Abstract
Background. Motor imagery (MI) is increasingly recognized as a treatment option after stroke, but not all stroke patients are able to perform MI. Objective. To examine if severe somatosensory deficits would affect MI ability. Methods. The Box and Block Test (BBT) was used to evaluate mental chronometry as 1 component of MI. Two groups of stroke patients and an age-matched healthy control group (CG) were studied. Patient group 1 (n = 10, PG1) had a severe somatosensory impairment on the affected side and PG2 (n = 10) had pure motor strokes. All subjects first performed the BBT in a mental and in a real version. The time needed to move 15 blocks from 1 side of the box to the other was measured. To compare the groups independently of their performance level, a (real performance − MI)/(real performance) ratio was calculated. Corticospinal excitability was measured by transcranial magnetic stimulation at rest and while the subjects performed an imagined pinch grip. Results. The CG performed the BBT faster than both patient groups, and PG1 was slower than PG2. MI ability was impaired in PG1 but only for the affected hand. Transcranial magnetic stimulation data showed an abnormally low MI-induced corticospinal excitability increase for the affected hand in PG1, but not in PG2. Conclusions. Severe somatosensory deficits impaired mental chronometry. A controlled study is necessary to clarify if these patients benefit at all from MI as an additional treatment.
Keywords
Introduction
Motor imagery (MI) can be defined as “a dynamic state during which the representation of a given motor act is internal rehearsal without any overt motor output.” 1 From sports literature it is well known that MI, when applied in addition to physical exercises, is more effective than MI or exercises only. 2 MI abilities have been studied with different approaches. MI vividness is examined by a questionnaire, hand rotation tests explore the subject’s ability to perform mental rotation, and mental chronometry is tested to examine temporal organization of simulated actions. 3 Tests of mental chronometry have been shown to be reliable in stroke patients. 4 An accurate timing of movements is an important feature of coordination and dexterity. Mental chronometry is influenced by several factors, for example, duration and complexity of the task, the type of MI (kinesthetic or visual imagery), and the instruction. 5
Approximately 10 years ago, the first studies that applied MI to patients with poststroke motor deficits were published. Most of them found MI to be beneficial as an add-on therapy.6-11 A systematic review concluded that MI-associated improvements of motor function could be demonstrated in tests such as the Fugl-Meyer Stroke Assessment and the Action Research Arm Test. 12 However, not all stroke patients are equally able to perform MI. Sirigu et al13,14 had reported that lesions of the left parietal cortex affected MI abilities. On the other hand, Johnson et al 15 found an intact ability to engage in MI in chronic stroke patients without premotor or parietal cortex lesion. De Vries and Mulder 16 reported that 40% of their stroke patients were impaired on motor and visual imagery.
Clinically, patients with a severe loss of somatosensory input often report that they feel unable to localize the position of their extremity in space without visual control. Therefore, we hypothesize that the ability to mentally simulate upper extremity movements is closely associated with intact afferent somatosensory input. To test this hypothesis, we studied 2 groups of stroke patients. In 1 group, sensory deficits were prominent; the other group had motor strokes without any somatosensory deficit. The patients were compared with an age-matched control group. Our hypothesis was that MI should be more difficult for patients with sensory deficits. To test our assumption, we employed the Box and Block test (BBT) to investigate mental chronometry and used transcranial magnetic stimulation (TMS) to detect MI-induced changes of corticospinal excitability. In normal subjects, the time to complete an imagined movement is similar to the time needed for actual execution of that movement. 16 Mental chronometry tests in stroke patients have also been used by other research groups.17,18
The TMS studies that explored MI-induced excitability changes in stroke patients are somewhat controversial. Two groups19,20 have described MI-induced task-specific increases of excitability in the affected hemisphere, another group 18 did not find a facilitation in the ipsilesional motor cortex. Whereas Cicinelli et al 19 reported excitability increases irrespective of lesion location, Battaglia et al 21 found an impaired motor facilitation during MI in patients with cerebellar stroke.
Methods
Patients
A total of 20 stroke patients were included after having given consent to participate. The study protocol was approved by the ethical committee of the University of Konstanz. Participants were recruited during inpatient neurological rehabilitation. They stayed in the hospital for 4 weeks and had 3 treatment hours per day, 5 days a week. Treatments focused on hand and arm function for approximately 2 hours per day and were similar for all patients regarding amount and duration of therapy. The amount of sensory stimulation was similar; both groups received training with the NOVAFON, a vibration device.
