Abstract
Introduction
It is now widely appreciated that adherence to antiretroviral therapy (ART) is a critical determinant of HIV treatment outcomes. Indeed, adherence to ART has been shown to be an important predictor of achieving adequate suppression of HIV replication,1–6 which is required to minimize the emergence of HIV drug resistance (DR), 7 slow disease progression, 8 and delay death.9–11
More recently, the importance of ART adherence was magnified in pre-exposure HIV prophylaxis (PrEP) trials, documenting that highly adherent patients had a higher likelihood of not acquiring HIV infection.12,13 Similar findings with respect to both test-and-treat and treatment as prevention (TasP) indicate the Achilles’ heel of any antiretroviral-based intervention—whether therapeutic or preventative—is suboptimal adherence.14,15 Such treatment and prevention strategies are critical to augment the ongoing scale-up of access to antiretroviral-based interventions around the world, which is a major global challenge in the fight against HIV/AIDS. Successful scale-up depends upon many factors, including political will and financial resources, as well as local infrastructure, and health care capacity. These factors are important for delivering services and for promoting, monitoring, and enhancing adherence.16,17 Furthermore, required aspects of infrastructure include not only mechanisms to obtain and dispense drugs but also to educate patients and health care providers (HCPs) about how to manage adverse effects, maintain adherence, and implement lifestyle modifications to improve treatment and prevention outcomes. 18
Only limited data are available on a global scale about patients' health literacy and patient–provider communication related to HIV treatment adherence and HIV DR. The availability of such information would enable development and implementation of simplified, standardized treatment adherence and monitoring guidelines to be used by HCPs in ART programs worldwide, 19 including in developed world settings, as appropriate. To fill this gap, we conducted the AIDS Treatment for Life International Survey (ATLIS) in 2010, with the aim of taking a worldwide snapshot of patients' reported ART adherence levels, patient health literacy, and HCP-patient communications on issues related to ART adherence.
Methods
Study Design, Sampling, Enrollment, and Settings
ATLIS was a multicountry, comparative, cross-sectional survey of peoples living with HIV/AIDS from 5 global regions: North America (United States), Latin America (Brazil), Europe (France, Germany, Italy, Russia, Spain, and the United Kingdom), Asia Pacific (Australia and Korea), and Africa (Côte d’Ivoire and South Africa). The project was initiated and coordinated by the International Association of Physicians in AIDS Care (IAPAC) and governed by an ATLIS Task Force composed of selected HIV/AIDS expert leaders from 5 continents. The ATLIS survey fieldwork was conducted from January to March 2010 by Kantar Health, an independent market research and global consultancy organization.
A standardized approach to participant recruitment, sampling frame, and recruitment setting was complemented by country-specific adaptations to account for local circumstances. Generally, potential participants were recruited via phone or in person through a proprietary database, community-based organizations and HIV/AIDS support groups, and health care facilities. Study eligibility included the age of 18 years or older; diagnosed with HIV or AIDS by an HCP; and being on ART in the past 5 years. Those who were eligible and provided written consent were interviewed for about 40 minutes, through 1 of the 3 approaches: (1) an online self-administered questionnaire; (2) a telephonic interviewer-administered questionnaire; or (3) a face-to-face interviewer-administered questionnaire. All interviews were conducted by an in-country trained interviewer in the participant’s preferred language. Face-to-face interviews took place in a location of the participant’s choice. Monetary incentives were offered to respondents for completion of interviews where customary and varied by country, depending on local practice. The ATLIS protocol, questionnaires, and patient recruitment materials were reviewed and approved by Essex Institutional Review Board, Lebanon, New Jersey, as well as by the research ethics review board in each of the other countries involved.
