Abstract
Microbiological reports of apical periodontitis have revealed that yeasts can be isolated from approximately 5-20% of infected root canals. They occur either in pure cultures or together with bacteria. Almost all isolated yeasts belong to the genus Candida, and the predominant species is C. albicans. Pheno- and genotypic profiles of C. albicans isolates show heterogeneity comparable with those of isolates from other oral sites. C. albicans expresses several virulence factors that are capable of infecting the dentin-pulp complex, including dentinal tubules. This causes, consequentially, an inflammatory response around the root apex, which suggests a pathogenic role for this organism in apical periodontitis. Yeasts are particularly associated with persistent root canal infections that do not respond favorably to conservative root canal therapy. This may be due to the resistance of all oral Candida species against a commonly used topical medicament, calcium hydroxide. However, other antimicrobial agents may offer alternative therapeutic approaches and improve the treatment of these persistent cases of apical periodontitis.
Introduction
Apical periodontitis is an inflammatory process of the periapical area caused by an infection of the dental root canal system. In most cases, chemo-mechanical preparation of the root canal and local medication with calcium hydroxide followed by filling of the root canal with gutta-percha and sealer result in elimination of the infection and healing of the lesion. Occasionally, apical periodontitis does not respond favorably to root canal therapy, and periapical inflammation caused by the root canal infection may persist months or even years despite treatment. Several factors may contribute to the failure of the treatment of persistent cases. Most commonly, these factors are related to difficulties in chemo-mechanical preparation of the root canals. Occasionally, micro-organisms resistant to conservative therapy may also be involved (Byström, 1986; Sirén et al., 1997). Literature on the microbiological findings in persistent apical periodontitis not responding favorably to conservative therapy is limited (Bender and Selzer, 1952; Grahnen and Krasse, 1963; Engström, 1964; Goldman and Pearson, 1969; Haapasalo et al., 1983; Ranta et al., 1988; Sirén et al., 1997; Molander et al., 1998; Hancock et al., 2001; Kalfas et al., 2001; Love, 2001). However, it is known that a few species are more frequently isolated from persistent cases compared with primary cases. These include the Enterococcus faecalis/faecium group, enteric Gram-negative facultative rods, i.e., coliforms, and Pseudomonas species (Engström, 1964; Haapasalo et al., 1983; Ranta et al., 1988; Sirén et al., 1997; Hancock et al., 2001; Love, 2001). Recently, there has also been increasing interest in the presence and role of yeasts in infections resistant to conservative root canal therapy (Nair et al., 1990; Sen et al., 1995, 1997a, b). This review presents the contemporary knowledge of the occurrence and biotypes of yeasts in endodontic infections, and susceptibility, in vitro, of yeasts to endodontic irrigants, local disinfectants, and antifungal agents.
Taxonomy and General Characteristics of Yeasts
Yeasts belong to a separate kingdom of living organisms, fungi. Contrary to bacteria, fungi are eukaryotic organisms, i.e., their genome is organized in a nucleus which is surrounded by a membrane. This membrane is continuous with the endoplasmic reticulum, and organelles, such as mitochondria, ribosomes, and different storage inclusions, are present. Fungal cell walls are rigid structures composed mainly of glucan, mannan, and chitin. For nutrition, fungal organisms are dependent on nitrogen and carbon compounds which are taken up through the cell wall (De Hoog and Guarro, 1995).
Yeasts are present in various sites in the human body as members of the normal flora. They occur, e.g., in the gastrointestinal tract, vagina, and perineal area (Jarvis, 1996). The oral cavity has suitable environmental conditions for yeast colonization. Oral yeasts belong to the division Ascomycota and class Endomycetes, which is divided further into four families: Saccharomycetaceae, Endomycetaceae, Dipodascaceae, and Lipomycetaceae. Clinically, the most important oral yeasts belong to the family Saccharomycetaceae and to the genus Candida. Reproduction of Candida is based on multilateral budding, which may take place anywhere on the mother cell (de Hoog and Guarro, 1995).
Oral Yeast Species
Candida albicans is the most dominant oral yeast species, followed by C. glabrata, C. krusei, C. tropicalis, C. guilliermondii, C. kefyr, and C. parapsilosis (Odds, 1988). Recent findings also suggest the occasional occurrence of C. dubliniensis, which is a species closely related to C. albicans (Hannula et al., 1997). Other yeast genera have also been isolated from the oral normal flora, e.g., Saccharomyces spp. and Geotrichum spp. (Tawfik et al., 1989; Stenderup, 1990; Heinic et al., 1992). Isolation of other fungi from the oral cavity has also been reported, but they are usually seen in association with systemic disease—for example, pulmonary cryptococcosis caused by Cryptococcus neoformans (Stenderup, 1990).
