was assessed using the Logical Memory (LM) delayed recall, a subtest of the Wechsler Memory Scale. 21 Participants were instructed to listen and remember the content of 2 short stories that were verbally presented to them. After a 30-minute delay, they were asked to repeat each story as accurately as possible. Visuospatial ability was assessed using the Block Design Test. 22 In this test, 4 to 9 blocks were presented as a model. Participants were instructed to arrange blocks to match a presented model as quickly as possible. Finally, executive function was assessed using the Digit Span forward-backward and Trail Making Test (TMT) B minus A (B-A). For the Digit Span forward-backward test, a sequence of numerical digits was presented verbally. Participants were asked to repeat the sequence either in the same order (forward) or reverse order (backward). 23 TMT consists of 2 parts: part A and part B in which participants were asked to draw a line to connect a sequence of 25 consecutive targets. For the TMT part A, they were instructed to draw a line to connect consecutive numbers in numerical order as quickly and correctly as possible (1-2-3- etc). For the TMT part B, they were asked to draw a line to connect consecutive numbers and letters in an alternating sequence as quickly and correctly as possible. 24 The difference between the time taken to complete part B and A (B-A) was used as a measure of task-switching ability.
The secondary outcome measures included plasma BDNF, TNF-α, and IL-10 levels. All blood samples were collected in the morning between 8 and 9
Statistical Analysis
All statistical analyses were performed using SPSS software (version 21.0, IBM Corporation, Chicago, IL). Tests of data normality were performed using the Shapiro-Wilk test. Demographic variables were analyzed using independent t-tests (for continuous variables) and χ2 tests (for nominal variables). All analyses were carried out using the intention-to-treat approach, with missing data calculated using multiple imputation. All outcome measure data at the end of the trial were expressed as normalized changes from baseline. Multivariate analysis of covariance (MANCOVAs) were conducted to determine the difference of the cognitive outcome measures between the 2 groups at the end of the trial, adjusting for age, gender, and education level. The plasma variables were log transformed to comply with multivariate analysis of covariance assumption. The significance level was set at P <.05, 2-sided.
Results