Survey Questionnaire
The standardized survey was adapted from the self-report adherence to antiretroviral medications instrument utilized by the Adult AIDS Clinical Trials Group (AACTG) in HIV clinical trials, and included a 30-day recall question of missing 1 or more tablets. 19
Statistical Methods
Data were entered in the study database via the Internet directly following the interviews. Data analyses were performed using STATA Release 10 (Stata Corporation, College Station, Texas). The main statistical analyses included proportions which were compared for statistical significance, where applicable, using chi-square test. Sample sizes varied by country (100-200) based on the overall national HIV/AIDS prevalence. For any independent variable, with at least 100 study participants evenly divided into 2 categories, there is 80% power to detect a difference of 20% when comparing 2 prevalence rates, with a type 1 two-sided error of 5%. All P values reported are nominal and 2-tailed. A P value of less than or equal to .05 was considered statistically significant.
Results
Sociodemographic Characteristics and ART Use
Of the 2035 participants, 63% were men and 37% were women. In the African region, 67% of participants were women, which was significantly higher than in any other region (P < .05). Overall, 40%, 53%, and 6% of participants were aged 18 to 39 years, 40 to 60 years, and >60 years, respectively. Respondents from Africa compared to North America were more likely to be <40 years old (63% vs 16%; P < .05). Thirty-seven percent of respondents have been living with HIV or AIDS for less than 5 years, while a similar proportion has been living with the disease for more than 10 years. Asia Pacific (Korea and Australia) and Africa participants had a higher proportion (49%, 36%, and 71%, respectively) of recent HIV diagnosis versus North America (11%), P < .05 for each.
Adherence Levels and Reasons for Suboptimal Adherence
Overall, 57% of respondents reported never having missed a dose of their HIV treatment in the past month, with highest levels of complete adherence in Latin America (89%) and Africa (73%) and lowest levels in North America (45%; P < .05 comparing Africa and Brazil to any other region). Country-specific adherence data showed that the proportion of participants who reported not missing a dose of their HIV treatment in the past month was highest in Brazil (89%) and South Africa (83%) and lowest in France (34%) (P < .05). Adherence was inversely related to the duration of HIV, with 49% of participants living with HIV for more than 15 years, missing no doses in the past month compared with 73%, 61%, and 57% of those who had been living with HIV for <1 year, 1 to 5 years, and 6 to 10 years, respectively (P < .05, for each comparison). Overall (74%) and across all regions, respondents reported that the most common reason for missed doses was forgetfulness. Running out of medication was more commonly mentioned in Côte d’Ivoire (25%) compared to France (16%), United States (13%), Germany (7%), Italy (10%), or Korea (1%; P < .05 for all comparisons, except United States). In addition, not refilling the prescription was more common in Africa (15%) and North America (12%) compared to Europe (6%) and Asia Pacific (2%; P < .05).
Knowledge about the Link between Suboptimal Adherence and HIV DR
Thirty-nine percent of responders from Korea mistakenly reported that there was no consequence of suboptimal ART adherence (P < .05 compared to all other countries); a substantial proportion of responders from Côte d’Ivoire (19%) and Brazil (18%) also were unaware of the consequences of suboptimal adherence to HIV regimens. Only 62% of respondents in Africa reported knowing what HIV DR is compared to significantly more in North America (82%), Europe (81%), Asia Pacific (90%), and Latin America (74%; P < .05 for each comparison). Overall, 18% of responders mistakenly identified HIV DR as a “good thing”: 35% in North America, 24% in Africa, 16% in Latin America, 15% in Europe, and 14% in Asia Pacific (P < .05 comparing North America Africa to Europe-Asia Pacific).
Patient-HCP Communication about Optimal Adherence and HIV DR
While the majority of participants (87%) agreed that their HCPs convey the importance of ART adherence, only 71% reported receiving practical recommendations from their HCPs to maintain optimal adherence (62% in North America vs 80% in Latin America or Africa; P < .05; Figure 1). Higher ART adherence rates were reported by participants who indicated they had good discussions with their HCPs about adherence issues versus those who had not (57% vs 50%; P > .05). These participants also had a better understanding of the fact that suboptimal adherence can result in medications losing their effectiveness (55% vs 36%; P < .05) and were more likely to understand the meaning of HIV DR and its detrimental health implications (83% vs 72%; P < .05).
