Abstract
We report a symptomatic case of HIV replication in CSF (Cerebro Spinal Fluid) despite low plasmatic viral load. The patient, known HIV positive since 3 years, under HAART (Highly Active Antiretroviral therapy), presented an acute meningoencephalitis which was improved by optimization of treatment for a better CNS (Central Nervous System) penetration according to the CHARTER's (CNS HIV Antiretroviral Therapy Effects Research) score. CNS can be considered as a specific compartment for HIV replication and can be involved in acute or chronic neurologic impairment. As showed in this observation, CHARTER score could be helpful to optimize HAART penetration in CNS, but indication and use remain uncertain in current practice.
Introduction
HIV is a neurotropic virus and HIV infection may be associated with neurological symptoms, 1 such as acute meningitis, myelitis, encephalitis, or AIDS-related dementia complex. 2,3 Before Highly active antiretroviral therapy (HAART), AIDS-related dementia complex was a common neurologic disease due to HIV replication in CNS. Since wide use of HAART for HIV-infected patients, occurrence of AIDS-related dementia complex has dramatically decreased and minor neurologic impairment with neurocognitive disorders has been now identified. The physiopathology of these neurocognitive symptoms remains unclear: the disorder can be due to inflammatory cascade effect due to HIV infection, drug toxicity, comorbidities, or persistent HIV replication in CNS. Antiretroviral drugs have different penetration in CNS, but the relationship with neurological symptoms is not well-known and could be a challenge for the physician in order to prevent neurological involvement. A penetration and efficacy scoring scale of antiretroviral drugs CHARTER (CNS HIV Antiretroviral Therapy Effects Research) score was recently published and updated in order to optimize clinical control of HIV infection when compartimentalized in CNS. 4, 5
We present a case of symptomatic HIV replication in the CNS despite successful reduction of plasmatic viral load under HAART, and usefulness of CHARTER’s score.
Case Report
A 38-year-old African woman was diagnosed with HIV-1 (subtype A1) infection in 2004. She had no opportunistic infection. CD4 count was 131 cells/mm 3 and plasmatic viral load (pVL) 135 000 copies/mL (5.13 log). HAART with zidovudine (ZDV), lamivudine (3TC), and indinavir/ritonavir (IDV/r; 400/100 mg × 2 per day) was introduced (CHARTER score 2010 = 10), with a rapidly good immune virological efficacy (CD4 cell count: 266 cells/mm 3 and undetectable pVL (<50 copies/mL) within 3 months. In November 2005, pVL was still undetectable and CD4 count raised to 413 cells/mm 3 .
In 2007, because of cutaneous pseudoretinoid syndrome and moderate renal failure (creatinine clearance = 50mL/min), IDV was switched to RTV boosted fos-amprenavir (100/700 × 2 per day) (CHARTER 2010 score=9).
One year later, the patient presented persistent headache, neck stiffness, fever, memory disorders, decreased consciousness, altered mental status, phono-photophobia, cerebellar ataxia, and dizziness. Usual plasma biological and hematological values were normal; cerebrospinal fluid (CSF) examination revealed mild pleiocytosis (74 cells/mm 3 with 94% lymphocytes) and albumin at 0.90 g/L. Magnetic resonance imaging (MRI) scan showed diffuse and huge brightness of the white matter on T2 weighted sequences and high apparent diffusion coefficient (ADC) values compatible with a leuco encephalopathy (Figure 1 ). Bacterial examination and PCR for CMV, EBV, HSV 1 and 2, VZV, enterovirus, and JC virus were negative, but HIV viral load in CSF was 12 400 copies/mL. Concomittent plasmatic VL was detected at 613 copies/mL and CD4 cells: 300 cells/mm 3 (21%). CSF HIV genotype revealed a new mutation 215 F on the reverse transcriptase gene, not present on plasma HIV genotype (other mutations present RT: 184I, PRO: 35D, 36I, 10V, 41K, 69K), effective (counting only the effective molecules) CHARTER 2010 score of this association was = 2.
