Abstract
Diseases of the dental pulp often have an infectious origin, and treatments are aimed to control infections of the root canal system. Endodontic treatment principles originally evolved on the basis of trial and error, and only in recent decades have scientific methods been adopted to support clinical strategies. Yet, relevant research on the disease processes, their diagnoses, and efficient treatment are rare in the endodontic literature. Hence, the advancement of biologically based knowledge significant to clinical endodontics has been slow. Therefore, many differences of opinion still prevail in this field of dentistry. This review highlights and analyzes the background of some of the more heavily debated issues in recent years. Specifically, it deals with disagreements regarding the clinical management of pulpal exposures by caries in the adult dentition, definitions of success and failure of endodontic therapy, and causes of and measures to control infections of the root canal system. Clearly, a most apparent gap in the published endodontic literature is the lack of randomized clinical trials that address the more significant controversial matters relating to the management of pulpal wounds, medication, and the number of appointments required for the treatment of infected root canals. However, trials in endodontics require extremely long follow-up periods if valid conclusions are to be generated. Therefore, it is not to be expected that there will be rapid solutions to these issues in the foreseeable future.
(I) Introduction
Lesions of the pulp are often infectious, and treatments involve several critical steps that are aimed to restrain infections of the root canal system in teeth. Originally, treatment methodologies in endodontics, like those in many other dental disciplines, evolved based on trial-and-error observations, and only in recent decades have scientific methods been adopted to support clinical strategies. Yet, research reports focusing on issues relevant to the disease processes of the pulp and how they can be diagnosed and managed effectively are rare in the endodontic literature. An assessment of articles appearing in leading endodontic journals reveals that technical aspects of the testing of instruments and filling materials, often by in vitro evaluations in extracted teeth, tend to be the dominating topics (Wu and Wesselink, 1993; Eriksen et al., 2002). As a consequence, the advancement of biologically based knowledge significant to clinical endodontics has been slow. Irving Naidorf (1972), in a critical review on contemporary issues in endodontics, observed: “The preoccupation of dentists with techniques has channeled dentistry (including endodontics) into a state of technical excellence that often is not accompanied by a biological awareness of the basic pathologic problems with which we are dealing or the biologic consequences of our therapy. The schism between clinicians and basic scientists is propagated by a tendency of each group to confer with themselves rather than with each other.” The past 30 years have brought little change, and only modest amounts of the accumulated biological knowledge have found clinical application in the endodontic arena. It is, therefore, not surprising that, in this field of dentistry, there are many contradictory views and opinions as to the proper management of endodontic disorders.
This review highlights and analyzes the background of some of the more significant issues that have been debated in the endodontic field in recent years. Specifically, this review discusses controversies regarding the clinical management of the disease processes associated with the vital and the non-vital dental pulp.
(II) Diseases of the Pulp
Teeth may be exposed to a wide range of injuries that may put the vital functions of the pulp at risk. Some of the adverse influences are non-infectious and include trauma from accidents that interferes with the tissue’s neuro-vascular supply. Other injuries and disease processes produce defects in the tooth structure. Common examples include dental caries, attrition, erosion, abrasion, and various forms of fracture. Also, as a consequence of dental treatment procedures for managing caries and periodontal disease or to replace lost teeth, tooth substance is frequently lost. While injuries per se may interfere with the normal function of the pulp, they may also open up pathways to the pulp for a variety of noxious agents present in the oral cavity of which the resident microbiota are the most important. Normally, the hard-tissue barriers of the tooth, together with the capacity of the pulp to mount an appropriate inflammatory response, allow the tissue, similar to other connective tissues, to sustain injury and bacterial challenges (reviewed by Bergenholtz, 2000). However, direct exposures to the oral environment are a threat to the pulp. Undeniably, the pulp has little self-healing capacity in such a situation because of lack of epithelia, which can bridge the defect and thereby offset deleterious effects of oral micro-organisms. Unless properly treated, even a minute pulpal exposure can provide oral micro-organisms with the opportunity to cause a severe inflammatory lesion that eventually may result in complete breakdown of the tissue. Once the pulp has lost its vital functions, the potential for regeneration is slim in the adult tooth as microbes readily take over the pulpal space and multiply into large numbers. The ultimate consequence of this development is a chronic release of bacterial products that lead to peri-radicular inflammatory lesions and possibly also to undesirable systemic health effects. It is in this clinical context that endodontic therapies are carried out, sometimes to prevent but often in attempts to cure painful and non-painful clinical expressions of infections of the vital and the non-vital pulp.
(III) Controversies in the Management of Exposed Pulp in the Adult Tooth
Nature of the controversy
The management of direct exposures of the pulp to the oral environment by caries or other forms of injury has intrigued the dental profession for hundreds of years. While for a long time it was considered a hopeless task to save it from breakdown by a conservative pulp-capping or pulpotomy procedure, it became apparent, some 70–80 years ago, that pulpal healing is indeed possible. This occurred when it was discovered that wound treatment with calcium hydroxide in a water vehicle resulted in the repair of the exposure site (Zander, 1939; Nyborg, 1955; Schröder, 1985; Yamamura, 1985). The hard-tissue-repair response, although it does not occur consistently (Nyborg, 1958), has since been considered a desirable outcome, since it indicates healing of the pulp.
Despite the fact that pulpal healing and repair have been reported at a high rate in both experimental and clinical follow-up studies, regardless of whether the tissue was injured by caries or accidental trauma (Nyborg, 1958; Cvek, 1978; Haskell et al., 1978; Hörsted et al., 1985; Caliskan, 1995), capping of the exposed pulp has remained a controversial treatment for the adult dentition (Lim and Kirk, 1987; Stanley, 1998; Maryniuk and Haywood, 1990; Ward, 2002). Disbelievers claim that the predictability of carrying out pulp-capping procedures successfully is inferior to the long-term success rate that endodontic therapy (pulpectomy and root filling) offers. Another compelling reason that has been put forth is that if treatment fails, the patient may have to face a rather severe painful condition. In the emergency situation, the dentist may also be challenged with the dilemma of having to identify the offending tooth among several candidate teeth. The subsequent endodontic treatment may furthermore become complex and more difficult to carry out than at the initial exposure, since the reparative phenomena, developing in the pulp during the healing phase, may result in a narrowing of the pulpal space. This is likely to have led many clinicians to uphold a skeptical attitude and to consider pulp capping a less-than-desirable therapeutic alternative (Lim and Kirk, 1987). Proponents of the treatment, on the other hand, allege that far too many pulps are extirpated that could have been saved by a conservative approach (Stanley, 1998; Bender 2000). From a socio-economic point of view, procedures less invasive than pulpectomy are certainly desirable, since they are less tissue-destructive and easier to carry out, thus saving effort, time, and money (Maryniuk and Haywood, 1990).
Between these two points of view are those who consider pulp capping appropriate only for exposures in healthy pulps occurring after accidental trauma. In particular, conservative treatment of exposures in young individuals is deemed suitable, since the pulp tissue volume in these individuals is large and rich in blood vessels and cells, making it apt to respond favorably to physical and microbiological challenges. Based on the analysis of a substantial number of clinical cases, Baume and Holz (1981) argued that direct capping with calcium hydroxide should not be used on pulps which have been exposed by caries. Treatment should therefore be reserved only for teeth which display minimal signs of pulpitis. Such a clinical strategy is currently still advocated (Christensen, 1998; Stockton, 1999; Ward, 2002).
Potential factors of significance to failure of pulp capping
Although often regarded as only one of many factors that can cause failure of pulp capping (Stanley, 1998), there is ample evidence to suggest that the major threat to the pulp is of bacterial origin. A most convincing study in this context is that by Kakehashi et al. (1965), who compared the tissue response to direct exposures of the pulp, after leaving them unprotected to the oral cavity, in molars of germ-free and conventional rats. While the pulps in the conventional rats, as expected, became necrotic after a period of time and obviously infected, as indicated by the development of periapical abscesses, no such responses took place in the germ-free animals. On the contrary, pulp tissue in these animals remained healthy, and, at the exposure site, hard tissue repair developed despite the presence of debris of various origins in direct contact with the tissue.
