Abstract
Identification of broadly neutralizing antibodies (NAbs) generated during the course of HIV-1 infection is essential for effective HIV-1 vaccine design. The magnitude and breadth of neutralizing activity in the sera from 46 antiretroviral treatment–naive HIV-1 clade C-infected individuals was measured in a single round infection assay using TZM-bl cells and multisubtype panel of env-pseudotyped viruses. Higher levels of NAb response (NAb titer 500 to >40 000) were measured in these patients against tier 1 and tier 2 viruses. The average magnitude of the NAb responses of chronically infected individuals against heterologous viruses was consistently higher than the response observed from individuals with long-term nonprogressor (P = .086). To conclude, high titers of HIV-1 cross-neutralizing activity were observed in the sera from a subset of HIV-1-infected individuals in Chennai, India. Additional studies of the epitopes recognized by these antibodies may facilitate the discovery of an effective vaccine immunogen.
Introduction
Broadly neutralizing monoclonal antibodies (NAbs) against glycoprotein 120 ([gp120]; immunoglobulin, 2G12, PG9, and PG16) and gp41 (4E10 and 2F5, [IgG]b12) have been described to have promising neutralizing ability against multiple circulating HIV-1 clades. However, measuring the length and breadth of broad neutralizing activity evolved during the course of HIV-1 infection is essential for vaccine studies. The antibodies that are capable of neutralizing heterologous viruses have been seen to target epitopes of highly conserved sites that might serve suitable for inclusion in vaccine immunogen. The quantification of these broadly NAbs in distinct geographical locations may provide information that facilitates the design of a vaccine that is effective worldwide. In such an effort, it was identified 1 that the subtype “C” envelope (env) sequences isolated to prepare pseudoviruses from the western part of India were highly complex and that the Indian HIV-1 gp120 subtype sequence’s genetic make up was distinct from 2 subtype “C” sequences from other parts of the globe. 3 –8 Also among the Indian subtype “C” sequences, it was observed 9 –11 that there is greater sharing of neutralization determinants, which can lead to an unvarying neutralization response in most of the specimens, and this should be explored. Hence, it is essential to study the autologous and heterologous neutralizing antibody (NAb) response from different parts of the country to identify the potent neutralizing monoclonal antibodies (mAbs).
Neutralization antibody response in different populations from San Diego, 12 La Jolla, 13 Paris, 14 Seattle, 15 San Francisco, 16 and Amsterdam 17 are available where the HIV-1 subtype “B” is prevalent, but the data on subtype “C” infected individuals is very limited, 18 –20 among the Indian population there are potent and broadly NAbs were described in recent studies 10,21 from recent 2,10,22 and chronically 1,11,21 infected individuals from the western part of the country. But there are no data on NAb response available from south Indian patients where the largest population 23 of HIV-1-infected individuals are being managed. The data about the NAb responses from long-term nonprogressors (LTNPs) are essential to find out the role of NAbs in controlling disease progression in the absence of antiretroviral (ARV) drugs for a longer duration of time. In the present study, we report the dynamics of neutralization response in a cohort of HIV-1-infected chronic and LTNPs from a tertiary referral center in Chennai, South India, with a panel of distinct molecularly cloned HIV-1 envelope pseudoviruses from different parts of the world, which is highly essential to influence future vaccine studies.
Methods
Study Design
The YRG Center for AIDS Research and Education (YRG CARE) of Chennai is the large community-based tertiary HIV-1 care institution in India. Since 1996, it has provided a continuum of care for more than 15 000 HIV-infected individuals. Individuals confirmed for HIV-1 infection and currently registered in YRG CARE for their HIV-1 disease management were recruited. All the study participants were never exposed to ARV drugs and had not encountered any opportunistic infections in their lifetime. Participants were assigned to 2 groups depending on their clinical status. (i) Individuals with CD4 count more than 500 cells/mm3 and diagnosed for HIV-1 infection at least 7 years earlier to the date of specimen collection were categorized as LTNPs (n = 16); (ii) individuals irrespective of their CD4 counts diagnosed for HIV infection within 7 years and at least 2 years earlier to the date of specimen collection were considered chronically infected individuals (n = 30). Informed consent was obtained, and whole blood was collected in a closed system of collection using serum separator tubes (BD Vacutainer SST, Becton Dickinson, New Jersey) containing clot activators. The study was approved by the institutional review board of YRG CARE.