A total of 212 potential participants were screened for eligibility, of whom 66 were enrolled in the study. The Consolidated Standards of Reporting Trials (CONSORT) flowchart that outlines participant flow through the study and reasons for dropout is shown in Figure 1. Before the end of the trial, 10 participants withdrew, resulting in a drop-out rate of 15.2%. There were no significant differences in participant demographic characteristics between the TC and control groups at baseline (Table 1). The TC group attended on average 60.2 ± 16.3 of the 72 sessions (83.6%). There were no study-related injuries or falls.
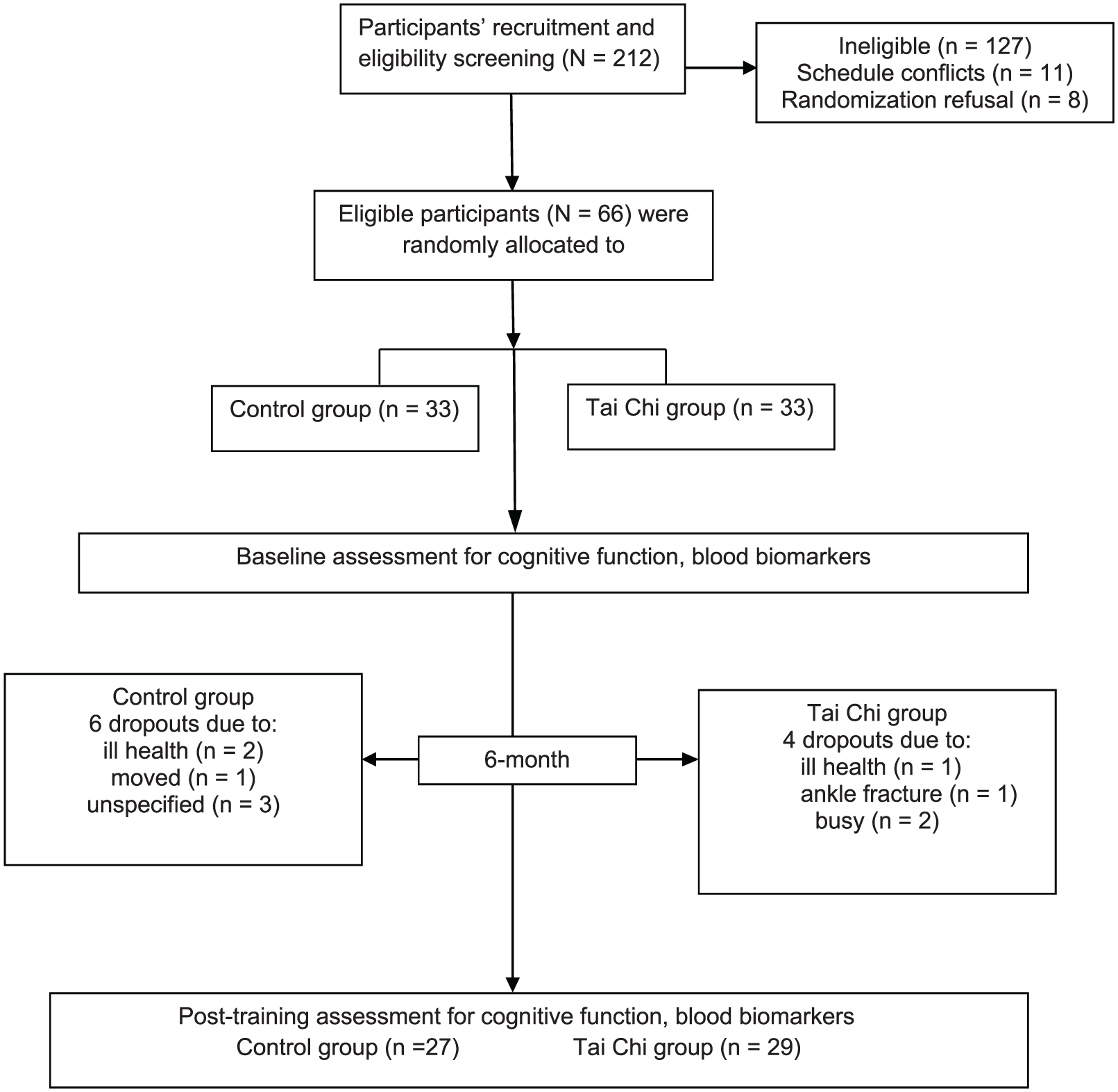
Flow of participants through the study.
Participant Characteristics. a
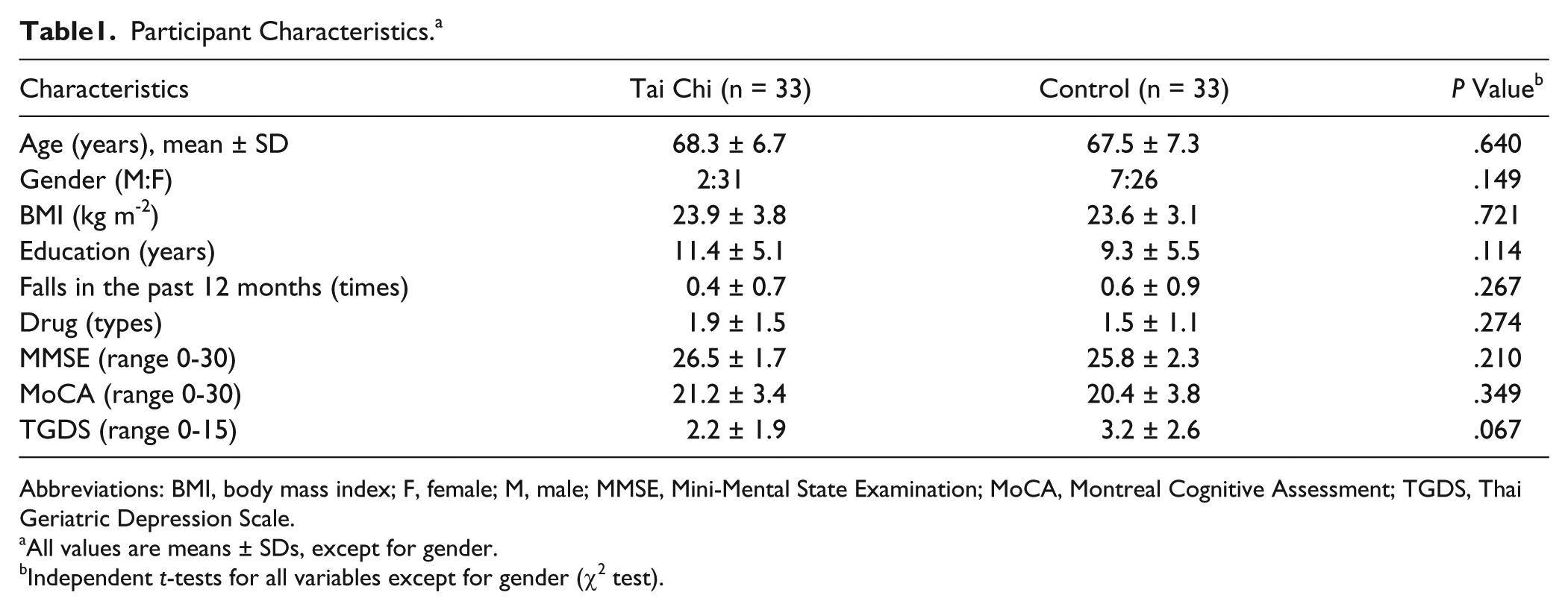
Abbreviations: BMI, body mass index; F, female; M, male; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; TGDS, Thai Geriatric Depression Scale.
All values are means ± SDs, except for gender.
Independent t-tests for all variables except for gender (χ2 test).
Baseline and post–6-month retest cognitive data for the 2 groups are presented in Table 2. The MANCOVA revealed that at the completion of the training period, the TC group performed significantly better in the LM-delayed recall and Trail Making B-A tests than the control group after adjusting for age, gender, and education level (P < .02). There was a trend indicating that the TC group had higher block design scores than the control group at retest (P = .06), whereas Digit Span forward/backward scores did not differ significantly between the groups at this time point (P > .05; Table 2).