A most obvious inference of the latter study and similar observations is that pulpal wounds should heal if wound infection can be controlled. In other words, critical to the maintenance of pulpal health after capping should be the integrity of the wound site and whether it can be protected over the course of time from adverse influences of the oral microbiota. Calcium-hydroxide-mediated hard tissue repair is thought to provide such protective functions (Schröder, 1985; Stanley 1998). Indeed, it has been reported that this hard tissue barrier can withstand microbial challenges if the surface restoration is lost (Caliskan, 1994). However, experimental observations in both humans and animals indicate that the new hard tissue, when formed, may not always serve as a dependable barrier (Ulmansky et al., 1972; Goldberg et al., 1984; Cox et al., 1985). In fact, it may become less than homogenous and may display porosities and defects, which can serve as pathways for microbial organisms to reach the pulp, should the integrity of the restoration margins or the entire restoration be lost (Cox et al., 1985). This mechanism of lesion development and pulpal breakdown may be one important reason why pulps show a declining rate of survival over time following capping, as reported by Hörsted et al. (1985) and Barthel et al. (2000). In the study by Hörsted et al. (1985), a retrospective analysis was carried out of 510 pulps that were previously capped because of accidental exposure or exposure due to excavation of deep caries. While some pulps were lost soon after treatment, there was a successive decrease of pulpal survival over time. In a life table analysis, pulpal deaths amounted to 20% at 5 years, a rate that continued to increase until the end of the 10-year follow-up period of the study (Fig. 1). The critical importance of the long-term wound site protection is also supported by the findings of Barthel et al. (2000). In a similar retrospective analysis of pulp-capped carious exposures, the failure rate increased from 45% of treated cases at 5 years to 80% at 10 years—a daunting high rate of pulp-capping failure.
Some failures, especially those that occur soon after the completion of the treatment, may be due to reasons other than a leaky surface restoration. The previous injury and the associated bacterial exposure or both may have impaired the condition of the pulp to such an extent that it is unable to survive, regardless of treatment measure. It is most challenging for a clinician encountering a clinical exposure of the pulp to make such an assessment. This is particularly difficult when there has been caries penetration. In these instances, the tissue may be more or less inflamed, depending on the extent of the bacterial invasion (Reeves and Stanley, 1966; Langeland, 1987; reviewed by Mjör, 2002). It is reasonable to assume that the condition of the pulp plays a decisive role in the outcome of a conservative, pulp-saving treatment. Unfortunately, as yet, there are no reliable means available to guide clinicians as to how far the inflammation has advanced in such cases (see below). Nevertheless, if the bacterial load has been reduced or completely eliminated by the caries excavation procedure, there should be an enhanced potential for recovery. Cox and Bergenholtz (1986) observed, in a study in monkeys, that fresh pulpal wounds challenged by a 24-hour oral exposure resulted in tissue destruction and intense but localized inflammatory cell infiltrates. Following pulp capping, however, the tissue underwent a sequential phase of resolution of the inflammatory lesion. This was followed by tissue re-organization and healing. Within two weeks, hard tissue repair had started to develop against the capping agent, a hard-setting calcium hydroxide compound. Even though extrapolation to clinical conditions from findings in animal models should be undertaken with care, a bacterially induced inflammatory lesion in an exposed pulp may thus not be irreversible. Similar to any infected wound, healing of a pulpal wound should be attainable, if the bacterial exposure is controlled.
In the treatment of deep caries, clinicians run the risk of displacing infected dentin chips into the pulp tissue proper. This is a distinct possibility unless the last layer of carious dentin covering the pulp is very carefully removed. While non-infected dentin debris may become integrated into the hard tissue to generate molecules of significance for hard tissue induction (Magloire et al., 2001; Smith et al., 2001), displaced infected debris may exacerbate the lesion or serve as a source of continual bacterial irritation of the tissue, or both. Consequently, the caries excavation procedure is potentially hazardous and could enhance the risk for an inflammatory breakdown of the pulp.
To circumvent this problem, Cvek (1978) has designed a procedure that has shown promise. The treatment, which he termed “partial pulpotomy”, utilizes a pulp-cutting technique described by Granath and Hagman (1971). The pulp tissue is atraumatically cut with an end-cutting diamond at high speed and with copious irrigation. The wound surface is smoothed and completely debrided and cleaned prior to being capped. Limited case series of clinical follow-ups of treatments using such measures have shown that the five-year pulpal survival rate in young teeth with penetrating caries might be as high as 90% (Mejàre and Cvek, 1993; Caliskan, 1995). It should be noted, however, that no such data exist on more mature adult teeth.
Claimed to be critically important for the outcome of a pulp-capping or partial pulpotomy procedure is the degree of pulpal bleeding upon mechanical exposure (Schröder, 1973; Matsuo et al., 1996; Stanley, 1998; reviewed by Stockton, 1999, and Schuurs et al., 2000). A profuse bleeding response that is difficult to stop is likely to indicate a more or less grave inflammatory involvement of the tissue. It may be equally important that unstoppable bleeding prevents proper management of the wound site and precludes its adequate closure by the capping agent. A blood coagulum may also be susceptible to wound infection (Schröder, 1973). On the other hand, if the infection is controlled, the coagulum may serve as a matrix or mold for tissue re-organization (Cox et al., 1982).
A potential, although not confirmed, risk of a bleeding reaction is that it may occur after the completion of the capping procedure, as the effects of the vasoconstrictor in the local anesthesia wear off. Such bleeding, if extensive, may destroy the tissue. This potential hazard may be reduced in young teeth, where the pulp chamber is large and where internal bleeding may be more readily absorbed. Yet, few studies have been able to confirm an effect of age on the outcome of pulp capping and partial pulpotomies (Hörsted et al., 1985; Zilberman et al., 1989). Hörsted et al. (1985), in their retrospective follow-up study, observed that the five-year survival for 10- to 30-year-olds was over 90%, while only 70% of the pulps had survived among the 50-to 80-year-olds.
Research directions
It is apparent from the published literature that use of pulp capping or pulpotomy procedures to preserve pulps exposed by deep caries will lead to a failure to maintain the pulp’s vital functions in a significant number of cases. Presumably, the reasons for this failure are both technical and biological. Even though treatment is carried out optimally with due consideration and care of wound-site bleeding and protection from the oral microflora, pulpal breakdown may nevertheless ensue. This has led researchers to direct efforts to find alternatives to calcium hydroxide as a pulp-capping agent. Hence, in recent years, considerable efforts have been directed to explore the potentials of dentin bonding systems (reviewed by Schuurs et al., 2000) and various minerals including mineral trioxide aggregate (MTA; PittFord et al., 1996). Numerous biologically active molecules and matrices have also been attempted and tested in a variety of animal models (e.g., Nakashima, 1990; Rutherford et al., 1993; Goldberg et al., 2001; Lovschall et al., 2001; Nakamura et al., 2002). The results so far are promising and certainly challenge the hegemony of calcium hydroxide as the sole agent for treatment of pulpal exposures. Also, new and interesting avenues for the management of pulpal wounds have opened. Yet these efforts will remain futile if one does not find tools that can accurately assess the pre-operative condition of the pulp (Bergenholtz, 2001). Therefore, high on the list of research objectives is improved diagnostic methodology that can predict how far infection and associated inflammatory lesions have advanced in the tissue. To date, clinicians have had to guess as to the true inflammatory status of the pulp, especially in instances of carious exposure. Indeed, there are no means yet available which can help to identify a cut-off point between a reversible and an irreversible inflammatory pulpal condition.
Spontaneous or inducible pain episodes of a lingering character, combined with percussion sensitivity, appear to be the best clinical predictors currently available to suggest an impaired prognosis for pulp capping (Nyborg, 1958; Baume and Holz, 1981). However, no correlation has been found in studies that have compared patients’ experiences of pain with the histological status of the pulp (degree of inflammatory involvement). While the presence of spontaneous, recurrent pain episodes of various durations and intensities were more often associated with partial necrosis and chronic inflammation, there was a poor correlation between the absence of pain and the pulp tissue status in the frequently cited study on extracted teeth (Seltzer et al., 1963). Many pulps had suffered a complete breakdown without having a previous history of pain. On examining records of well over 2000 maxillary anterior teeth that had been treated endodontically, Michaelson and Holland (2002) recently confirmed that diseased pulps are often silent and progress to pulpal necrosis without producing a painful condition and do so more often in elderly individuals. Per se, it is not surprising that the number or extent of inflammatory cells in a given pulp does not correlate with the patient’s report of pain, given that such cells do not stimulate pain fibers directly. Instead, it is the local release of mediators produced during inflammation that seems to regulate the initiation of pain. In fact, observations in patients with the use of a micro-dialysis technique (Hargreaves et al., 1994) have shown significantly higher extracellular levels of bradykinin (Lepinski et al., 2000) and substance P (Bowles et al., 2003) in pulp tissue diagnosed as irreversible pulpitis than that found in normal pulps. However, such observations do not explain why pain is present in some patients and not in others with similar lesions. Inter-individual differences may exist that have an underlying genetic background (Mogil, 1999; Mogil et al., 2000). Therefore, to gain better insights into the complex mechanisms underlying the mediation of pulpal pathology and pain, investigators ought to explore, in future pulp biology research, the potential of advances in molecular biological technology allowing for the cloning, mapping, and sequencing of relevant genes as well as genetic epidemiological studies.
