Abstract
External apical root resorption (EARR) is a common sequela of orthodontic treatment, although it may also occur in the absence of orthodontic treatment. The degree and severity of EARR associated with orthodontic treatment are multifactorial, involving host and environmental factors. Genetic factors account for at least 50% of the variation in EARR. Variation in the Interleukin 1 beta gene in orthodontically treated individuals accounts for 15% of the variation in EARR. Historical and contemporary evidence implicates injury to the periodontal ligament and supporting structures at the site of root compression following the application of orthodontic force as the earliest event leading to EARR. Decreased IL-1β production in the case of IL-1B (+3953) allele 1 may result in relatively less catabolic bone modeling (resorption) at the cortical bone interface with the PDL, which may result in prolonged stress concentrated in the root of the tooth, triggering a cascade of fatigue-related events leading to root resorption. One mechanism of action for EARR may be mediated through impairment of alveolar resorption, resulting in prolonged stress and strain of the adjacent tooth root due to dynamic functional loads. Future estimation of susceptibility to EARR will likely require the analysis of a suite of genes, root morphology, skeleto-dental values, and the treatment method to be used—or essentially the amount of tooth movement planned for treatment.
(I) Introduction
Basic descriptors of root resorption are based on the anatomical region of occurrence—i.e., internal root resorption and external root resorption (cervical root resorption and external apical root resorption). Additional classification may involve two types of internal resorption: root canal (internal) replacement resorption and internal inflammatory resorption. External resorption can be classified into four categories according to its clinical and histologic manifestations: external surface resorption, external inflammatory root resorption, replacement resorption, and ankylosis. External inflammatory root resorption has been further categorized into cervical resorption with or without a vital pulp (invasive cervical root resorption) and external apical root resorption (EARR) (Ne et al., 1999).
This paper reviews EARR and its association with orthodontic treatment, and examines a new paradigm for its multifactorial etiology. EARR is a frequent iatrogenic outcome associated with orthodontic treatment, especially in the maxillary incisors, and may also occur in the absence of orthodontic treatment (Harris and Butler, 1992; Harris et al., 1993). Depending on the methodology, the incidence of EARR without orthodontic treatment has been reported to range from zero to 90.5% (Brezniak and Wasserstein, 1993). From 7% to 13% of individuals who have not had orthodontic treatment show some EARR on radiographs (Rudolph, 1936; Harris et al., 1993), presumably as a function of occlusal forces. There is an association of EARR in those who have not received orthodontic treatment with missing teeth, increased periodontal probing depths, and reduced crestal bone heights (Harris et al., 1993). Individuals with bruxism, chronic nail biting, and anterior open bites with concomitant tongue thrust may also show an increased extent of EARR before orthodontic treatment (Harris and Butler, 1992). Dental trauma, especially with re-implantation of an avulsed tooth, is also associated with increased EARR (Donaldson and Kinirons, 2001). For the most part, EARR is asymptomatic unless substantial tooth structure is affected, so early detection is unlikely unless radiographs are used (Brezniak and Wasserstein, 1993, 2002b).
(II) Root Resorption and External Apical Root Resorption
EARR is the loss of root structure involving the apical region to the extent that it can be seen on standard radiographs (Fig. 1). EARR is distinct from root resorption (RR). Microscopic areas of resorption lacunae hallmark root resorption. These microscopic lesions lack clinical significance and are not detected by standard radiographs (Brezniak and Wasserstein, 1993). The resorption lacunae develop on the cementum root surface and can be visualized by histological techniques (Fig. 2). Although EARR and RR during orthodontic tooth movement are believed to be related conditions, a distinction should be made between these two conditions when incidence and prevalence are studied. Orthodontic force applied to teeth over a short period of time can produce resorption lacunae in the absence of EARR (Kvam, 1972). An increase in RR can be accomplished with increased duration of orthodontic force application and force, with the higher magnitude of moments producing exposure of root dentin (Casa et al., 2001).