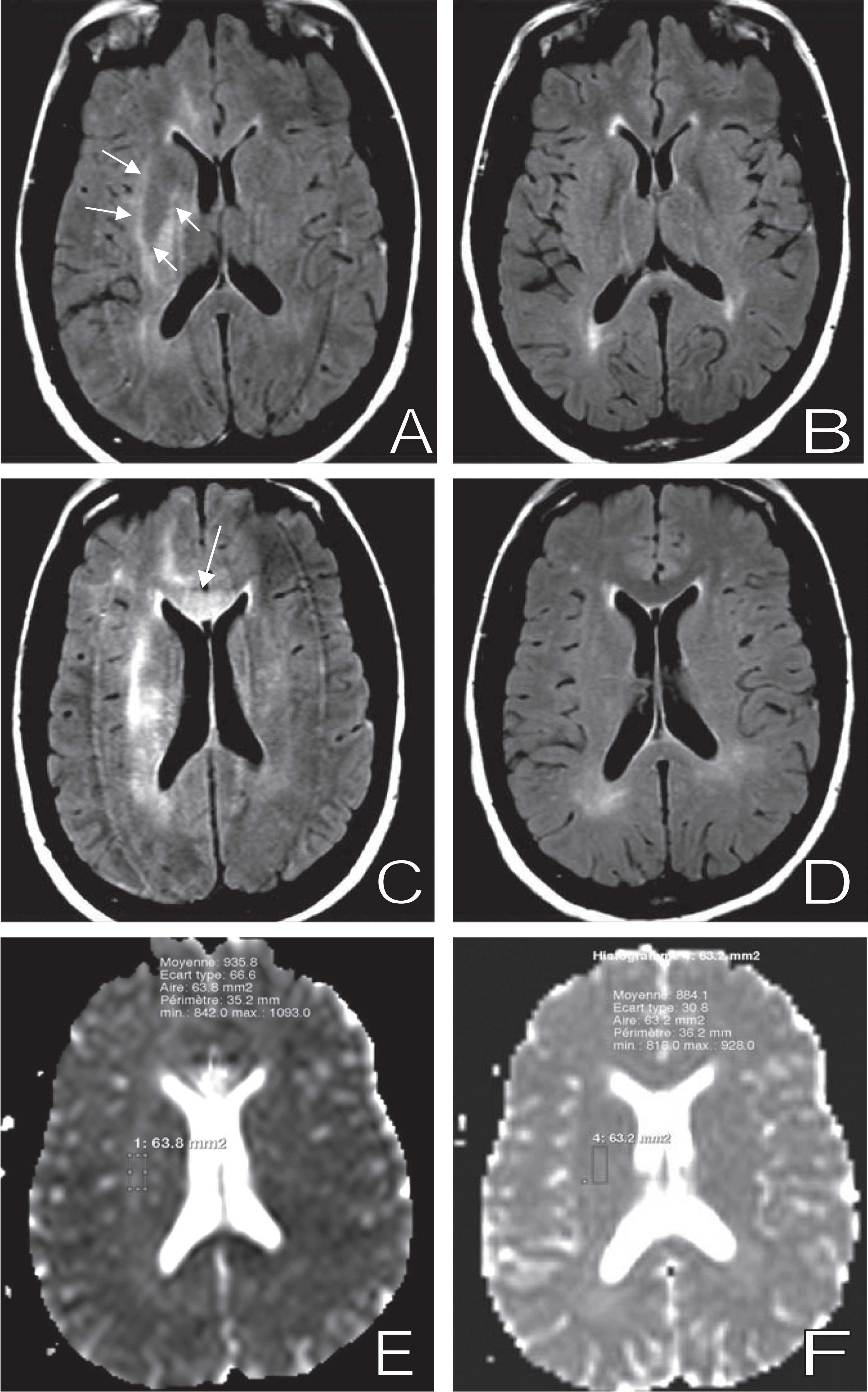
Initial magnetic resonance imaging (MRI) examination performed at time of meningoencephalitis and HIV cerebrospinal fluid (CSF) elevated viral load on the left column (A, C, E) and control after improvement on the right (B, D, F). On FLAIR images (A-D), there is a diffuse white matter bright signal on the initial examination (A-C) particularly visible into corpus callosum (white arrow) and internal and external capsules (double white arrows), which heavily decreased on the control MRI (B,D). On apparent diffusion coefficient (ADC) maps brightness and measured ADC into capsule decreased between initial (E) and follow up (F) examination.
The diagnosis of meningoencephalitis due to HIV 1 was assessed and antiretroviral therapy modified for abacavir (ABC), lamivudine (3TC), efavirenz (EFV), IDV/r to optimize the CNS penetration according to effective CHARTER 2010 score (=10).