HIV-1 Single-Round Neutralization Assay
Neutralization assay
Neutralization was measured by quantifying luciferase reporter gene expression in TZM-bl cells after a single round pseudovirus infection as described earlier. 24 Briefly, tissue culture of 50% infectious dose of each virus preparation was determined on TZM-bl cells. Patient serum was heat inactivated at 56°C for 30 minutes prior to assay. Serum samples were diluted with Dulbecco-modified Eagle medium (Gibco, Carlsbad, CA, USA) 10% fetal bovine serum (Hyclone Laboratories, South Logan, UT, USA) with the starting dilution of 1:20 by 3-fold serially diluted. Then 50 µL of env pseudovirus was added and kept for 45 to 90 minutes at 37°C in 5% CO2 environment. After 1 hour of incubation, the TZM-bl cell monolayer was trypsinized, and the cells were seeded in 10 000 cells per well in 100 µL of growth media and incubated for 48 hours at 37°C in 5% CO2 environment. A virus control (pseudovirus and cells) and a cell control (only cells) well were assigned for the validation of assay. After 48-hour incubation, 150 µL of supernatant was discarded from each well and 100 µL of substrate (BriteLite plus Reporter Gene Assay System, PerkinElmer Life and Analytical Sciences, Shelton, Connecticut) was added to each well and incubated for 2 minutes. Then 150 µL of lysed suspension was transferred into a 96-well black solid plate (Costar Corning, New York) for measurements of relative luminescence units (RLU) using luminometer (VICTOR X3 Multilabel Plate Reader, PerkinElmer Life and Analytical Sciences, Turku, Finland). Neutralizing antibody titers were expressed as the reciprocal of the serum dilution required to reduce RLU by 50% compared to those of the virus control wells after subtraction of background RLUs. Fifty percent inhibitory dilutions (ID50) were calculated based on a curve fit to each neutralization assay in the Atlas Macro online software tool. 25
Cell stocks
The TZM-bl cells were obtained from John Kappes and Xiaoyun Wu through the National Institutes of Health AIDS Research and Reference Reagent Program. These were HeLa cell clones genetically engineered to express CD4, CXCR4, and CCR5 and contained integrated reporter genes for firefly luciferase and Eschericia coli–galactosidase under control of an HIV-1 long terminal repeat. HIV-1 pseudoviruses: Molecularly cloned env pseudoviruses used in this study were obtained from Dr David Montefiori and Hagen von Briesen, Fraunhofer Institut für Biomedizinische Technik IBMT, Sulzbach, Germany. Monoclonal antibodies: the human anti-gp120 carbohydrate–specific mAb 2G12, the human anti-gp41 mAb 2F5 and 4E10, recombinant sCD4, and HIV immunoglobulin G were kindly provided by Dr David C. Montefiori, Duke University Medical Centre, Durham, North Carolina.
Statistical Analysis
Nonparametric tests were used for all analyses. Difference in continuous variables between participant groups was analyzed with Mann-Whitney U test. All the correlations were determined using Spearman rank correlation and Wilcoxon rank test. All statistical analyses were done using SPSS software, version 13.0 Chicago, Illinois. A P value of <.05 was considered statistically significant.
Results
A total of 46 individuals were included in this study. The clinical characteristics of these participants are given in Table 1. Among these study participants, 20 were men and 26 were women. The HIV-RNA levels of these individuals ranged between 1.6 and 6.8 log10 copies/mL (interquartile range [IQR]: 3.7-5.1), and the CD4 count was between 283 and 1617 (IQR: 363-647).
Clinical Characteristics of Study Participants.