EARR may occur preferentially in the apical region, since more than three-quarters of resorption lacunae occur in the apical region of the root (Henry and Weinmann, 1951), a fact that could be explained by the following: (1) Forces are concentrated at the root apex because orthodontic tooth movement is never entirely translatory, and the fulcrum is usually occlusal to the apical half of the root (Harris, 2000); (2) periodontal fibers assume a different direction in the apical end, which might explain the increased stress in the region (Henry and Weinmann, 1951); and (3) the apical third is covered with cellular cementum, whereas the coronal third is covered with acellular cementum. The active cellular cementum depends on a patent vasculature; accordingly, periapical cementum is more friable and easily injured in the case of trauma and concomitant vascular stasis (Henry and Weinmann, 1951; Baumrind et al., 1996; Harris, 2000).
(III) Incidence of EARR in Association with Orthodontic Treatment
Depending on the methods, EARR associated with orthodontic treatment has been noted to range between zero and 100% (Brezniak and Wasserstein, 1993). Although orthodontic treatment is associated with some maxillary central incisor EARR in most patients, and more than one-third of those treated experience greater than 3 mm of loss, severe EARR (more than 5 mm) occurs in 2% to 5% of the population (Taithongchai et al., 1996; Killiany, 1999).
(IV) The Effect of Tooth Movement and the Role of the PDL
The amount of orthodontic movement is positively associated with the resulting extent of EARR (DeShields, 1969; Sharpe et al., 1987; Parker and Harris, 1998). Orthodontic tooth movement, or “biomechanics”, has been found to account for approximately one-tenth to one-third of the total variation in EARR (Linge and Linge, 1991; Baumrind et al., 1996; Horiuchi et al., 1998), although, in one study (Parker and Harris, 1998), up to 90% of the variation has been attributed to the extent of tooth movement.
In turn, the required amount of tooth movement is a function of severity of malocclusion. For example, the greater the incisor overjet, the greater the amount of retraction during treatment, and the greater the amount of incisor resorption (Beck and Harris, 1994; Harris et al., 1997). Moreover, it was found that the deeper the overbite, the greater the incisor intrusion and resorption, and the greater the root resorption of the distal root of the maxillary first molar (Beck and Harris, 1994). Extraction patterns can influence the degree of EARR because of the increased tooth movement, compared with non-extraction cases, required to close extraction spaces (Blake et al., 1995; McNab et al., 2000). For example, cases in which 4 first premolars were extracted have more EARR than cases with no extractions, or only extractions of the 2 maxillary first premolars (Sameshima and Sinclair, 2001b).
The amount of time spent in orthodontic treatment can be a factor in EARR (Taithongchai et al., 1996), but not necessarily (Beck and Harris, 1994; Taner et al., 1999). Even when duration of treatment is a factor, it, along with several significant dentofacial structure measurements, does not account for enough of the observed variability to be useful as a predictor of EARR by itself (Taithongchai et al., 1996).
The suitability of the rat as a model for studying tooth movement was demonstrated almost 50 years ago (Macapanpan et al., 1954; Waldo and Rothblatt, 1954). Macapanpan and co-workers (1954) positioned elastic materials between the first and second maxillary molars, tipping the first molars mesially and the second and third molars distally. Tipping the teeth created sites of compression, where cellular death and hyalinization occurred, and tension, where cellular proliferation was observed in the periodontal ligament (PDL). Subsequent histological studies in rodents support an association between root resorption and the presence and active removal of the hyalinized tissues at the sites of compression following experimental tooth movement (Rygh, 1977; Williams, 1984; Brudvik and Rygh, 1993a,b, 1994; Hellsing and Hammarström, 1996). Normal healthy PDL may modulate the cascade of root resorption. Cultured primary dentition PDL fibroblasts have been shown to inhibit the differentiation of osteoclast-like cells in mouse bone marrow cultures (Wu et al., 1999). Root resorption therefore may require damage to the cementoblastic layer in combination with necrosis or inflammation. Interestingly, when orthodontic force is stopped, root resorption continues until a functional PDL is established (Brudvik and Rygh, 1995a,b).
(V) Influence of Dental Anomalies and Root Morphology
While Kjaer (1995) suggested, as risk factors, the increased prevalence (as compared with published population frequencies) of dental anomalies-such as tooth agenesis, peg-shaped or small maxillary lateral incisor crown, crown invaginations, ectopic eruption, and taurodontism-in a sample of 107 orthodontic cases presenting with “excessive” (more than one-third of one or more roots resorbed during treatment) EARR, Lee et al. (1999) did not find increased EARR in 84 orthodontic cases exhibiting at least one dental anomaly, compared with 84 matched cases in which there were no dental anomalies. The presence of more than one dental anomaly did not appear to be associated with a greater risk of EARR. The divergent outcomes may be due to the difference in the ascertainment of the study sample as well as other aspects of the two investigations.