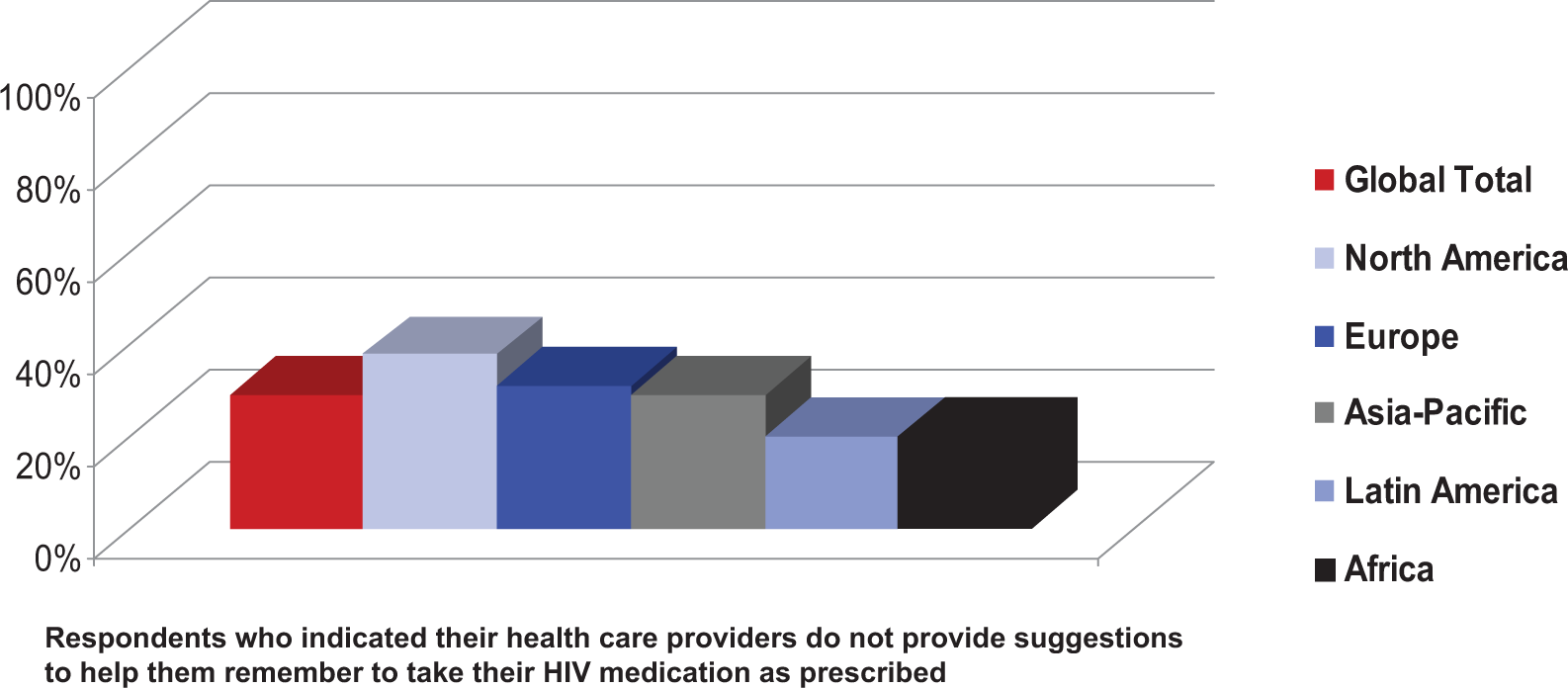
Gaps in communication between health care providers and patients about antiretroviral therapy adherence.