Abbreviations: IQR, interquartile range; LTNP, long-term nonprogressor.
Cross-Neutralizing Activity against Heterologous Viruses
Of these 46 sera tested, higher neutralization response was observed in 23 specimens (Figure 1) against distinct HIV-1 pseudoviruses. Notably, many serum specimens have neutralized subtype “C” viruses from Indian and 1 South African strain. The geometric mean ID50s ranged between 20 and 3839. As expected, there was a higher response for tier 1 viruses than that against tier 2 viruses. Among nonprogressors, 5 participants showed higher NAb responses against multiple HIV-1 subtypes, with ID50 neutralization titers of 500 to >40 000. Among chronically infected individuals, 10 participants showed high NAb titers >1000 and up to 43 000 against tier 1 viruses from the United States (SF162LS, Bal.26, and SS1196.1) and Malawi (MW965.26). Against tier 2 viruses, higher neutralization titers were observed against viruses of Indian origin (HIV-00836-2.5, HIV-001428-2.42, HIV-0013095-2.11, HIV-16055-2.3, HIV-16845-2.22, HIV-16936-2.21, HIV-25710-2.43, HIV-25925-2.22, and HIV-26191-2.48). Low-level responses with titers ranging between 1:20 and 1:100 were observed against tier 2 viruses from other countries, such as Zambia (HIV-ZM249M.PL1), Kenya (Q259.d2.17, Q842.d12, Q461.e2, Q769.d22), and South Africa (TV1.21). The average magnitude of the NAb responses of chronically infected individuals against heterologous viruses was consistently higher than the response observed from the individuals with LTNP (Figure 2; P = .086). Also the magnitude of the NAb response against heterologous viruses of Indian origin ranged between 20 and 5446 (median [IQR] 96 [32-352]); this was consistently higher than the response observed against heterologous viruses of African origin ranging between 20 and 43 720 (median [IQR] 37 [20-165]; P < .0001; Wilcoxon rank test). This trend was also observed within 2 different populations studied—both chronic (P < .001) and LTNP (P = .029). Also analysis of the magnitude of NAb response between subtype “C” and nonsubtype “C” African viruses found that the response to African subtype “C” viruses was significantly higher than that of African nonsubtype “C” viruses (P = .003). The same significance was found when the analysis was limited only with chronically infected individuals (P = .011).

Magnitude and breadth of neutralization by subtype C serum specimens. A panel of chronically infected individuals and long-term nonprogressors (LTNPs) are depicted. The tier 2 and tier 1 viruses are given in top and bottom halves, respectively. The values from the HIV-negative control specimens are given on the right side of this table; The pseudovirus clades and their graphic origin are described in the third column. Fifty percent inhibitory dilutions (ID50) titers are color coded as follows: titer <20 yellow boxes, titer <100 dark yellow boxes, titer <500 golden brown boxes, titer <1000 dark brown boxes, titer >1000 red boxes, the viral load and CD4 values of these specimens are given at the bottom.