Root length and shape have also been variables that have been studied for their association with EARR. It has been suggested that teeth exhibiting relatively short roots prior to orthodontic treatment tend to develop more resorption during orthodontic treatment (Ketcham, 1929; Becks, 1936; Massler and Malone, 1954; Massler and Perreault, 1954; Jakobsson and Lind, 1973; Goldson and Henrikson, 1975; Newman, 1975; Kjaer, 1995; Taithongchai et al., 1996; Harris et al., 1997; Thongudomporn and Freer, 1998). Even though some have inferred that this is due to increased root resorption activity prior to the orthodontic treatment, it is not clear that all relatively short roots prior to orthodontic treatment are the result of active pre-treatment resorption.
Although it has been held that a valid prognosis can be made by the clinician as to the amount of EARR that could be expected in the majority of cases on the basis of a careful analysis of relatively short pre-treatment root lengths on the radiographs taken before treatment (Massler and Malone, 1954), it has also been said that EARR does not increase in teeth with short roots (Levander and Malmgren, 1988; Goldin, 1989), or that the tendency for EARR increases with increasing tooth length (Mirabella and Årtun, 1995; Sameshima and Sinclair, 2001a). The latter finding may be related to longer teeth needing stronger forces to be moved, and the fact that the actual displacement of the root apex is larger during tipping or torquing movements of longer teeth, although the apical shortening that occurs in the shorter root is of greater concern (Mirabella and Årtun, 1995; Sameshima and Sinclair, 2001a).
The two-dimensional shape of the root as delineated on radiographic film appears to be associated with various amounts of EARR. The tendency for EARR was found to be greater in teeth with pipette-shaped roots and apical bends (Newman, 1975; Levander and Malmgren, 1988; Kjaer, 1995), bottleneck roots (McFadden et al., 1989), abnormal (pointed, eroded, blunt, bent, and bottle shape) root shape (Mirabella and Årtun, 1995), thin or pipette-shaped roots (Thongudomporn and Freer, 1998), and dilacerated, bottle-shaped, or pointed roots (Sameshima and Sinclair, 2001a), although so far there is no systematic way to estimate the likelihood of EARR based upon root shape other than to say it is increased. Teeth with blunted roots have been found to be associated with both an increased (Thongudomporn and Freer, 1998) and a decreased (Sameshima and Sinclair, 2001a) occurrence of EARR. Overall, the shape of the root does appear to be associated with the likelihood of EARR, and is best examined on periapical rather than panoramic radiographs (Sameshima and Asgarifar, 2001).
(VI) Cellular and Molecular Mechanisms of Odontoclast/Osteoclast Regulation
Multinucleated cells referred to as “odontoclasts” resorb three dental hard tissues, i.e., cementum, dentin, and enamel. These cells have morphological and functional characteristics similar to those of bone-resorbing osteoclasts (Sahara et al., 1994, 1998). Osteoclast and odontoclast precursors originate from hemopoietic cells in the bone marrow. Osteoclast formation from hemopoietic precursors is induced by the cytokines such as macrophage colony-stimulating factor (M-CSF) and TRANCE (also called RANKL, ODF, and OPGL), a membrane-bound ligand expressed by bone marrow stromal cells (Lean et al., 2000; Tsurukai et al., 2000). Osteoclast precursors may also be recruited from the blood via activated endothelium (McGowan et al., 2001). As circulating mononuclear cells (monoctyes), these precursors can be induced to proliferate and differentiate into osteoclasts (Quinn et al., 1998; Massey and Flanagan, 1999; Fujikawa et al., 2001). Receptor activator of nuclear factor kappa B (RANK) and its ligand RANKL have been localized in odontoblasts, pulp fibroblasts, periodontal ligament fibroblasts, and in single odontoclasts, the latter finding suggesting an autocrine/paracrine role (Lössdörfer et al., 2002a,b). RANK is coded for by the TNFRSF11A gene.