Virulence Factors of Candida
The transition of C. albicans from a harmless commensal to a pathogenic organism appears to be dependent on minor changes in predisposing conditions which cause the expression of a variety of virulence factors (Shepherd, 1992; Sweet, 1997). These factors include adherence, hyphal formation, thigmotropism, protease secretion, and phenotypic switching phenomenon.
Adherence of micro-organisms is a complex, multifactorial process involving several types of cell-surface adhesins which are essential for colonization and infection of the host. The main adhesin molecules of C. albicans responsible for adhesion to host cells seem to be cell wall mannoproteins (Sweet, 1997). However, several other factors also contribute to the adherence of yeasts, e.g., cell-surface hydrophobicity, environmental pH, and concentrations of iron, calcium, zinc, and carbon dioxide (Ener and Douglas, 1992; Klotz, 1994; Samaranayake et al., 1995; Sohnle et al., 2001). Furthermore, environmental proteins from saliva and gingival crevicular fluid as well as extracellular matrix components affect the complex adherence of Candida to host cells and tissues (Calderone et al., 2000; Holmes et al., 2002). C. albicans is a pleomorphic micro-organism demonstrating different growth forms such as germ tubes, yeasts (blastospores), pseudo- and true hyphae, and chlamydospores (Odds, 1988; de Hoog and Guarro, 1995). All growth patterns except chlamydospores may show conversion to each form of growth, depending on the environmental conditions. Therefore, the term ‘dimorphic’, often used in the literature, is semantically inaccurate to explain C. albicans morphogenesis. Although hyphal formation is not a prerequisite for pathogenicity of C. albicans, biopsies of candidal infections often reveal hyphal adherence to and penetration through epithelial tissues, indicating increased pathogenicity in comparison with ovoid yeast forms (Sweet, 1997). It seems that the hyphal penetration into tissues is enhanced by thigmotropism, i.e., contact sensing by hyphae to find intracellular junctions or microscopic breaks on mucosal surfaces (Sherwood et al., 1992; Gow et al., 1994; Sweet, 1997). One of the key virulence determinants of Candida species is their ability to produce and secrete aspartyl proteases which digest a variety of host proteins. The virulence of these proteases has been demonstrated with animal experiments showing that the amount of protease is directly comparable with the pathogenicity of the strain (MacDonald and Odds, 1983; Kwong-Chung et al., 1985; Okamoto et al., 1993; Togni et al., 1994). Therefore, the higher rate of protease activity of C. albicans in comparison with other Candida species also suggests higher virulence. In addition to these major virulence factors, C. albicans has a tendency to phenotypic alteration, which contributes to environmental adaptation. Phenotypic alterations include change of colony morphology and protease activity (Slutsky et al., 1985; White and Agabian, 1995). This genetically controlled phenomenon is known as phenotypic switching, and it may occur relatively frequently, especially under stress (Soll, 1988). Phenotypic switching may assist in survival of and colonization by the yeasts, and it may also lead to genetic selection of adaptive strains (Sweet, 1997). Virulence factors and their possible contributions to apical periodontitis are listed in Table 1.
Oral Yeast Infections
A characteristic feature of yeast infections is that they develop when the host provides the environmental conditions and nutrients essential for attachment, growth, and reproduction of fungi. In other words, yeasts are opportunistic pathogens. Thus, local or general predisposing factors are required for yeast infection to develop (Shepherd, 1992). These factors can be classified into four categories: (i) host factors, such as normal and pathological changes in physiological status of the host; (ii) dietary factors, such as carbohydrate-rich diets and vitamin deficiencies; (iii) mechanical factors, such as denture-wearing; and (iv) iatrogenic factors, such as administration of broad-spectrum antibiotics and corticosteroids (Odds, 1988). The clinical form of candidosis is often related to a predisposing factor, e.g., acute pseudomembraneous candidosis (thrush) is often associated with natural factors such as immunological and microbiological instability at birth, angular cheilitis with dietary factors, chronic atrophic candidosis (denture-associated stomatitis) with mechanical factors, and acute atrophic candidosis with iatrogenic factors (Lynch, 1994). In dental practice, chronic atrophic candidosis (denture stomatitis) is perhaps one of the most frequently encountered oral Candida infections (Wilson, 1998). There has also been an increasing interest in the presence of yeasts in infected periodontal pockets and their possible role in the pathology of different forms of periodontitis (Slots et al., 1988; Zambon et al., 1990; Rams and Slots, 1991; Dahlén and Wickström, 1995; Hannula et al., 1997, 2001).
Microbiology of Apical Periodontitis
Apical periodontitis is a host defense response to infection of necrotic pulp (Miller, 1894; Kronfeld, 1939; Kakehashi et al., 1965). The host has an array of defense mechanisms consisting of several types of inflammatory cells, such as polymorphonuclear leukocytes and lymphocytes, intercellular messengers, such as cytokines, and chemical weapons such as proteolytic enzymes (Nair, 1997). Despite these defenses, the body cannot eliminate the micro-organisms residing in the necrotic root canal, and therefore the inflammatory process does not result in healing. The interaction between root canal infection and the host defense mechanisms eventually cause destruction of periapical tissues and formation of apical periodontitis (Nair, 1997).