Another significant research target is the pursuit of measures which can promote regeneration rather than repair of diseased pulpal tissue. Except for the imminent risk of a breakdown, the inflammatory process induced by either caries or other microbial sources is known to trigger a number of reparative phenomena. Well-known is the formation of reparative dentin on the root canal walls facing the site of irritation. Also, other more or less undesirable phenomena may occur in a rather uncontrolled manner, especially during tissue healing. Such changes include narrowing of the pulpal chamber by hard tissue deposition on the root canal wall as well as in the soft tissue per se (e.g., pulp stones and dystrophic mineralizations), making a subsequent endodontic treatment precarious (Langeland, 1987). While tissue changes of this nature have been attributed to the calcium hydroxide treatment (Lim and Kirk, 1987), they are more likely to have a link to the inflammatory status of the pulp at the time of capping, since they are rarely seen in experimental treatments of healthy pulps. Increased fibrosis at the expense of cellularity and neuro-vascular supply is another example of scarring. Tissue changes of this nature are likely to impair a pulp’s capacity to withstand iterated microbial challenges and may explain why breakdown suddenly occurs without an obvious cause long after the completion of treatment (Hörsted et al., 1985; see also Bergenholtz, 2000). Therefore, it seems that not only should infection control be of concern in the treatment of pulpal wounds, but also how the inflammatory lesion is monitored to minimize adverse scar tissue formation. No doubt, for optimal function, healing of an injured tissue such as the pulp should ideally be in the form of regeneration rather than repair.
Glucocorticoids (steroids) combined with antibiotics were frequently attempted in the 1960s and 1970s to control pulpal pain and suppress pulpal inflammation (Watts and Paterson, 1988). Indeed, steroids would seem useful as adjuncts to pulpal wound treatment, since they retard and suppress many of the events associated with the production and release of inflammatory mediators (Hargreaves and Seltzer, 2002). However, the use of steroids in this context never gained universal acceptance, since reports of poor wound healing and even pulpal necrosis started to emerge (Paterson, 1976; Langeland, 1981; Watts and Paterson, 1988). Glucocorticoids seem to have come into disuse as an adjunct to treatment in pulpal wounds.
Given the recent gain in new knowledge of the cellular and molecular basis of the inflammatory and repair processes of the pulp (Tziafas 1997; Jontell et al., 1998; reviewed by Byers and Närhi, 1999; Tziafas et al., 2000) and the potential for the application of modern pharmacological strategies, there are now many possibilities for the development of improved and predictable treatment methods for infected and inflamed pulps. Rutherford and Fitzgerald (1995) proposed a principle for the treatment of damaged and diseased pulps which may prove useful in this context. They suggested the placement of bioactive substances bound to a carrier onto amputated pulp tissue for stimulation of wound healing. As the carrier substance gradually resorbs, new tissue is expected to grow into the newly available space. They proposed this model to be suitable for the stimulation of reparative dentin formation. However, soft tissue regeneration followed by regeneration of regular dentin would be a more ideal outcome.
Indirect pulp capping —alternative to direct pulp capping?
Indirect pulp capping as a method for management of deep caries lesions has drawn heated discussions over the years. A more favorable approach might be step-wise caries removal at re-entry operations (Björndal, 2002). However, both procedures rest on the principle of avoiding the pulp tissue by leaving the deepest portions of the carious dentin untouched. While indirect pulp capping is finished by a permanent restoration in a one-step treatment, in the step-wise mode, various amounts of carious dentin are sealed off by a temporary restoration for a period of time. In anticipation of the remineralization of softened non-infected dentin over the pulp and recovery of any prevailing pulpal lesion with hard tissue repair, a re-entry operation is carried out to complete excavation of remaining caries (for review, see, e.g., Björndal, 2002).
This latter mode of treatment has been severely criticized (Nygaard-Östby, 1971; Langeland, 1981). By leaving caries behind to avoid pulp exposure, the clinician is unable to assess the depth of the caries lesion and the status of the pulp. Nygaard-Östby (1971) has drawn attention to the different scenarios that might exist in a case when caries has progressed to near the pulp tissue. On the one hand, an unbroken layer of primary dentin may separate the pulp from the caries lesion. In such an instance, the inflammatory involvement of the pulp is normally small if present at all (Reeves and Stanley, 1966; Langeland, 1987; Kamal et al., 1996) and thus is inconsequential. On the other hand, caries may have invaded reparative dentin and even advanced to the pulp tissue proper. Pulpal inflammation is then evident due to bacterial invasion of the tissue (Reeves and Stanley, 1966; Langeland, 1987), a response which can progress to an abscess and partial necrosis. In both of these scenarios, leaving infected dentin permanently is not likely to change the preconditions for a successful outcome of a pulp preservation effort. Similarly, in the case of a healthy layer of remaining dentin, carious dentin in situ is of no benefit, and only impairs the prospect of a good restoration and a healthy pulp being retained in the long term. In a situation where bacteria have invaded the tissue, it remains to be shown that partial excavation of caries can actually suppress the infection. No such documentation is yet available. In this context, Jordan et al. (1978) reported results from a follow-up of 24 molars which had signs of periapical involvement (small periapical lesion or widened apical periodontal space). These teeth had responded as vital to testing and had no history of spontaneous or prolonged pain. Of interest is that 11 teeth showed resolution of the periapical lesion and were judged clinically healthy at the final examination, from 10 months to 7 years following treatment. But 13 of the 24 treatments failed. Spontaneous recurrent pain occurred in 12 instances, and one tooth developed an acute swelling after 3 days. For the reasons stated above, and the fact that convincing clinical data in support of the procedure have not surfaced over the years, indirect pulp capping as an attempt to preserve a functional and asymptomatic pulp tissue should be discouraged.
Step-wise excavation, on the other hand, has the advantage that the clinician can evaluate, on re-entry, whether vital pulpal conditions prevail and at the same time can check the integrity of the hard tissue covering the pulp. The method has been spurred by promising results in randomized clinical trials (Magnusson and Sundell, 1977; Leksell et al., 1996). In these studies, complete caries removal in one step was compared with the step-wise procedure as to the number of pulpal perforations. Significantly fewer pulpal exposures were recorded in primary (Magnusson and Sundell, 1977) and young permanent teeth (Leksell et al., 1996). Notably, regardless of direct or stepwise excavation, the teeth with no pulpal exposure presented with normal clinical and radiographic conditions at the final clinical check-up. Björndal and Thylstrup (1998) confirmed these findings in an uncontrolled follow-up of step-wise treatments carried out by general practitioners. Only 5 of 94 teeth with deep caries lesions, which the clinicians believed would result in pulp exposure if excavated in a single session, resulted in exposure. On the basis of these findings, it is reasonable to conclude that the procedure of excavating caries in a step-wise fashion may avoid inadvertent (iatrogenic) exposure of pulps. It is believed that by closing off the caries lesion from the oral environment for a period of time, acid-producing bacteria will be deprived of their nutritional supply. As a result, less acid will be generated, which in turn may allow for remineralization of softened but not infected dentin. Support for this potential comes from numerous reports of reduced microbial counts in caries lesions sealed off from the oral environment (Fisher, 1972; Fairbourn et al., 1980; Leung et al., 1980; Weerheijm et al., 1999; Björndal and Larsen, 2000; Maltz et al., 2002).
In assessing the evidence in support of the step-wise excavation procedure, one has to take into consideration that, in the clinical follow-ups so far published, the cases were carefully selected. It is not unreasonable to assume that, in many of these instances, there was a favorable pulpal condition because of the absence of or limited bacterial invasion of the tissue, owing to a more or less intact layer of primary dentin. So far, there are no data to show that more pulps are actually being saved by such a measure in comparison with a direct capping procedure. Consequently, there is an obvious need to validate whether step-wise excavation is a worthwhile clinical procedure. Controlled prospective clinical trials with due consideration of relevant clinical parameters are therefore called for.
