Abstract
The extracellular matrix (ECM) of bone and dentin contains several non-collagenous proteins. One category of non-collagenous protein is termed the SIBLING (
Introduction
Bone and dentin are mineralized tissues that closely resemble each other in composition and mechanism of formation. Both are composed of organic matrices and mineral contents of plate-like apatite crystals. They are formed when osteoblasts and odontoblasts secrete type I collagen-rich unmineralized extracellular matrices (ECM), termed osteoid and predentin, respectively, which are subsequently mineralized when apatite crystals are deposited (Figs. 1a, 1b). Formation of predentin and its transformation to dentin as mineralization takes place are highly controlled, orderly processes. The predentin width is rather uniform, indicating that the rates of synthesis of unmineralized matrix and its conversion to dentin at the predentin-dentin (PD) border are equal (Fig. 1b). These considerations indicate that several unspecified, controlling mechanisms must be involved. The importance of the correct collagen structure is clearly seen in patients with osteogenesis imperfecta caused by mutations in type I collagen gene (Cole, 2002). In addition to type I collagen, the ECM of bone and dentin contains several non-collagenous proteins (NCPs). These NCPs are believed actively to promote and control mineralization of collagen fibers and crystal growth within osteoid and predentin, when these tissues are converted to bone and dentin. This belief is strongly supported by the fact that mutations in their genes (Xu et al., 1998; Xiao et al., 2001; Zhang et al., 2001; Feng et al., 2002a; Sreenath et al., 2003) are associated with dramatic phenotypic abnormalities in the mineralization process of bone and dentin. One category of the NCPs is termed the SIBLING (Small Integrin-Binding LIgand, N-linked Glycoprotein) family that includes osteopontin (OPN), bone sialoprotein (BSP), dentin matrix protein 1 (DMP1), dentin sialophosphoprotein (DSPP), and matrix extracellular phosphoglycoprotein (MEPE) (Fisher et al., 2001). Although the known sequences for SIBLING proteins have little similarity except in limited regions of the molecules, they share the following common features (Fisher et al., 2001). (1) SIBLING proteins are principally found in bone and dentin and are secreted into the ECM during the formation and mineralization of these tissues; the expression of BSP, OPN, DMP1, and DSPP is qualitatively similar but quantitatively different between the two tissues (Qin et al., 2001a). (2) They display an Arg-Gly-Asp (RGD) motif that mediates cell attachment/signaling via its reaction with cell-surface integrins. (3) Their gene organizations are similar, with the last one or two exons being the largest and containing the RGD tripeptide. (4) Their genes share common localization on human chromosome 4q21-23. (5) They share similarities in post-translational modifications (PTMs) such as phosphorylation and glycosylation. Some biological functions of these polyanionic SIBLING proteins are dependent on the nature and extent of the PTMs, including phosphorylation, glycosylation, proteolytic processing, sulphation, and transglutaminase cross-linking.
This brief review will provide information relative to the post-translational modifications of SIBLING proteins and the roles of these PTMs in osteogenesis and dentinogenesis. We are focusing mainly on OPN, BSP, DMP1, and DSPP, since little is known about the PTMs of MEPE, the newest member of this family that was discovered recently by cDNA cloning (Rowe et al., 2000). The PTMs of OPN and BSP have been recently discussed and summarized in several excellent reviews (Butler et al., 1996; Ganss et al., 1999; Sodek et al., 2000). For comparative purposes and for inclusion of newer data, we have included in this review a brief summary concerning these two proteins. Information relative to sulphation and transglutaminase cross-linking of OPN and BSP is excluded in this review, since no newer data have been reported since the publication of two former reviews (Ganss et al., 1999; Sodek et al., 2000). The naturally occurring, full-length forms of DMP1 and DSPP have not been isolated or characterized; these proteins appear as proteolytically processed fragments in the ECM of bone and dentin, probably cleaved by the same enzyme (Qin et al., 2003a). We have included a detailed discussion about the proteolytic processing of these two SIBLING members because the data are relatively new, and because we believe that this process may represent an important step that plays a significant role in osteogenesis and dentinogenesis.
For clarity, this review on the PTMs of OPN, BSP, DMP1, and DSPP has been organized in the following manner. A short general introduction (A) for each of them is given, followed by presentation of data relative to each type of PTMs, i.e., (B) phosphorylation, (C) glycosylation, (D) proteolytic processing. Finally, some ideas about potential future research (E) with regard to PTMs of the SIBLING proteins are included.
The Table is a summary of the PTMs for OPN, BSP, DMP1, and DSPP. The data presented in this review are based mainly on results obtained from rats when available. If the data from rats were unavailable, findings are from other species, such as cows and humans.
(A) General Introduction
(1) OPN
OPN was first described as a secreted 60-kDa transformation-specific phosphoprotein (Senger et al., 1979). Subsequently, it was independently discovered by several groups with different approaches and in different fields (Butler, 1989). The name ‘osteopontin’ was introduced to reflect the potential of this protein in bone to serve as a bridge between cells and hydroxyapatite through RGD and polyaspartic acid motifs in its primary amino acid sequence discovered by cDNA cloning (Oldberg et al., 1986). The secreted rat OPN contains 301 amino acids. Although OPN is present in bone in relatively large quantities, it is also expressed in a variety of tissues and cells (Butler et al., 1996; Sodek et al., 2000). The broad expression of OPN indicates a multiplicity of functions in diverse biological events (Butler et al., 1996; Giachelli and Steitz, 2000; O’Regan and Berman, 2000; Sodek et al., 2000; Denhardt et al., 2001). Some in vitro studies have shown that OPN is an effective inhibitor of apatite formation and growth (Boskey et al., 1993; Hunter et al., 1994; Goldberg and Hunter, 1995; Wada et al., 1999). Data from OPN null mice strengthen the conclusion that this protein may be a major inhibitory factor on mineralization (Boskey et al., 2002). Although there is a lack of a clear phenotype in OPN knockout mice, several studies have demonstrated that OPN deficiency affects the functions of osteoclasts (Rittling et al., 1998; Rittling and Denhardt, 1999; Ishijima et al., 2001; Suzuki et al., 2002; Chellaiah et al., 2003), consistent with our recent findings that OPN is involved in regulation of osteoclast activity (Razzouk et al., 2002).
(2) BSP
BSP was initially isolated from bovine cortical bone (Herring, 1972) and subsequently characterized by Fisher et al.(1983, 1987). The primary sequence of BSP was first deduced from a rat cDNA sequence (Oldberg et al., 1988). The secreted rat BSP contains 303 amino acids. Unlike OPN, the tissue distribution of BSP is restricted, being found almost exclusively in mineralized tissues including bone, dentin, mineralizing cartilage, and cementum (Oldberg et al., 1988; Bianco et al., 1991; Somerman et al., 1991; Chen et al., 1993). The quantity of BSP in bone is similar to that in dentin (Qin et al., 2001a). However, the biological functions of BSP in mineralized tissues are not known. Analysis of some data suggests that BSP acts as a nucleator of the initial apatite crystals; then, as this mineral grows on the collagen matrix, it acts as an inhibitor in directing the growth of the crystals (Hunter and Goldberg, 1994). BSP may also contribute to the activity of osteoclasts by interacting with cell-surface integrins (Ross et al., 1993; Razzouk et al., 2002).