Discussion
The survey results show that globally nearly half (43%) of patients admitted to missing at least 1 dose of their ART regimen in the past month, suggesting that they may not fully understand the impact on their health. Of concern, respondents in Korea, the United Kingdom, Brazil, and Côte d'Ivoire believed that there are no consequences to missing medication doses. We found a striking heterogeneity of ART adherence by regions, with optimal self-reported adherence highest in Latin America (89%) and Africa (73%) and lowest in North America (45%). These data are in agreement with a meta-analysis by Mills and colleagues who reported pooled levels of adherence as measured in a variety of ways (pill counts, pharmacy refill data, medication event monitoring system, and self-report) to be on average; 77% of African patients met the standard definition 20 of good ART adherence (>80%). Similar to a few reported studies in Africa,21,22 our data also demonstrate a dramatic inverse duration-adherence effect and provide results for the longest duration (>10 years) on HIV treatment to date (73% excellent adherence for participants living with HIV for <1 year, vs 61% for those with HIV for 1 to 5 years, vs 49% for those with HIV for >15 years; P < .01 all comparisons).
Our data are in conflict with those reported from a South African study showing ART adherence levels as assessed by pill count to be sustained at a median of about 95% at all time points during a 2-year follow-up. 23 However, most previous studies reporting high levels of ART adherence in resource-limited settings were carried out in a research setting in which eligibility criteria (such as HIV status disclosure to at least 1 other individual), research interventions (such as social support, psychosocial counseling, and the opportunity to join peer support groups), and material support (such as pillboxes, drug identification charts, and daily schedules) could positively influence adherence. Our survey, in contrast, was more likely to determine natural, “real-life” adherence levels.
In all regions, participants reported that the most common reason for missing ART doses was forgetfulness (74%). These data are in agreement with a systematic review by Mills and colleagues, which showed that prevalent factors associated with suboptimal treatment adherence in both resource-rich and -limited settings included individual level adherence barriers such as untreated depression, active substance abuse, poor insight into disease and treatment, being an adolescent or young adult, and forgetfulness.24,25 Forgetfulness as a barrier suggests that patients could use more support, tools, and tips for remembering to take their medications as prescribed. Furthermore, the fact that in the present study participants mainly from middle- and low-income countries (Brazil and Côte d’Ivoire) are failing to take their medications because they simply ran out or did not refill their prescription suggests a need for better structural support of the pharmacy system in the ART supply chain. This barrier has also been identified in previous studies and has underscored the importance of local infrastructure, including transportation, in ensuring ART adherence during worldwide scale-up. Our data and that from other groups underscore the critical need to address structural barriers related to poverty, including HIV treatment cost (in settings where ART is not free of charge), food insecurity, and lack of transport money to attend the clinic, as well as health care facility–related factors such as pharmacy stock outs.26–31 On the other hand, previously reported social-related barriers to ART adherence such as HIV status nondisclosure and stigma32–36 were reported among our study participants in all regions.
Interestingly, while participants overall seemed to grasp the idea that ART adherence has serious implications for their health and the effectiveness of their HIV medications, it is concerning that a substantial proportion of respondents in countries such as Korea, the United Kingdom, Brazil, and Côte d’Ivoire appear to be unaware of the consequences of suboptimal adherence or even believe there are none. It is important that patients understand HIV DR and when/why it occurs in order for them to fully realize the relationship of treatment adherence to outcome, and their own role in that dynamic. In our survey, patients who understand HIV DR were more likely to report high levels of adherence to their HIV medications. Eighty-seven percent of respondents reported that their HCP stressed the importance of adherence, but many reported that their HCPs did not provide them with practical tips to help maintain optimal adherence. Responders who agree strongly that their HCPs explained adherence in a way they could understand appear to be more cognizant of the need to take their HIV medications as prescribed every day. These data are in agreement with Bezreh and colleagues who did report frequent noncommunication about medication adherence issues, 37 as well as a Canadian study which showed that the probability of completely defining HIV DR increased from 15.8% to 63.9% if respondents had discussed their HIV regimen with both a physician and a pharmacist. 38 These data strongly indicate a need for more HCP-facilitated discussions about how to sustain high levels of ART adherence.