Inclusion criteria included occurrence of a first stroke, sufficient strength in the affected hand to grasp and release an object, and the ability of TMS to evoke responses in a hand muscle. Contraindications included a history of epileptic seizures, inability to understand the instructions, intake of drugs known to affect brain excitability, pregnancy, and metallic implants. The vibration sense was measured with a tuning fork that was scaled from 0 to 8, thus allowing some quantification. “0” means no sense of vibration and “8” means that vibration is still felt just before the input ceases. Stereognosis was tested by touching a finger while the subject had eyes closed. Three different fingers were studied in each hand. If the subject was wrong in 1 finger, it was called “slightly reduced,” if he or she was wrong in 2 or 3 fingers, stereognosis was defined as being strongly reduced. Light touch was tested with a cotton carrier. If the subject did not feel the mere touch but felt it when applied with a gentle pressure, the sense was called “slightly reduced.” If the patient did not feel it with that pressure, we called it strongly reduced. “Loss” means that the subject did not feel it with a higher pressure. Light touch was tested at 3 fingers per hand. Motor strength was evaluated clinically by applying the Medical Research Council Scale. All patients had a similar degree of strength, ranging between 4− and 4+. Lesion location was determined by brain imaging. In 18 patients, a magnetic resonance imaging of the brain was available, and in 2 patients a computed tomography scan was done.
Measures
In 18 patients, median nerve somatosensory evoked potentials (SSEPs) were recorded to demonstrate a more objective measurement of sensory nerve conduction. The median nerve was stimulated at the wrist (pulse duration, 0.1 ms; frequency, 5.6 Hz; 150 stimuli); surface electrodes for recording were positioned 3 cm behind and 5 cm lateral of Cz.
Box and Block Test
To explore the ability of MI and to test motor functions, patients and healthy subjects were asked to perform the BBT first mentally and then as a practical execution.22-26 We chose to use the BBT even though it has not yet been validated as a test of MI ability, because it has a standardized size, is well known, is easy to administer, and includes the most important aspects of upper extremity motor functions (grasping, moving, and releasing an object). We tested the BBT in a group of 79 healthy subjects (Greiner et al, unpublished data). These were subdivided into 3 age-groups. The group with the oldest subjects (56-83 years) performed slightly but significantly worse in the mental chronometry task than the other 2 groups. The time difference between imagination and execution of the task was 1.4 ± 1.2 seconds in the 2 younger age-groups and 2.4 ± 1.8 seconds in the oldest age-group, indicating that there is a small decline in mental chronometry with increasing age. There were no sex differences.
We decided to use the BBT rather than an MI questionnaire because the former measures MI abilities more objectively than by self-report.
Transcranial Magnetic Stimulation
Our reasons for using transcranial magnetic stimulation (TMS) measurements in the patients are the following: First, patients had to perform an MI task. This makes it highly probable that TMS as a method to probe the motor system can demonstrate MI-associated excitability changes. Second, pure sensory deficits can alter motor cortex excitability. 27 Therefore, we were interested whether the motor system could still be activated by MI despite the sensory deficit.
Recordings with surface electrodes (belly-tendon montage) were taken simultaneously from the first dorsal interosseus muscle (FDI) on both sides to study motor excitability in both hemispheres.
TMS was performed with a circular coil (outer diameter 14 cm; The Magstim Company, Dyfed, UK), which was connected to a magnetic stimulator (Magstim 200 HP). The optimal coil position where MEPs could be evoked with the lowest stimulus intensity from both FDI was marked with ink on the scalp to ensure an exact repositioning of the coil throughout the experiment. At this coil position the motor threshold (MT) was determined. MT was defined as the stimulus intensity needed to produce MEPs with a size of 50 to 100 µV in 5 out of 10 consecutive trials during complete muscle relaxation. MT for the affected side was higher than for the healthy side. We therefore decided to apply TMS single pulses with an intensity of 120% of the affected side’s MT at rest.
Experiment 1: Box and Block Test
Prior to task performance, subjects kept 1 block in their hand for 10 seconds in order to get an impression of the weight and the surface. They were asked to keep their eyes open and to look at the box where 15 blocks were positioned (3 blocks per row in 5 rows). Then they were asked to imagine picking up each block 1 by 1 and moving it to the other side of the box. They were encouraged to keep a sequence, for example, starting with the first row and finishing with the last row, but were free to decide where to start. They were instructed to imagine performing the movements as fast as possible. Thus, they had to imagine from a first-person perspective and take the impairment of the paretic hand into consideration. Afterward, they were asked if they had felt to be able to perform the task. All subjects stated that they had been able to do so.