(3) DMP1
DMP1 was first discovered by cDNA cloning with the use of a rat odontoblast mRNA library (George et al., 1993). The full-length DMP1 secreted by the rat consists of 473 amino acids, containing an unusually large number of acidic domains, a property consistent with DMP1 being a possible participant in regulating matrix mineralization. The roles of DMP1 in mineralization are supported by observations that transgenic MC3T3-E1 cells overexpressing DMP1 show earlier onset of mineralization and produce mineralized nodules of significantly larger sizes when compared with the non-transgenic cells (Narayanan et al., 2001). More recently, Narayanan et al.(2003) showed that, in addition to its role in the regulation of mineral formation, DMP1 may act as a transcriptional component for activation of osteoblast-specific genes like osteocalcin. The conclusion that DMP1 plays an important role in mineralization of bone and dentin is further strengthened by the manifestations in DMP1 knockout mice. Although these DMP1-deficient mice appear normal at birth, delayed conversion of osteoid to bone and predentin to dentin occurs at three weeks, and this condition grows more severe by three months (Feng et al., 2002a). Since native DMP1 has not been isolated and characterized, properties resulting from its PTMs have not been tested. Recently, we obtained clear evidence suggesting that DMP1 is proteolytically processed into 37K and 57K fragments, present in the ECM of bone and dentin (Qin et al., 2003a), as discussed later. DMP1 was originally postulated to be dentin-specific, but later, its expression was observed in bone and brain (Hirst et al., 1997; MacDougall et al., 1998).
(4) DSPP
This precursor protein, predicted from the cloning of a cDNA (MacDougall et al., 1997; Gu et al., 2000; Ritchie et al., 2001; Yamakoshi et al., 2003), gives rise to two proteins, dentin sialoprotein (DSP) with coding sequences in the 5′ end and dentin phosphoprotein (DPP) representing the 3′ end of cDNA. The two proteins were independently identified as ECM components extracted from dentin when it is demineralized. DPP, discovered by Veis and his colleagues (Veis and Perry, 1967; Dickson et al., 1975; DiMuzio and Veis, 1978), is the most abundant NCP in the ECM of dentin. It contains large amounts of aspartic acid (Asp) and phosphoserine (Pse), largely present in repeating sequences of (Asp-Pse-Pse)n and (Asp-Pse)n. A sizeable data pool indicates that DPP is an important initiator and modulator of dentin apatite crystal formation (Linde et al., 1989; Boskey et al., 1990; George et al., 1996; Saito et al., 1997; Butler, 1998). DSP, discovered by Butler and his colleagues (Butler et al., 1981, 1992; Butler, 1987), is the second most abundant NCP in dentin ECM. The functions of DSP or a recently discovered isoform of DSP, HMW-DSP, that is present in dentin ECM (Qin et al., 2003b) are not known. The occurrence of one gene transcribing a single mRNA encoding DSP and DPP indicates that the translated product is proteolytically processed into DSP and DPP by unidentified proteinases. Analysis of recent data suggests that the proteolytic processing of rat DSPP to DSP and DPP may involve the same proteinase that cleaves DMP1 (Qin et al., 2003a; see below). The importance of DSPP in dentinogenesis is supported by experiments showing the association of mutations in the Dspp gene with dentinogenesis imperfecta in humans (Xiao et al., 2001; Zhang et al., 2001) and by defective mineralization of dentin in DSPP knockout mice (Sreenath et al., 2003). Originally, the expression of DSPP was thought to be tooth-specific, but recent studies have demonstrated its expression in bone and osteoblasts at a much lower level than in the tooth (Qin et al., 2002, 2003c).
(B) Phosphorylation
(1) OPN
Phosphorylation of OPN occurs mainly on serine residues and is principally catalyzed by casein kinases (Lasa et al., 1997). The amino acid sequence alignment from seven species indicates that OPN has a high level of serine conservation and that about half of the conserved serine residues are potential phosphorylation sites for casein kinases I and II (Butler et al., 1996). Rat bone OPN has an average of 12 phosphoserines and 1 phosphothreonine (Prince et al., 1987), with considerable heterogeneity (Neame and Butler, 1996), whereas 27 phosphorylated serines and one phosphorylated threonine have been identified in bovine milk OPN (Sorensen et al., 1995). Notably, different forms of OPN that vary in their phosphorylation have been reported in transformed osteogenic cells (Kasugai et al., 1991; Safran et al., 1998a). The level of phosphorylation of OPN was shown to be modulated by 1,25-dihydroxyvitamin D3 in a non-transcriptional mechanism (Safran et al., 1998a). The addition of this hormone to cultures of ROS17/2.8 cells lowers the level of OPN phosphorylation prior to transcriptional up-regulation by 1,25-dihydroxyvitamin D3.
Although data are available showing that phosphorylation of OPN is important for its function, the exact effect of phosphates on this protein is unknown. In vitro mineralization studies with a gelatin gel diffusion system used for the study of its effects on the formation and growth of hydroxyapatite have shown that OPN is a very effective inhibitor of these processes, but this activity was lost after removal of phosphate groups (Boskey et al., 1993). Likewise, the phosphorylation of OPN has been shown to be necessary for the inhibitory effect of this protein on smooth-muscle-cell calcification, since native (phosphorylated) OPN inhibits calcium deposition in these cells (Wada et al., 1999), whereas the dephosphorylated and the bacteria-derived recombinant OPN display no inhibitory effect (Jono et al., 2000). These results indicate that phosphorylation of OPN is essential for its inhibitory role on mineralization. Two studies have suggested that phosphorylation is necessary for OPN-stimulated osteoclast attachment (Ek-Rylander et al., 1994; Katayama et al., 1998). More recently, experiments performed with primary osteoclast cultures on bone slices have demonstrated that phosphates on OPN may be involved in regulating the resorptive activity of osteoclasts (Razzouk et al., 2002).
(2) BSP
Like OPN, phosphorylation in BSP occurs mostly at serine residues (Franzén and Heinegård, 1985). The cDNA sequence indicates that rat BSP has 10 serines and 8 threonine residues that are potential phosphorylation sites for casein kinases I and II (Oldberg et al., 1988). Based on consensus sequences in the primary structure of mammalian BSP, there are 5 potential sites for serine phosphorylation by protein kinase C, 9-11 sites for Ser/Thr phosphorylation by casein kinase II, and 1–2 sites for Tyr phosphorylation by tyrosine kinase (Ganss et al., 1999; Fig. 2). A recent study carried out on bovine BSP indicated that 5.8 phosphates are incorporated into the protein, predominantly by casein kinase II but not by casein kinase I (Salih, 2003). A detailed study using mass spectrometry combined with dephosphorylation indicated that human BSP contains 5 phosphates, 4 of which are located in the central region of its primary sequence (Zaia et al., 2001). The functions of phosphate groups of BSP are unknown; removal of the phosphates has no apparent influence on the ability of BSP to promote crystal formation, but they may affect crystal growth (Hunter and Goldberg, 1994).
(3) DMP1, 37K and 57K Fragments
The cDNA-deduced sequence indicates that full-length rat DMP1 has 65 Ser/Thr that are potential casein kinases I and II phosphorylation sites (George et al., 1993), with the NH2-terminal (37K) portion containing 22 and the COOH-terminal (57K) region having 43. Phosphate analysis indicates that the 37K fragments have 12 phosphates/mol, while the 57K fragments have 41 (Qin et al., 2003a). Although the functions of phosphates on DMP1 are unknown, they may serve as sequestering groups for recruiting calcium ions, similar to the roles of phosphates on DPP (see below).
(4) DSPP, DSP, and DPP Fragments
Rat dentin DSP has 6.2 phosphates/mol, about half of the 13 Ser/Thr potential casein kinases I and II phosphorylation sites (Qin et al., 2003b). DSP also shows heterogeneity in phosphorylation (Qin et al., 2001b). HMW-DSP has 10.3 phosphates/mol (Qin et al., 2003b). The functions of phosphates relative to DSP are unknown.
