Abstract
Keywords
Introduction
Ischemic stroke (IS) is one of the main causes of death and disability in the adult population worldwide.1-3 Thrombolytic therapy, as the only treatment proven to improve the outcome of such patients, is accessible in only a minority of clinical settings.4-6 Even in the presence of this medication, more than half of IS patients still remain disabled.7,8 Despite the limited effectiveness of current standard physical rehabilitation therapies, the possibility of recovery from stroke using pharmacological agents has always been attractive. These medications act through different mechanisms and have the potential to improve functional outcome in many survivors of acute IS.9,10 Pharmacological strategies in neurorehabilitation are mainly based on neuroplasticity, 11 which is an intrinsic capacity of the cerebral nervous system enabling continuous reorganization of the damaged neuronal network and remodeling of its function in conditions such as stroke.9,11,12 Several medications are currently under study for this function.4,11,13 In recent years, some trials have proposed a role for selective serotonin reuptake inhibitors (SSRIs) in motor and functional recovery of IS patients, independent of their antidepressant effects.4,9,11,14 Although among this class of drugs fluoxetine is broadly studied, there are positive promising data for citalopram in improving the motor function of IS patients.4,15 Nevertheless, enough confirmatory findings for this concept are missing in clinical studies.16-18 The aim of the present trial was to evaluate the effect of early citalopram administration on 3-month outcome of nondepressed acute IS patients.
Materials and Methods
Patients and Outlines
This randomized, double-blind, placebo-controlled trial was conducted in the Department of Neurology, Razi Hospital (a university and tertiary center) of Tabriz University of Medical Sciences in north-west Iran from May 2012 to January 2014. The inclusion criteria were as follows: (1) confirmation of acute IS diagnosis within the first 7 days after symptoms onset, (2) age between 18 and 95 years, and (3) willingness to participate in the study. Participants were excluded from the study for the following reasons: (1) having severe IS (National Institutes of Health Stroke Scale [NIHSS] score > 20); (2) previous history of taking citalopram, monoamine oxidase inhibitors, other antidepressants, neuroleptics, or benzodiazepines during the past month prior to inclusion; (3) having current or previous disabling diseases; (4) having residual disability from the previous stroke; (5) being pregnant or lactating; (6) having severe loss of consciousness (stupor or coma); (7) having a clinically diagnosed depression or Montgomery Asberg Depression Rating Scale (MADRS) score of more than 19 or a positive history of depression during the recent months; (9) having received thrombolytic therapy; (10) history of endarterectomy; (11) desiring to withdraw from the study (after enrollment); (12) having any condition interfering with completing the follow-up periods; and (13) frailty likely to cause death within 3 months or any fatal disease such as cancers.
Enrollment and Randomization
A total of 144 patients were randomized through an allocation sequence based on 2 blocks with size of 72, generated with a computer random number generator (Randlist version 11 software package; DatInf GmbH, Tübingen, Germany), into 2 groups; the first group received oral citalopram 20 mg once daily, and the other group received placebo of the same shape and full packaging during the first day after hospital admission. Citalopram and placebo tablets were manufactured by Sobhandaru Pharmaceutical Company, Rasht, Iran. Allocation was concealed using the sequentially numbered black envelopes. Written informed consent was obtained from all patients (if they were able to understand and sign the consent)/their surrogates before the onset of the study. After baseline evaluations, citalopram or placebo was given on the first day of inclusion and continued for 90 days. The variables of the study, including demographics, baseline risk factors and characteristics of patients, in-hospital events, and clinical status of acute IS using modified Rankin Scale (mRS) and NIHSS scores were assessed by certified neurologists and neurology residents on admission, day 30, and day 90. Measurement of lipid profile, hematology markers, fasting blood glucose levels, and coagulation markers was performed on the first day of admission (Table 1). All patients underwent brain imaging with brain computed tomography scan on the first day of admission and then brain magnetic resonance imaging (MRI) during the first 3 days of their hospitalization. Determination of the stroke subtypes classification (according to Trial of Org 10172 in Acute Stroke Treatment [TOAST]), 19 the territory of involved vessels, and the anatomical location of lesions were assessed based on clinical evaluation and paraclinic information obtained from neuroimaging, vascular imaging with duplex echocardiography, and data from laboratory assessments. Neurological impairment was measured at baseline and on days 30 and 90 using NIHSS and mRS scores. Also, to better understand the changes of motor or speech condition, the mean changes in NIHSS parts, including motor arm (part 5), motor leg (part 6), best language (part 9), and dysarthria (part 10), were compared between groups.
Comparison of Demographic and Baseline Variables.