Mean ± SD and Normalized Mean Change in Cognitive Measures Between Baseline and 6-Month Retest and Between-Group Differences at Retest. a
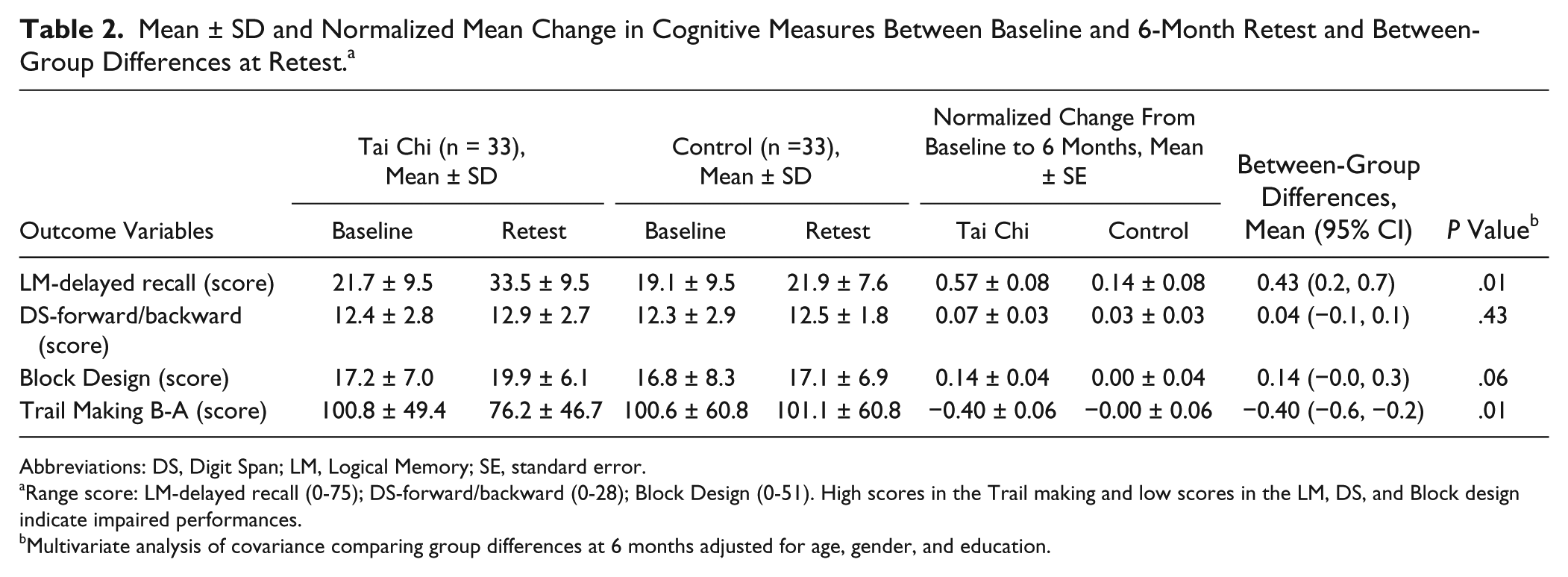
Abbreviations: DS, Digit Span; LM, Logical Memory; SE, standard error.
Range score: LM-delayed recall (0-75); DS-forward/backward (0-28); Block Design (0-51). High scores in the Trail making and low scores in the LM, DS, and Block design indicate impaired performances.
Multivariate analysis of covariance comparing group differences at 6 months adjusted for age, gender, and education.
Plasma data are presented as medians, ranges, and normalized change values from baseline (Table 3). At the completion of the 6-month home exercise, plasma BDNF level was significantly increased for the TC group compared with controls after adjusting for age and gender (P < .05), whereas the TNF-α and IL-10 levels were not significantly different between the 2 groups (P > .05).
Median (Range) and Normalized Change in Plasma Variables Between Baseline and 6-Month Retest and Between-Group Differences at Retest.
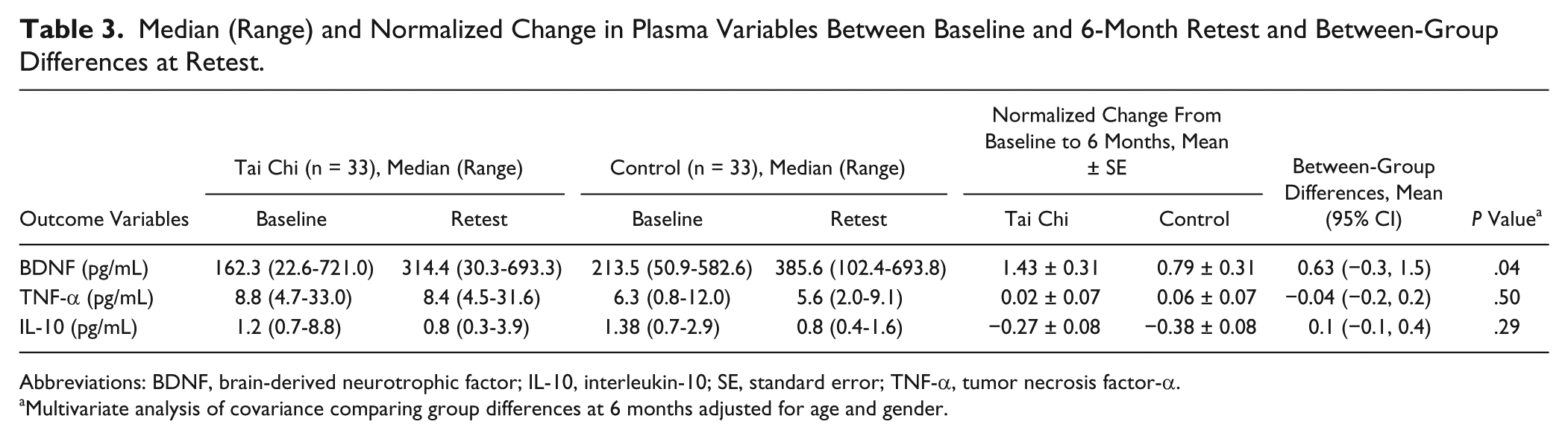
Abbreviations: BDNF, brain-derived neurotrophic factor; IL-10, interleukin-10; SE, standard error; TNF-α, tumor necrosis factor-α.