The outcome was favorable with clinical improvement: apyrexia, disappearing of headache, and consciousness restoration. Intellectual tests performed 1 month later showed improvement with no major neurological impairment but still neurological slowing down and some perseverations. Learning, attention, and executives functions were moderately affected. Radiological evolution was also favorable on MRI with partial regression of white matter brightness and decrease of ADC values and virological control with undetectable plasmatic and CSF viral load maintained 2 years later.
Discussion
In this observation, HIV replication in the CSF was considered as the cause of meningoencephalitis for the following reasons: the CSF investigation performed in the presence of clinical symptoms showed active HIV-1 replication and meningeal inflammation, no other infectious etiology for these disorders have been found, and neuroloradiological evaluation showed leucoencephalopathy concordant with HIV encephalitis. Clinical presentation of neurological disorders due to CSF HIV replication varies from simple headache to coma. 6
One striking feature is the discordance between viral replication in CSF and its control in plasma probably due to the poor CNS penetration of some drugs used under HAART. This could be due to the poor compliance, underlined by the 184I mutation or poor penetration through the blood-brain and blood-CSF barriers resulting also in low drugs concentrations in the CSF. The ARV drug CSF concentration cannot be used as direct marker of efficacy because CSF penetration may not be equivalent to parenchymal penetration, drug levels may be different in CSF and brain extracellular fluid and brain parenchyma. 7
Furthermore, the finding of different profiles of resistance between plasma and CSF isolates support the hypothesis that CSF and plasma are virologically distinct compartments as previously suggested. 8,9 Compartmentalized HIV replication, despite successful suppression of plasmatic viremia, with development of CSF HIV resistance can result in acute or subacute neurological manifestations. 6,7,9 The efficacy of HAART on CSF HIV replication, regardless of penetration, is remarkable at introduction of treatment in drug-naive patients, but in few patients, persistent CNS low replication in this compartment occurs and can promote acute or chronical neurological disorders. 10
The CHARTER Cohort group defined a median CNS penetration effectiveness rank to characterize drug penetration in the CNS and the correlation with CSF HIV replication. 4,5 In the updated 2010 CHARTER score, the most considerable concentrations in CSF are observed with ZDV, nevirapine, and ritonavir/indinavir. But, in our case, the lower efficacy of ARV drug in CNS after switch from IND/r to fos-Amp/r was not reflected in the 2010 CHARTER’s score, despite rising clinical neurological symptoms. It could be due to the poor compliance to this drug or to the methodology of CHARTER’s score based on neurological efficacy in patients with variable level of HIV plasma replication, some of them naïve of therapy. Furthermore, we should take account of viral resistance to calculate an effective CHARTER score.
Indeed, CSF HIV replication could not always be explained by insufficient drug penetration in the CNS compartment. 6 A low CD4 nadir count suggests a longer duration of viral replication, a lower immune status, and, therefore a higher risk for the presence of HIV in the CNS, 11 other risk factors could be involved.
Finally, for this meningoencephalitis the modification of HAART selected to achieve better virological efficacy on CSF HIV RNA, according to CHARTER score, led to clinical improvement and subsequent long-term control of viral replication in both plasma and CSF.
We can hypothesize that controlled HIV replication in the CSF is one of the factors that could prevent neurocognitive decline. 12 It underlines the necessity of attaining complete control of HIV replication in the CSF as well as in the plasma. Furthermore, in the case of moderate virological failure, it could be useful to investigate the CSF compartment to look for residual replication and potential resistance. 12 Integration of virological and pharmacological characteristics could provide a better means to predict virological response and can be helpful in determining therapeutic strategies, especially for patients with neurological impairment. The reduced ability of some drugs to achieve adequate CSF concentrations and the resistance profile in the CSF may need to be considered, especially if CNS manifestations are present. Treatment decision for CNS manifestations may require knowledge of drug penetration and the susceptibility of HIV in CSF, but the optimal antiretroviral regimen for treating HIV infection in the cerebral compartment has not been yet defined despite CHARTER’s score. 11
Furthermore, a better CHARTER’s score is not necessarily associated with neurocognitive improvement. 13 Physicians should be aware of the possibility of HIV-related CNS disorders, even in the presence of minor neurological complaints that should prompt a CSF evaluation with the determination of viral replication and genotypic resistance testing.
There is a specific need for a better understanding of the dynamics of viral replication in the CNS compartments and its consequences on potential neurological or neurocognitive dysfunctions.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