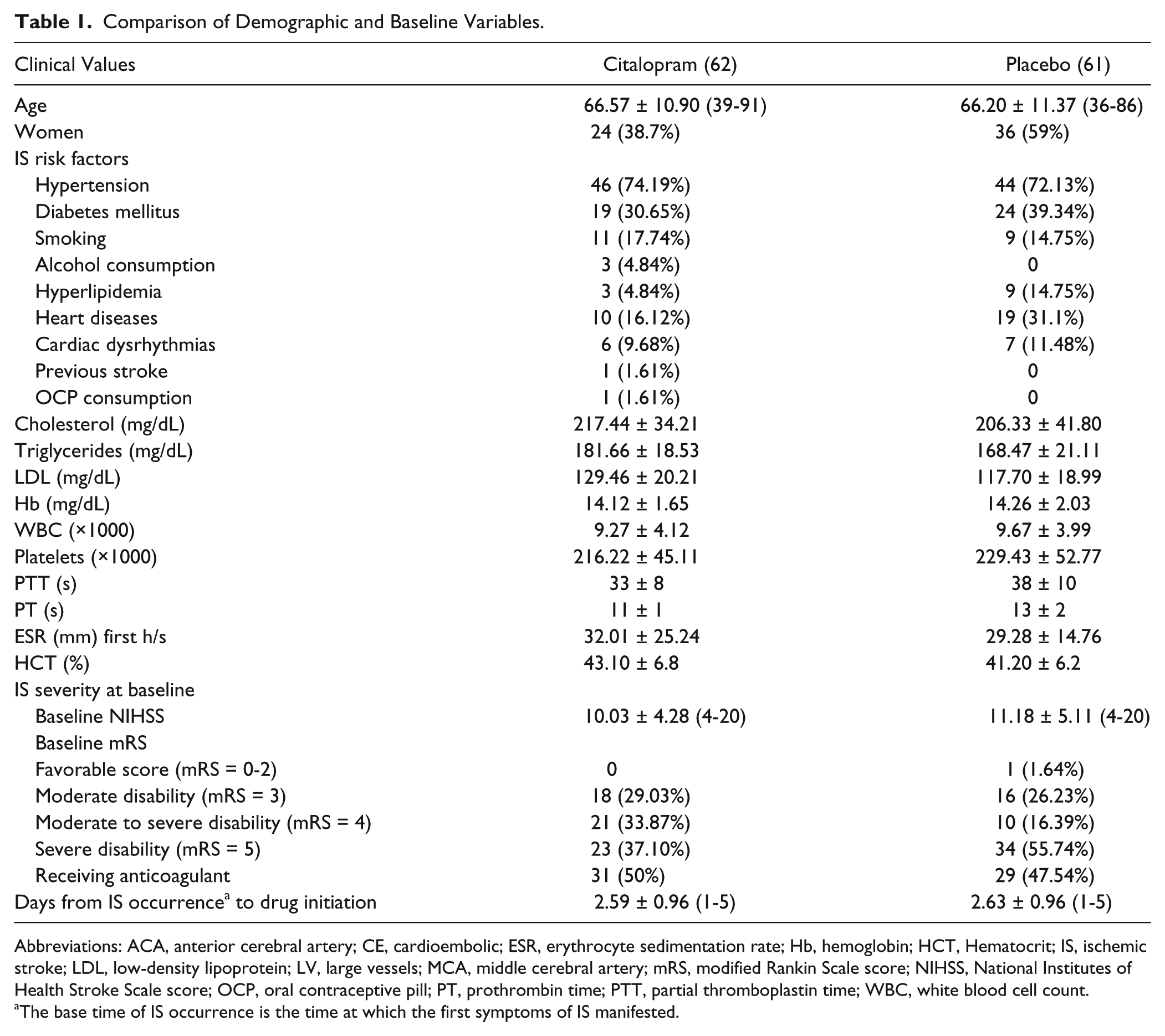
Abbreviations: ACA, anterior cerebral artery; CE, cardioembolic; ESR, erythrocyte sedimentation rate; Hb, hemoglobin; HCT, Hematocrit; IS, ischemic stroke; LDL, low-density lipoprotein; LV, large vessels; MCA, middle cerebral artery; mRS, modified Rankin Scale score; NIHSS, National Institutes of Health Stroke Scale score; OCP, oral contraceptive pill; PT, prothrombin time; PTT, partial thromboplastin time; WBC, white blood cell count.
The base time of IS occurrence is the time at which the first symptoms of IS manifested.
Interventions
All patients received standard protocol of care ordered by an expert stroke care team, whereas the main pharmacological treatment protocol of all patients remained unchanged during the study follow-up. Regular daily physiotherapy and rehabilitation sessions throughout the total period of study follow-up (if necessary) were scheduled for all patients in addition to pursuing the standard therapeutic protocol, which mainly included Aspirin (325 mg/d in the first week followed by 80 mg/d until the end of the follow-up period), atorvastatin 40 mg/d, suitable hydration with IV normal saline, control of risk factors (diabetes mellitus, malignant hypertension, cardiac problems, etc), appropriate feeding care, speech and swallow therapy, and deep-vein thrombosis prophylaxis. The patients and their surrogates were trained to take the medications correctly and to confirm the correct and timely use of the medicines; the drug-free boxes were counted during follow-up in both groups. A standard protocol was used for the evaluation of all patients. Accurate registration of deaths and reoccurrence of cerebrovascular or adverse events was performed during the study. The blinding code remained confidential until the end of the study. All patients, except for those who died and those who could not or did not want to continue the study, completed their clinical 90-day follow-up. At each monthly visit, clinical evaluation for depression and completing the MADRS was performed by psychologists.