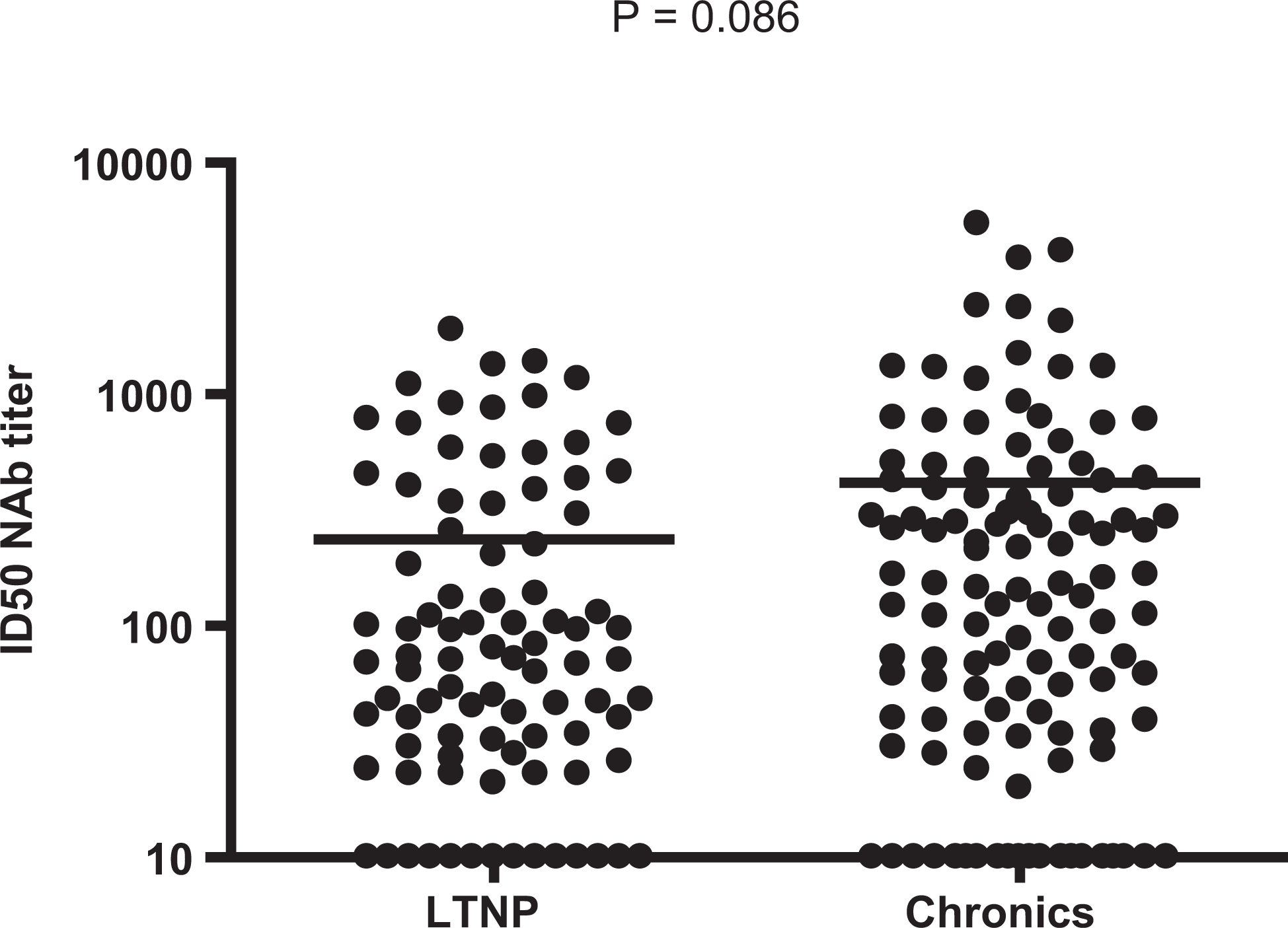
Neutralizing antibody titers of participants with long-term nonprogressor ([LTNP] n = 16) and chronically HIV-1 infected individuals (n = 30) against Clade C pseudoviruses derived from Indian HIV-1 strains.
When all the study participants are considered, there was a consistently higher response to the heterologous viruses than to the non-HIV envelope control virus (murine leukemia viruses), suggesting the heterologous neutralizing ability of these individuals against HIV-1 pseudoviruses. There was a negative correlation observed between the neutralizing titers and the plasma HIV-1 RNA levels against different pseudoviruses which is depicted in Table 2. However, there was a positive association found between the magnitude of neutralizing responses against 1 heterologous virus (HIV-1 16936-2.21) and the level of viremia (ρ = .628; P < .001; Figure 3A). This effect was also seen when the analysis was carried out separately in chronically infected individuals (ρ = .618; P = .024; Figure 3B) and LTNPs (ρ = .785; P < .007; Figure 3C). There was no observed association between NAb response and CD4 counts.
Correlation between Neutralizing Antibody Titer and Plasma HIV-1 RNA Levels for Different Molecularly Cloned Envelope Pseudouviruses.