Our study has several important clinical and public health implications: (1) there is a critical and urgent need for expanding patient-HCP conversations about HIV treatment adherence and HIV DR; (2) practical, evidence-based interventions and tools should be provided to HCPs and their patients to increase and sustain ART adherence over time; (3) national and international efforts are needed to ensure that in resource-limited countries, structural barriers to ART adherence are overcome; (4) investing in monitoring and increasing ART adherence is likely to be cost saving, and even cost effective in the long term, and thus these efforts should be intensified; (5) multifaceted (to account for the range of stigmatizing conditions) and multilevel (to account for individual and structural levels of stigma and discrimination) approaches should be explored to fight HIV/AIDS stigma,39–43 approaches might include education (mass media such as television, radio, print, as well as social media), school education programs, self-help and support groups, HIV/AIDS organizations, government education programs and policies, education in local community (health fairs, etc), religious or spiritual center education programs, workplace education programs, and legal interventions; and (6) there is a need for targeted research to evaluate the effectiveness and cost-effectiveness of practical community-based interventions (to include patient-HCP communication/education) to increase and sustain ART adherence levels globally.
The primary strength of the ATLIS study is its global nature. To our knowledge, ATLIS is the first global snapshot of ART adherence and health literacy by self-report. Our study also adds to the literature another example of a dramatic decrease in ART adherence with duration of HIV infection, and it does so for the longest duration, that is for more than 10 years reported to date. Finally, these are among the few data showing a link between health literacy and suboptimal adherence to HIV medications. Our study also has possible limitations. Because of its cross-sectional nature, our study is subject to reverse causality which limits proper inference. In addition, our study’s ART adherence data were generated by self-report, which has been shown to tend toward an overestimation of adherence levels. However, to date there is no gold standard tool for measuring adherence, and each approach has advantages and disadvantages. Self-report is the most commonly used ART adherence measurement tool in resource-limited settings and can be useful beyond its utility in alerting providers to the need for adherence intervention. Despite the known social desirability of providing the “right answer,” self-reported adherence has good specificity, can be robust, and has been shown to correlate well with virologic suppression and other clinical outcomes, suggesting that it can be a valuable tool. 44
We conclude that varying levels of ART adherence across geographic regions and limitations in health literacy regarding the detrimental effects of suboptimal adherence require greater educational, behavioral, and clinical interventions to attain and maintain optimal adherence levels and thus to achieve and maintain successful treatment outcomes, including long-term virologic suppression.
Footnotes
Acknowledgments
The authors wish to thank the following organizations in each country that made possible the collection of data. South Africa: Beatit; Moretele Sunrise Hospice; Bophelong MES Impilo Hospice; Sicelinceba Health Organisation; Tswaragang; Hermanus Rainbow Trust; SAMAREC. Australia: NAPWA—National Association of People Living with HIV/AIDS; PLWHA—(Vic) Inc—People Living With HIV/AIDS (in Victoria); Queensland Positive People; Western Australian AIDS Council; PLWHA (SA)—People Living With HIV/AIDS (in South Australia); Positive Life (NSW) Inc; Positive Women's Network. Korea: KANOS HIV/AIDS Network of Solidarity; Love4one. Russia: Human Action (‘Gumanitarnoye Deystviye'); Regional Center for Prophylaxis and Treatment of AIDS and Infectious Diseases; Social and Psychological Center “Doveriye”; Center for Prophylaxis and Treatment of HIV of Sverdlovsk Region; Jasen. United Kingdom: Positively Women (local branches); Positive Action local branches; Terence Higgins Trust (local branches); AB Plus (local Branches); Body Positive (local branches). Côte d'Ivoire: Lumiere Action; Club Des Amis (Association Des Personnes Vivant Avec Le Vıh); Aıbef (Association Pour Le Bien Etre Familial); Le Mıssı; Cip Cames; Fsu Com; Chu De Yopougon; Hopital General Yopougon Attie (Port Bouet 2); Fsu Com Abobo Avocatier; Fsu Abobo Baoule; Hopital General D'Anyama; Centre plus de yopougon.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: ATLIS 2010 was funded by Merck & Co., Inc., Whitehouse Station, New Jersey, USA, which operates in many countries as Merck Sharp & Dohme. Merck & Co. had no input in the study design, data collection, data analysis, or presentation of the results reported in this manuscript.