More than 300 species of micro-organisms colonize the human oral cavity, but only a limited number of these have been isolated from infected root canals with apical periodontitis (Moore, 1987). Several factors contribute to the selection of micro-organisms. Primarily, the selection takes place among those micro-organisms entering the root canal, which depends on the pathway to the pulp. For example, a deep caries lesion may serve as a pathway and limit the number of possible microbial species. In addition, the host defense mechanisms in the infected but still vital pulp reduce the number of surviving species. Furthermore, environmental factors of the necrotic root canal, e.g., redox-potential and source of nutrients, give an advantage to species with proteolytic activity and ability to survive in anaerobic conditions. Finally, microbial interactions—either negative (such as competition for nutrients, secreted toxic metabolites, and specific bacteriocins) or positive (i.e., symbiosis of different species)—regulate the microflora of the infected root canal (Sundqvist, 1994).
Apical periodontitis is a polymicrobial infection dominated by obligate anaerobes (Bergenholtz, 1974; Kanz and Henry, 1974; Sundqvist, 1976; Byström et al., 1985; Haapasalo, 1986; Sundqvist et al., 1989; Baumgartner and Falkler, 1991). Usually, the number of isolated species is between two and eight, and monoinfections are rare (Kanz and Henry, 1974; Sundqvist, 1976, 1994; Haapasalo, 1986). Before root canal therapy, the most frequently isolated micro-organisms are: Gram-negative anaerobic rods, such as Prevotella spp., Porphyromonas spp., Fusobacterium nucleatum, Campylobacter rectus, and Selenomonas spp.; Gram-positive anaerobic cocci, such as Peptostreptococcus spp.; Gram-positive anaerobic and facultative rods, such as Eubacterium spp., Propionibacterium acnes, Actinomyces spp., and Lactobacillus spp.; and Gram-positive facultative Streptococcus species (Sundqvist, 1976; Haapasalo, 1986). The microbiology of root canal infections is still not clear in many regards, e.g., the data concerning the occurrence of uncultivable species such as spirochetes are scarce (Dahle et al., 1993).
The literature on microbiological findings in persistent root canal infections is also relatively limited (Bender and Selzer, 1952; Grahnen and Krasse, 1963; Engström, 1964; Goldman and Pearson, 1969; Haapasalo et al., 1983; Ranta et al., 1988; Sirén et al., 1997; Molander et al., 1998). However, it is known that a few species are frequently isolated from persistent cases. These include the Enterococcus faecalis/faecium group, enteric Gram-negative facultative rods (i.e., coliforms), and Pseudomonas species (Engström, 1964; Haapasalo et al., 1983; Ranta et al., 1988; Molander et al., 1998; Sundqvist et al., 1998; Hancock et al., 2001; Love, 2001). Micro-organisms commonly associated with chronic periodontitis, apical periodontitis, and persistent root canal infections are listed in Table 2.
Yeasts in Apical Periodontitis
Microbiological investigations of apical periodontitis during the past 50 years have revealed that yeasts can be isolated from infected root canals (Grossman, 1952; Slack, 1953, 1957; Macdonald et al., 1957; Hobson, 1959; Goldman and Pearson, 1969; Matusow, 1981; Nair et al., 1990; Najzar-Fleger et al., 1992; Sen et al., 1995, 1997a, b; Waltimo et al., 1997; Molander et al., 1998). Slack (1953, 1957) reported that yeasts exist in about 5% of cases of apical periodontitis. According to Grossman (1952), as many as 17% of infected root canals may contain Candida species. Hobson (1959) reported that Candida albicans was often isolated from root canal infections, although their pathogenicity in the root canal was unclear. However, according to a case report, a pure culture of Candida albicans caused acute pulpal-alveolar cellulitis (Matusow, 1981).
In another case report, C. albicans was found in root canals and in periapical granulomas of a patient suffering from chronic urticaria (Eidelmann et al., 1978). The complete cure of the patient was achieved only after the extraction of the infected teeth. Histological examination revealed that the granuloma exhibited an invasive Candida infection composed of acute and chronic granulation tissue along with hyphae and yeast cells. The root canal surfaces were covered by dense masses of yeast cells, and dentinal tubules were totally filled with hyphae. In addition to these cases, Damm et al. (1988) described two cases of cancer patients having dentinal candidosis. In the first case, carious dentin of the patient’s deciduous teeth contained numerous oval to filamentous Candida cells. The teeth exhibited either acute irreversible pulpitis or acute apical abscess. Complete healing was accomplished after extraction of all deciduous teeth. The second case demonstrated exposed coronal dentin with heavy colonization by C. albicans. Pseudohyphae and yeast cells were present not only in pulp tissue but also in cervical and apical soft tissues. As seen in these cases, extensive invasion by fungi seems to be mostly associated with the immunocompromised state of the patients. However, Kinirons (1983) described a similar clinical case with no systemic illness.