Osteoclastogenesis is modulated by an inhibitor, osteoprotegerin (OPG, also called TNFRSF11B). OPG is a soluble (decoy) receptor for TRANCE and a member of the TNF receptor superfamily. Osteoclast formation and survival require and are enhanced by transforming growth factor-beta (TGF-beta), which is abundant in bone matrix. TNF-alpha can also induce osteoclast formation in vitro from bone-marrow-derived mononuclear phagocytes, especially in the presence of TGF-beta. Chambers (2000) suggests that the osteoclast is a mononuclear phagocyte directed toward a debriding function by TGF-beta, activated for this function by TRANCE, and induced to become specifically osteoclastic by the characteristics of the substrate or signals from bone cells. Interleukin 1-beta (IL-1β), a potent bone-resorptive cytokine, is a component of the complex pathways leading to root resorption. A balance between IL-1β activity and interleukin receptor antagonist (IL-1RA) may be crucial in the development of periapical lesions (Shimauchi et al., 1998). Interleukin 1 alpha (IL-1α) is also a potent bone-resorptive cytokine and has been found along with TNF-alpha in periapical lesions (Fouad, 1997).
Orthodontic force leads to microtrauma of the PDL and activation of a cascade of cellular events associated with inflammation. Orthodontically induced inflammatory root resorption (OIIRR) has been suggested (Brezniak and Wasserstein, 2002a,b).
(VII) Systemic Factors
The intimate relationship between tooth movement and root resorption suggests that factors affecting tooth movement also affect root resorption. Tooth movement and changes in periodontal tissue in response to orthodontic force in rats vary depending on the time of day the force is applied (Miyoshi et al., 2001). Other systemic factors—such as nutritional factors, metabolic bone diseases, age, and use of drugs—affect orthodontic tooth movement (Tyrovola and Spyropoulos, 2001). Cyclic changes in the estradiol level may be associated with the estrous-cycle-dependent variation in tooth movement through its effects on bone resorption (Haruyama et al., 2002), and estrogen deficiency can cause rapid orthodontic tooth movement (Yamashiro and Takano-Yamamoto, 2001). Calcitonin can inhibit odontoclast activity (Wiebkin et al., 1996). The action of calcitonin on osteoclasts occurs at later stages of osteoclast development, and it inhibits the fusion of committed pre-osteoclasts to form mature multinucleated cells. Bisphosphonates include potent inhibitors of bone resorption used to treat osteoporosis and other bone diseases. Bisphosphonates directly or indirectly induce apoptosis in osteoclasts, which may play a role in inhibition of bone resorption (Reszka et al., 1999). Odontoclasts also undergo apoptosis following exposure to bisphosphonates (Watanabe et al., 2000).
(VIII) Effects of Treatment and Technique
Although treatment and, specifically, the amount of tooth movement may contribute to EARR, a comparison of maxillary central incisor EARR among cases treated with the Tweed standard edgewise technique, the Begg lightwire technique, and the Roth-prescription straightwire technique found no difference among the techniques (Parker and Harris, 1998). However, another study did find more EARR in patients treated with Begg appliances than edgewise appliances (McNab et al., 2000). No difference was also found between patients treated with the “Speed” appliance system and those who received the edgewise straightwire appliance (Blake et al., 1995). Comparisons of different techniques may be confounded by the treatment protocols or decisions of different practitioners, since there is also variation of EARR seen among different practitioners using fixed edgewise appliances (Sameshima and Sinclair, 2001b).
(IX) Effect of Sex
Although some questionable studies have found that orthodontically treated females had a greater incidence of EARR than males (Massler and Perreault, 1954; Kjaer, 1995), several studies have found no difference in EARR between treated males and treated females (Beck and Harris, 1994; Blake et al., 1995; Parker and Harris, 1998; Harris et al., 2001; Sameshima and Sinclair, 2001a).
(X) Interindividual Variability in EARR
Historically, there has been appreciable variability among orthodontic patients in susceptibility to EARR, which might be due to a systemic or innate predisposition to resorption in permanent as well as primary teeth (Becks, 1936; Massler and Malone, 1954; Massler and Perreault, 1954; Reitan, 1957; Newman, 1975). It was proposed that when extreme susceptibility exists, severe EARR would occur even in the absence of any demonstrable causes (Massler and Perreault, 1954). An ethnic dichotomy has been reported, with Asian patients having significantly less EARR than Caucasian or Hispanic patients (Sameshima and Sinclair, 2001a). Familial clustering of EARR has been reported, although the pattern of inheritance was not clear (Newman, 1975; Harris et al., 1997). This implies that there may be a genetic component for susceptibility to EARR.