The primary outcome of this trial was defined as a 50% reduction in the 3-month NIHSS after IS follow-up compared with the baseline scores, and we defined the secondary outcome as achieving favorable scores in the 3-month mRS compared with the mean of controls, 50% reduction in the 3-month motor parts of NIHSS, 50% reduction in the 3-month language parts of NIHSS, and comparison of mortality rate during the study follow-up between groups.
The study protocol was approved by the ethical board committee of Tabriz University of Medical Sciences, Tabriz, Iran, and it was conducted in accordance with the Declaration of Helsinki. This trial has been registered in the Iranian Ministry of Health and Medical Education and World Health Organization International Clinical Trials Registry Platform Search Portal under ID IRCT201203192150N2 (http://www.irct.ir and http://apps.who.int/trialsearch)
Statistical Analysis
For sample size estimation and statistical analysis of the data, STATA 11 software (STATA Corp, College Station, TX) was used. Using this software, a total sample size of 116 patients in 2 groups was calculated to detect at least 25% difference in the proportion of patients achieving the primary outcome at a 50% standard treatment success, with 80% statistical power and 95% confidence level. Yet we decided to enroll 144 patients to improve the precision. The general strategy for analyzing the main outcome in the current clinical trial was intention-to-treat analysis; however, per-protocol analysis results were also reported. Relative risk (RR) and number needed to treat (NNT) with a CI of 95% were used. To calculate CI for RR and NNT, the exact method was used.
20
In bivariable analysis for the numerical scales, according to their distribution, an independent
Results