(IV) Controversies in Endodontic Therapy
Nature of the controversy
Possibly one of the strongest factors contributing to the controversies often encountered in the endodontic field is the lack of understanding that the disease processes of the pulp generally have a microbiogical etiology. While the critical steps to accomplish effective treatments from a microbiological point of view have been outlined and tested over many years of research, the acquired knowledge is often overlooked or disregarded (Spångberg, 1998). Indeed, there is a widespread reliance on the technical performance of endodontics, and there are strong beliefs that once a technically satisfactory result is attained, as judged by the radiological appearance of a root filling, successful treatment has been achieved. This view has been spurred in recent years by a remarkable improvement of the technological aids in this field of dentistry. Clinicians can now carry out intracanal instrumentation more predictably than in the past without straightening or blocking out root canals or causing other procedural errors that were prevalent with the old armamentarium. Also, improved methods for effective filling of root canals have been developed. Per se, this is not an argument to lessen the important role of technical skills in endodontics. As a matter of fact, surveys carried out in many countries (recently reviewed by Kirkevang and Hörsted-Bindslev, 2002) have shown that substandard technical performances are more often associated with persistent inflammatory lesions in the peri-radicular area, than root fillings of high technical standard. However, seemingly adequate fillings are no absolute guarantee of a successful healing result. Moreover, from a technical perspective, an ideal treatment may not always be feasible. Yet, the result may turn out to be biologically excellent.
While recent technological developments have been valuable and most welcome, the strong emphasis on techniques in modern endodontics has no doubt detracted attention from the primary aim of endodontic therapy, which is to prevent or treat endodontic infections. As a result, less weight appears to have been placed on the means by which root canal infections can be controlled. The fact that endodontic instrumentation today involves less time than in the past has also brought forward the view that endodontic treatment should preferably be completed in one session. Consequently, we have found it pertinent to review some of the biological background and the critical measures aimed to reduce or eliminate root canal infections, measures which have gained support in many clinical trials and experimental studies.
Success and failure of endodontic therapy
Definition of success and failure is critical in the discussion of clinical validity of endodontic treatment alternatives. However, over the years it has been difficult for clinicians to agree upon unifying criteria for success and failure (Kvist, 2001).
In simple terms, success of a given therapy in medicine or dentistry may be defined as the result obtained which achieved the initial treatment goal. The term “failure” consequently may be defined as a treatment that did not reach the objective or fell short of the acceptance level—for example, in terms of tissue function, elimination of pathology, comfort, or even survival. From a biological perspective, an endodontic failure is associated with an inflammatory process in the peri-radicular support structures of a tooth. Such a lesion is often evident in radiographs as an osteolytic bone lesion in the periapical region. Micro-organisms residing in the root canal system and, less often, organisms in the peri-radicular tissue compartment support the pathological process. The lesion may have developed after endodontic treatment of a tooth with a vital pulp. It may also be an expression of an expansion or an exacerbation of a peri-radicular lesion that existed when the treatment was initiated. The treatment failure is due, in the overwhelming number of cases, to an infectious process which has either been caused by a breach of asepsis during the treatment of a vital pulp or has resulted from lack of success in eliminating an existing root canal infection. Leakage of bacterial elements from the oral environment is another significant cause of endodontic treatment failure (see below).
"Treatment failure" is a loaded expression. Thus, success or failure may be measured in terms of completing the treatment without post-operative pain, or in retaining the tooth for a time period, or in treating a root canal infection. Some definitions are broad and consider success to be the retention of a tooth. Others may include lack of painful symptoms. To this may be added certain radiographic criteria, such as the absence or decrease in size of a periapical radiolucency. It is clear to most clinicians that the development of a peri-radicular osteolytic bone lesion after endodontic treatment of a tooth with a vital pulp is an untoward reaction. Yet, from a clinical practice point of view, it may sometimes be considered a successful treatment if the patient experiences no pain and if the lesion does not continue to grow in size. In a pre-existing lesion, the mere size decrease, or lack of expansion, may also be considered success.
Often, the attainment of a pain-free tooth overrides all other considerations. Thus, in many cases the definition of success is in the eye of the beholder. In that sense, from a patient’s perspective, the terminology contains a value component, which serves as a normative guide as to the kind of clinical action that ought to be taken (Reit and Kvist, 1998).
This wide definition of success is incompatible with a scientific approach to outcome measurements. For scientific purposes, reproducible and stringent criteria, which carry little margin for interpretation, are necessary. The often-used strict criteria established by Strindberg (1956) rely heavily on radiographic observations, although they also consider subjective and objective clinical signs of root canal inflammation and infection. The observer, however, has been proven to be a highly significant variable which must be carefully controlled for in outcome studies (Goldman et al., 1972, 1974; Reit and Gröndahl, 1983; Reit and Hollender, 1983; Molven et al., 2002).
It is obvious that scientists and clinicians apply different meanings to success and failure of endodontic therapy. Although a persistent apical lesion after root canal treatment may represent a failure in a biological sense, it may not require further attention in the clinical setting. Personal values, risks for a variety of adverse complications in conjunction with either a conventional or surgical re-treatment, or monetary considerations are but some of the significant factors in treatment decisions. To better guide clinical decision-making, more knowledge is needed to establish when serious consequences of a residual lesion are to be expected. If risks for worsening of the condition in terms of a subsequent flare-up and adverse systemic effects turn out to be low, the concept of success and failure in endodontic therapy may have to be re-assessed from a biological point of view.
Coronal leakage in endodontic treatment failures
In recent years, the concept of coronal leakage has taken on a life of its own in various attempts to explain endodontic treatment failures. There are numerous in vitro studies that have used radioisotopes, dyes, and microbes to demonstrate the presence of leakage along root fillings (Marshall and Massler, 1961; Swanson and Madison, 1987; Torabinejad et al., 1990). Considering the fact that practically all of the numerous studies in vitro consistently show leakage along fillings, it is surprising that endodontic treatment ever succeeds. The few in vivo studies on this issue, however, show a slightly different picture. Madison and Wilcox (1988), in an experimental study in the monkey, found that all samples—including the negative controls—leaked.
Another extension of the coronal leakage issue is the attempt to correlate outcome of endodontic treatment to the quality and completeness of the coronal restoration. In a survey of full-mouth radiographs on patients seeking treatment at a US dental school, Ray and Trope (1995) came to the conclusion, disquieting for the endodontic profession, that the technical quality of the coronal restoration was more important for the result than the technical quality of the endodontic treatment. It was found that poor restorations—e.g., those with radiographic signs of overhangs, open margins, or recurrent decay, even in combination with good endodontic fillings (canals filled without voids and to within an ideal 0–2 mm off the radiograph apex)—had a negative impact on outcome. Thus, the findings suggested that leakage of bacteria and their products along the margins of the restoration and the root filling could have induced or maintained apical inflammatory lesions. More understandable was the observation that a very high failure rate was seen when both coronal restoration and endodontic fills were defective.
This study generated considerable upheaval by challenging established endodontic treatment concepts. It has long been held that thorough and high-quality management of the root canal, including filling the canal properly, is a highly desirable feature. The study further seemed to corroborate the numerous in vitro observations referred to above, which had shown that even under the most ideal conditions, i.e., in extracted teeth, leakage-free root fillings may not be obtained (Wu and Wesselink, 1993). However, the findings of Ray and Trope (1995) have, although only to some extent, been corroborated by other similarly designed studies (Kirkevang et al., 2000; Tronstad et al., 2000; Hommez et al., 2002). Kirkevang et al. (2000) suggested that both the quality of the endodontic treatment and the coronal restoration affect the health of the peri-radicular tissues in a synergistic way. Tronstad et al. (2000) attached some importance to the coronal restoration in cases where the endodontic treatment appeared satisfactory. However, if the quality of the endodontic treatment was poor, the quality of the restoration was of no significance. In addition to radiographic criteria for signs of marginal leakage or decay, Hommez et al. (2002) scored the restorations clinically using modified Ryge’s criteria. They observed that both the quality of the coronal restoration (judged radiographically) and the length and homogeneity of the root-canal fillings significantly influenced periapical health.
Although providing interesting data, it is far from clear if these studies measure the correct interrelationship, that is, the importance of coronal restoration to periapical health. What this kind of analysis may simply reveal is the impact of both suboptimal endodontic work and deficient restorative work. When root canal fillings are so poor that lateral voids are detectable, the issue of coronal leakage is unimportant. Consequently, radiographically detectable defects in a root canal filling are so large compared with the sizes of micro-organisms that the question of leakage is a moot point. Another factor often used to assess root-filling quality is the extension of the filling in relation to the apex. It is true that extensive over- or underfilling of the root canal is associated with an increased rate of failure. It is doubtful, however, that failure in these instances is in any way associated with coronal leakage. With regard to the quality of coronal restorations, it is not clear how this factor was adequately controlled for in the studies by Ray and Trope (1995), Kirkevang et al. (2000), Tronstad et al. (2000), and Hommez et al. (2002). Indeed, there are few restorations that do not leak clinically. At best, a radiographic assessment of restoration quality may be a qualitative measure rather than a quantitative measure.