They received a signal when to start this imagination and gave a stop signal as soon as they had completed the task mentally. The time between “go signal” and “stop signal” was measured. Then, subjects performed the task. The time difference between the imagination task (MI time) and the actual task (real time) was chosen as a measure of MI ability. Both hands were studied consecutively. In the patient group, the affected hand was tested first, and the nonaffected hand was tested approximately 10 minutes later. In the healthy subjects, the order (right or left hand first) was randomized. To eliminate the difference in performance (patients being slower than healthy controls) data were normalized by the formula (real performance − MI/real performance).
Experiment 2: Transcranial Magnetic Stimulation
Subjects were allowed to hold a clothes peg between thumb and index finger for 10 seconds prior to initiation of the experiment. All stated that they were familiar with a clothes peg and had a vivid image of using it. Then the patients were instructed to imagine a tonic pinch grip by pressing a clothes peg with thumb and index finger. They kept their eyes closed while performing MI. The experiment started only after the patient announced that he felt able to imagine this motor task. The reason for choosing an MI task with a tonic motor activity was that a reliable timing of the TMS pulse is impossible during a dynamic MI task. We did not study more proximal muscles, since these muscles usually have higher MTs. We wanted to avoid high stimulus intensities in order not to elicit any feeling of discomfort in the patients.
Two seconds after the command to start the imagery the TMS pulse was given. After each TMS pulse, subjects were asked to stop imagery for a few seconds in order to avoid mental fatigue. The task was repeated 8 times. The frequency of TMS was approximately 0.1 Hz. Auditory feedback via a loudspeaker ensured that, during imagery, no muscle contraction occurred. Recording of each trial started 30 ms prior to the TMS pulse and finished after 200 ms. Trials with EMG contamination were excluded from further analysis. As a control, 16 TMS pulses were applied during rest. The sequence (affected hand first or unaffected hand first) was randomized. Recordings were stored on a Viking IV (Nicolet, Kleinostheim, Germany) and analyzed off-line. Motor evoked potential (MEP) amplitudes were measured peak-to-peak. Amplitudes of MEP during MI were expressed as percentage of the mean MEP amplitude obtained during the rest condition. For analysis, a laterality index (MEP amplitude of the FDI ipsilateral to the imagined pinch grip/MEP amplitude of the FDI contralateral to the imagined pinch grip) was calculated. The higher the value, the more the increase of excitability is directed toward the hand the imagination is focused on.
Statistical Analysis
In experiment 1, we used a 2-factorial analysis of variance (ANOVA) with the factor GROUP (3 levels; 2 patient groups and the control group) and the factor SIDE (2 levels, affected side and unaffected side) to compare the durations for BBT real performance. This test was also applied for comparing the normalized ratios (real performance − MI/real performance). Ratios were expressed as absolute differences, irrespective of whether MI time was shorter than performance time or vice versa. In experiment 2, ANOVA tests were used to compare laterality indices between the patients groups. For each patient group, 2 laterality indices exist: MEP size affected side/MEP size unaffected side for MI of the affected side and MEP size unaffected side/MEP size affected side for MI of the nonaffected side. A 2-factorial ANOVA with GROUP (2 levels) and LATERALITY (2 levels) was run. In both experiments, the level of significance was assumed at 5%. In case of a significant result, post hoc t tests were performed to identify the differences in detail.
Results
Patient group 1(PG1) included 10 stroke patients (mean age = 64 ± 10 years with mean duration of symptoms 14.9 ± 13.8 weeks. Patient group 2 (PG2) consisted of 10 stroke patients with a pure motor stroke (mean age, 62 ± 14 years) with mean duration of symptoms 7.4 ± 3.4 weeks. Further details are presented in Table 1. The control group (CG) had 10 healthy subjects (mean age, 65 ± 11 years). All subjects were naïve to MI prior to inclusion in the study. None had a profession that might have used MI training, such as professional musicians or high-level athletes.