DPP, the most abundant NCP in dentin, is highly phosphorylated: About 45% of the total sequence is serine residues, and most of them are phosphorylated (Richardson et al., 1978; Butler et al., 1983). Rat dentin DPP is present in three isoforms—HP, MP, and LP—with different levels of phosphorylation (Butler et al., 1983; Butler, 1998). HP, the most highly phosphorylated and also the most abundant form of DPP, has 209 phosphates/mol (Butler et al., 1983). DPP contains a large number of repeating sequences of (Asp-Pse-Pse) and (Asp-Pse). The predicted structures for these repeating sequences depict extended backbone structures, with long ridges of carboxylate and phosphate groups alternating on opposite sides of the stretched-out polypeptide chain (George et al., 1996). Assuming that such a structure is formed on a collagen matrix, one can visualize an orientation that structurally fits its purported function, with calcium phosphate crystals growing on this lattice (Butler, 1998). Several in vitro studies have consistently demonstrated that immobilized DPP initiates mineralization and interacts with growing apatite crystals, thereby affecting their growth (Linde et al., 1989; Boskey et al., 1990; Saito et al., 1997). The large number of phosphates is vital to the functions of DPP, since removal of phosphate groups results in the loss of its role in the nucleation of apatite onto collagen (Saito et al., 1997).
(C) Glycosylation
(1) OPN
Significant glycosylation occurs on OPN in most tissues (Sodek et al., 2000). The cDNA sequence indicates that rat OPN has one Asn-X-Ser/Thr motif that is a potential site for N-linked glycosylation and ~ 26 Ser-X-Glu sequences that are potential sites for O-linked glycosylation (Oldberg et al., 1986; Sodek et al., 2000). In rat bone OPN, a single N-linked and 5 or 6 O-linked oligosaccharides have been identified, which consist of about 16.6% of the protein weight (Prince et al. 1987). In bovine milk OPN, 3 O-linked oligosaccharides have been identified (Sorensen et al., 1995). Although OPN has a considerable amount of carbohydrates, their precise functions are unclear.
(2) BSP
The rat BSP sequence contains 3 Asn-X-Ser/Thr motifs that are putative sites for N-linked glycosylation (Oldberg et al., 1988), 2 of which are conserved in all of the mammalian species (Fisher et al., 1990; Ganss et al., 1999). It has been estimated that BSP synthesized by UMR 106-01 cells (a rat osteosarcoma cell line) has 3 N-linked and 21-24 O-linked oligosaccharides (Midura et al., 1990). A recent study with mass spectrometry combined with a series of deglycosylation procedures has indicated that human BSP contains 33.8% oligosaccharides, with 12.3% being N-linked and 21.5% O-linked (Zaia et al., 2001), while another study with similar approaches demonstrated that oligosaccharides made a mass contribution of 30% to BSP extracted from human bone (Wuttke et al., 2001). Although the precise roles of attached carbohydrates are unknown, the extensive glycosylation of BSP may affect cell attachment activity (Fujisawa et al., 1995), but does not seem to be important for nucleation and the formation of apatite (Hunter and Goldberg, 1994; Goldberg et al., 1996).
(3) DMP1, 37K, and 57K fragments
The cDNA-deduced sequence indicates that a full-length rat DMP1 has 3 putative Asn-X-Ser/Thr N-linked glycosylation sites; all are located in the 57K fragment portion, COOH-terminal to the RGD motif (George et al., 1993). However, information with regard to DMP1 glycosylation is unavailable. In the later, high [Cl-] region of the DEAE chromatogram of rat bone and dentin ECM extract, a series of protein bands with Mr of 150–200 kDa contains components that are immunoreactive to anti-57K antibodies (Qin et al., 2001a). The identity of these DMP1-related, high-molecular-weight components is unknown. They may be highly glycosylated forms of DMP1 fragments.
(4) DSPP, DSP, and DPP fragments
The cDNA-deduced sequence indicates that rat DSP has 6 Asn-X-Ser/Thr potential sites for N-linked glycosylation (Ritchie et al., 1994). DSP isolated from rat dentin has 29.6% carbohydrates that include sialic acids and N- and O-linked oligosaccharides (Butler et al., 1992; Qin et al., 2003b). The functions of carbohydrates on DSP are unclear. HMW-DSP is larger and more acidic than DSP, and deglycosylation analysis indicates that the slower migration rate of HMW-DSP on sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) is due to its higher level of carbohydrate modifications (Qin et al., 2003b). The unusually slow migration rate and extreme acidity of HMW-DSP indicate that it may contain GAG chains and/or considerable amounts of sulphate, in addition to oligosaccharides. However, the exact nature of the carbohydrate compositions and their structures, as well as biological functions, is unknown. It has been demonstrated that the ~ 100-kDa DSP has little or no effect on in vitro mineralization (Boskey et al., 2000). Perhaps the HMW-DSP with extremely large amounts of carbohydrates may fulfill some special function different from that of the ~ 100-kDa form of DSP.
Although the cDNA-deduced sequence indicates that the DPP region of DSPP has some potential Asn-X-Ser/Thr N-linked glycosylation sites (MacDougall et al., 1997), the lack of detection of glucosamine and galactosamine in hydrolysates on the amino acid analyzer indicates that little or no carbohydrate is present on rat dentin DPP (Butler, unpublished observations).
(D) Proteolytic Processing
(1) OPN
Two fragments migrating at 20 kDa and 23 kDa on 15% SDS-PAGE have been reported for OPN isolated from porcine bone (Zhang et al., 1990). The NH2-terminal sequence analysis revealed that these 20- and 23-kDa proteins were derived from the COOH-terminal end of OPN and appeared to be generated by trypsin-like cleavage at Arg-Ser and Lys-Ala bonds. The rapid generation of the 23-kDa fragment, and its presence in different bone tissues at different developmental stages suggested that the fragmentation of porcine OPN might be important in bone formation (Zhang et al., 1990). A 14-kDa protein from fetal rat calvarial cell cultures, although not reactive with anti-OPN antibodies, was thought to be a fragment of OPN (Nagata et al., 1991). This conclusion was primarily based on its susceptibility to thrombin cleavage (Nagata et al., 1991). However, since the initial description, no further information has been reported concerning the 14-kDa fragment. OPN isolated from rat long bone displays Mr of ~ 65 to ~ 75 kDa on 5–15% gradient SDS-PAGE (Prince et al., 1987; Qin et al., 2001a), while on 15% SDS-PAGE, it migrates at ~ 45 kDa (Prince et al., 1987). It is worth noting that rat bone OPN with different levels of phosphorylation displays variations in migration rate (Kasugai et al., 1991; Razzouk et al., 2002). No fragmentation has been noticed for rat bone OPN; with Western immunoblots, only the ~ 65- to ~ 75-kDa bands on 5–15% SDS-PAGE were immunoreactive to the highly specific anti-OPN antibodies, without any trace of lower-molecular-weight fragments (Qin et al., 2001a). Although it is unclear why fragmentation was observed in extracts of porcine but not rat bone, these variations could be due to species differences. It is worth noting that the 20- and 23-kDa fragments were much less abundant than the ‘full-length’ porcine OPN migrating at 67 kDa on 15% SDS-PAGE (Zhang et al., 1990).
(2) BSP
There has been no information about the proteolytic processing of BSP. The 23-kDa bovine bone sialoprotein originally described by Herring (1972) was later considered to be a fragment of BSP produced by proteolytic activity during isolation (Fisher et al., 1983).