A recent clinical follow-up study has challenged the significance of the coronal leakage concept (Ricucci et al., 2000). In this study, a sample of 55 root-filled teeth, optimally treated by the same dentist, had lost their restorations or were affected by extensive caries, or both. These teeth were carefully matched, one to one, with well-restored root-filled teeth with regard to pulpal and periapical diagnosis, tooth type, age of patient, and observation period. The observation period after endodontic treatment was at least 3 years. The time for bacterial exposure was months or years. Although there was a three-fold increase in the odds ratio for lesions to occur in teeth with poor coronal seal or lack of seal, the difference did not reach statistical significance, which could have occurred in a larger sample. However, the overall number of periapical osteolytic lesions in the material was small, and in most pairs the periapical structures appeared normal, suggesting that coronal leakage might be of minimal importance in well-treated endodontic cases.
In conclusion, it is difficult to completely ignore the numerous observations in vitro on the topic of coronal leakage. However, more clinical evidence is required to substantiate its role in long-term failure in properly treated endodontic cases.
Location of micro-organisms
Vital to the discussion on the efficacy of antimicrobial measures undertaken to combat root canal infections during endodontic treatment is the location of the micro-organisms once the pulpal tissue has become necrotic. On rare occasions, pulpal necrosis may initially be aseptic, but the necrotic tissue will sooner or later become infected (Bergenholtz, 1974). The infected necrotic tissue initiates both innate and acquired immune responses (Kakehashi et al., 1965; Barnes and Langeland, 1966; Sundqvist, 1976; Torabinejad et al., 1985; Stashenko et al., 1992). As a result, bone is resorbed to provide space for the host defense activities (Stashenko et al., 1998). The ensuing inflammatory lesion has long been regarded as the first line of defense against an all-out invasion of micro-organisms into the peri-radicular bone. Conceptually, it is in the expanding phase that micro-organisms may invade the periapical tissue compartment. However, once the lesion has entered a more established form, it is believed that bacteria are eliminated from the soft tissue lesion. Indeed, careful microscopic examinations have failed to identify micro-organisms in inflammatory periapical lesions associated with necrotic pulps in more than occasional instances (Nair, 1987). Consequently, organisms are then confined to the root canal space and thus are accessible for conventional endodontic treatment measures. Yet, numerous publications have reported the presence of micro-organisms in seemingly quiescent periapical inflammatory tissue lesions (Sundqvist and Reuterwing, 1980; Borssén and Sundqvist, 1981; Nair and Schröder, 1984; Happonen, 1986; Tronstad et al., 1987; Sjögren et al., 1988; Iwu et al., 1990; Wayman et al., 1992; Abou-Rass and Bogen, 1998), and these findings question the validity of the concept of the sterile apical granuloma. There are clear clinical implications associated with this issue, in that a persistent chronic extra-radicular infection would not be treatable by conventional non-surgical means and therefore would require surgical intervention or chemotherapeutic systemic medication, or both.
An important question in this context is when and how often an extra-radicular infection may pose a clinical treatment problem. Unfortunately, prevalence data have not been established. Therefore, to gauge the significance of the problem, it is important to distinguish under which conditions bacteria may be present in the extra-radicular tissue. In the expansion phase, before organization of the host tissue defense, or through an exacerbation of an established lesion, bacteria are no doubt able to invade the peri-radicular tissues and form an abscess, which, if untreated, may drain to a skin or mucosal surface. An exacerbation of this nature may be due to either increased pathogenicity of the root canal microbiota or decreased host tissue response (Sundqvist et al., 1979, 1989). Numerous studies have confirmed that during the development of these lesions, several micro-organisms, normally residents of the infected pulp space, will be found in the abscessed tissue (e.g., van Winkelhoff et al., 1985). After drainage, the lesion enters into a chronic stage, and a tissue cavity with a sinus tract may remain that can allow for the continuous presence of micro-organisms in the peri-radicular tissues and on the exposed root surface (Tronstad et al., 1987). A chronic sinus tract will allow bacteria to colonize the tract as well as the root surface around the apical foramen where the abscess originated. Such a sinus tract is not much different from a deep periodontal pocket and is in many instances only a few millimeters deep. The fact that plaque and calculus have been reported in such cases is not surprising (Tronstad et al., 1990), but it must not be interpreted that bacteria prevail in solid periapical tissue granulomas.
Some studies on bacteria in periapical tissue lesions have questioned the procedure of sampling the periapical region via surgical access (Tronstad et al., 1987; Iwu et al., 1990; Wayman et al., 1992; Abou-Rass and Bogen, 1998). Such an approach involves poor control of the risk for contamination with extraneous organisms (Möller, 1966). In addition to the nearly impossible task of obtaining sterile access, it is easy, when sampling, to scrape or suck out bacteria from the apical foramen. It is also impossible during the surgical operation and the subsequent sampling procedure to observe if the tissue surrounding the apical foramen has a small abscess cavity or pocket cyst that would allow bacterial presence in the tissue as a continuum of bacterial outgrowth from the root canal space.
Immunohistochemical markers have provided good evidence for the presence of both Actinomyces israelii and Proprionibacterium propionicum in confirmed well-organized but treatment-resistant periapical tissue lesions (Sundqvist and Reuterwing, 1980; Borssén and Sundqvist, 1981; Nair and Schröder, 1984; Happonen, 1986; Sjögren et al., 1988). Both organisms are not uncommon colonizers of infected root canals and may have entered the lesion during the expansion phase. The presence of these organisms in treatment-resistant cases may also be explained by their displacement into the peri-radicular tissue during the treatment procedure (Sundqvist and Reuterwing, 1980; Sjögren et al., 1988).
Although the debate has been heated at times, the concept of the sterile peri-radicular granuloma must still be regarded as valid, although certain bacterial species, such as the aforementioned Actinomyces israelii and Proprionibacterium propionicum, may sometimes survive in the tissue. This concept is still compatible with the presence of micro-organisms in peri-radicular tissue with surface contacts such as sinus tracts, pocket cysts, periodontally compromised teeth, and root fractures. Therefore, the main targets for treatment are the organisms located within the confines of the root canal space, and one can conclude that rarely is there a need for systemic use of antimicrobials to remedy treatment failures.
Antimicrobial agents for intracanal disinfection
While often prevented by the host defense from entering the peri-radicular tissues, micro-organisms in the root canal space will rapidly invade the dentinal tubules and create a dentin infection (Chirnside, 1957, 1958). Dentin infection is a significant treatment problem and needs to be addressed during the treatment phase. Although a good root canal filling may entomb many micro-organisms remaining in the root canal dentin, root fillings do not invariably provide a complete seal of root canals. Hence, if defects in the root canal filling exist that communicate with the periapical tissue, tissue fluid may enter the voids and boost bacterial re-growth. Subsequently, bacterial inflammatory mediators will be produced and released periapically to initiate or maintain a tissue lesion. This is the primary reason for the use of antimicrobial agents in endodontic therapy.
Antimicrobial agents are applied during two different stages of the endodontic treatment. First, a chemically active irrigation solution is used during the instrumentation of the pulp space, and second, an antiseptic medication is often applied in the root canal space between treatment sessions. There has been little debate regarding the most effective antimicrobials. For irrigation purposes, sodium hypochlorite has remained the chemical of choice, and calcium hydroxide is the most commonly accepted inter-appointment antiseptic agent. The debate has raged for years, however, over the appropriate concentration of sodium hypochlorite. Recently, the need for calcium hydroxide medication has also been questioned.
Irrigation solutions
Sodium hypochlorite is an effective antimicrobial agent. Enterococci, Actinomyces, and Candida, proven to be some of the more difficult micro-organisms to eliminate from root canals, are indeed sensitive to this chemical. In recent years, E. faecalis in particular has attracted considerable interest as a potential pathogen, in that it is a common isolate in root canals of failing treatments (Molander et al., 1998; Sundqvist et al., 1998) and obviously one of the hardest organisms to eliminate once it has established itself in root canals (Chávez de Paz et al., 2003). Sodium hypochlorite, at a concentration of 0.0005%, effectively kills E. faecalis within 3 minutes following direct exposure (Zehnder et al., 2002). A 0.25% concentration was required to completely kill E. faecalis in infected dentin blocks within 15 minutes (Zehnder et al., 2002). A. israelii were killed within 1 minute by 1% sodium hypochlorite (Barnard et al., 1996). However, sodium hypochlorite may be less effective in killing Candida albicans within the normal treatment time available for root canal therapy. Ferguson et al. (2002) reported killing of C. albicans within 48 hours at less than 10 μg/mL concentration of sodium hypochlorite. In contrast, Sen et al. (1999) found that Candida albicans inoculated in root canals in vitro were killed within an hour following exposure to 1% sodium hypochlorite. These differences in findings may be due to the isolation of the micro-organisms in the dentinal tubules and/or to an inhibiting effect of the dentin tissue components (Haapasalo et al., 2000).