Nair et al. (1990) studied therapy-resistant root canal infections and found micro-organisms in 6 of 9 specimens. Bacteria were shown in 4 of the 6 cases, while yeast-like organisms were found in 2 cases as judged by electron microscopy. The presence of intraradicular fungi in the endodontically treated human teeth was associated with periapical lesions that persisted after treatment. Sen et al. (1995) observed bacteria and fungi with scanning electron microscopy in infected root canals and dentinal tubules associated with periapical lesions. They found that 4 out of 10 root canals were heavily infected with yeasts, confirming the association between yeasts and root canal infections. They formed dense but separate colonies, and, in one specimen, hyphal elements were also present. Since the patients in this study did not have any systemic disease, the presence of yeasts in root canals may be attributed to poor oral hygiene.
In a report by Waltimo et al. (1997), the occurrence of yeasts was studied in 967 microbiological samples taken from cases of apical periodontitis not responding favorably to conventional treatment. Micro-organisms were found in 692 (72%) samples, whereas 275 (28%) showed no growth. Forty-eight fungal strains were isolated from 47 samples, which represented 7% of the culture-positive samples. The fungi were endomyceteous yeasts, and they were isolated either in pure culture (6 cases, 13%) or together with bacteria (41 cases, 87%). The identification of yeasts was carried out with conventional clinical laboratory procedures, showing results comparable with those of earlier studies. Almost all isolates belonged to the genus Candida, and C. albicans was the most common species. C. glabrata, C. guilliermondii, C. inconspicua, and Geotrichum candidum were also isolated.
In studies of the initial microbial flora of root canal infections, yeasts have usually not been found (Haapasalo, 1989; Sundqvist et al., 1989). However, according to a recent study of randomly selected patients with periapical radiolucencies, C. albicans was detected in 5 out of 24 samples (21%) taken from infected root canals by means of the polymerase chain-reaction-based (PCR) molecular detection technique (Baumgartner et al., 2000). The PCR was carried out conventionally with a detection limit of 10-4 ng of DNA. Although the material was limited, the high percentage may be due to the higher sensitivity of the method in comparison with detection of micro-organisms by conventional culture procedures. However, the finding indicates that yeasts may be present in low numbers at the start of root canal treatment, and that they may reach higher proportions during conventional treatment procedures. In another recent study, intact root canals with pulp necrosis were examined microbiologically (Lana et al., 2001). C. tropicalis and S. cerevisiae were recovered from two canals (7.4%) before root canal therapy. C. guilliermondii and C. parapsilosis were cultivated in the second and third collections, respectively, of root canal contents. According to these findings, it is also possible that yeasts which are common opportunistic pathogens of the oral cavity gain access to the root canal as contaminants during endodontic therapy (Sirén et al., 1997). This emphasizes the importance of aseptic treatment procedures in the prevention of persistent infections. Regardless of the source and means of entry for yeasts into the root canal, their presence in cultivable numbers may have clinical importance in persistent cases.
Influence of Necrotic Root Canal on Strain Selection
Necrotic root canals provide harsh ecological conditions for micro-organisms in comparison with other oral sites. The influence of these conditions for yeast strain selection was examined in a recent study that compared the phenotypes and genotypes of C. albicans isolates from root canals and periodontal crevices (Waltimo et al., 2001). Briefly, the phenotyping was based on the presence of 5 enzymes (valine arylamidase, phosphoamidase, alpha-glucosidase, beta-glucosidase, and N-acetyl-beta-glucosaminidase), ability of the strains to assimilate 11 carbohydrates (glycerol, L-arabinose, xylose, adonitol, xylitol, sorbitol, methyl-D-glucoside, N-acetyl-D-glucosamine, sucrose, trehalose, and melezitose), and the resistance to boric acid (Williamson et al., 1987). Genotyping was based on randomly amplified polymorphic DNA (RAPD) profiles obtained with the use of two different primers.
A total of 14 different phenotypes was found among the 37 root canal isolates of C. albicans. The majority of the isolates (26) were classifiable into three major phenotypes, described by Williamson et al., (1987): 16 isolates (43.2%) belonged to phenotype A1R, 6 (16.2%) to A1S, and 4 (10.8%) to B1S. Interestingly, C. albicans phenotype A1R, which was predominant, has been associated mainly with patients with symptomatic C. albicans infections but not with asymptomatic carriers. This may indicate a higher virulence of this phenotype in comparison with other phenotypes (Xu and Samaranayake, 1995). The genotypic characterization with use of the combination of the two different primers yielded 32 different profiles for the 37 C. albicans strains, demonstrating high genotypic divergence of the isolates.