Considering the inclusion criteria, 178 patients were primarily selected, but eventually, considering the full criteria, 144 patients were enrolled. During the follow-up, 15 patients died (4 in the citalopram and 11 in the placebo group), 13 patients could not or did not want to continue the study (8 in the citalopram and 5 in the placebo group), and 8 patients with depression (2 in the citalopram and 6 in the placebo group) were excluded from the study. The other patients completed their clinical 90-day follow-up. Furthermore, drop-out number was 10 in the citalopram and 11 in the placebo group (Figure 1).
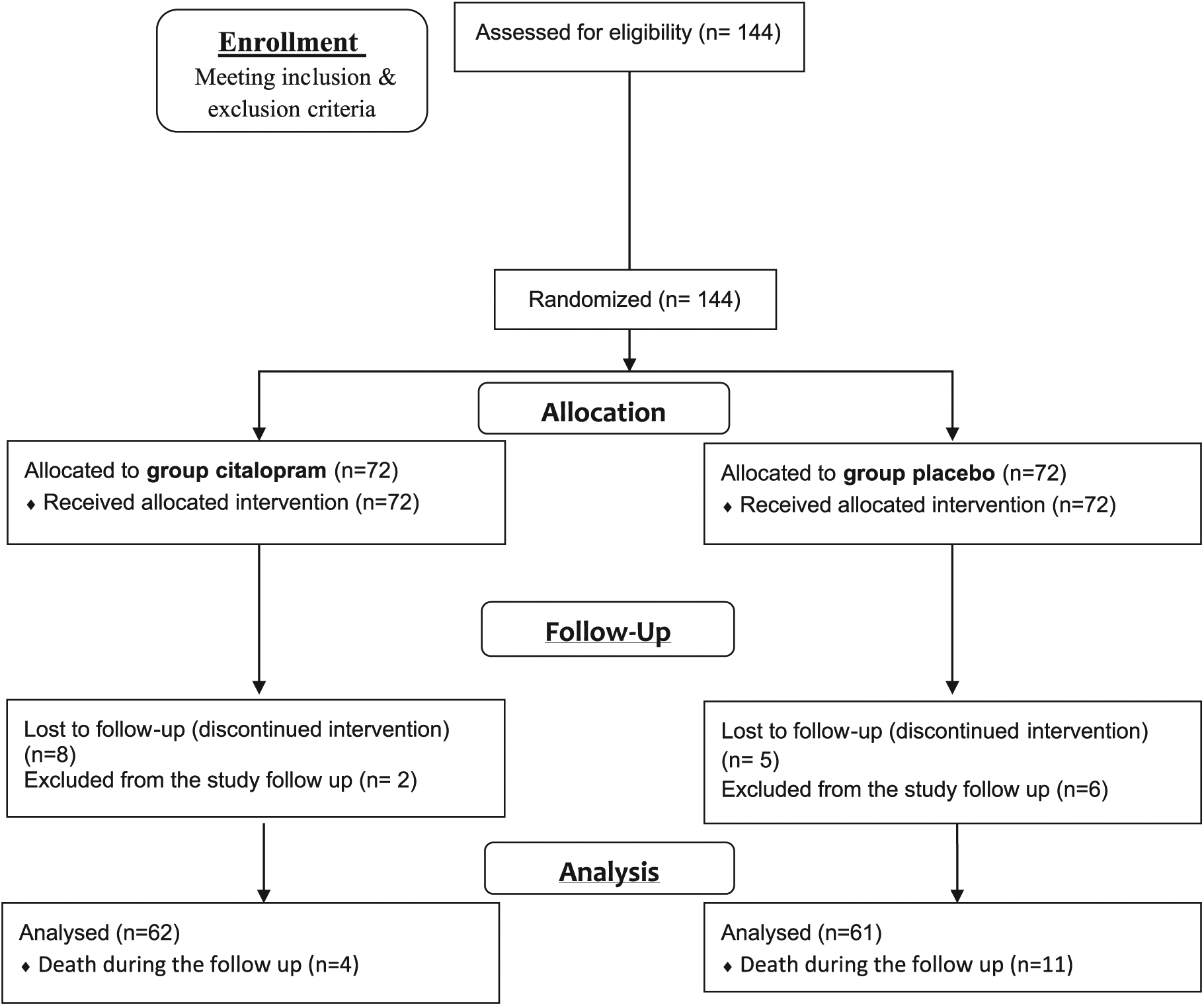
Flow diagram: randomization and patient flowchart.
Finally, considering the drop-out number, 123 patients (62 and 61 patients in citalopram and placebo groups, respectively) with a mean age of 66.4 ± 11.10 years and age range of 36 to 91 years were analyzed. Demographics and background variables were not significantly different between the 2 groups, except for sex distribution (
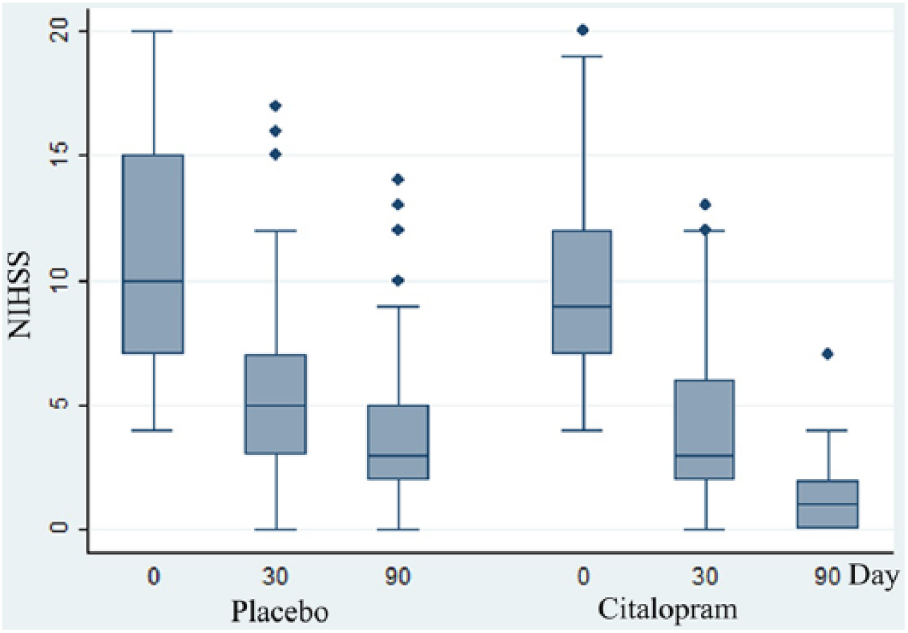
The changes in mean National Institutes of Health Stroke Scale (NIHSS) scores on days 0, 30, and 90; the median is indicated by the horizontal line inside the box. The interquartile range is represented by the height of the box. The upper and lower whiskers represent scores outside the middle 50%, and the dots show the extreme values.
Furthermore, a comparison of the improvement in different subscales of the NIHSS, including motor arm, motor leg, dysarthria, and best language, between the study groups revealed score reductions that were significantly more prominent in the citalopram group. In addition, higher rates of achieving at least 50% improvement in each subscale score after 90 days was observed (Table 2). Achievement of the final outcome (at least 50% improvement in NIHSS score) was not statistically different between stroke subtypes, sex, and age subgroups (less than 55, 55-65, and older than 65 years) and vasculature regions in the citalopram group.
Change in National Institutes of Health Stroke Scale (NIHSS) Components During 3 Months of Follow-up.