(X1) Application of Genetic Analyses to EARR
Before undertaking DNA-based studies to elucidate genetic determinants of development or disease expression, one would ideally like to infer as much as possible about the role of genetic factors in phenotypic expression on the basis of development or disease patterns within families and populations (Lander and Schork, 1994). Heritability (h 2) is defined as the proportion of the phenotypic variance attributable to genetic, as opposed to environmental, variance (factors), and its estimation is one of the first objectives in the genetic study of a quantitative trait such as EARR.
Exploration of the hypothesis of genetic influence on EARR according to the sib-pair model found moderately high heritability (h 2) for EARR. The h 2 estimate averaged about 70% for the maxillary incisors and mesial and distal roots of the mandibular first molars, and this accounts for approximately half of the total phenotypic variation (Harris et al., 1997). This means that siblings experience similar levels of EARR in response to orthodontic treatment. Analysis of variable DNA markers is required to indicate which areas of the genome contain genes that are at least partly responsible for the variation seen in EARR associated with orthodontic treatment. Before embarking on DNA analysis related to EARR, we confirmed the heritability of the trait in a sample from the Graduate Orthodontic Clinic at the Indiana University School of Dentistry (unpublished observations) similar to the findings of Harris et al. (1997) (Table). The heritability estimates were significantly above zero in 3 out of the 4 roots examined, at α = 0.05. Excluding the lower incisors, where h 2 = zero, the heritability estimates for EARR ranged from approximately 50% and 60% for the distal and mesial roots of the mandibular first molars, respectively, to 84% for the maxillary central incisors. These estimates are comparable with those determined by Harris et al. (1997), who suggested that a low heritability estimate for the mandibular central incisor could be due to little variation in EARR experienced in the anterior mandibular teeth. Alternatively, since the power to detect heritability decreases with increasing measurement error, it is possible that the difference in heritability estimates between the various roots could be the result of differences in measurement error among teeth. It could be that the measurement error is too high in the case of the lower incisor tooth to permit significant heritability to be detected. This is supported by the mandibular central incisor root apex being the most difficult to identify, due to the superimposition of many teeth in that region on cephalographs in our study. In either case, the lack of heritability for EARR of the mandibular incisors does not necessarily indicate that these teeth are behaving differently from the roots of the other teeth where a genetic component has been documented.
Calculation of heritability estimates is a preliminary step that should be followed by tests for causative agents. With clinical orthodontics, the preliminary goal has been to define the relative contributions of genetics and the environment.
(XII) Genetic Factors Influencing EARR
EARR is likely influenced by a combination of environmental and host factors in a multifactor cascade. Efforts to investigate host factors have focused on a possible genetic component. The intrinsic value of the laboratory mouse as a model stems from several reasons, including: linkage studies; the availability of a dense and detailed genetic map that makes gene mapping in mice practical and efficient; synteny or the genomic conservation of gene order with humans (regions of many mouse chromosomes show conservation of both linkage and gene order with various segments of human chromosomes); and high degrees of homology with human gene sequences (Meisler, 1996; Ehrlich et al., 1997; Nadeau and Dunn, 1998). In addition to the large number of available mutants and inbred strains, mice are excellent hosts for genome manipulation (i.e., transgenic and gene inactivation via gene targeting/homologous recombination). Finally, from a practical standpoint, mice can be easily and economically raised in relatively small facilities and have a short gestation and lifespan, which allows large-scale and longitudinal studies to be performed.
Al-Qawasmi et al. (unpublished observations) subjected genetically disparate inbred strains of mice to standardized orthodontic force magnitude and duration where age, gender, food, and housing were controlled and found variation in the susceptibility or resistance to root resorption attributed to orthodontic force (RRAOF), indicating that genotype is an influencing factor. Mice were grouped into resistant (A/J, C57BL/6J, and SJL/J), intermediate (C3H/HeJ and AKR/J), and susceptible (BALB/cJ, DBA/2J, and 129P3/J) strains. That study identified a seven-fold difference in susceptibility between the DBA/2J (susceptible) and A/J (resistant) mouse strains. It is anticipated that in-depth study of susceptible and resistant mouse strains will facilitate the dissection of precise molecular pathways and genetic mechanisms involved in the pathogenesis of EARR in mice and could provide new information that may contribute to our understanding of mechanisms underlying the cause of EARR in humans by orthodontic force.