Clinical Characteristics of the 2 Patient Groups a
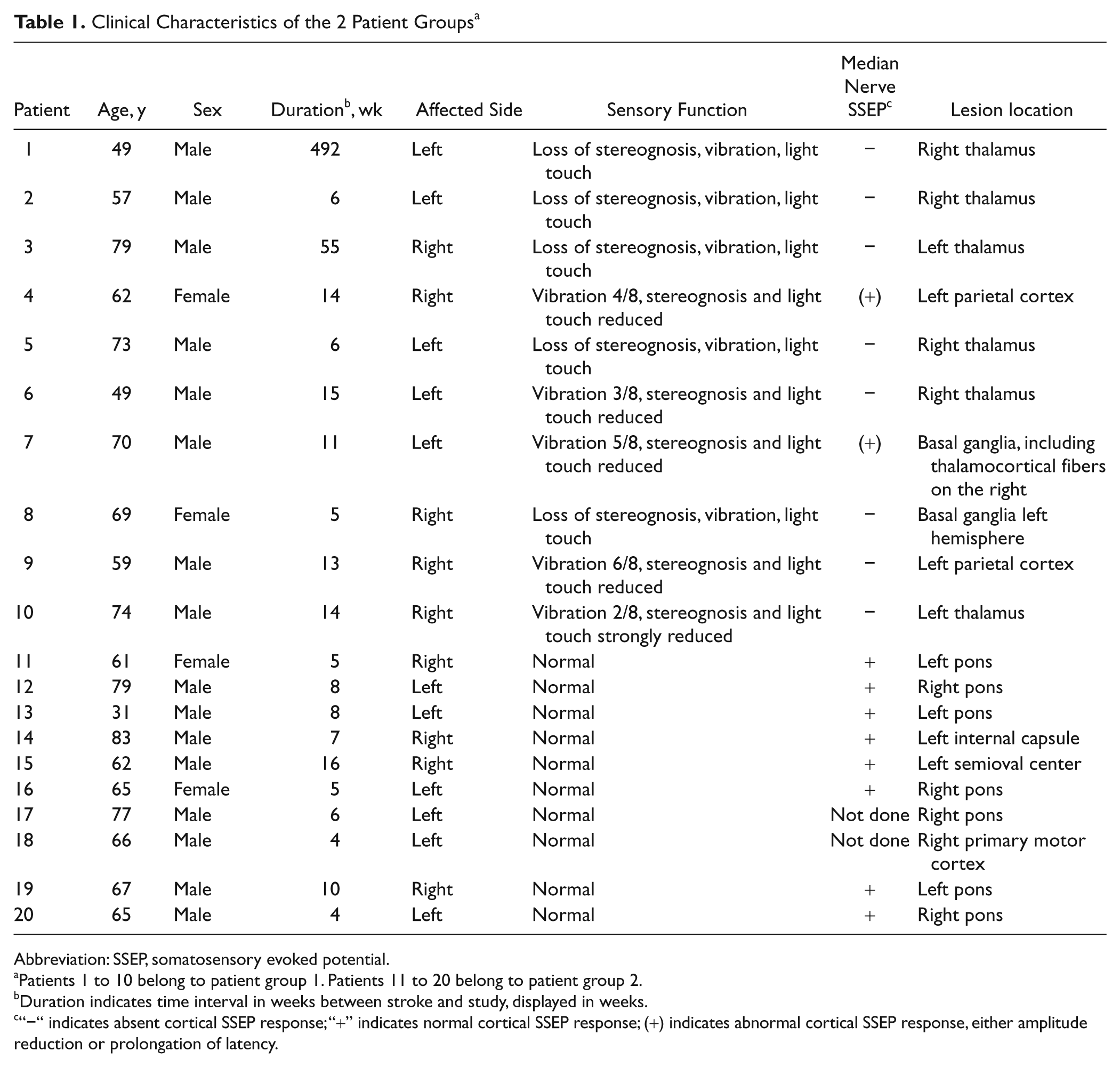
Abbreviation: SSEP, somatosensory evoked potential.
Patients 1 to 10 belong to patient group 1. Patients 11 to 20 belong to patient group 2.
Duration indicates time interval in weeks between stroke and study, displayed in weeks.
“−“ indicates absent cortical SSEP response; “+” indicates normal cortical SSEP response; (+) indicates abnormal cortical SSEP response, either amplitude reduction or prolongation of latency.