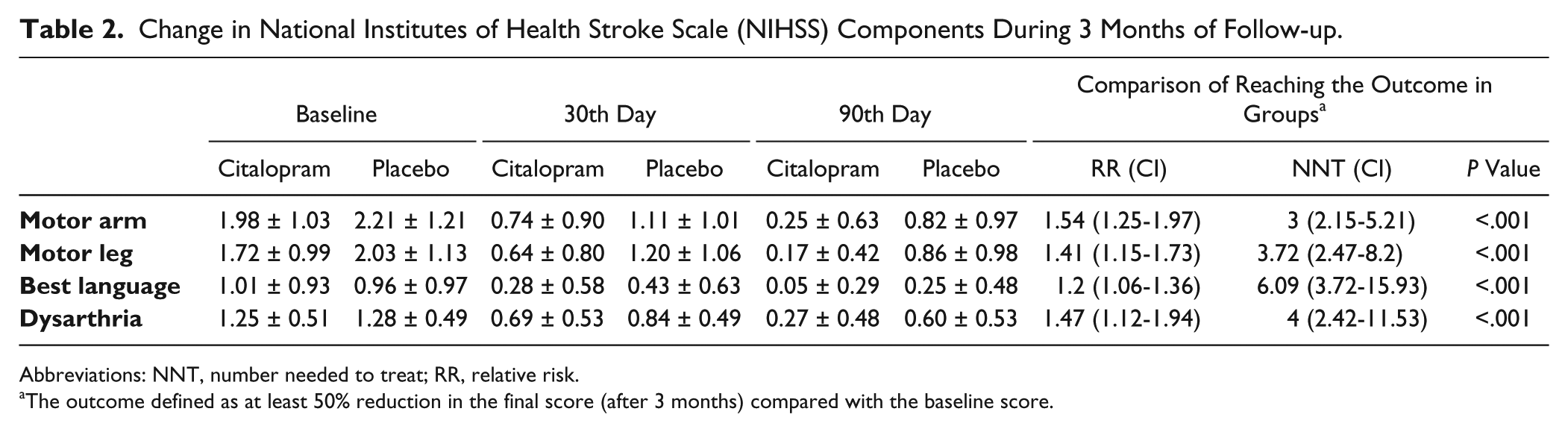
Abbreviations: NNT, number needed to treat; RR, relative risk.
The outcome defined as at least 50% reduction in the final score (after 3 months) compared with the baseline score.
Reaching favorable mRS (score = 0-2) as the secondary outcome was also associated with significant difference between the groups on both days 30 and 90 after onset of the study: 55 (94.8%) versus 22 (43.1%); RR = 2.2 with CI = 1.6-3.03; NNT = 2 with 95% CI = 1.93-2.71 (
Rates of Achieving Favorable mRS Score as the Secondary Outcome at Days 30 and 90 After Starting Citalopram Versus Placebo.
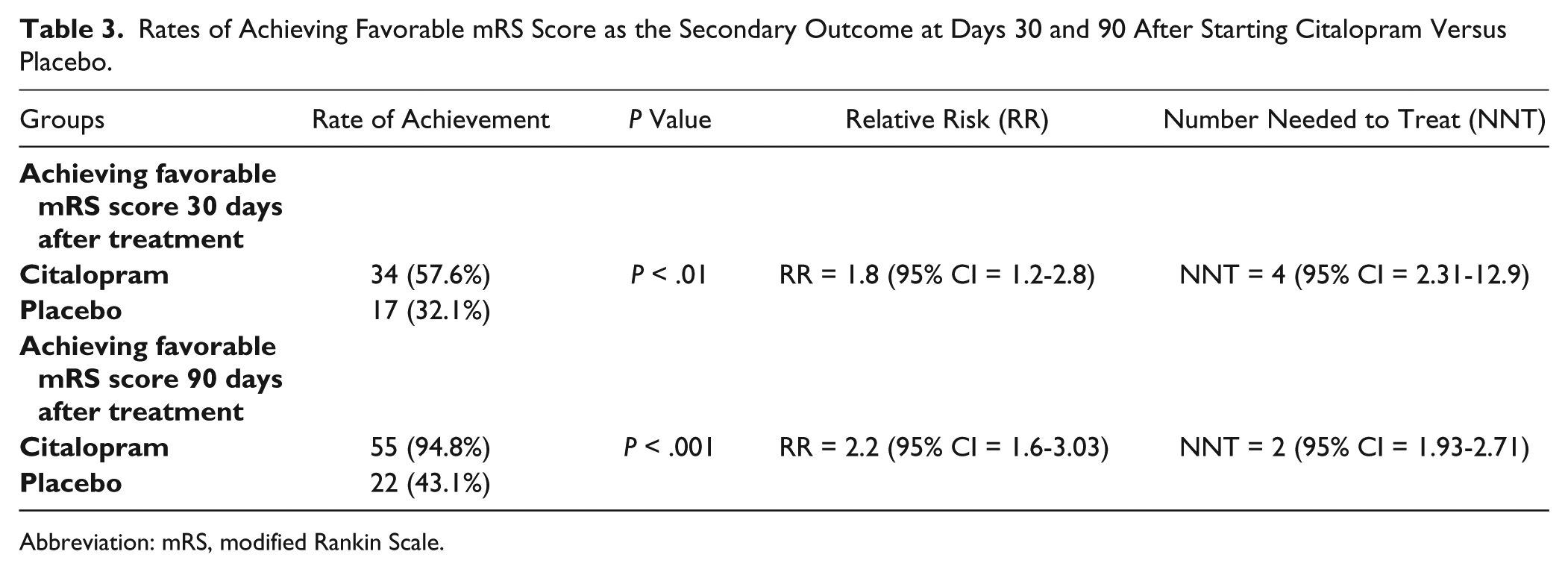
Abbreviation: mRS, modified Rankin Scale.