Analysis of the current data implies genotypic heterogeneity of C. albicans isolates from root canals in humans. However, frequently encountered phenotypes were similar to the ones reported from other oral and non-oral sources (Bostock et al., 1993; Tsang et al., 1995; Xu and Samaranayake, 1995; Matee et al., 1996). This implies that phenotypically unusual strains of C. albicans are not frequently involved in root canal infections. Therefore, it seems that the root canal, an ecologically harsh niche with regard to redox-potential and nutrients supply, may not have an impact on strain selection that differs from those of other oral sites. Thus, it seems that a general characteristic of C. albicans is its ability to tolerate a wide variety of different environmental conditions.
Accompanying Bacteria in Yeast Infections
Recent studies have shown that yeasts can survive as a monoinfection of the root canal (Matusow, 1981; Waltimo et al., 1997). However, they are usually found in mixed cultures together with bacteria. Yeasts may often be isolated together with facultative Gram-positive bacteria such as a- and non-hemolytic Streptococcus species, whereas Gram-negative isolates are rare (Waltimo et al., 1997). The dominance of the facultative Gram-positive accompanying bacteria may be due to the special ecological conditions of the root canal during prolonged treatment, which could favor yeasts and streptococci. There may also be synergism between these micro-organisms. However, no or only negative association of Streptococcus spp. with other bacteria has been reported in root canals (Sundqvist, 1992). However, it has been reported that C. albicans may prolong the viability of β-hemolytic streptococci (Burnet and Sherp, 1968). Furthermore, C. albicans co-aggregates with a variety of streptococci such as S. gordonii, S. mutans, and S. sanguis (Holmes et al., 1995; Nikawa et al., 2001). This may promote their colonization and thus explain the concomitant occurrence of these microbial species. Further investigations concerning microbial interactions are needed for a better understanding of the dynamics and ecological succession of microflora in root canal infections.
Yeast Infection of Dentin
There are a few studies that have reported the capability of yeasts to infect dentin (Sen et al., 1997a,b; Waltimo et al., 2000a). In the first study by Sen et al., (1997a), the root canals without smear layer were infected with C. albicans in a nutritionally stressed medium. Yeast cells and hyphal structures formed dense but separate colonies on the root canal walls. They exhibited strong attachment to dentinal walls and microfibrils on the cell walls. Since hyphal branches grew typically into dentinal tubules, budding yeast cells were also observed in the tubules. The investigators concluded that C. albicans may be considered as a dentinophilic micro-organism with the capability of invading dentinal tubules with different forms of growth, and can use dentin as a source of nutrition for a long period. In a subsequent study, Sen et al. (1997b) demonstrated colonization of C. albicans on dentin with or without the smear layer. When the smear layer was present, the colonies were not distinct and demonstrated the presence of a thick biofilm with different growth forms. In the absence of the smear layer, a network of branching pseudo-hyphae was present on dentinal walls, but biofilm formation was not evident. The tips of the pseudo-hyphae always tended to penetrate dentinal tubules. It was proposed that, by this contact-sensing (thigmotropism) ability, the invasion of dentin by C. albicans is almost inevitable. In addition, the presence of the smear layer demonstrated a more suitable substrate for active C. albicans growth and attachment. On the other hand, dentin treatment with ethylenediamine-tetraacetic acid (EDTA) and NaOCl decreased the amount of all available organic and inorganic dentin which apparently did not favor biofilm formation by C. albicans (Figs. 1, 2 ).
An in vitro method was developed to study the comparative penetration of dentin by C. albicans and E. faecalis (Waltimo et al., 2000a). Both macroscopic recording of penetration through the dentin disc and microscopic examination of micro-organisms in the dentinal tubules were carried out. C. albicans growth was relatively weak in comparison with that of E. faecalis. However, both organisms were capable of penetrating the 2-mm-thick human dentin disc. Histological preparations showed penetration of yeast cells and hyphae into a few dentinal tubules, but most of the tubules remained free of cells. Continuous penetration could be seen up to 60 μm, whereas single budding cells in low numbers could be found throughout the dentin specimens.
There are case reports presenting heavy yeast infection of carious and normal dentin (Kinirons, 1983; Damm et al., 1988). A thick biofilm of C. albicans with different growth forms was always found on exposed coronal dentin. Pseudo-hyphal structures were found in all dentinal tubules penetrating toward the pulp. C. albicans was also shown in root canals (Eidelman et al., 1978). The root canal surfaces were covered by dense masses of yeast cells, and dentinal tubules were totally filled with hyphae.