Experiment 1
BBT performance
The ANOVA indicated a significant difference between the 3 groups, F(1, 59) = 86.3; P < .001, a significant difference between affected and unaffected side, F(1, 59 = 22.8; P < .001, and a significant interaction GROUP × SIDE, F(1, 59) = 42.3; P < .001. Post hoc tests showed that healthy subjects performed significantly better than both patient groups with the affected side. Patients in PG2 completed the task significantly faster than those in PG1. For the unaffected side, motor function was similar in the patient groups. Both patient groups were slower than the CG (Figure 1).
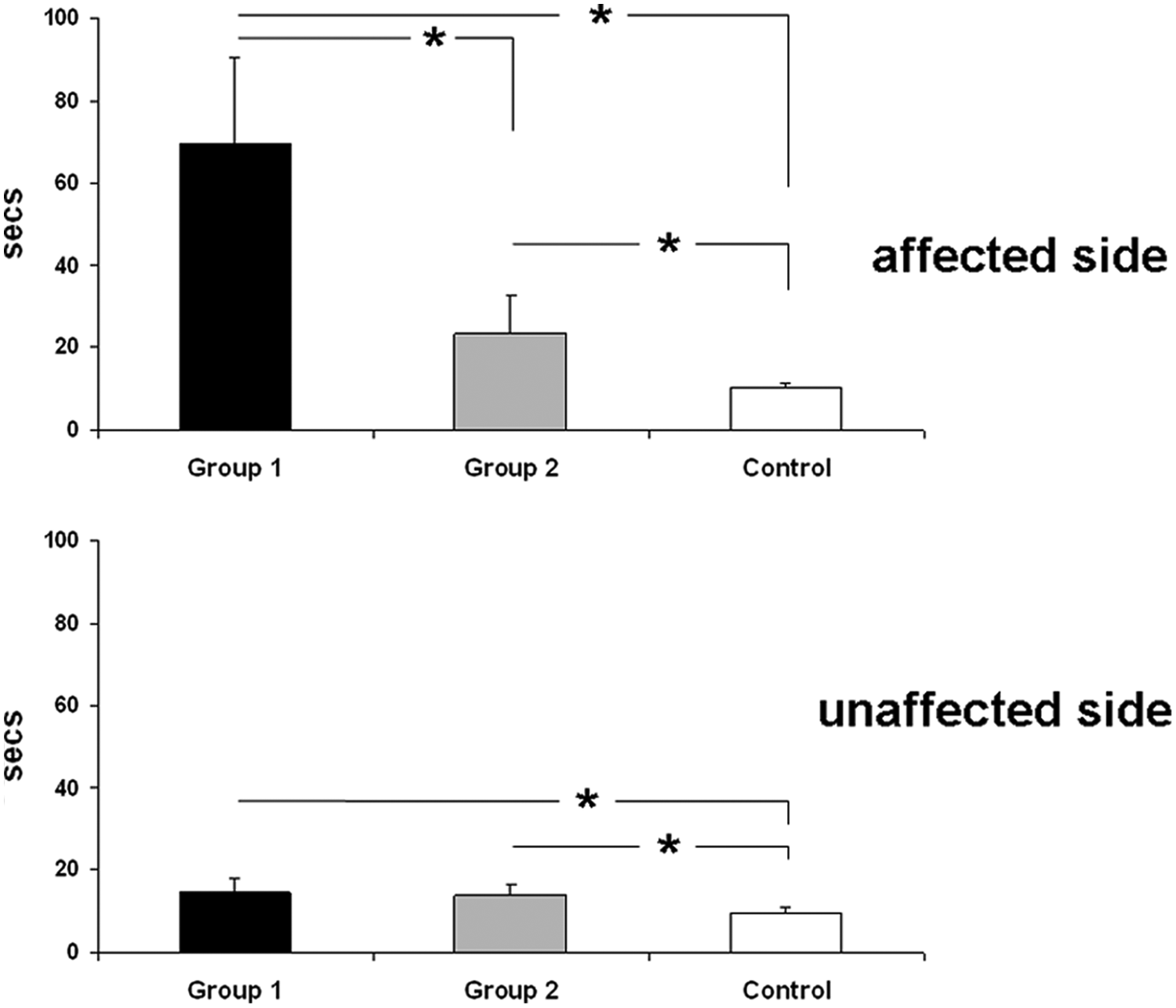
Time needed to complete the Box and Block Test measured in seconds for the 2 patient groups and the control group. Error bars show standard deviations. *P < .05.