There were 4 cases of mortality in the citalopram group (2, 1, and 1 cases as a result of coronary artery disease, aspirative pneumonia, and intracerebral hemorrhage [ICH], respectively). In the placebo group, there were 11 cases of mortality (6, 2, 2, and 1 cases as a result of coronary diseases, ICH, aspirative pneumonia, and sepsis, respectively); of note, more than half of the patients (53.3%) in this group died from coronary diseases. However, the difference in mortality between both groups after adjusting for age and sex was statistically insignificant. Based on the log-rank test, survivor functions compared between both groups revealed no statistical significance (
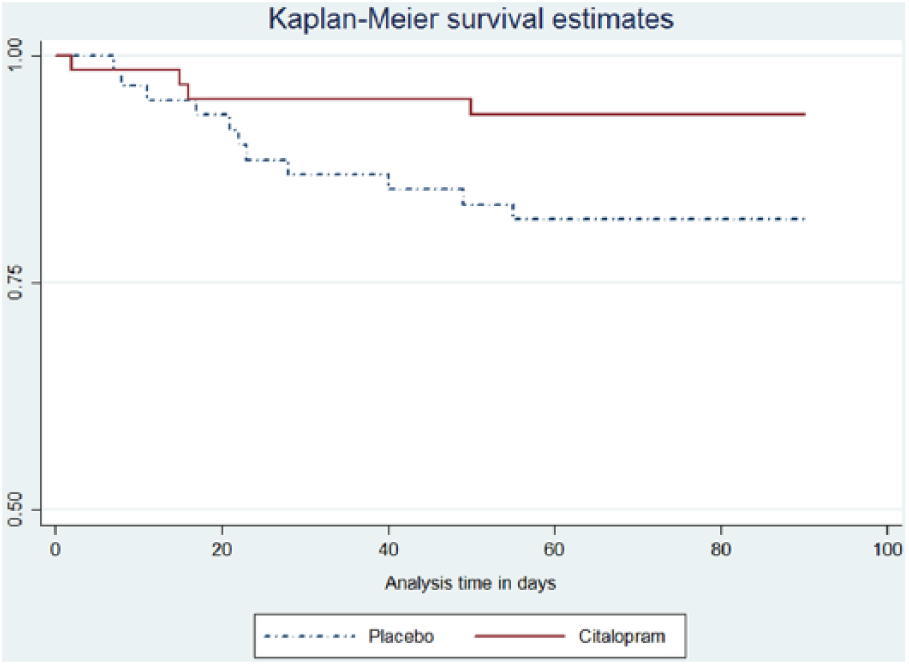
Kaplan-Meier curve of survival probability after stroke compared between citalopram and placebo groups.
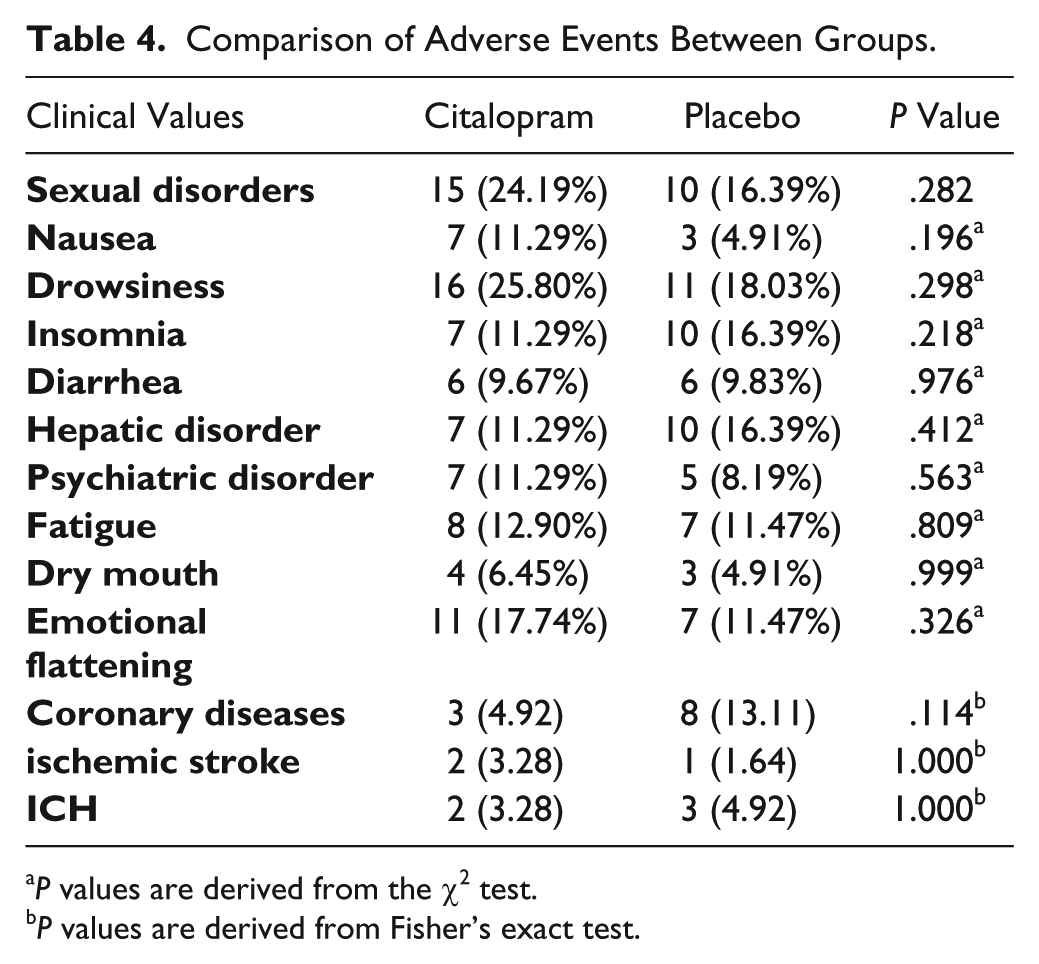
Comparison of Adverse Events Between Groups.
Discussion
In the present study, citalopram proved to have positive effects on reducing the stroke severity of IS patients within its 3-month administration period following acute IS. These results were obtained in terms of minimum reductions of 50% in NIHSS scores of patients compared with their baseline values. Furthermore, the scores of motor and language parts of NIHSS were significantly lower in patients who received citalopram compared with the control group. Improvement toward an independent life, considered as achieving mRS scores of 2 to 0, was also more frequent in the citalopram group.
The main objective of neurorehabilitation after stroke is the reorganization of the ischemia-injured neuronal networks in the brain through neuroplasticity procedures. 11 Very little study has been done, however, of the different effects of medications on the functional improvement of these patients.9,11
The study by Ofek et al 21 on an animal model, investigated the effect of fluoxetine on the brains of rats that had undergone induced acute IS. The authors suggested that fluoxetine can induce arteriolar vasodilation through comodulating NO-muscarinic signaling. Espinera et al, 22 in another animal study on male C57/B6 mice with induced acute IS, reported that thanks to neurogenesis, their subjects treated with citalopram produced more neurons and small vessels in the peri-infarct region and, therefore, experienced further accelerated and enhanced microvessel support, leading to sensory and motor improvements. 22 Furthermore, in another animal study, Sas et al 23 evaluated the effect of SSRI medications (citalopram and fluoxetine) on corticocerebral blood flow in rabbits that had undergone induced stroke. The authors suggested that although fluoxetine was associated with minor improvements in the ischemic cortex cerebral blood flow (cCBF), citalopram administration led to a strong dose-dependent increase in cCBF. 23