(XIII) Candidate Gene Analysis
Evidence of EARR heritability in humans and genetic background in an animal model, along with the present understanding of the cellular events and molecular networks/pathways implicated in EARR, provides a starting point for the investigation of candidate genes. Linkage disequilibrium methods are becoming increasingly more important in the genetic dissection of complex traits. They facilitate evaluation of candidate polymorphisms (Spielman et al., 1993) and the fine-mapping of linked regions (Risch and Merikangas, 1996). In addition to linkage studies, one of the most common means for the evaluation of evidence of an association, or linkage disequilibrium, between a candidate gene and a phenotype of interest is the case-control design. This approach involves the collection of a sample of affected and control individuals whose allele frequencies at the polymorphism in a candidate gene are then compared. A common concern in the case-control design is the spurious detection of association due to population stratification. To avoid the pitfalls of population-based association studies, investigators developed a family-based association test, the transmission disequilibrium test (TDT) (Spielman et al., 1993). The primary advantage of the TDT is that it avoids the necessity of collecting a matched control sample. As originally proposed, the TDT analyzes a nuclear trio consisting of an affected individual and his/her parents. These three individuals are genotyped at a marker in or near the candidate gene. The alleles transmitted by the genotyped parents to the affected offspring are the “affected” sample, and the alleles not transmitted from these two parents are then used as “control” alleles. Through the use of a within-family design, the control sample of alleles is perfectly matched to the affected sample of alleles, since they are transmitted from the same two parents. Thus, spurious association results due to population stratification are avoided. This approach has been extended to allow for the analysis of linkage disequilibrium with the use of quantitative rather than qualitative phenotypes (Allison, 1997; Rabinowitz, 1997; Abecasis et al., 2000; Monks and Kaplan, 2000).
Highly significant (p = 0.0003) evidence of linkage disequilibrium of an IL-1B polymorphism with the clinical manifestation of EARR has been recently reported (Al-Qawasmi et al., 2003a). Individuals homozygous for the IL-1B (+3953) allele 1 have a 5.6-fold (95% CI, 1.9–21.2) increased risk of EARR > 2 mm as compared with individuals who are not homozygous for the IL-1B (+3953) allele 1. The IL-1B gene diallelic variation at +3953 is associated with variable IL-1β protein production (Pociot et al., 1992). Cells from individuals homozygous for IL-1B (+3953) allele 1 have reduced production of secreted IL-1β compared with individuals homozygous for IL-1B (+3953) allele 2 and even individuals heterozygous for IL-1B (+3953) alleles 1 and 2 (Pociot et al., 1992). The IL-1B polymorphism described above accounts for 15% of the total variation of maxillary incisor EARR (Al-Qawasmi et al., 2003a). Rossi et al. (1996) examined IL-1 beta and TNF-alpha production in monocytes from a group of orthodontic patients with severe root shortening and found no significant differences in mean levels between resorption and non-resorption groups. This supports the likelihood that EARR is genetically heterogeneous.
In another linkage disequilibrium study (Al-Qawasmi et al., 2003b), no evidence of linkage was found with EARR and the TNF α and TNSALP genes. Non-parametric sibling pair linkage analysis with the microsatellite marker D18S64 (tightly linked to TNFRSF11A) identified evidence of linkage (LOD = 2.5; p = 0.02) of EARR affecting the maxillary central incisor (Al-Qawasmi et al., 2003b). This indicates that the TNFRSF11A locus, or another tightly linked gene, is associated with EARR. The TNFRSF11A gene codes for the protein RANK.
(XIV) Working Hypothesis for Predisposition to EARR
IL-1β is a potent stimulus for bone resorption and osteoclastic cell recruitment during orthodontic tooth movement (Alhashimi et al., 2001). Expanding on the possible mechanism of the association of the IL-1β allele 1 and EARR, a relatively decreased IL-1β production in the case of allele 1 may result in relatively less catabolic bone modeling (resorption) at the cortical bone interface with the PDL. Stress analysis of orthodontically stimulated rat molars suggests that the initiation of mechanically induced bone resorption is due to fatigue failure within the bone itself (Katona et al., 1995; Roberts, 1999). This would suggest that a deficiency of IL-1β inhibits the resorptive response to orthodontic loads. A slower rate of bone resorption may result in a prolonged stress that is concentrated in the root of the tooth, which in turn could trigger a cascade of fatigue-related events leading to root resorption (Ketcham, 1929).