Ratios
The data showed a normal distribution, so we applied ANOVA calculations. The ANOVA that compared MI of the affected hand in the 2 patient groups with the first MI of the healthy subjects showed a significant difference between the groups, F(1, 59) = 5.9; P = .005. Affected and unaffected sides also showed a significant difference, F(1, 59) = 6.2; P = .014. However, the interaction GROUP × SIDE was not significant, F(1, 59) = 1.75; P = .192. Post hoc tests demonstrated that MI in PG1 was significantly different from MI in PG2 and the CG. There was no difference between healthy subjects and patients without sensory deficits (Figure 2).
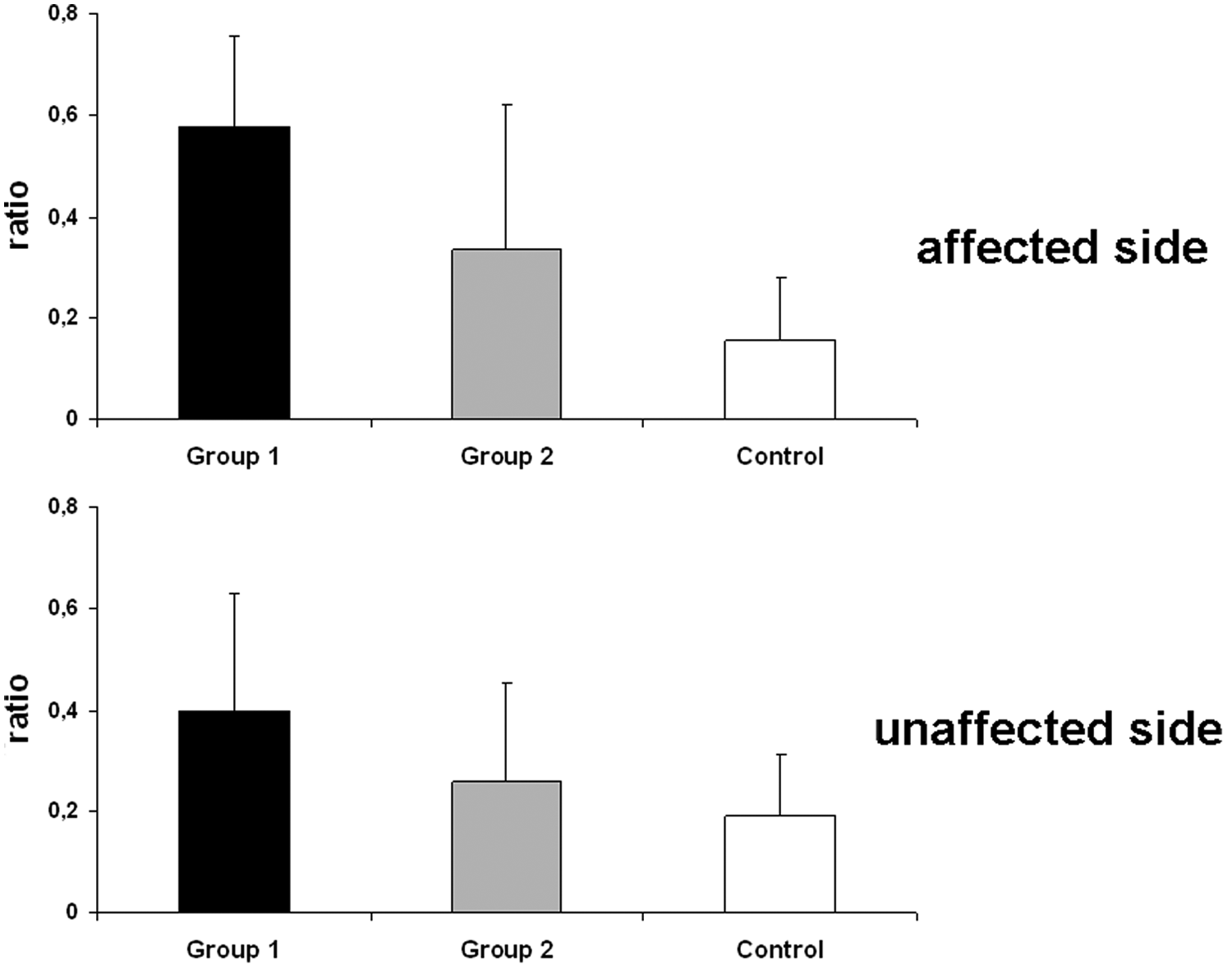
Ratios, (real performance − motor imagery)/(real performance), in the 2 patient groups and the healthy control group. Error bars show standard deviations. Significant differences are described in the Results section. Note that the lower the ratio, the smaller the difference between motor imagery and real task execution.