Numerous studies have focused on the efficacy of physiotherapy sessions in combination with the required medications in patients with acute IS.24-27 In this study, we scheduled regular physiotherapy and rehabilitation sessions for all patients. Furthermore, stroke risk factors were carefully checked throughout the study by the stroke team.
Engelter et al 28 in a prospective multicenter study reported that SSRIs, serotonin-norepinephrine reuptake inhibitors, and levodopa have no specific efficacy in poststroke rehabilitation. However, Mead et al, 14 in a review study, concluded that SSRIs are efficient in improving poststroke disability, dependence, neurological injuries, anxiety, and depression; yet they mentioned that most of their evaluated studies had methodological limitations.
An evaluation of valid medical databases suggested that very few clinical trials have been conducted on the efficacy of SSRIs in poststroke patients.4,9 In a study, Dam et al 29 divided 52 stroke patients with hemiplegia and severe disability into 3 therapeutic groups: placebo, maprotiline 150 mg/d, and fluoxetine 20 mg/d. All the studied patients received daily rehabilitation sessions for 3 months. Fluoxetine was associated with greater improvement in functional outcome and motor status after 3 months. Pariente et al, 30 in a study carried out on 8 acute lacunar stroke patients with pure motor involvement, concluded that the fluoxetine group demonstrated greater improvements in the motor function of the affected side; functional MRI of the patients also revealed hyperactivation in the ipsilesional primary motor cortex. 30 In another study, Loubinoux et al 31 evaluated the dose-dependent effect of paroxetine in comparison with placebo in 6 healthy participants; paroxetine (20 mg/d) led to further activation of the entire motor pathways and production of motor output. Acler et al 32 evaluated the motor area excitability effect of oral citalopram in 20 stroke patients through transcranial magnetic stimulation. The authors suggested that oral citalopram 10 mg/d improved NIHSS scores of the patients after 1 month and decreased motor excitability significantly. Zittel et al 15 reported that a single dose of citalopram significantly improved motor function in the affected rather than the unaffected limbs of patients with chronic IS. In similar studies, fluoxetine has been reported to improve the activity of agonist and antagonist muscles of the affected limbs significantly. 33 Overall, although most of these studies indicate the efficacy of SSRIs, they have mostly been conducted on a limited number of patients over a limited time span. In contrast, our study was carried out for 3 months on a larger number of patients focusing on the administration of a continuous regimen of the medication within the first days after acute IS in addition to the standard regime and physiotherapy sessions.