It is apparent from all studies, in vitro, and case reports that C. albicans is capable of invading dentin. The ability of C. albicans to grow into dentin indicates expression of several virulence factors in necrotic root canals: hyphae formation, thigmotropism, and secretion of proteolytic enzymes (Kaminishi et al., 1986; Hagihara et al., 1988; Sweet, 1997; Naglik et al., 1999). Therefore, the invasion of the dentinal tubules by candidal pseudohyphae resembles the penetration of hyphae deep into the epithelium in cases of acute pseudomembranous candidosis (Kinirons, 1983).
Susceptibility of Candida Species to Disinfectants
All oral Candida species have been shown to be highly resistant to calcium hydroxide in vitro (Waltimo et al., 1999a,b). E. faecalis, which has served as a reference organism, is more resistant than other oral bacteria tested in this respect (Byström et al., 1985). It was therefore interesting that, compared with E. faecalis, all oral Candida species showed at least as high or higher resistance to calcium hydroxide. This resistance may explain its frequent isolation from persistent root canal infections (Byström, 1986; Sirén et al., 1997). Similarly, the ability of yeasts to survive in harsh ecological conditions and in high alkalinity is likely to be an important factor that accounts for their presence in persistent root canal infections after conventional root canal therapy.
In the same series of studies (Waltimo et al., 1999b), sodium hypochlorite showed the highest efficacy against C. albicans in therapeutic concentrations. It is already a commonly used irrigant in endodontics. Concentrations below the minimum inhibitory concentration of sodium hypochlorite may be effective on potentially pathogenic traits of Candida species (Webb et al., 1995). However, in the environment of necrotic root canal, the significance of this finding is questionable. Chlorhexidine acetate was also effective in killing C. albicans strains at therapeutic concentrations after short periods of exposure. However, there are also a few papers reporting clinical resistance of yeasts to chlorhexidine (Sharon et al., 1977; Addy and Hunter, 1987). Iodine compounds are efficient against a variety of micro-organisms (Scott et al., 1986) and may also be considered for elimination of yeasts in endodontic treatment (Waltimo et al., 1999b).
In a recent study, Sen et al. (2000) demonstrated antifungal properties of several endodontic disinfectants and irrigating solutions. They found that EDTA showed the highest antifungal activity, while povidone-iodine had the weakest capacity. Chlorhexidine (CHX) concentrations of 0.2% demonstrated lower effectiveness as compared with EDTA and sodium hypochlorite solutions. They concluded that EDTA showed its high antifungal activity against C. albicans by interacting with the calcium in the medium and in the cell walls and, hence, limiting growth and affecting nutritional conditions.
In conclusion, recent studies have shown that sodium hypochlorite, EDTA, iodine, potassium iodide, and chlorhexidine are more effective against C. albicans than calcium hydroxide, in vitro (Table 3). Therefore, the use of these disinfectants may provide an alternative local medication in endodontic yeast infections. On the other hand, calcium hydroxide has a long-lasting alkaline effect, it is relatively easy to apply, and it forms an effective physical barrier to the nutrients of inflammatory exudates from the periapical area. Therefore, a combination of these disinfectants with calcium hydroxide may result in medicaments with a long-lasting and broad-spectrum antimicrobial efficacy, and good biological and physical properties of calcium hydroxide (Waltimo et al., 1999b; Sirén et al., unpublished data).
Factors Inhibiting Disinfectants
Recent studies, in vitro, have demonstrated that there are several factors that interfere with the clinical efficacy of disinfectants. These factors include, e.g., dentin, smear layer, and fungal biofilms which consist of a mono- or multilayer of micro-organisms embedded in an extracellular matrix. The protein and mineral components of dentin can bind and inhibit activity of disinfectants, and smear layers and biofilms may serve as barriers which prevent the exposure of micro-organisms to effective concentrations of the disinfectants (Sen et al., 1999; Portenier et al., 2000; Haapasalo et al., 2000). In addition, it is likely that inflammatory exudates from periapical areas dilute and inhibit the local medication in apical parts of the root canal system.
Sodium hypochlorite in 5% and 0.5% concentrations has been shown to be highly effective in optimal conditions, in vitro (Waltimo et al., 1999b). In addition, this commonly used solution showed complete antifungal activity in a short period, from 15 sec to 5 min (Smith and Wayman, 1986; Harrison et al., 1990). In contrast, antifungal efficacy of NaOCl and CHX on C. albicans biofilms in root canals was shown to be very limited (Sen et al., 1999). Micro-organisms living in the surface layers of root canal biofilm are directly affected by the disinfectant or irrigating solution. However, the extracellular matrix may protect the micro-organisms in the deeper layers from the full-strength activity of the endodontic disinfectants. Hence, the strains may become more resistant, and the infection may therefore persist in the root canal. This finding demonstrates the importance of local inhibitory factors, and it emphasizes the need for effective mechanical preparation during root canal therapy. Endodontic instrumentation “disorganizes” and exposes biofilm cells to endodontic disinfectants/irrigating solutions.
