Abstract
We describe correlates of reduced antituberculous serum drug concentrations (SDCs) in HIV-infected patients receiving treatment for active tuberculosis (TB). Cross-sectional analysis of individuals diagnosed with HIV and active TB in Northern Alberta, Canada, was performed. Of the 30 HIV-TB cases, 27 underwent measurement of SDCs. Rates of low SDCs were 9 of 26 (34%) for isoniazid (INH) and 16 of 25 (64%) for rifamycins. Increased weight and elevated body mass index (BMI) correlated with low SDCs for rifampin (P < .05) and increased weight also correlated with reduced SDCs for INH (P < .05). This suggests that conventional antituberculous dosing may be too low and consideration should be given to increase the maximum initial weight-based doses in HIV-infected patients.
Introduction
Patients with TB-HIV coinfection are thought to experience higher rates of treatment failure, relapse, and acquired drug resistance. 1 -6 A complication in TB treatment unique to HIV-coinfected patients is acquired rifamycin monoresistance. 1 -4 Pharmacokinetic data shows an association between acquired rifamycin resistance and low serum drug concentrations (SDCs) observed with intermittent rifamycin dosing. 5,6 HIV infection itself, through involvement of gastrointestinal lymphoid tissue, may lead to malabsorption of oral medications. 1
Routine therapeutic drug monitoring (TDM) of isoniazid (INH) and the rifamycins (rifampin [RIF) and rifabutin [RFB]) became standard practice in the management of individuals with HIV-TB coinfection, in Northern Alberta, Canada, in 2002. The purpose of our study was to estimate the frequency of reduced antituberculous SDCs in HIV-infected patients receiving treatment of active TB and to identify clinical correlates of low SDCs.
Methods
We performed a cross-sectional analysis of all individuals diagnosed with both HIV and active TB, in Northern Alberta from 1998 to 2009. This study received ethical approval from the University of Alberta Health Ethics Research Board. Patients were included if antituberculous SDCs were obtained for either INH or a rifamycin. Demographic and clinical characteristics were obtained from retrospective chart review.
The SDCs for INH and rifamycins were performed in HIV-coinfected individuals during the first month of treatment. After administration of standard daily doses of antimycobacterials, blood samples were collected in vacutainers at 1, 2, and 3 hours for INH, RIF, and RFB, respectively, and at 6 hours (for delayed absorption) for all. Samples were transported to the laboratory of Dr Charles Peloquin (Infectious Disease Pharmacokinetics Laboratory at the University of Florida, Gainsville, Florida; previously Denver, Colorado) for standardized high-performance liquid chromatography. Clinical interpretation of SDCs was provided based on peak concentration (Cmax), with usual Cmax breakpoints of 3 to 6 μg/mL for INH at 1 to 2 hours, 8 to 24 μg/mL for RIF at 2 hours, and 0.3 to 0.9 μg/mL for RFB at 3 to 4 hours. 7 Adequate peak SDCs were defined based on the SDCs achieved after the standard daily dosing of TB drugs in fasting, healthy volunteers. 7
Correlates of low Cmax were assessed using chi-square and Fischer exact test for categorical outcomes and using Student t test (2-tailed) for continuous outcomes. Regression analysis was also used for multivariate analysis. Statistical analysis was done using Stata/IC 10.0 software for Macintosh (College Station, Texas).
Results
Between 1998 and 2009, there were 30 cases of HIV-TB coinfection. Clinical and demographic features are shown in Tables 1 and 2. Of these cases, 28 patients achieved TB cure while 2 patients died secondary to TB before completing treatment. Cause of death in these 2 patients was deemed to be secondary to HIV-related complications with TB as a contributing factor. Twenty-seven patients underwent measurement of SDCs (26 INH, 15 RIF, and 10 RFB). Rates of low Cmax were 9 (35%) of 26 for INH and 16 (64%) of 25 for rifamycins (9 [60%] of 15 for RIF and 7 [70%] of 10 for RFB). A low SDC for at least one drug was documented in 20 (74%) of 27 patients.