Under clinical conditions, the use of sodium hypochlorite has been proven less effective. Thus, several clinical studies (Cvek et al., 1976a; Byström and Sundqvist, 1983, 1985) have shown that, even at a high concentration, nearly one-third to one-half of root canals remain infected after instrumentation and irrigation. It has also been demonstrated that lower concentrations are as effective—or ineffective—in eliminating bacteria (Cvek et al., 1976a,b; Byström and Sundqvist, 1983, 1985; Dalton et al., 1998; Shuping et al., 2000).
Various ramifications of the main root canal, including branching, lateral canals, and fins, often compromise effective mechanical cleansing of the root canal space. Owing to its ability to dissolve necrotic tissue, sodium hypochlorite is certainly helpful in these instances and, by getting into unreachable spaces, is likely to enhance disinfection. From various in vitro studies, it is obvious that the higher the concentrations, the better the tissue-dissolving effect (The, 1979; Zehnder et al., 2002). This enhanced effectiveness, however, is obtained at the high price of increased tissue toxicity (Spångberg et al., 1973; Pashley et al., 1985), with no proven clinical gain in bacterial killing (Cvek et al., 1976a; Byström and Sundqvist, 1983, 1985). At very low concentrations, sodium hypochlorite induces an inflammatory response when it comes into contact with vital tissue (Rutberg et al., 1977) (Table). At higher concentrations, the tissue irritation may be substantial (Pashley et al., 1985). Hence, displacing highly concentrated sodium hypochlorite into the periapical tissue may cause severe tissue damage (Hülsmann and Hahn, 2000). Consequently, the combination of an efficient microbicidal effect at low concentration and a proportional increase of toxicity with a higher concentration, but with no demonstrable enhanced clinical effectiveness, suggests that sodium hypochlorite at concentrations higher than 0.5–1.0% offers little therapeutic value.
Calcium hydroxide
Calcium hydroxide has extensive use in endodontic therapies. Hermann (1920) introduced this agent for treatment of infected root canals on the basis of in vitro as well as in vivo studies. As mentioned above, calcium hydroxide also became frequently used in vital pulp therapies (Hermann, 1930), and, before the Second World War, European immigrants (Zander, 1939) introduced dentists in the USA to its use. Even though Matsumiya and Kitamura (1960) clearly demonstrated, in the dog, that calcium hydroxide packed into infected root canals eliminated or reduced micro-organisms, it took many years before the agent gained universal acceptance in endodontics. For quite a while, calcium hydroxide was solely recognized for its ability to induce hard tissue repair in the apical portion of root canals, especially of immature teeth with an infected pulp necrosis (Cvek et al., 1976b). Although calcium hydroxide has been used routinely in Scandinavia, for more than 30 years, for the dressing of root canals between treatment sessions, it was not until the mid-1980s that Byström and Sundqvist (1985) promoted it as an antimicrobial agent. The clinical value of calcium hydroxide in endodontic therapy is now linked to its beneficial antimicrobial effects without a simultaneous risk for severe tissue damage.
Byström et al. (1985) found that infected root canals treated with calcium hydroxide for one month were successfully disinfected in 97% of the cases. In a later study, from the same clinic, the effectiveness was confirmed even when the dressing was retained in the root canal for only one week (Sjögren et al., 1991). However, data from other studies cast doubt on the effectiveness of calcium hydroxide as an intracanal disinfectant (Reit and Dahlén, 1988; Ørstavik and Haapasalo, 1990; Barbosa et al., 1997; Peters et al., 2002). In these studies, not as many bacteria-free root canals were recorded after treatment as those in the Byström et al. (1985) and Sjögren et al. (1991) studies. Cvek et al. (1976b), in a study of 141 traumatized incisors, of which 95 showed initial growth of bacteria, found that 76 teeth (80%) still harbored micro-organisms after initial instrumentation and irrigation. After 3 months of calcium hydroxide dressing, 11 teeth (12%) still exhibited growth of cultivable bacteria. This number did not change after an additional 3 months of calcium hydroxide dressing.
The effectiveness of calcium hydroxide in vivo may vary depending on the initial composition of the microbial flora and the effectiveness of the cleansing procedure carried out. The antimicrobial effectiveness is believed to be due to an increase of pH in the bacterial environment. However, some micro-organisms are less susceptible to high pH. E. faecalis tolerates pH up to 11.5 (Byström et al., 1985), which may be one explanation as to why this organism survives antimicrobial treatment with this agent. Although the calcium hydroxide slurry will have a pH of about 12.5, this level may not be reached in all parts of the root canal space and is unlikely to be attained in the dentinal tubules, where bacteria may be lodged. Tissue remnants and dentin debris may also modify the calcium hydroxide effect (Haapasalo et al., 2000). Consequently, calcium hydroxide may not be as effective an antimicrobial agent as once believed (Byström and Sundqvist, 1985; Peters et al., 2002). Yet, it must still be regarded as a most effective intracanal medicament by virtue of its antimicrobial effects combined with its ability to suppress nutritional supply for growth and multiplication of any remaining organisms.
Apical terminus
Although debated for decades, the apical extension of root canal instrumentation and root filling is still the focus of much disagreement in the endodontic field. To understand the biological basis for a decision as to where instrumentation should be terminated in a case of vital although inflamed pulp, it is important to understand the preconditions for tissue healing in the apical part of the root canal. A most ideal outcome of an endodontic treatment is hard tissue closure, which permanently separates the root canal content—the root filling—from the periapical tissues and thus prevents chronic irritation and foreign body reactions by material components (Ricucci and Langeland, 1998). Clinical studies have indicated that, following an aseptically performed partial pulpectomy, where some millimeters of the apical pulp tissue remain, the natural healing process takes place (Ketterl, 1955; Mayer, 1964; Engström and Lundberg, 1965). If an appropriate filling material is used and/or dentinal filings have been packed against the pulp stump, healing will occur, with the formation of cementum-like tissue (Engström and Spångberg, 1967; Tronstad, 1978) (Fig. 2). Consequently, it is hard to rationalize why the apical foramen should be pierced with a patency file, as is sometimes advocated.
The necrotic infected pulp presents with a different challenge than the vital pulp, due to the potential presence of micro-organisms at the apical foramen (Nair, 1987; Nair et al., 1990). Therefore, the length of instrumentation may be more critical (Molven, 1976), since cleansing and disinfection ought to be carried out for the entire length of the infected tissue. Antimicrobial agents, with their limited in situ effect, are not likely to kill microbes in the apical portion of the root canal effectively without the synergistic action of mechanical cleaning. There are few published long-term outcome studies where the apical preparation has been carefully recorded. Sjögren (1996) established an optimal outcome for an apical terminus of 0–2 mm off the radiographic apex. However, the exact determination of where the root canal ends is a difficult if not an impossible clinical task. When the apical foramen is intact and located close to the anatomical apex of the root, the root canal ends with a constriction of about 0.5 mm, on average, inside the canal (Stein et al., 1990). Yet, there is great variability, and in cases of apical periodontitis, the apical foramen may often be open due to resorptive processes (Fig. 3). A defined apical constriction is then usually absent. In many cases, the very apical part of the root canal may contain an extension of inflammatory tissue (Nair, 1987). Since this tissue will prevent micro-organisms from reaching the most apical part of the root canal, this condition is certainly beneficial from a treatment aspect. On the other hand, removal of this tissue to obtain an effective filling is often precarious. Ideally, the entire root canal should be instrumented in teeth with infected pulp necrosis, and the clinician must, in each case, make an individual assessment of where the root canal ends. Yet, the instrumentation must be tempered by the need to avoid over-instrumentation and lateral transportation of the canal exit, so-called “apical zipping”. An over-prepared canal easily promotes overfilling. Even though a slight excess fill may not bring serious consequences, an over-prepared canal is often difficult to fill properly. This might be the reason why overfilled root canals more often result in impaired treatment outcomes than when fillings end within the confines of the root (Bergenholtz et al., 1979).