In summary, EARR may be mediated through impairment of alveolar resorption, resulting in prolonged stress and strain of the adjacent tooth root due to dynamic functional loads (Katona et al., 1995). This scenario contradicts the hypothesis that increased severity of root resorption after orthodontic treatment is related to an increase in alveolar bone resorption (Engström et al., 1988). On the contrary, root resorption may be related to reduced rates of bone resorption at the PDL interface of the root and the alveolar socket. This would result in a prolonged inductive (lag) phase associated with compressed necrotic areas in the PDL prior to alveolar bone resorption. In any event, it is likely that the genetic factors that influence EARR are heterogeneous, with different mechanisms operating in affected individuals, or even site-specific responses in the same individual.
Currently, there are no reliable markers to predict either which patients will develop EARR or the severity of EARR following orthodontic tooth movement (Vlaskalic et al., 1998). The association of the IL-1B (+3953) allele 1 and EARR, which accounts for approximately 15% of the total EARR variation seen in orthodontic patients, has emerged as a potential genetic marker (Al-Qawasmi et al., 2003a). Although the presence of particular root shapes, genetic markers, marked overjet, or the need for extractions may all be associated with an increase in the likelihood of EARR, none is sufficient, alone, to predict EARR reliably. Future analysis of all of these factors (which will also require further discovery of genetic factors such as one or more genes flanking the D18S64 polymorphism, perhaps TNFRSF11A) together should increase the validity of risk estimation, and may even, within reasonable confidence intervals, result in a prediction.
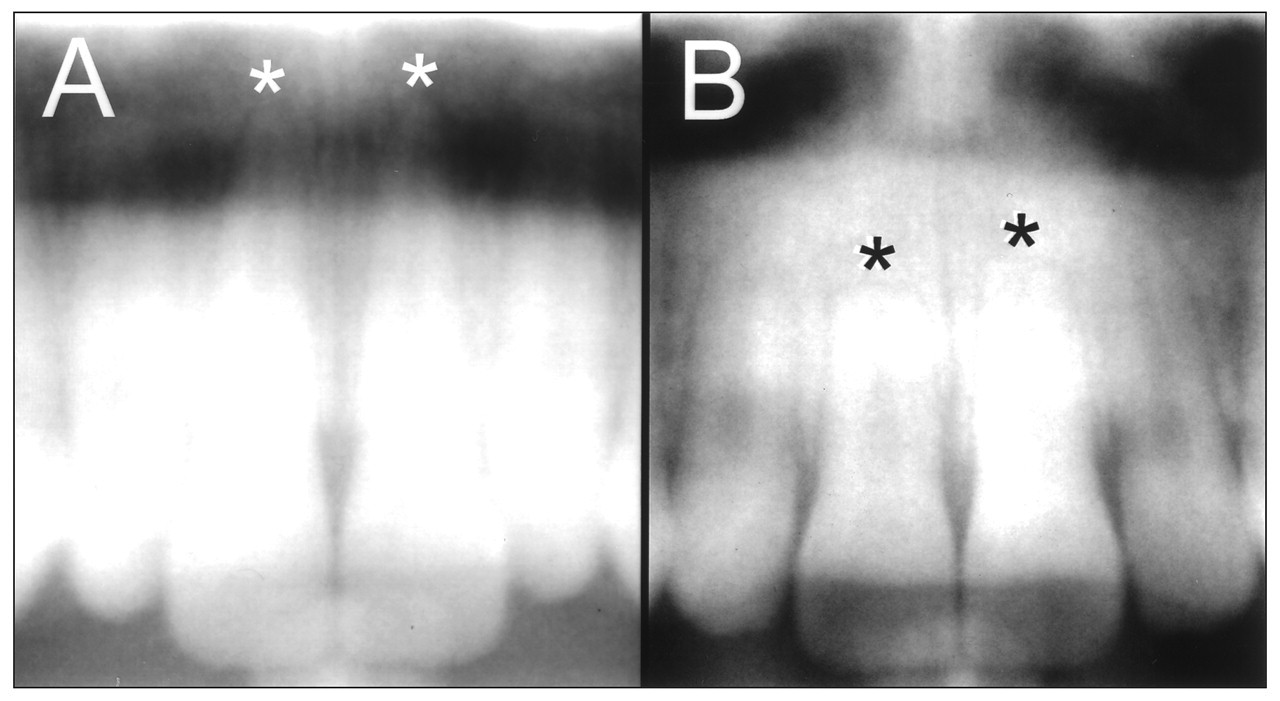
Pre- and post-treatment radiographs (
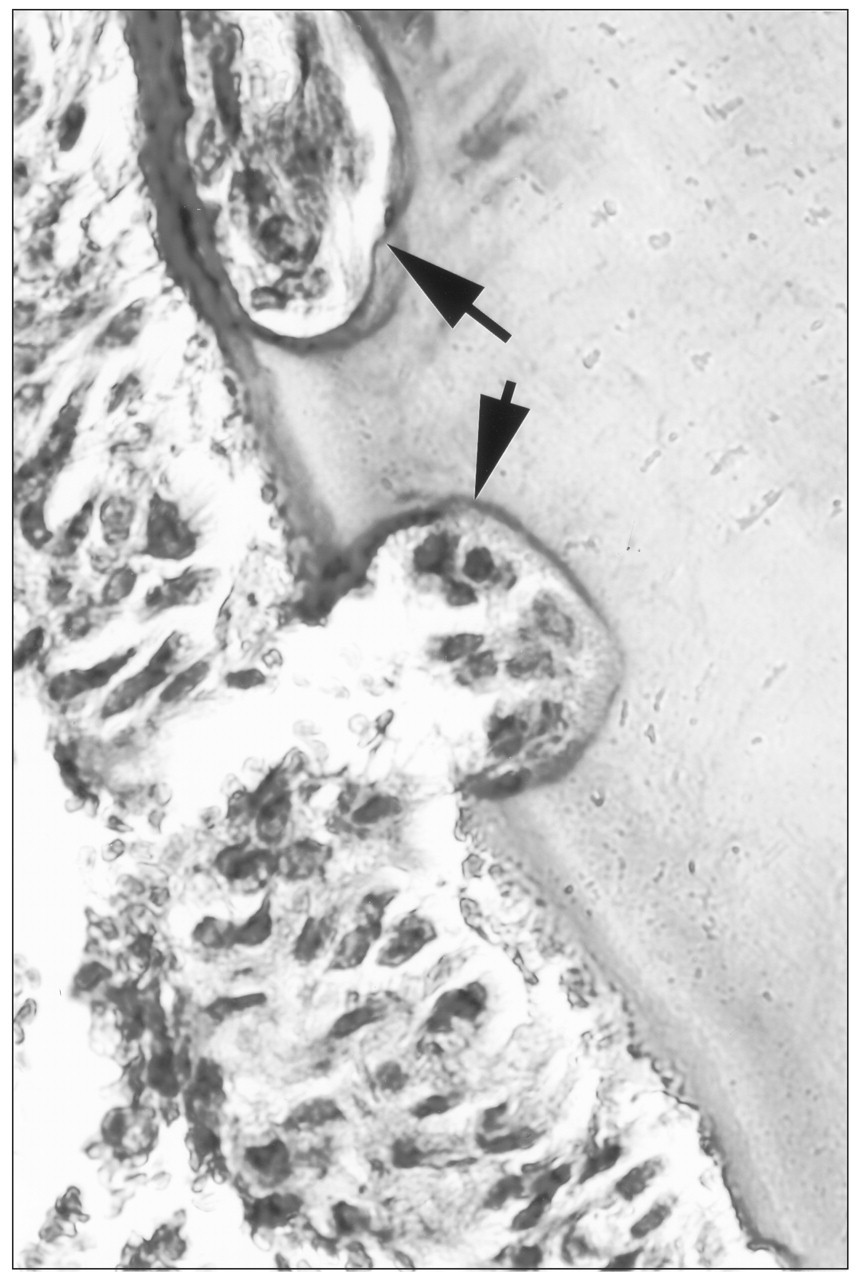
Histological section through a mouse maxillary first molar root tipped mesially with 25 g of orthodontic force for 9 days. Resorption lacunae are indicated by arrows.
Footnotes
Acknowledgements
An American Association of Orthodontists Foundation Biomedical Research Award to JKH supported this work.