A, Relationship between HIV-1 RNA levels and the neutralization titers against heterologous virus (HIV-1 16936-2.21) with respect to all the study participants. B, Relationship between HIV-1 RNA levels and the neutralization titers against heterologous virus (HIV-1 16936-2.21) with respect to chronically infected individuals. C, Relationship between HIV-1 RNA levels and the neutralization titers against heterologous virus (HIV-1 16936-2.21) with respect to long-term nonprogressors (LTNPs).
Discussion
There is no information available about the neutralizing properties of serum from HIV-1-infected individuals from South Indian patients. Detection of magnitude and breadth of NAbs among distinct geographical population is important in the discovery of possible targets for vaccine design. This is the first cross-sectional study measuring NAb response among patients from the tertiary referral center in Chennai. These samples are representative of the South Indian HIV-1-infected population, as these are collected from a tertiary referral center, in Chennai, India, where patients from surrounding states are being referred to and taken under care for the management of HIV-1 disease. A considerable higher neutralization breadth was observed against Indian and African subtype C tier 2 viruses, suggesting the ability of cross-neutralization; this could be due to the sharing of neutralization determinants among these individuals. The results of these selected specimens are depicted in Figure 1; however, results from all the specimens were considered (i) to compare the magnitude of neutralization response between chronic individuals and individuals with LTNP, (ii) to compare the magnitude of NAb response between pseudoviruses of Indian and African origin, and (iii) to compare the NAb response between African subtype C and African nonsubtype C viruses. Also, a significant and higher neutralization responses were seen among chronically infected individuals, suggesting that there is a consistent increase in NAbs over a period of time from the time of infection and shows their ability in controlling HIV-1 disease progression in the absence of ARV drugs; however, this response was declining in individuals with HIV-1 infection for more than 10 years (LTNPs). This is consistent with the findings in Mombasa, Kenya, 15 that NAb response is higher in early infection with high viral load set point and higher HIV-1 env diversity and may decline with disease progression. Hence, it is evident that NAbs are controlling disease progression for about 5 to 10 years. The study also observed that many of the LTNPs (with low NAb response) had started on ARV drugs (data not shown) in recent days; this could be due to the fact that these NAbs are losing potency in controlling viral replication and disease progression. The reason for waning antibody response may be due to the increasing genetic variation and the losses of proliferative CD4 count over a period of time. The potency of neutralization antibodies in controlling disease progression for several years is consistent with previous reports from California, 26 Paris, 14 Shenyang, 27 and Durham. 28 This finding is crucial and interesting as most of the Indian population lives under the poverty line, and it would be hard for them to obtain and take drugs even if they were available at subsidized rates and/or free of cost 29 in the country because of other extraneous factors such as the cost of traveling to an HIV care center and compromising wages for that day. Many people may not be sharing their infection status with their family members or with their neighbors, since it would be difficult for them to take medication without the knowledge of their family members, which can lead to compromised adherence. In every aspect, it is highly essential to prolong the time of disease from the time of infection up to the day of antiretroviral treatment (ART) initiation. Because of these factors and with the added burden of primary and secondary drug resistance issues, the control of disease progression by NAbs is highly critical and essential for these individuals to control HIV disease without ART for a maximum number of years.
There was a negative association found between the neutralization breadth and viral load levels of the study specimens, supporting previous findings from Germany, 30 San Diego, 31 and Mumbai 2 specimens. However, positive association with 1 specimen was found in agreement with the studies from Senegal 18 and Mombasa. 15 This could be due to the high viral load set point and HIV-1 env diversity as discussed earlier 15 and the factors such as continued viral replication. Increased exposure of specific epitopes in the presence of high viral load may also be responsible for higher production of specific neutralization antibodies.