In recent years, electronic length-measurement devices have come into frequent use for determination of the root canal terminus and seem to be valuable tools to prevent over-instrumentation. Commercially available devices, able to analyze several impedance values at different frequencies concurrently, have been shown, in several combined in vitro and in vivo studies, to be able to indicate the location of the apical foramen accurately, regardless of pulpal diagnosis (Mayeda et al., 1993; Shabahang et al., 1996; Pommer et al., 2002). Furthermore, the use of a combination of electronic measurements and radiographs of a trial file has been demonstrated both to be more accurate than the use of trial file radiographs alone (Brunton et al., 2002) and to improve the length quality of the final filling (Fouad and Reid, 2000). The performances of these devices, however, are limited by canal obstructions such as intracanal mineralizations and iatrogenic blocks by dentinal shavings and ledges.
One vs. several visits
In recent years, a most heated discussion has emerged as to the need for more than one treatment session in endodontics. Traditionally, treatment has been divided into two or more appointments before the placement of a permanent filling, allowing the clinician to enhance root canal disinfection, to increase patient comfort, and to observe the progress of healing (Trope and Bergenholtz, 2002). Unfortunately, the arguments for the number of patient visits are often unclear. When one carefully considers the case selection criteria from advocates of one-visit treatments, it becomes clear that there are many exclusion criteria which are not articulated in the debate. Ashkenaz (1984) suggested that multi-rooted teeth should be excluded. Pekruhn (1986) limited the inclusion criteria to “teeth in my practice, which could be conveniently treated in a single visit”. Roane et al. (1983) stated that decisions on the use of single- or multiple-visit treatment were based solely on the time available for treatment. This type of case selection does not lend itself to scientific scrutiny and should, of course, not serve as a basis for a treatment strategy. As in all branches of medicine, the choice among endodontic treatment approaches should be based on a proper diagnosis.
Endodontic diagnosis is simple in most cases. An important first step is to distinguish between teeth with vital pulp and those with non-vital pulp. As discussed previously, the clinical diagnosis of vital pulp cannot differentiate between various stages of severity of the inflammatory process. In a somewhat arbitrary fashion, the clinical judgment of reversible and irreversible pulpitis has been accepted. Although “irreversible pulpitis” is a poorly defined diagnosis, it is well-established that as long as the pulp tissue is vital, the infection is limited to the superficial portions of the tissue, while the larger portion is not infected. This means that the primary focus of the endodontic procedure is to prevent microbial infection of the root canal space. Appropriate procedures to be applied in this respect are well-established and will not be reviewed here. Pulp extirpation and the placement of wound dressing—the root-filling implant—are best completed in a one-treatment visit if aseptic conditions are to maintained. Consequently, endodontic treatment of the vital pulp is a perfect one-visit case. If the surgical procedure is carefully carried out and is truly aseptic, a successful outcome should be expected in nearly 100% of the cases.
When the pulp tissue deteriorates to necrosis, the common diagnosis is necrotic pulp and, later, pulp necrosis with apical periodontitis. Another important treatment situation is the presence of a failing, previously root-filled tooth, where apical periodontitis has emerged or prevailed. Obviously, there are many other more rare pathological conditions that can be confused with endodontic disease processes, but the few listed here include the overwhelming number of lesions that occur in general practice.
From a pathological point of view, pulp necrosis and its sequelae are very different from the diseases of the vital pulp. They are almost all infectious. It is therefore reasonable to believe that teeth with necrotic infected pulp tissue should require a different treatment regimen than teeth with vital pulp.
The proper control of root canal infections by biologically compatible means has pre-occupied the endodontic research profession for many years. It has been estimated that the pulpal space may harbor from 107 to 108 bacterial cells (Byström and Sundqvist, 1983; Sjögren et al., 1991). The use of saline as an irrigant in combination with mechanical instrumentation may reduce this number 1000-fold, but it will not predictably eliminate all the organisms. The use of an antimicrobial agent, such as sodium hypochlorite, will further reduce the bacterial number—in some cases, to such an extent that cultivable bacteria are not recovered. There is no evidence yet available that instrumentation and antimicrobial irrigation can predictably reduce the bacterial count to zero during a single treatment session (Byström and Sundqvist, 1983, 1985; Byström et al., 1985; Sjögren et al., 1997; Cvek et al., 1976a,b; Shuping et al., 2000; Sundqvist et al., 1998). When the presently available best regimens of treating infected root canals are used, about one-third remain infected after one treatment. The number of remaining bacterial cells is then often small, in the range of 102 to 104 cells (Sjögren et al., 1991, 1997; Dalton et al., 1998; Shuping et al., 2000). Despite these small numbers, the effect on outcome can be significant (Sjögren et al., 1997). There are conflicting opinions, however, regarding the need for a complete disinfection of the pulpal space before the root filling is placed. Peters and Wesselink (2002) found no difference in treatment outcome when root canals were filled, despite the presence of residual microbes. Weiger et al. (2000), in a randomized clinical trial, observed no difference in outcome between teeth treated in one session and teeth filled after calcium hydroxide medication. They concluded, “One-visit root canal treatment is an acceptable alternative to two-visit treatment for pulpless teeth associated with an endodontically induced lesion.” Yet, both studies may have lacked sufficient statistical power for valid conclusions to be drawn.
Other clinical studies suggest that the rate of successful treatment outcome will decrease if the treatment is concluded with a root filling before the root canal is free of micro-organisms. Such data are available from both prospective (Sjögren et al., 1997) and retrospective studies (Engström et al., 1964; Heling and Shapira, 1978). It is presently unknown if the numbers or types of micro-organisms remaining at the time of root filling may modify this conclusion. Therefore, if root canal disinfection remains the goal, the treatment of a tooth with an infected necrotic pulp may require at least a two-visit procedure.
(V) Concluding Remarks
The analysis presented here suggests that, if many of the different opinions prevailing in the endodontic field are to be resolved, there is considerable room for properly designed clinical studies. The most apparent gap in the published literature is the lack of randomized clinical trials, which are needed to resolve issues that relate to proper management of pulpal wounds, proper medication, and number of appointments for treatment of infected root canals. Such studies must be based not only on sufficient numbers of cases but also on due consideration of confounding factors. Trials in endodontics also require very long follow-up periods, if valid conclusions are to be generated.
In recent years, molecular biology techniques for the study of microbiology have advanced significantly to permit the identification of many new micro-organisms. The classic phenotypic identification has been replaced, in many situations, by genotypic identification. This has provided new observations of significance for future research. Thus, in addition to the identification of new micro-organisms in the pulp space, it has also been possible to establish reliable information about their prevalence (Conrads et al., 1997; Gonçalves and Mouton, 1999; Machado de Oliveira et al., 2000; Xia et al., 2000; Hashimura et al., 2001; Jung et al., 2001; Roças et al., 2001; Rolph et al., 2001; Siqueira et al., 2001, 2002). This advance in endodontic microbiology is promising, and molecular biology techniques may, in the future, have a significant role to play in developing a better understanding of the still-complex pathological process associated with tissue healing of apical periodontitis. Research in this area is in its infancy, however, and much work still needs to be done.
It is not to be expected that within a foreseeable future there will be rapid solutions to the issues reviewed here. Therefore, in teaching endodontics, academic institutions and continuing education courses must rely on science and supporting clinical evidence. While future sound scientific work may result in protocols that are effective from an outcome point of view, translation of this knowledge to the clinical profession, and the proviso under which they can be applied, will remain a challenging task for many years.