All patients of PG1 estimated that they would perform the task faster than they actually did (range of difference between MI and real performance: from −11 to −69 seconds). In PG2, 6 subjects were faster in their MI than in real performance; in the other subjects it was vice versa (range of difference between MI and real performance: from −20.9 to +17.8 seconds). In the control group, MI was shorter than real performance in 6 subjects and vice versa in the other 4 subjects (range of difference between MI and real performance: from −4 to +3 seconds).
Experiment 2
The ANOVA showed a significant interaction between GROUP and LATERALITY, F(1, 39) = 8.73; P = .005. Post hoc tests demonstrated that MI affected in PG1 was significantly different from the other 3 conditions (MI unaffected in PG1, MI affected in PG2, MI unaffected in PG 2; Figure 3). MTs at rest did not differ between the 2 patient groups: PG1, affected side, 53.6 ± 7.7; nonaffected side, 52 ± 5.9; PG2, affected side, 56.1 ± 10.9; nonaffected side, 51.6 ± 5.8. Results are expressed as percentage of maximum stimulator output intensity. However, MTs expressed as laterality indices (MT affected/MT nonaffected) were significantly different between the 2 groups, F(1, 19) = 9.6; P = .006. PG2 had a higher laterality index (1.16 ± 0.12) than PG1 (1.03 ± 0.05), indicating relatively higher MTs in the affected hemisphere of patients with pure motor strokes.
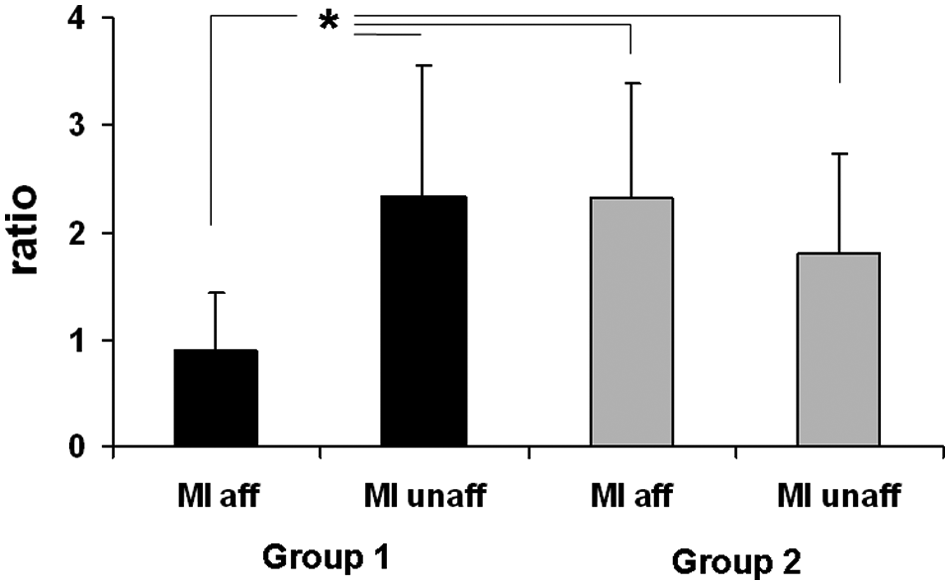
Laterality indices (MEP amplitude of the side the imagery is directed to/MEP amplitude of the nonimagined side) in the 2 patient groups. Abbreviations: MEP, motor evoked potentials; MI, motor imagery. Black columns, patient group 1; gray columns, patient group 2. Error bars show standard deviations. *P < .05.
Discussion
A main result of this study is that stroke patients with somatosensory deficits are more impaired in their ability to perform a mental chronometry task as one aspect of motor imagery ability than stroke patients with pure motor deficits. All patients with impaired somatosensory functions underestimated the severity of their functional deficit and expected to complete the BBT much faster than they actually did. This phenomenon cannot be attributed to a lack of experience, since the stroke had occurred at least several weeks before and all patients had already participated in a rehabilitation program, which included exercises with the affected upper limb. Also, it cannot be because of an impaired ability to imagine motor acts in general, since the nonaffected hand was not significantly different from the control group. Therefore, we suggest that MI per se is still intact. The impairment of mental chronometry is induced by the lack or reduction of somatosensory input from the affected upper limb. Probably, the cortical representation of the affected hand is impaired, making it much more difficult to involve the extremity in a mental motor program.