Susceptibility of C. albicans to Antifungal Agents
Systemic or local antifungal agents may be considered in some acute or persistent cases after a microbiological diagnosis of a root canal yeast infection. In a recent study, Waltimo et al. (2000b) compared the susceptibility to antifungal agents of 35 endodontic isolates with an equivalent number of periodontal strains. The medicaments studied were: amphotericin B, 5-fluorocytocine, and 3 azole-group agents (fluconazole, miconazole, and clotrimazole). Although amphotericin B has been extensively used for more than 30 years, the emergence of resistant strains is rare. As expected, all strains in this study, from both the gingival crevice and the root canal, showed susceptibility to this polyene. In contrast to amphotericin B, strains resistant to 5-fluorocytosine are frequently isolated (McGinnis and Rinaldi, 1985). However, in this study, all strains also showed susceptibility to 5-fluorocytosine. This may be due to the infrequent use of 5-fluorocytosine in dentistry. Varying susceptibility of the tested isolates to the azole-group antifungal agents is in accordance with recent reports of oral yeast isolates (Martins et al., 1997; Monteil et al., 1997; Hunter et al., 1998). In addition, in this study, a few oral isolates showed cross-resistance to the azoles. The relatively frequent resistance to azole-group antifungals seems to be an increasing problem with Candida species (White et al., 1998). Another recent report describes the susceptibility of oral C. albicans isolates to ketoconazole and nystatin, which were found to be effective (Sen et al., 2000). However, with treatment strategies based on disinfectants, the use of antifungal agents should be limited to acute cases and, particularly, to medically compromised patients. Furthermore, if development of resistant strains is to be avoided, susceptibility testing should be done before administration of these medicaments.
Concluding Remarks
Yeasts can be isolated from an infected root canal both in pure culture and together with bacteria. The occurrence in pure culture in cases of apical periodontitis indicates the pathogenic potential of yeasts in the root canal environment. The ability of Candida species to survive in a nutrient-limited environment and to withstand the action of commonly used endodontic medicaments may provide ecological advantages for this group of micro-organisms in the root canal. Several virulence factors seem to contribute to its capability to infect pulp tissue and dentin. In conclusion, Candida species may have important clinical implications, particularly in persistent cases of apical periodontitis. However, administration of root canal medicaments effective against Candida may improve treatment results in these cases. There is a need for controlled clinical trials to evaluate the efficacy of potent treatment strategies.
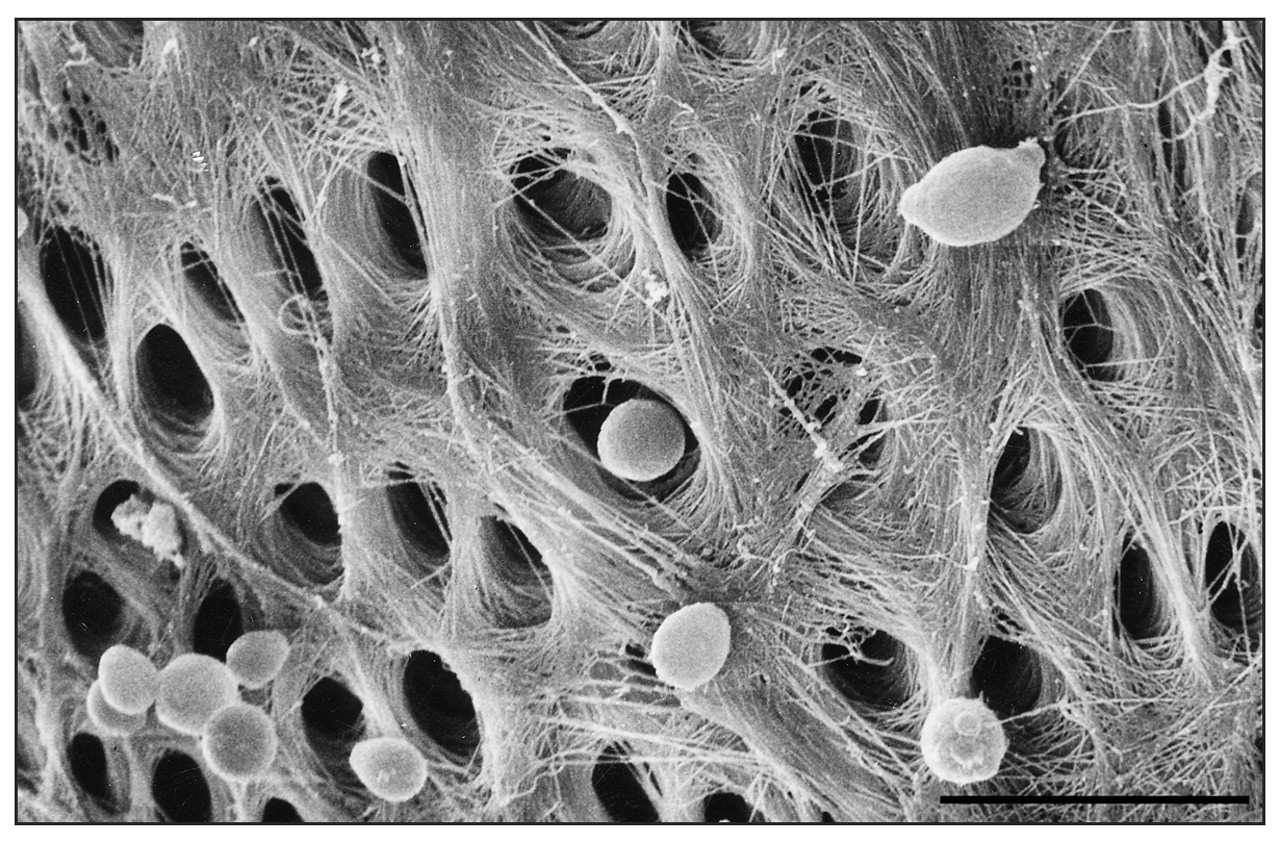
Scanning electron micrograph of C. albicans blastospores on root canal surface in vitro. The bar indicates 10 μm.