Multivariate analysis of covariance comparing group differences at 6 months adjusted for age and gender.
Discussion
This randomized controlled trial provides evidence that TC training can improve cognitive performance, specifically memory and executive function, in older adults with multiple-domain a-MCI. Furthermore, cognitive improvements were accompanied by increased plasma BDNF levels, suggesting that BDNF may be partially responsible for cognitive improvement as a result of TC training.
Memory impairment is a hallmark feature of a-MCI. 4 In the present study, we found that TC training was effective in improving memory of older adults with this condition. Such improvement might be a result of the learning of new skills and memorization of movement patterns engaged during TC practice. This finding is consistent with findings from previous studies19,25 and suggests the potential of TC as an exercise regimen to improve memory in older adults with a-MCI. Our findings of improved performance in the Trail Making but not the Digit Span test confirm the findings of previous studies that have demonstrated improved performance in the Trail Making after TC training26,27 and further suggest that the training effect of TC may be specific to certain executive components. Specifically, TC appears to benefit the task switching component of executive function but not the updating and monitoring facility required for working memory. 28 Cognitive activities, including movement recall, switching, and visuospatial orientation, that require attention and executive control devoted to multisegmental movement during TC training might account for the improved cognitive abilities found in the current study. Improvement of memory and executive function after TC training has been shown to be associated with structural and functional changes of cortical areas related to memory and executive control processes.29-31 For example, a cross-sectional study using high-resolution magnetic resonance imaging showed that highly experienced TC practitioners had better executive controls and increased prefrontal and temporal cortex thickness compared with controls. 31 Recent studies found that 12 weeks of TC training significantly improved memory performance and increased resting-state functional connectivity (rs-FC) between the hippocampus and medial prefrontal cortex, 29 whereas it decreased rs-FC between the dorsolateral prefrontal cortex and left superior frontal gyrus and anterior cingulate cortex, suggesting different modulation effects of TC on these cognitive networks. 30
Although results on memory and executive function at the end of the trial replicated our interim 15-week analysis, 17 the difference in block design scores at the end of the trial did not reach statistical significance. It is possible that the effect of TC on visuospatial ability is more prominent at the beginning of TC training and reaches a plateau over time. An alternative explanation is that the effect of the present training program might reach a ceiling at 15 weeks, with the training period after serving as the maintenance phase. Indeed, the time response analyses revealed no significant differences between cognitive performance at 15 and 24 weeks of TC training. A lack of training progression and a decline in adherence (from 87.5% to 83.6%), which may indicate a reduction in participant motivation, might account for no further progression after 15 weeks of training. Thus, a program with training progression and a strategy to boost adherence may be needed for cognition to continue to improve in long-term training.
The role of vascular endothelial growth factor, nerve growth factor, and BDNF signaling pathways in neuroprotection has been well recognized. These neurotrophic factors have been shown to facilitate neurogenesis and promote synaptic plasticity in the hippocampus and cerebral cortex.9,32 Among these trophic factors, BDNF is considered a good candidate for mediating the long-term benefits of exercise on the brain because its upregulation is long lasting and more robust than that of other trophic factors. 9 Several studies have demonstrated that exercise facilitates the synthesis and release of BDNF.10,12,33 BDNF is transported in both directions across the blood-brain barrier; thus, circulating BDNF levels can be used as an index of BDNF levels in the brain. 8 Previous studies found significant elevation of peripheral BDNF levels and improved cognition following multimodal exercise.10,12 The present study is one of the first to demonstrate that TC training was effective in improving peripheral BDNF levels along with cognitive function in individuals with a-MCI. Together, these findings suggest that the production of BDNF may be one mechanism responsible for cognitive improvement after exercise. Furthermore, our findings that nonvigorous exercise such as TC could improve BDNF levels have important implications for exercise regimens in elderly populations, who often have health conditions that preclude vigorous exercise. Previous studies that have reported positive effects of exercise on BDNF have utilized moderate to high-intensity aerobic exercise protocols. 1 However, exercise type could also influence BDNF levels, in that it has been suggested that a combination of mental and physical training has greater effects on neurogenesis and survival than either training alone. 34 Thus, TC, a mind-body exercise, may be particularly beneficial for BDNF upregulation.