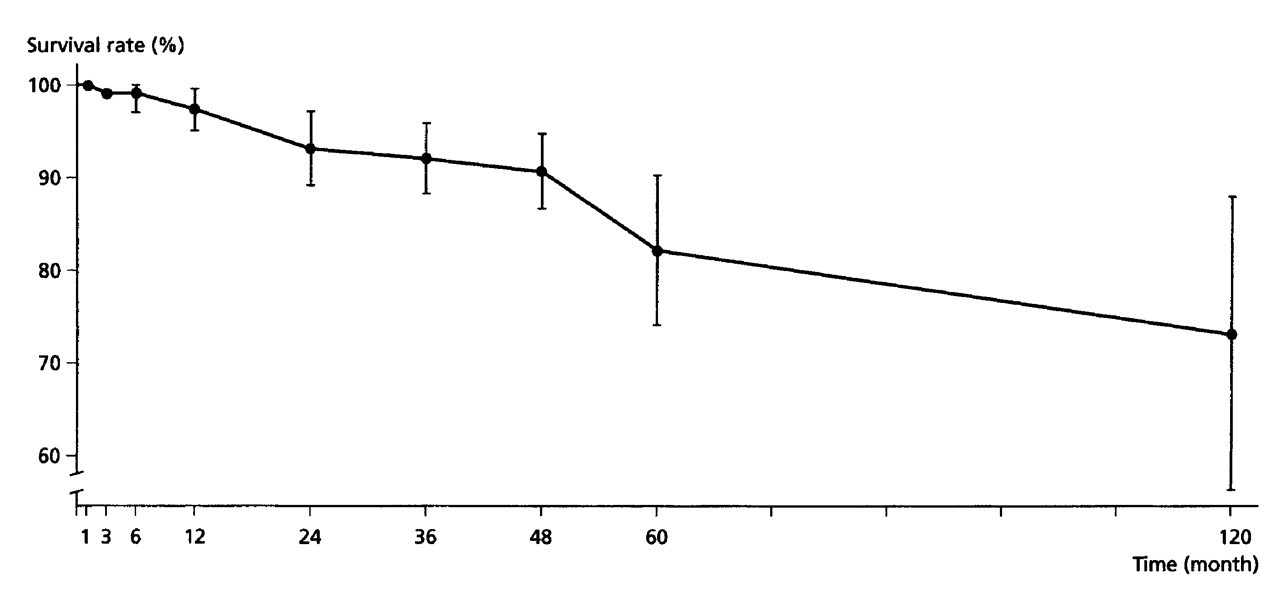
Graphic representation of the findings in the study by Hörsted et al. (1985). While the overall rate of pulpal survival was high, there was a decline over time. Bars indicate 95% confidence intervals.
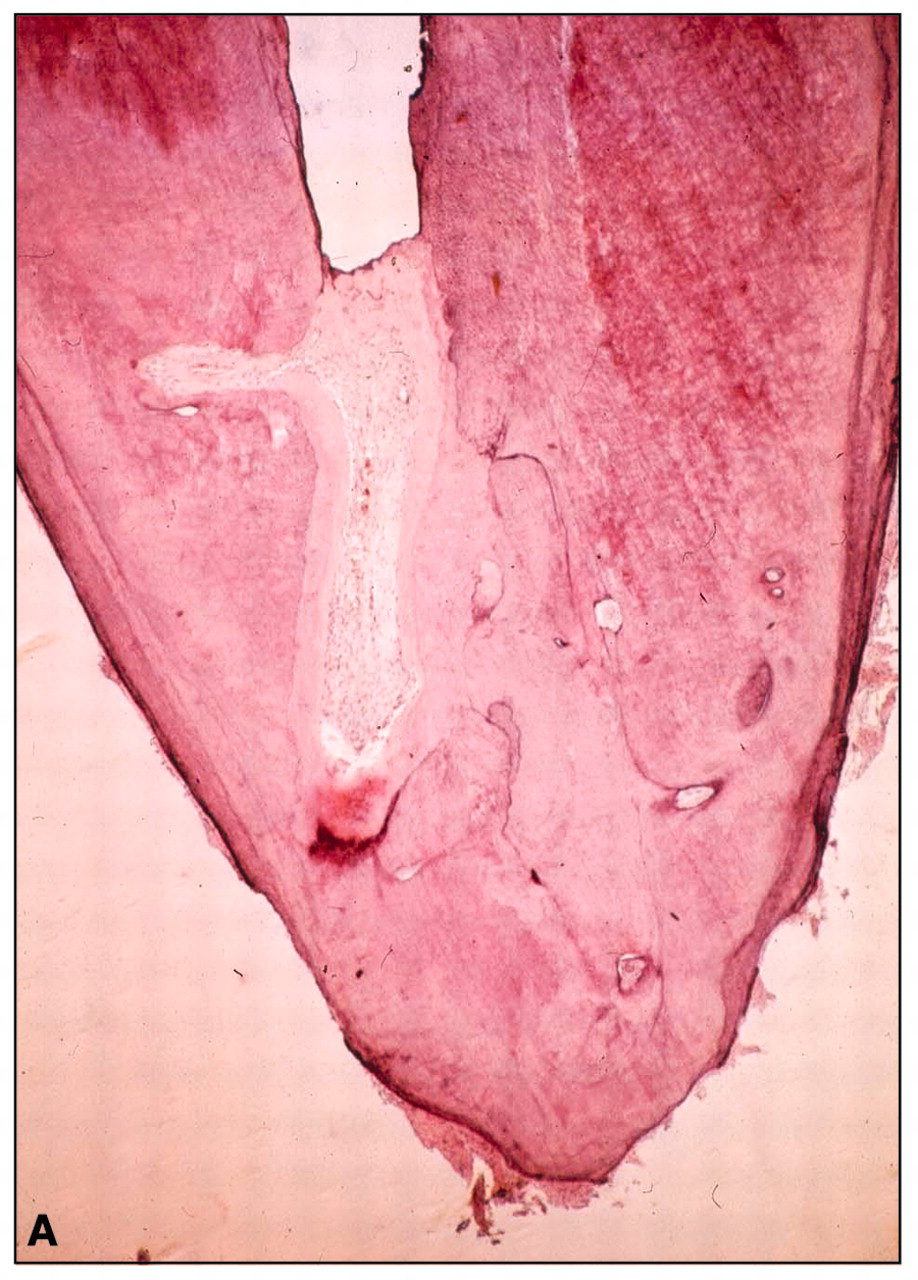
Tissue specimen demonstrating hard tissue repair in the apical portion of a root canal subsequent to pulpectomy and dressing with calcium hydroxide to close off residual non-inflamed pulp tissue
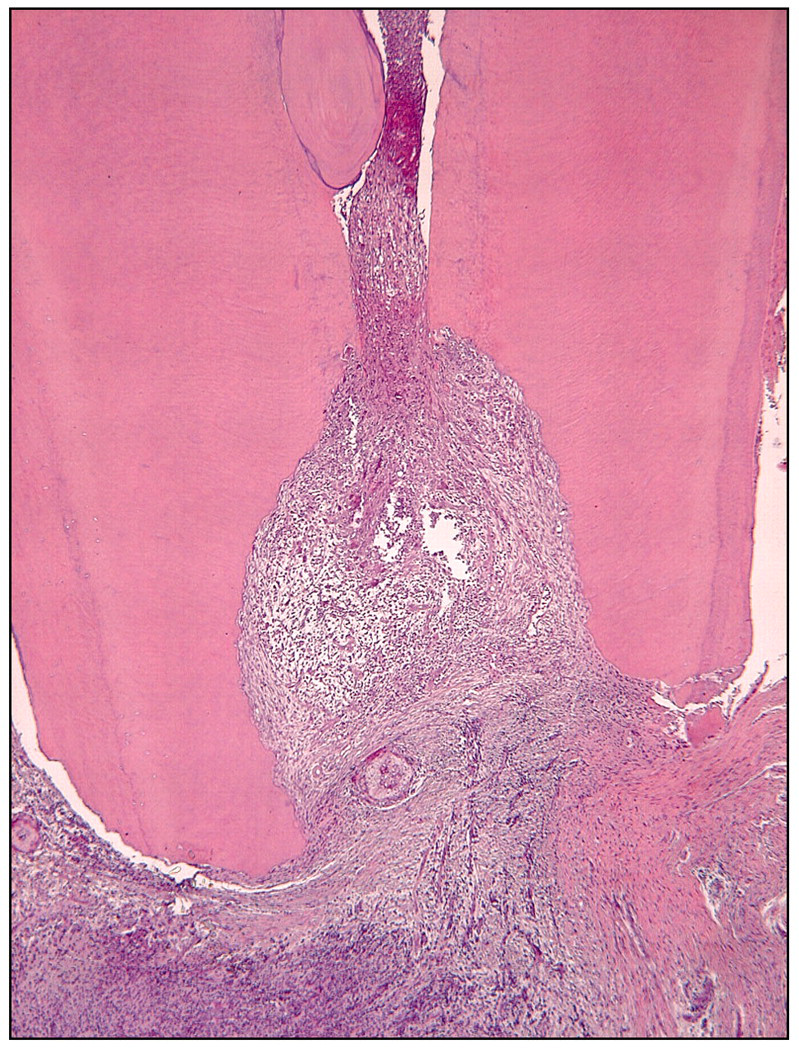
Tissue specimen of an extracted tooth with inflammatory tissue attached to the root tip. Extensive apical root resorption has destroyed original apical canal configuration. Courtesy of Dr. D. Ricucci.