(3) DMP1
Full-length cDNA coding for DMP1 mRNA has been cloned and sequenced in several mammalian and non-mammalian species, but the corresponding complete protein has not been isolated or characterized. Originally, a 61-kDa protein band from dentin extracts was recognized by immunoblotting with an antibody monospecific to recombinant rat DMP1; this 61-kDa protein was thought to be a full-length DMP1 (George et al., 1993). Later, the same group reported that the molecular weight of the bacteria-derived recombinant rat DMP1, determined by SDS-PAGE, was approximately 90–95 kDa, which is higher than that of the purported naturally occurring 61-kDa form of DMP1 (Srinivasan et al., 1999). We now believe that this 61-kDa band is a processed fragment(s) of DMP1, possibly equivalent to the 57K fragment (Qin et al., 2003a). In bone and dentin ECM extracts, there are two clusters of proteins (37K and 57K fragments) that elute in the same (D4b) fraction of the DEAE-Sephacel chromatogram as OPN and BSP (Qin et al., 2001a, 2003a). When the mixture from this fraction of bone was cleaved with trypsin, sequences originating from DMP1 (as well as OPN and BSP) were obtained (Safran et al., 1998b). With a combination of several chromatographic approaches, these rat bone DMP1 fragments were purified. Extensive sequencing and analysis of tryptic peptides derived from these highly pure materials (37K and 57K fragments, respectively) by Edman degradation and mass spectrometry have yielded some significant discoveries (Qin et al., 2003a). First, analysis of these data shows unequivocally that 37K fragments originate from the NH2-terminal region, and that the 57K fragments are from the COOH-terminal region of the DMP1 amino acid sequence, strongly suggesting that DMP1 in bone is proteolytically processed (Fig. 2a).
Second, from 37K fragments, 2 COOH-termini, Phe173 and Ser180, were identified, and from 57K fragments, 2 NH2-termini, Asp218 and Asp222, were detected. These findings, along with comparison with DMP1 sequence deduced from cDNA, indicate that this protein is proteolytically cleaved at 4 bonds, Phe173-Asp174, Ser180-Asp181, Ser217-Asp218, and Gln221-Asp222 (Fig. 2b). It should be noted that the sequences around the cleaved Phe173-Asp174 and Ser180-Asp181 bonds are highly conserved, but those surrounding Ser217-Asp218 and Gln221-Asp222 are not conserved.
Third, the uniformity of cleavages at the NH2-terminal peptide bonds of select aspartyl residues suggests that a single proteinase is involved (see below). Fourth, phosphate analysis indicates that the 57K fragments are more highly phosphorylated than the 37K. Additionally, dentin ECM also contains 37K and 57K fragments. The HPLC chromatographic profile of tryptic peptides derived from dentin 37K fragments was identical to that obtained from bone 37K fragments, while selective sequencing of tryptic peptides indicated that the NH2-termini and COOH-termini of dentin 37K fragments were identical to those of bone (Qin et al., 2003a). Attempts to obtain pure dentin 57K fragments for sequence analysis have thus far been unsuccessful.
(4) DSPP
The two acidic proteins, DSP and DPP, are individual, distinctive proteins that are found in abundance in the ECM of dentin (Dickson et al., 1975; DiMuzio and Veis, 1978; Butler, 1987; Butler et al., 1992). It appeared that they had no relationship to each other, an idea that changed when results from cDNA cloning and sequencing of a mouse mRNA library showed that DSP and DPP are derived from the same precursor, DSPP (MacDougall et al., 1997). Subsequently, results from cDNA cloning and sequencing in human, rat, and porcine samples consistently demonstrated that the 5′ end of DSPP mRNA contains the sequences coding for DSP, followed by the DPP sequences on the 3′ end, in agreement with results obtained from cDNA cloning in the mouse (Gu et al., 2000; Ritchie et al., 2001; Yamakoshi et al., 2003). The occurrence of one gene transcribing a single mRNA encoding DSP and DPP indicates that the translated product, DSPP, is cleaved by a specific proteinase(s), giving rise to the individual proteins, DSP and DPP (Fig. 2c). This conclusion is supported by the fact that DSP and DPP, but not DSPP (the protein representing the entire sequence), are found in dentin ECM. The presence of HMW-DSP (Qin et al., 2003b) may be one of the reasons why there is more DPP than DSP in dentin ECM. To clarify proteolytic cleavage sites, we have isolated and characterized tryptic peptides from the COOH-terminal region of rat DSP (Qin et al., 2001b). Two COOH-termini were identified and characterized. The major peptide ended at Tyr421 and the minor one at His406. Analysis of these data, along with comparison with the DSPP sequence deduced from cDNA and the established NH2-terminus of DPP, shows that the proteolytic processing of rat DSPP to form DSP and DPP involves a proteinase(s) that cleaves Tyr421-Asp422 (giving rise to the COOH-terminus of DSP) and Gly430-Asp431 (the NH2-terminus of DPP) and, to lesser extent, His406-Ser407 (a second, minor COOH-terminus). The fact that the two major cleavages occur at conserved sequences at the NH2-terminus of an aspartic acid residue (Fig. 2b) indicates that DSPP may be cleaved by the same proteinase involved in the proteolytic processing of DMP1 (possibly PHEX protein; see below).
In summary, DMP1 and DSPP are proteolytically processed, and they appear to be processed similarly. We hypothesize that full-length DMP1 and DSPP are inactive precursors and that the proteolytic processing is an activation step, releasing their functional fragments which play significant roles in the mineralization of bone and dentin. This view predicts that if these precursors are not properly processed, defective mineralization will result (see below).
(5) PHEX protein, a possible proteinase that cleaves DMP1 and DSPP
This protein, encoded by PHEX (phosphate-regulating gene with homologies to endopeptidases on X chromosome), is expressed predominantly in osteoblasts, osteocytes, and odontoblasts (Du et al., 1996; Guo and Quarles, 1997; Lipman et al., 1998; Ruchon et al., 1998, 2000; Thompson et al., 2002). It is a cell-membrane-associated glycoprotein with structural homologies to members of the M13 family of metalloendopeptidases (The HYP Consortium, 1995; Lipman et al., 1998; Boileau et al., 2001). The M13 endopeptidases are zinc-containing type II integral membrane glycoproteins with a relatively short cytoplasmic NH2-terminal region, a single transmembrane domain, and a long extracytoplasmic domain, which contains the active site of the enzyme (Crine et al., 1997). Mutations of PHEX are associated with X-linked hypophosphatemic rickets (XLH) in humans, as well as with the murine homolog, Hyp (The HYP Consortium, 1995; Holm et al., 1997).
The PHEX protein is likely to be the enzyme responsible for the proteolytic processing of DMP1 and DSPP (Qin et al., 2003a). This conclusion is based on the following observations: (a) Several studies have shown that the PHEX protein has a strong preference for cleavage of peptide bonds at the NH2-terminus of aspartyl residues (Lipman et al., 1998; Boileau et al., 2001; Sabbagh et al., 2003; Campos et al., 2003); as described above, DMP1 and DSPP are cleaved similarly (see Fig. 2b). (b) PHEX is expressed in osteoblasts, osteocytes, and odontoblasts (Du et al., 1996; Guo and Quarles, 1997; Lipman et al., 1998; Ruchon et al., 1998, 2000; Thompson et al., 2002), as are DMP1 (Butler et al., 2002; Feng et al., 2002b) and DSPP (Butler et al., 1992; D’Souza et al., 1992; Bègue-Kirn et al., 1998; Qin et al., 2002, 2003c). (c) The bony and dental defects of DMP1 knock-out mice at post-natal stage (after the age of 3 wks; Feng et al., 2002a) are similar to those of XLH and Hyp, while the dental defects in the DSPP null mice (Sreenath et al., 2003) show similarities to those of XLH and Hyp.