Clinical and Demographic Characteristics of HIV-TB Patients with Reduced Serum Drug Concentrations of INH in Northern Alberta
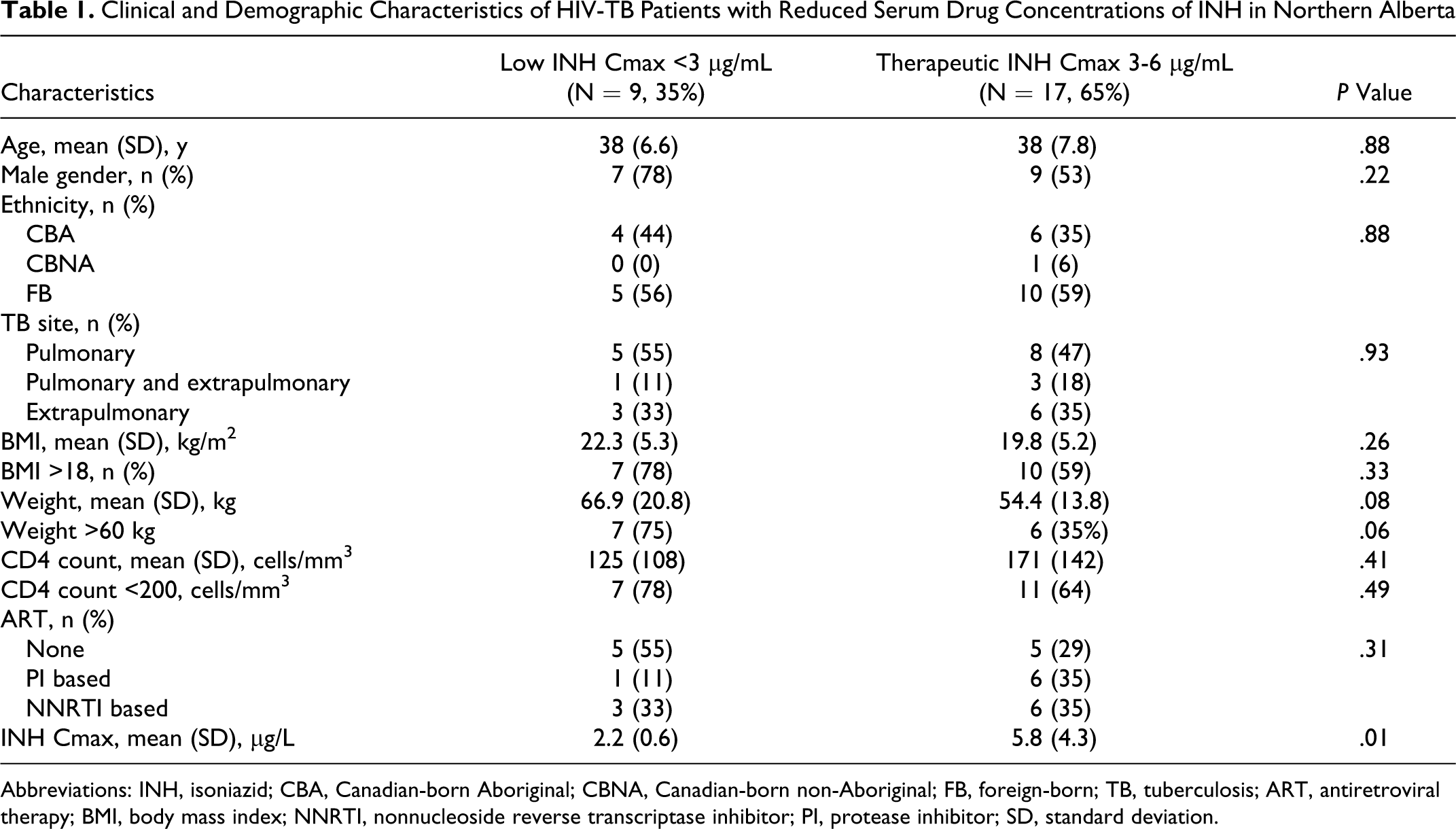
Abbreviations: INH, isoniazid; CBA, Canadian-born Aboriginal; CBNA, Canadian-born non-Aboriginal; FB, foreign-born; TB, tuberculosis; ART, antiretroviral therapy; BMI, body mass index; NNRTI, nonnucleoside reverse transcriptase inhibitor; PI, protease inhibitor; SD, standard deviation.
Clinical Characteristics of Patients with HIV-TB with Reduced Serum Drug Concentrations of RIF or RFB in Northern Alberta

Abbreviations: RIF, rifampin; RFB, rifabutin; CBA, Canadian-born Aboriginal; CBNA, Canadian-born Non-Aboriginal; FB, foreign-born; ART, antiretroviral therapy; BMI, body mass index; NNRTI, nonnucleoside reverse transcriptase inhibitor; PI, protease inhibitor; SD, standard deviation.
Clinical characteristics of patients with HIV-TB coinfection with reduced SDCs are shown in Tables 1 and 2. Although patients with reduced drug absorption were more likely to have advanced HIV infection indicated by low CD4 counts (mean 125 cells/mm3 and 155 cells/mm3 for INH and RIF, respectively) and not be on antiretroviral therapy ([ART], 55% and 44% for INH and RIF, respectively), this did not achieve statistical significance. Increased weight correlated with low SDCs for INH (mean weight 66.9 kg) and RIF (mean weight 69.6 kg; P < .05) and with body mass index (BMI) >18 for RIF (P < .05). There was a nonstatistically significant trend between weight >60 kg and low SDCs for INH and RIF. Additional factors including hepatitis C seropositivity, serum albumin level, and history of heavy alcohol abuse did not correlate with reduced SDCs although the sample size was small.
Four patients experienced elevated SDCs, 3 of which had INH SDCs greater than 10 μg/mL and 1 patient had an RIF SDC greater than 11 μg/mL. In these cases, clinical toxicity was not documented.