Task execution was particularly impaired for the affected hand of PG1. However, patients were also slower than the control group when performing the BBT with the unaffected hand. This result confirms other reports showing an impaired dexterity of both hands after unilateral stroke. 28
Our TMS results also show differences in MI-associated excitability increases between the 2 patient groups. It is difficult to draw conclusions from the TMS results to explain the BBT results because of the different tasks in experiments 1 and 2. Usually, MI induces a rather selective increase of corticospinal excitability for the target muscles involved in the MI task.29-33 The laterality indices used in this study indicated that this is also the case for the nonaffected hand in patients with somatosensory deficits and for both hands in the patients with pure motor strokes. In contrast, the laterality index <1 that was found for the affected hand in PG1 demonstrates that the excitability increase was more pronounced on the nonaffected side during the MI task involving the affected hand. The finding cannot be explained by an ipsilesional reduction of motor excitability in general since, in earlier studies, we showed that thalamic lesions and a lesion in the primary somatosensory cortex are associated with an intracortical disinhibition in the primary motor cortex that induces an increase of motor excitability.27,34 We suggest that patients with somatosensory deficits had more difficulties imagining the tonic pinch grip and therefore could not focus their attention on the affected side. A relationship between attention and corticospinal excitability has been described in healthy subjects. 35 If attention was diverted away from a tactile discrimination task, the increase in motor excitability was much smaller than if attention was directed toward the task. As usual in studies with MI, we cannot completely exclude the possibility that the result is because of a lack of deliberate cooperation. However, we consider this to be improbable, since the results obtained from the nonaffected side were as expected.
Cicinelli et al 19 included patients with different lesion locations, for example, internal capsule and parietotemporal regions. They did not find location-associated differences in MI-induced excitability increases. However, the article does not describe the presence or absence of somatosensory deficits, so we cannot compare their results with our study. Stinear et al 18 reported the absence of MI-induced facilitation in the lesioned hemisphere. However, they described a hemispheric asymmetry. In right-hemisphere lesions, MI did not facilitate MEPs in either hand. In contrast, patients with left-hemisphere lesions had MEP increases in the left hand during MI of the right hand. The latter finding resembles our result in the patients with somatosensory deficits. Stinear et al 18 included patients with different lesion locations (mainly basal ganglia, internal capsule, parietal cortex, and insula) but does not describe the presence or absence of sensory deficits. In contrast to their study, we did not find a hemisphere-specific effect, since right- and left-hemispheric strokes were equally distributed in our patient groups, and neither BBT results nor TMS results could be associated with one particular hemisphere. In a recent study, 36 TMS demonstrated that auditory and visual cuing of MI are both effective on the paretic side of chronic stroke patients, but sensory impairment was not reported.
Our study does not cover all aspects of motor imagery abilities, for example, the vividness of MI was not taken into consideration. 37 The relevance of different MI tests and their correlations with each other are currently being explored. We started the experiment with the mental chronometry task. Task execution was performed afterward. This sequence does not allow development of a motor engram, which may have limited the mental chronometry results. However, this effect was similar in all groups and should therefore not account for the differences between groups.
The crucial question is whether patients with severely reduced somatosensory input might not be suitable for MI exercises as an add-on therapy during rehabilitation. Of course, although our results suggest that these patients have greater difficulties in performing MI tasks, a controlled prospective study in which MI is used as a treatment will be necessary to answer the question. Preliminary results from 2 stroke patients undergoing constraint-induced movement therapy and mental practice also showed that the patient with a parietal lesion had less benefit from the treatment than the patient with a nonparietal damage. 38 Our ultimate goal should still be to become able to define for individual patients a priori what the most effective treatment will be. Knowledge about the intactness of somatosensory input may become a highly relevant issue for the application of a MI intervention in stroke patients.
Footnotes
Acknowledgements
We thank Professor Schoenfeld and the reviewers for helpful comments and suggestions.
The author(s) declared no potential conflicts of interests with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