A more recent study (Steiglitz et al., 2004) described cleavage of recombinant full-length DMP1 by bone morphogenetic protein-1(BMP-1)/Tolloid-like proteinases that are widely expressed in mesenchymal tissues and that have broad substrate specificity. Two DMP1 fragments generated by the BMP-1/Tolloid-like proteinases are similar to those previously isolated from bone (Qin et al., 2003a). Based on the known roles of BMP-1/Tolloid-like proteinases in the biosynthetic conversion of precursor proteins into their mature functional forms, it is believed that proteolytic processing of DMP1 is an activation step, i.e., converting full-length DMP1 precursor into functional fragments. Although recombinant DMP1 was shown to be cleaved by BMP-1/Tolloid-like proteinases in vitro, it was stated that the possibility that PHEX is involved in DMP1 processing is not excluded (Steiglitz et al., 2003). It should be noted that the expression of BMP-1/Tolloid-like proteinases begins at a rather earlier stage of embryonic development, whereas DMP1 is expressed in a much later stage (Feng et al., 2002b) within the same time window of PHEX expression (Thompson et al., 2002). Additionally, BMP-1/Tolloid-like proteinases cleaved only one (Ser180-Asp181) of the 4 cleavage sites that have been identified in rat bone DMP1 processing (Qin et al., 2003a). A combination of information (also see below) seems to suggest that PHEX protein is the major proteinase responsible for processing DMP1, functioning synergistically with BMP1/Tolloid-like proteinases in the proteolytic processing of DMP1 precursors.
(6) Relationship of XLH and Hyp to DMP1 and DSPP
XLH, the most common inherited form of rickets, is a dominant disorder characterized by defective calcification of bone and dentin, growth retardation, impaired renal tubular re-absorption of inorganic phosphate (Pi), and resistance to phosphorus and vitamin D therapy (Winters et al., 1958; Burnett et al., 1964; Rasmussen and Anast, 1983; The HYP Consortium, 1995; Grieff et al., 1997; Holm et al., 1997). As stated earlier, mutations of PHEX are associated with XLH in humans and the murine homolog, Hyp. PHEX encodes for a cell-surface metalloprotease. Since the physiologically relevant PHEX substrates have not been identified (Guo et al., 2001; Quarles, 2003), several different mechanisms have been considered to explain how inactivation of an enzyme results in both phosphaturia and defective mineralization. Currently, one widely discussed theory is that hypophosphatemia (phosphaturia) and defective mineralization are caused through different pathogenetic pathways (Bai et al., 2002; Liu et al., 2002; Quarles, 2003). Because the expression of PHEX is predominantly in bone and tooth tissues, and it is expressed by osteoblasts, osteocytes, and odontoblasts, locally produced substrates in the skeleton are thought to be involved in the defective mineralization of XLH (Xiao et al., 1998; Bai et al., 2002; Quarles, 2003). Since PHEX is also expressed less abundantly at extraskeletal sites, additional substrates in these non-mineralized tissues may be involved in the pathogenesis of hypophosphatemia (Quarles, 2003). In this review, we will discuss only possible mechanisms relative to the defective mineralization in XLH and Hyp.
Several lines of evidence support the view of intrinsic skeletal factor(s) responsible for impaired mineralization, independent of the hypophosphatemia condition: First, the bone defect in Hyp mice is more severe than can be explained by hypophosphatemia alone (Quarles, 2003). Second, transplantation of osteoblasts from Hyp mice into normal animals produces abnormal bone with impaired mineralization but with the absence of hypophosphatemia (Ecarot et al., 1992). Third, osteoblasts derived from the Hyp mouse have an impaired capacity to mineralize ECM in vitro; compared with cells from normal mice, osteoblast cultures from the Hyp mouse display diminished 45Ca accumulation into the ECM and reduced formation of mineralization nodules (Xiao et al., 1998). Fourth, deletion of the renal sodium-dependent phosphate transporter in Npt-2 null mice displays both hypophosphatemia and abnormal skeletal development at 21 days of age, but with increasing age, there is a dramatic reversal and eventual overcompensation of the skeletal phenotype without ‘normalization’ of hypophosphatemia (Beck et al., 1998). The clearly age-dependent pattern of skeletal changes in Npt-2 null mice suggests that adaptive responses occur in osteoblasts, when PHEX is active, to minimize the effects of hypophosphatemia on bone (Quarles, 2003).
In summary, it is likely that protein substrates for the PHEX enzyme produced by osteoblasts, osteocytes, and odontoblasts are involved in the normal mineralization of bone and dentin. Analysis of amino acid sequencing data establishing cleavages at X-Asp bonds strongly suggests that DMP1 and DSPP are proteolytically processed by the PHEX protein, since this enzyme has a strong preference for these bonds. This endopeptidase, expressed predominantly in bones and teeth, is an integral membrane protein with a relatively large extracytoplasmic domain containing the catalytic site. This type of structure would imply that the proteolytic processing would occur during or after secretion. In this way, if the cleavage is an activation step, one would visualize activation in a controlled, timely manner, as part of a stepwise series of events. In the cases of DMP1 and DSPP, the conversion to 37K and 57K fragments and to DSP and DPP releases components that promote mineralization of osteoid and predentin (Fig. 3). Thus, we envision that mineralization of bone and dentin requires that DMP1 and DSPP be proteolytically processed by PHEX protein and that if these precursors are improperly processed, defective mineralization in XLH and Hyp results. This view regarding the proteolytic processing of DMP1 and DSPP by the PHEX protein resembles the activation of matrix metalloproteinase precursors (proMMPs) by membrane type MMPs (MT-MMPs). Through pericellular proteolysis, MT-MMPs process proMMPs, and the activated MMPs are secreted into the ECM, where they act on substrates remote from the producer cells (Seiki and Yana, 2003). For example, MT1-MMP has been identified as possessing the ability to activate proMMP-2 (Sato et al., 1994), the activated form of which is secreted into the ECM, where it plays an important role in the degradation of type IV collagen.
(E) Future Research
This review shows that PTMs of SIBLING family members may play vital roles in their functions. Despite excellent progress in this field, this review of the literature indicates that several areas relative to the PTMs of SIBLING proteins need further investigations. The roles potentially played by the carbohydrate moieties have not been studied. The nature of glycosylation bringing about heterogeneity of DSP and other SIBLING proteins warrants further studies. Phosphorylation of OPN clearly plays a role in the inhibition of mineralization and in osteoclast functions, but their exact roles have not been elucidated. This specific function of phosphorylation is accentuated by the realization that the seryl residues that are phosphorylated are highly conserved. The importance of post-translational proteolytic cleavage of DMP1 and DSPP must be clarified. The strong possibility that PHEX protein catalyzes this cleavage must be examined by a series of careful experiments. The hypothesis that this proteolytic cleavage of X-Asp bonds is an activation process needs further testing. Other proteins with X-Asp cleavage sites should be sought. One major objective in all the investigations will be to uncover the basic mechanistic steps in the mineralization process. The questions surrounding the unknown pathways that lead to the conversion of osteoid to bone (Fig. 1a) and predentin to dentin (Fig. 1b) must be uncovered. These answers will lead to better methods for the diagnosis of malformations of these tissues as well as interventions that are scientifically based.