The most prominent clinical trial conducted on the efficacy of SSRIs on the motor function of acute stroke patients is the FLAME study, which was conducted by Chollet et al 4 on 118 acute IS patients with depression to evaluate the efficacy of fluoxetine on 3-month motor recovery. In this multicenter study, patients who received fluoxetine experienced significantly improved motor function, which was evaluated using the Fugl-Meyer Motor Scale (FMMS). In our study, in contrast to the FLAME study, patients with depression were excluded from the study throughout the follow-up period; in the FLAME study, despite significant changes in FMMS score being the main outcome of the study, 3-month NIHSS scores of the fluoxetine group was not significant compared with the placebo group. 4 However, in contrast to the FLAME study, we set a 50% reduction in the 3-month NIHSS score compared with the baseline scores of the patients as the primary outcome of the study, which resulted in an NNT of 4; that is, nearly 1 out of 4 patients receiving citalopram could benefit from reaching the set outcome and significant decrease in their IS severity.
In the present study, occurrence of depression was less in the citalopram group, which was in line with the results of previous studies, highlighting the efficacy of these medications in preventing depression in patients with stroke.16,34-36 Furthermore, as regards drug intolerance, 13 patients could not complete the follow-up (5 and 8 in the placebo and citalopram groups, respectively); the difference of these values between the 2 groups was not statistically significant. Also, no significant difference was detected with regard to medication adherence by patients in both groups.
Some studies have highlighted the probable complications associated with the administration of SSRIs, such as stroke, hemorrhage, mortality, and morbidity, especially in the elderly.37-40 For instance, Shin et al 37 reported an increased risk of stroke incidence in those taking these medications, especially in the hemorrhagic type. In contrast, other studies found these medications to be safe, disproving the results of the previous studies.41-43 For instance, Lee et al 41 studied a large number of patients using SSRIs and Tricyclic antidepressants (TCAs) and concluded that the risk of stroke was significantly lower in patients using SSRIs. Ried et al 42 also showed that SSRIs could decrease poststroke mortality and, therefore, could be used as an element of a therapeutic regimen for patients with stroke. Furthermore, Marquez-Romero et al 44 have started a randomized, double-blind, placebo-controlled, multicenter clinical trial to evaluate the effect of fluoxetine: Fluoxetine for Motor Recovery After Acute Intracerebral Hemorrhage (FMRICH).
In the present study, the incidence of recurrence of stroke in the 3-month follow-up period was low in both groups (2 and 1 cases in the citalopram and placebo groups, respectively). In addition, ICH was reported in 2 and 3 cases in the citalopram and placebo groups, respectively. No case of postischemic hemorrhage was observed. Of note, the mortality rate was lower in the citalopram than placebo groups (4 vs 11 cases, respectively); this, however, was not of statistical significance. Also, no considerable complications were observed, and the difference between both groups regarding the complications was insignificant. Throughout the 3-month study period, cardiovascular events were observed in 3 and 8 cases in the citalopram and placebo groups, respectively, which could be explained by the higher number of cases with preexisting cardiac diseases in the placebo group. Interestingly, a significant improvement in the speech elements of NIHSS score was observed in the citalopram group, suggesting a positive effect of these agents; however, further, more specific, verbal tests are required to confirm this hypothesis.
In conclusion, citalopram seems to be an efficient medication in improving the stroke severity of patients with acute IS. Considering the lack of major complications and low intolerance to this medication, its administration in patients with acute IS would facilitate the rehabilitation of these patients. However, further multicenter studies with larger sample sizes and longer follow-up periods are recommended in order to study better the medical aspects of this medication in the motor recovery of and outcome for stroke patients.
Footnotes
Acknowledgements
We would like to acknowledge Dr Hajimohammad Soleimani, Dr Ghader Borji, Dr Hannaneh Aliyar, Sara Gholamzadeh, and Gity Baybordi for their assistance during this study.
Authors’ Note
This clinical trial conforms to consort statement.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Neurosciences Research Center (NSRC) of Tabriz University of Medical Sciences was the source of funding in the conduct of this study.
Supplementary material for this article is available on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