Inflammatory processes in the brain have been implicated in chronic neurodegenerative disease such as AD, and the cytokine IL-1 is a known key mediator.35,36 Intact IL-1 signaling is important for the brain’s ability to adapt to acute and chronic neuroinflammation. IL-1 upregulates a wide array of genes in astrocytes, which encode not only neurotoxic mediators, including proinflammatory cytokines such as IL-6 and TNF-α, but also survival-promoting factors such as nerve growth factor. 36 Local expression of IL-1 has been implicated in impairment of hippocampal-dependent memory processing. 37 Although several studies have demonstrated that regular exercise can modulate inflammatory cytokines,12,16 we found no difference between groups in inflammatory cytokine levels at the end of the trial. A growing number of studies have examined the effects of mind-body training such as TC, Qigong, yoga, and meditation on inflammatory cytokines. Studies that examined genomic markers consistently demonstrated reduction in inflammatory signaling after mind-body exercise, whereas mixed effects were found for circulating inflammatory markers.38-40 It has been suggested that alterations in molecular signaling pathways may be more sensitive to this type of intervention. Changes in circulating inflammatory markers may take longer to emerge and require more intensive practice. 38 Thus, insufficient training duration and/or intensity may have been a factor for the nonsignificant improvement of IL-10 and TNF-α levels in the present study. A review of randomized controlled trials noted that training durations longer than 6 months are required for the reduction of systematic inflammatory markers. 16 Although TC is considered to be a moderate-intensity exercise, intensity was tailored to suit each participant’s physical ability because of safety concerns. Thus, exercise intensity varied among participants, which may account for the lack of improvement in inflammatory markers following TC training.
The main strengths of this study are the assessment of specific cognitive domains (ie, memory, attention, executive) often shown to be impaired at an early stage of cognitive impairment and the assessment of physiological parameters that may provide insight into the modulating factors linking TC training and cognitive improvement. However, we also acknowledge certain study limitations. First, the TC and control groups were not attention matched; the TC group had greater interaction with the research staff than control participants from attending TC classes, although this was the case only at the beginning of the trial. Nevertheless, we cannot rule out the potential influence of the additional attention and/or social interaction on the improvement of cognitive function and BDNF levels that we observed in the TC group. Second, because all participants were instructed by the same instructor, the findings may not fully generalize to studies or clinical practice that uses multiple instructors for their interventions. Third, given the small sample size and the fact that this sample size was not determined for the plasma data, the study may not have sufficient power to evaluate the effects of TC on these plasma outcomes. Together, our findings should be considered preliminary and interpreted with caution. A larger randomized controlled trial with an attention-matched control group and long-term follow-up is needed to confirm these findings. In addition, the “dose” (ie, intensity, duration) of training required to unveil the effect of TC on circulating inflammatory cytokines is yet to be determined.
In conclusion, the present study demonstrates that TC training improved memory and the mental switching component of executive function in older adults with multiple-domain a-MCI. The link between cognitive improvement and elevation of plasma BDNF levels observed in this preliminary trial suggests the potential for TC, a mind-body training, to modulate cognition via an upregulation of BDNF. Our findings provide justification for the use of circulating markers to examine the physiological factors mediating TC-induced cognitive improvement.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by The Thailand Research Fund (TRF), RSA5680020 (SS) and MRG5980222 (SK), and The NSTDA Research Chair Grant from the National Science and Technology Development Agency Thailand (NC).