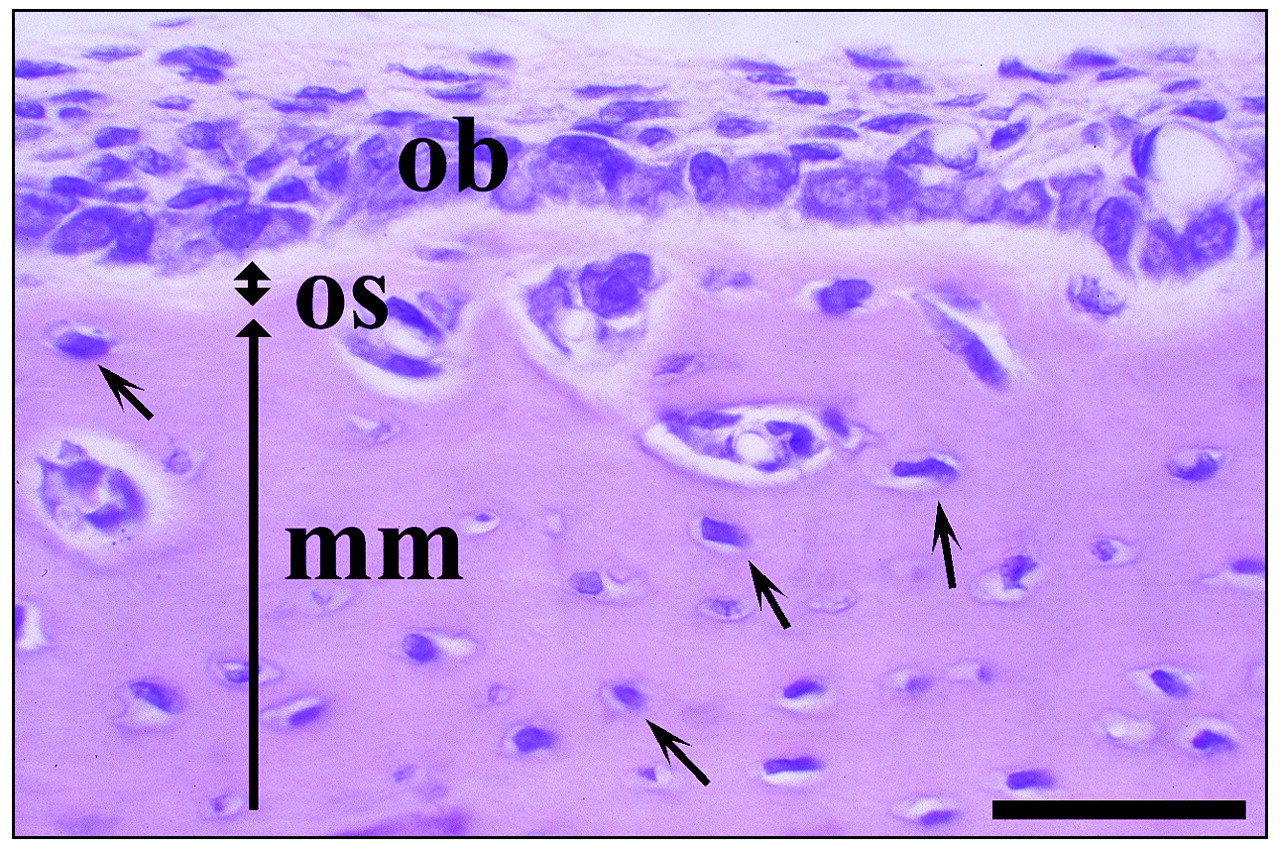
H&E staining of a bucco-lingual section from the mandible of a three-week-old rat. Osteoblasts (ob) secrete unmineralized bone matrix, termed ‘osteoid’ (os, doubleheaded arrow), which is subsequently mineralized and becomes bone (mm, long arrow) when apatite crystals are deposited. Small arrows indicate osteocytes buried in the mineralized bone matrix. Bar = 40 μm.
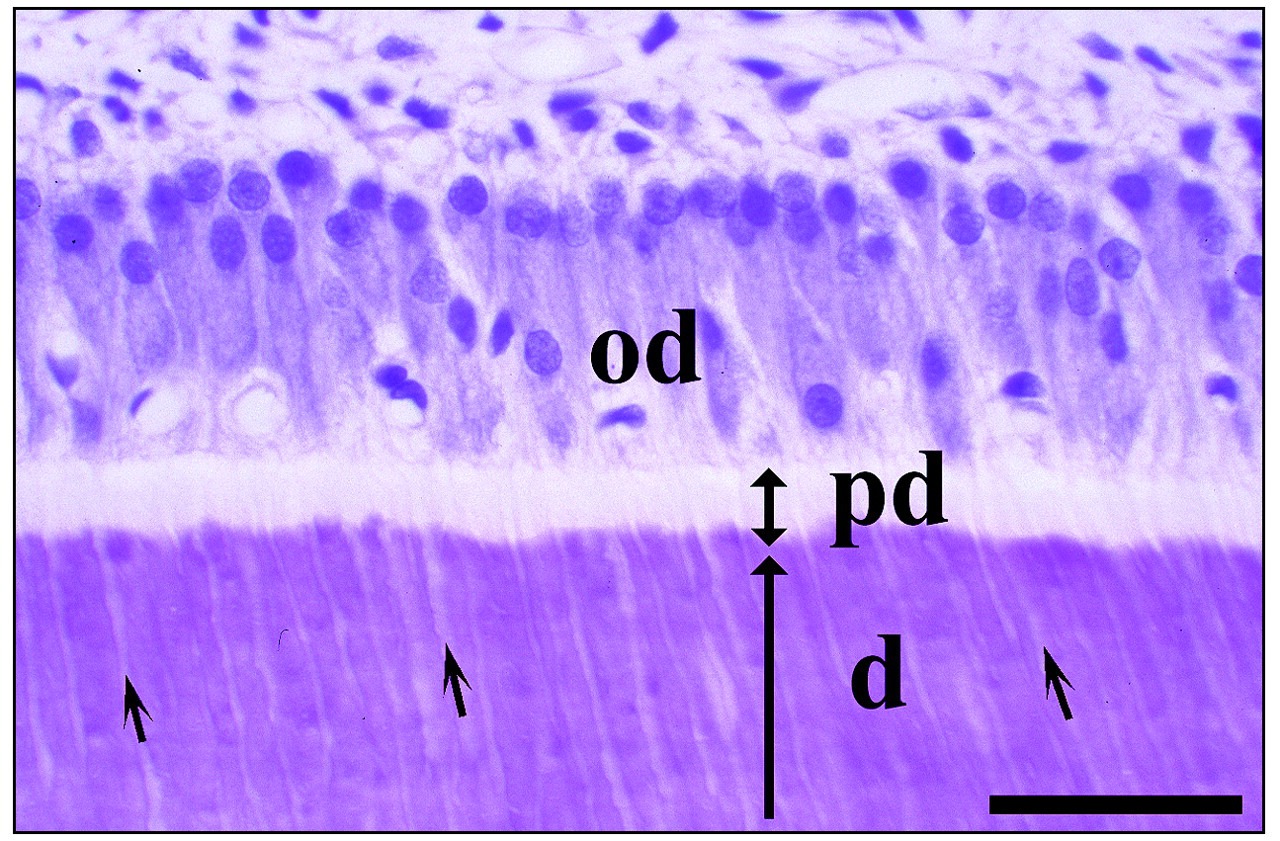
H&E staining of a bucco-lingual section from a mandibular incisor of a five-week-old rat. Similar to osteoblasts, odontoblasts (od) secrete unmineralized predentin (pd, doubleheaded arrow), which is subsequently mineralized and becomes dentin (d, long arrow) when apatite crystals are deposited. The long columnar odontoblasts form processes that extend into dentin. The short arrows indicate dental tubules that house odontoblast processes. Bar = 40 μm.