Discussion
Inadequate absorption of first-line TB drugs, including INH and/or a rifamycin, is common in patients with HIV-TB coinfection and was detected in nearly three-quarters of our population. Our findings are supported by previous studies describing an association between reduced antituberculous SDCs and HIV. 8 –11 In a cohort of patients with HIV-TB coinfection, in Botswana, 30% to 37% of patients had reduced INH serum concentrations and 78% to 84% had reduced serum RIF concentrations. 8 In another predominantly HIV-TB cohort from Botswanna, low INH and RIF concentrations occurred in 37% and 84%, respectively. 9 In the latter study, authors found poorer TB treatment outcomes associated with reduced pyrazinamide concentrations but not with reduced INH or RIF concentrations.
Low serum concentrations of anti-TB drugs have been associated with treatment failure, relapse, and acquired drug resistance in HIV-infected and noninfected TB patients, and adjustments of drug doses after TDM have been related to clinical improvements. 5,6,10,12 –15 These findings support the need to monitor anti-TB drug SDCs in this population to facilitate the early detection of a possible therapeutic failure. There has been concern that patients with advanced HIV infection may have more extensive malabsorption as the basis for reduced SDCs. Our finding that CD4 count and concomitant use of ART were not associated with reduced anti-TB drug SDCs is reassuring and has been documented in other HIV-TB-coinfected populations. 8,9
A novel finding of this study is that patients with increased weight and elevated BMI were more likely to experience low SDCs. This is in contrast to the findings in another study of patients with HIV-TB coinfection in which low weight corresponded to reduced SDCs. 8 One possible explanation for this discrepancy is the latter study used fixed dose combinations (FDCs), while our study used standard dosing of INH 300 mg/d and RIF 600 mg/d. At lower weights, the population receiving FDCs would have only received dosages of INH 150 mg/d and RIF 300 mg/d for weight of 30 to 37 kg, INH 225 mg/d and 450 mg/d for weight of 38 to 54 kg, INH 300 mg/d and RIF 450 mg/d for weight of 55 to 70 kg and thus were likely underdosed in comparison to patients in our cohort. At higher weights of >70 kg, the population receiving FDCs would have received INH 375 mg/d and RIF 750 mg/d and thus the dosage would have been higher than that used in our cohort. Thus, our data actually support the finding that FDCs for lower weight bands are suboptimal and were likely underdosed.
It has previously been suggested that rifamycin dosing in particular may be suboptimal and that efficacy of higher dosing regimens needs further evaluation. 16,17 We further speculate, based on the findings of our exploratory analysis, that recommended initial doses are too low for patients with HIV-TB coinfection weighing greater than 60 kg. Although the clinical significance of low concentrations warrants further study, it is prudent to optimize the drug dosages as guided by TDM. It has been suggested that median doses based on TDM required to achieve normal concentrations in an HIV-TB cohort were INH 600 mg/d (range, 300-1500 mg/d), RIF 1050 mg/d (range, 600-1200 mg/d), and RFB 300 mg (range, 150-450 mg) 3 times per week. 9 We recommend increasing the starting dose as follows: INH 5 mg/kg up to a maximum of 450 mg/d (rather than 300 mg/d); RIF 10 mg/kg up to a maximum of 750 mg (rather than 600 mg); RFB 450 mg/d (rather than 300 mg/d). Therapeutic drug monitoring would also be useful to monitor whether further dose adjustments are required. In the case of RFB, increased dosing may be especially important within the context of coadministration of a ritonavir (RTV)-boosted protease inhibitor-based ART regimen as this interaction may not be as pronounced as initially anticipated. 18 Instead, an RFB dose ranging from 300 mg thrice weekly up to 450 mg 5 times weekly (guided by TDM) may be needed to achieve the Cmax and area under the curve (AUC) values associated with clinical and microbiological success. 18
Limited literature exists regarding the safety of higher antituberculous dosing; however, preliminary data suggest that such dosage adjustments are possible without affecting the incidence of serious adverse effects. 9,19 Further study is needed to confirm that higher antimycobacterial doses would achieve adequate SDCs, that relapse and acquired-drug resistance would be reduced, and that higher rates of drug toxicity would not result. As well, ensuring timely turn around time of less than 1 week for SDC results within the laboratory would be imperative in operationalizing higher dosing regimens.
Although our findings are of clinical interest, several limitations exist. First, the small sample size reduced our statistical power. However, given the low incidence of HIV-TB coinfection in our population, it may not be feasible to generate a larger sample size in a reasonable time frame. Second, the timing of drug administration and food intake, which may have affected drug pharmacokinetics, was not documented. Third, we were not able to measure the impact of reduced SDCs on clinical outcomes as drug dosages were generally increased within the first 2 months of TB treatment due to low SDCs. Finally, we could not assess whether dosage adjustments achieved normal SDCs as follow-up measurements were not routinely obtained.
Conclusion
In summary, HIV-infected patients receiving treatment for TB experience reduced antituberculous drug SDCs of TB drug. Further study is required to assess the role of higher initial doses of TB drugs in this population.
Footnotes
Acknowledgments
The authors would like to acknowledge the nurses and staff of the Edmonton TB Clinic, Northern Alberta Program, and the TB isolation unit at the University of Alberta Hospital. We would also like to acknowledge the work of the Level III Provincial Laboratory staff as well as the significant contribution of Dr Sheela Vijay.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