The HIV-1 viral load dynamics and envelope diversity must also be studied from this particular specimen. This observation is in contrast with the majority of our specimens where the ID50 titer and viral load levels are negatively associated. Hence it is difficult to interpret the association between viral load and neutralizing response in these asymptomatic patients, since usually the viral replication in expected to be inversely correlated with NAb heterologous response. 30,31 We found higher neutralization response even against subtype “C” viruses from African origin, which is consistent with a previous Indian report 1 that demonstrated that there was higher neutralizing sensitivity to subtype “C” viruses from Africa when compared to non-“C” HIV-1 subtypes of African and non-African countries. This could be attributed to sharing of neutralizing determinants and antibody specificities against conserved regions in subtype “C” strains. This information is vital in choosing the epitopes from subtype “C” strains from different parts of the world to prepare effective immunogen in vaccine design. Additional neutralizing magnitude and breadth must be studied with envelopes from different subtypes to find out the complete cross neutralizing ability of these specimens.
Even though this study gives a snapshot of the current neutralizing ability of HIV-1-infected individuals in south India, this deserves a comment on its limitations. We were unable to obtain specimens from recently infected individuals to study NAbs response. This subset of individuals would be more essential as during the initial course of infection there will be the monoclonal NAb response in the absence of mutants, quasi-species as the mAbs have shown protection of macaques against HIV-1 viruses, 32 which is highly needed for vaccine design. We were unable to obtain tier 3 viruses and hence could not measure the responses against them. Even though there is a considerable higher neutralizing response against tier 1 viruses (eg, MW965 and SF162), similar neutralization titers were not observed for other tier 1 viruses. Also, in some specimens very low neutralization response (including Ch3 as shown in Figure 1) was observed. The NAbs seen in chronically infected individuals have to be further studied over a period of time (at least after 3 years) and the responses must be assessed, as it would be useful to know the neutralizing response and relevant clinical markers in the natural history of HIV-1 disease. In accordance with the previous studies, 15,18 we did not observe any association between CD4 counts and ID50 titers. While this demonstrates that there is no clinical relevance between CD4 count response and NAbs, however, the in-depth studies on the T-cell responses and their relevant humoral immune expression need further exploration. Another major limitation of this cross-sectional study is that the data about autologous responses from these individuals are lacking. It is crucial to see the exact in vivo neutralizing ability, viral evolution, and the difference with heterologous responses. Sequencing of envelopes from these specimens is required and must be studied with envelopes from western parts of the country to find out genetic similarity that would eventually give a clear picture about the different neutralizing determinants available in the country. There was comparatively less NAb response in LTNPs than chronically infected individuals, most likely due to reasonably higher levels of cytolytic T cells and Th1 response, restriction of cell-associated viruses in LTNPs, and a relatively similar number of viruses in the extracellular compartment to elicit a neutralizing response. It is also ideal to study the levels of cytolytic T cells in these LTNPs.
To conclude, high titers of HIV-1 cross neutralizing activity were observed in sera from a subset of HIV-1-infected chronic and LTNPs patients, suggesting a role of these responses in the durable control of HIV-1 replication among the HIV-1 infected individuals in Chennai. These responses were associated with lower plasma HIV-1 RNA levels. Additional studies concerning the epitopes recognized by these antibodies may facilitate the discovery of an effective vaccine immunogen for India and other parts of the world. More robust analysis of NAb response with autologous and heterologous viruses with additional specimens including recently infected individuals is required to study the NAb response during the initial course of HIV-1 infection, which will be helpful in finding out neutralization-specific targets for use in vaccine immunogen.
Footnotes
Acknowledgments
We would like to thank Dr David C. Montefiori and his laboratory team for providing an opportunity to obtain training in NAb assay and pseudovirus preparation at Duke University Medical Centre, Durham, North Carolina. We would like to express our gratitude to Hagen von Briesen, Fraunhofer Institut für Biomedizinische Technik IBMT, Sulzbach, Germany, for providing pseudovirus stocks. We also would like to thank the physicians and clinical staff of YRG CARE for helping in participant recruitment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Bill & Melinda Gates Foundation's Collaboration for AIDS Vaccine Discovery/Comprehensive Antibody—Vaccine Immune Monitoring Consortium, Grant number 38619.