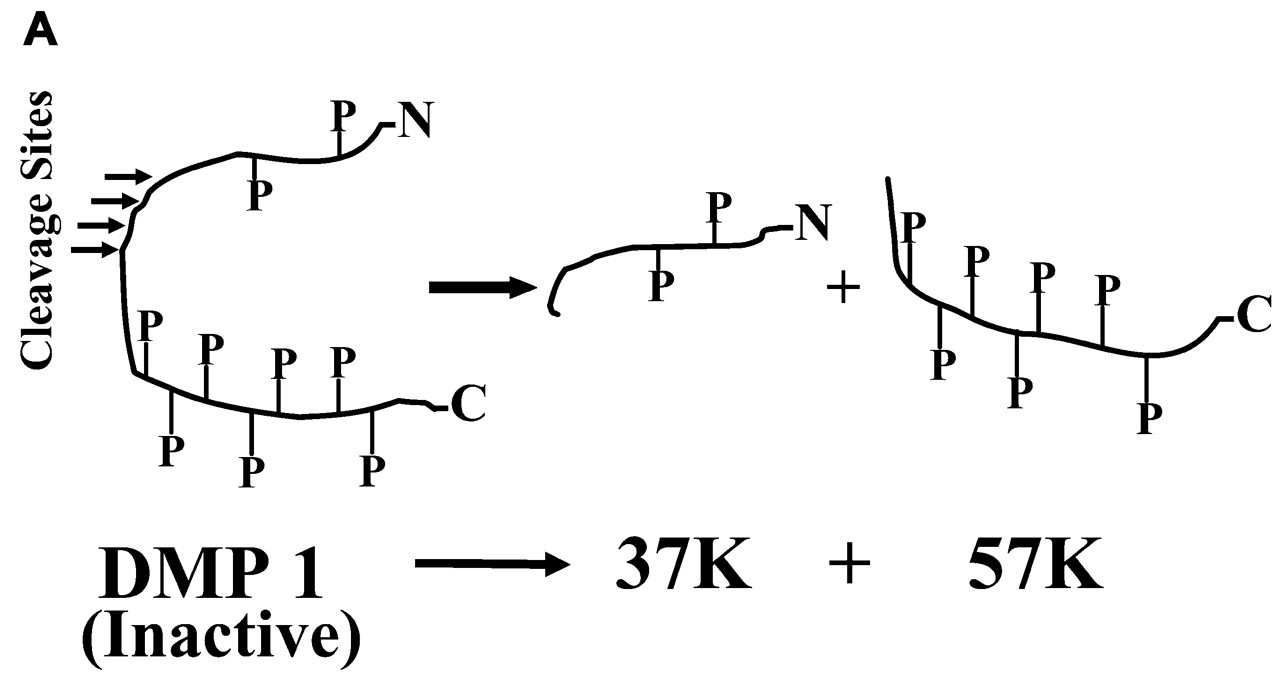
Full-length DMP1 is an inactive precursor. It is proteolytically processed into 37K and 57K fragments, originating from the NH2-terminal and COOH-terminal regions of the DMP1 amino acid sequence, respectively. The 57K fragments contain more phosphates (P) than the 37K fragments.
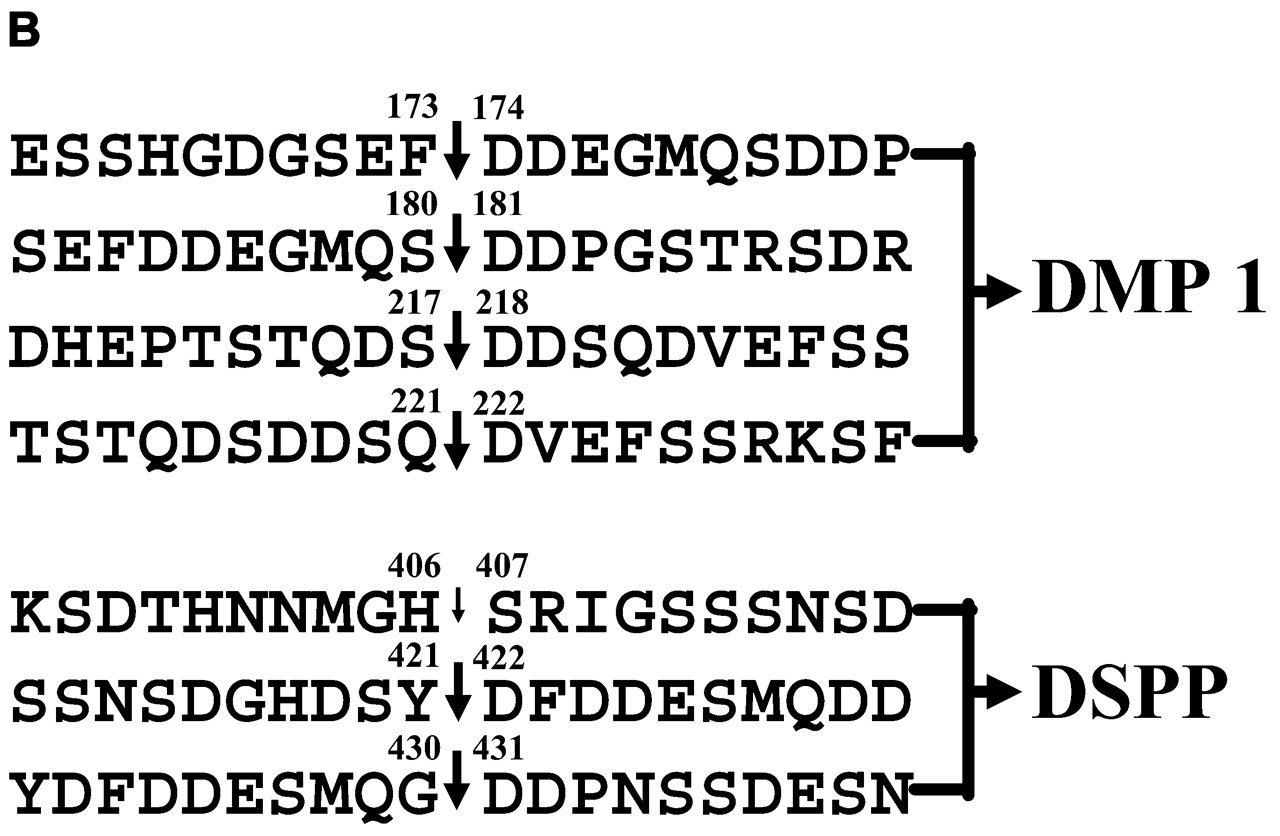
Amino acid sequences surrounding the cleavage sites of DMP1 and DSPP. Vertical arrows indicate cleavages sites. In DMP1, all 4 cleavages occur at the NH2-terminus of the Asp residue (Qin et al., 2003a). Among the 3 cleavage sites in DSPP, 2 major ones are at the NH2-terminus of the Asp residue, while the minor one, as indicated by a smaller arrow, is at the His-Ser bond (Qin et al., 2001b). Amino acid residues are numbered (written on the top of corresponding residues), starting from the NH2-terminus of full-length DMP1 or DSPP, excluding signal peptide.