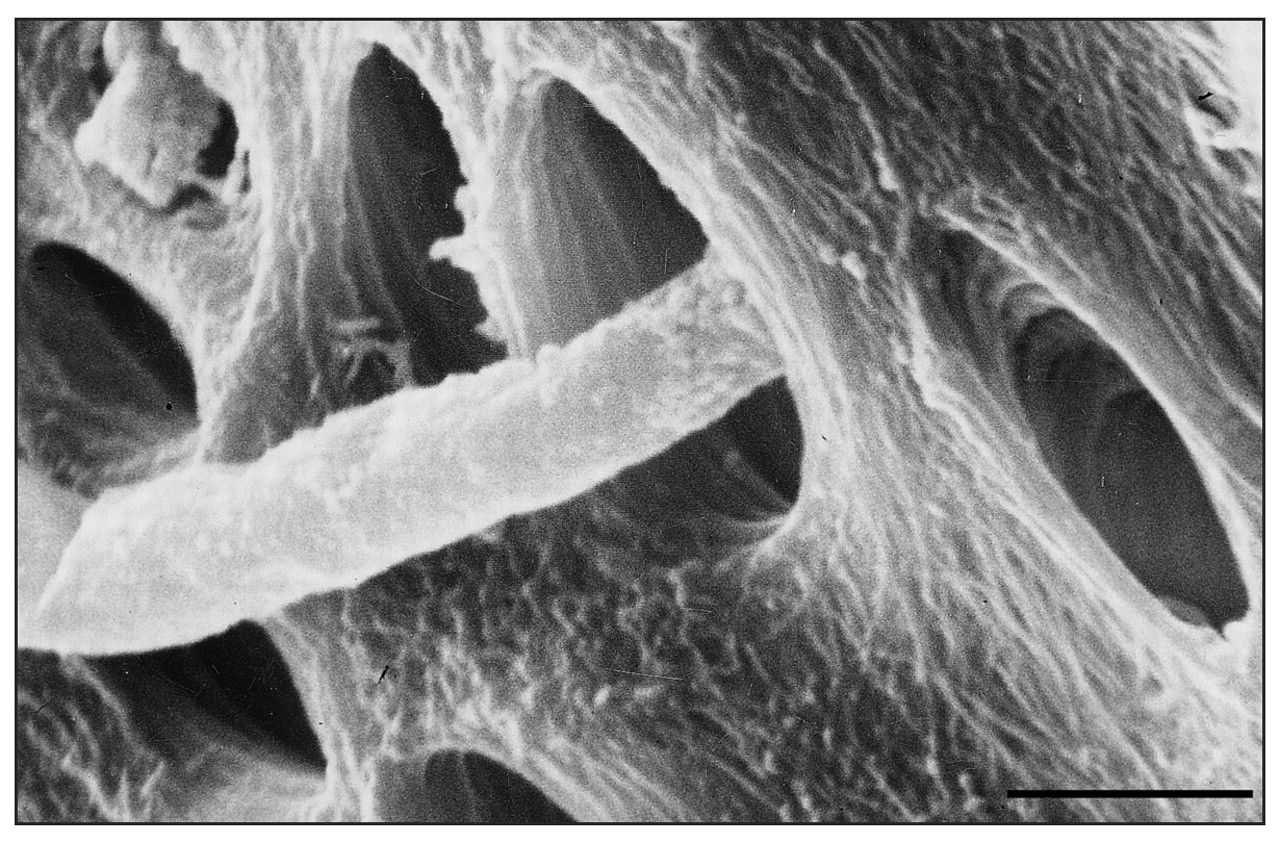
Scanning electron micrograph of C. albicans hyphae penetrating a dentinal tubule. The bar indicates 2 μm.