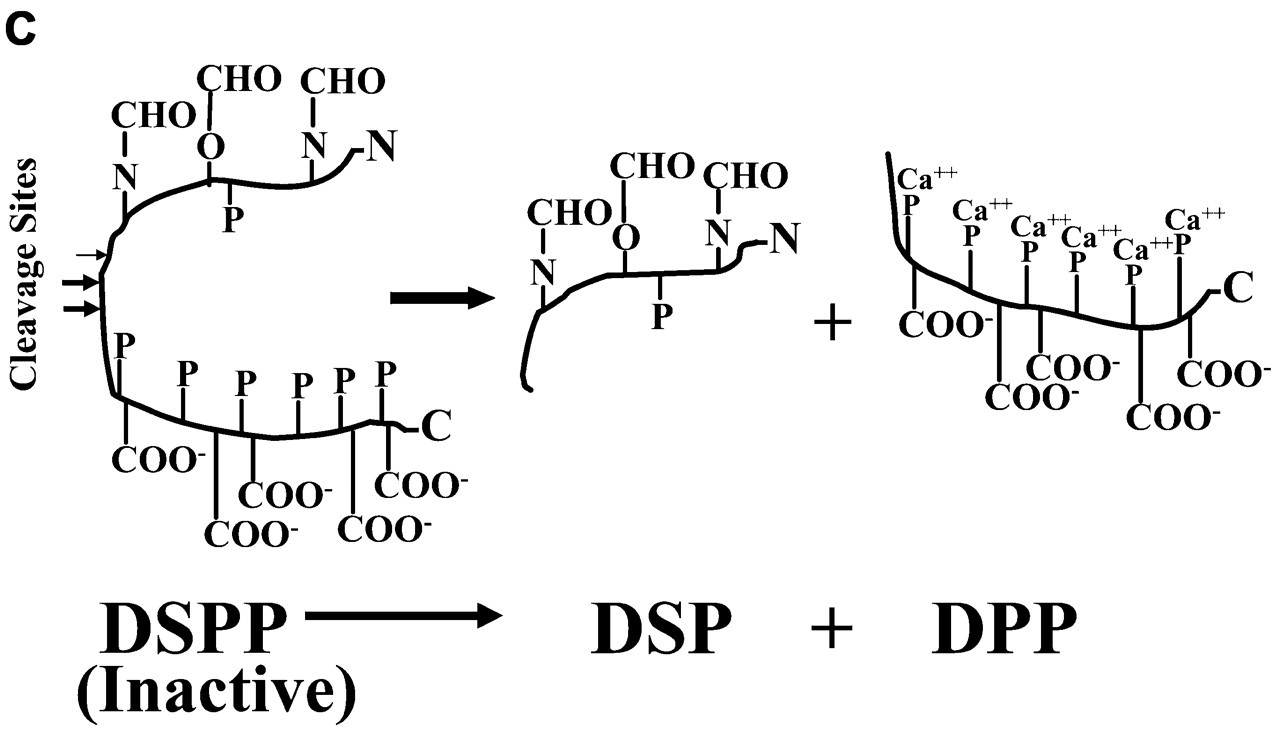
Full-length DSPP is an inactive precursor. It is proteolytically processed into DSP and DPP that are from the NH2-terminal and COOH-terminal regions of DSPP, respectively. DSP is rich in carbohydrates (CHO) but contains fewer phosphates (P), whereas DPP is devoid of glycosylation but contains an unusually large number of phosphates. The phosphates on DPP are mainly present on one side of the protein backbone (George et al., 1996) and are postulated to sequester calcium ions (Ca++).
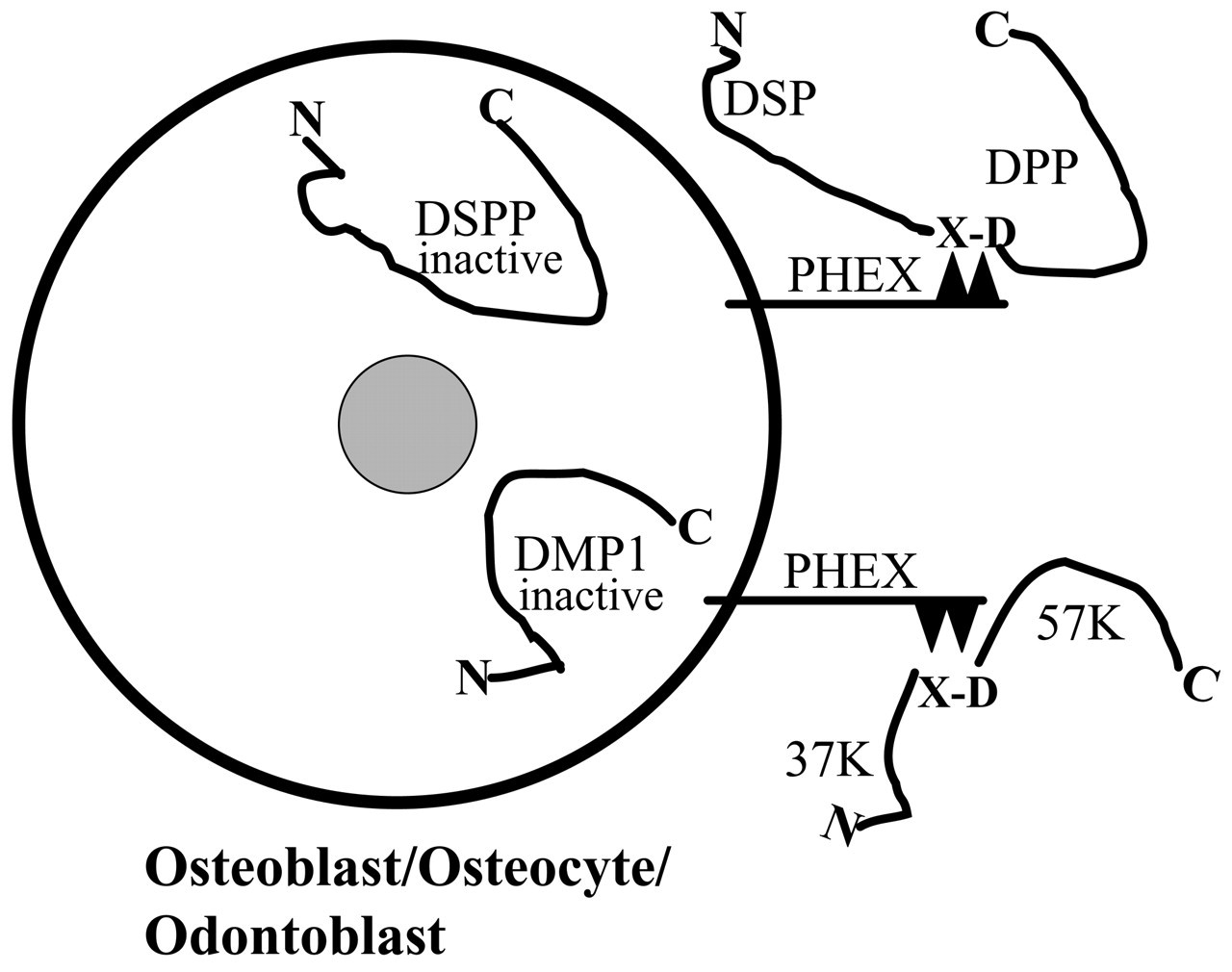
Hypothetical processing of DMP1 and DSPP by PHEX protein. DMP1 and DSPP are converted to functional fragments by PHEX enzyme on the surfaces of osteoblasts, osteocytes, and odontoblasts. PHEX protein, one of the M13 endopeptidases, has a short cytoplamic NH2-terminal region, a single transmembrane domain, and a long extracytoplasmic domain that contains the active site of the enzyme (Crine et al., 1997). After DMP1 and DSPP are processed by cleavage of selected X-Asp bonds, their fragments—namely, 37K, 57K, DSP, and DPP—are transported to the ECM of bone and dentin, where they influence mineralization.
Footnotes
Acknowledgements
We are grateful to our colleague, Jan Brunn, for his excellent work relative to the purification of OPN, BSP, DSP, DPP, and DMP1 fragments from bone and dentin. Much of our data about the PTMs of SIBLING members has been obtained through analyses on the proteins he purified. We thank Richard Cook and Ralph Orkiszewski at the Protein Chemistry Core Laboratory, Baylor College of Medicine, for their continuous, wonderful collaboration in analyzing peptide samples of OPN, DSP, and DMP1 fragments. We are also thankful to Dr. Adele Boskey of the Hospital for Special Surgery, New York, for her continuous collaboration in testing the effects of SIBLING proteins on in vitro mineralization. We deeply appreciate consultations with Dr. Darryl Quarles at the Center for Bone and Mineral Disorders of Duke University Medical Center for giving us insights about PHEX. This work was supported by NIH Grant DE 05092 to William T. Butler.
