Abstract
The oral cavity and pharynx are anatomically separate but functionally integrated regions of the head. The two regions are involved in complex motor responses that include feeding, chewing, swallowing, speech, and respiration. The multiple sensory receptors that innervate these two regions provide the first link in reflexes that control muscles of the entire head, upper gastrointestinal tract, and airway. Most of the reflexes affect the diversity of muscles that compose the tongue, which is vital to all stages of feeding and which continually affects the patency of the airway. Oral-pharyngeal reflexes are evident in the mammalian fetus and continually emerge as the animal or human matures. Some of the first reflexes in the oral region are geared toward nourishment. As the central nervous system matures and the oral and pharyngeal regions develop morphologically, new reflexes develop. Many of these reflexes are protective both of the tissue in the oral cavity, such as the tongue, and of the upper airway in preventing aspiration. While simple reflexes can be evoked in isolation, most reflexes combine with more complex oral and pharyngeal responses such as chewing and vocalization. Oral-pharyngeal reflexes demonstrate a range in complexity. Some sensory stimuli will evoke a series of responses, as is often evident in the infant, and other stimuli will evoke a complex multiple-level recruitment of muscles in a sequence, as in pharyngeal swallowing. Certain sensory inputs evoke an entire motor behavior pattern, such as taste avoidance or facial expression. The oral-pharyngeal reflexes are critical to maintaining life and ultimately serve functions that the oral and pharyngeal regions have in common, such as communication, feeding, and breathing.
(I) Introduction
(A) The oral and pharyngeal regions
The oral and pharyngeal regions are anatomically and functionally distinct but are well-integrated into certain motor responses, such as speech and swallowing. Both regions are composed of striated muscle. The oral region encompasses the anterior portal of the gastrointestinal tract and possesses the hardest articulating surfaces of the body in the dentition of the two arches. The lips are functionally integrated within the oral region and provide the most exposed tissue. The two dental arches of the oral cavity surround a complex organ—the tongue—that has exquisite ability and fine motor control of multiple muscles. The tongue changes its shape and form so that it can assist in mixing food, holding the food between the teeth for chewing, and then transporting the bolus of food into the pharynx, where the bolus is recruited in the pharyngeal swallow. The oral cavity transforms anatomically into the pharynx through the oropharynx. The pharynx is divided into the nasopharynx and oropharynx. The oral cavity and pharynx are innervated by several cranial nerves: trigeminal (V), glossopharyngeal (IX), vagal (X), accessory (XI), and hypoglossal (XII).
To use both the tongue and the dentition effectively, the oral cavity and the lips possess some of the richest sensory modalities of any region of the body. Somatosensory input from the oral region provides the central nervous system (CNS) with the ability to perceive extremely sensitive two-point discrimination, texture, and shape. Sensory receptors of the periodontal region provide feedback related to loads developed on the dentition and the direction of that touch/pressure. Temperature and nociceptive sensory fibers innervate much of the oral region, including the palate, tongue, and pillars of fauces. The oral cavity also possesses the highest density of sensory receptors capable of responding to multiple types of chemicals and that provide the perception within the CNS of myriad tastes. The anterior tongue is more receptive to sour and bitter substances, which are often associated with poisonous or harmful food.
The pharynx consists of the nasopharynx (also called the epipharynx) and the oropharynx. The most caudal region of the oropharynx, immediately above the larynx, is often referred to as the hypopharynx. The nasopharynx composes that part of the pharynx above and behind the soft palate. The oropharynx extends from the nasopharynx caudally to the larynx. The pharynx, while anatomically joined to the oral region, really serves as a conduit for two different but interacting functions: (1) moving boluses of air to and from the lungs, and (2) transferring boluses of food and liquids from the oral cavity toward the stomach through the esophagus. The pharynx must coordinate respiration and swallowing. The pharynx has two rostral portals, with the oral cavity usually providing the bolus of food and the nasopharynx providing passage for air. Under certain conditions, the oral region will become a second passage for moving air to complement the nasopharynx (i.e., oral respiration). Most of the time, the pharynx serves as part of the respiratory pathway that includes the nose, nasopharynx, oropharynx, larynx, trachea, bronchi, and smaller divisions, to terminate in small alveolar sacs. The palatal muscles, the extrinsic tongue muscles, and the hyoid muscles maintain the patency of the pharyngeal airway. The muscles of the tongue are pivotal to the control of the pharynx. While these muscles assist in maintaining the pharyngeal passageway, the pharyngeal constrictors support the walls of the pharynx.
The sensory input from the pharynx provides reflexes that (1) protect the pharynx from substances or mechanisms that impair the pathway, (2) transport substances to and from the esophagus, and (3) induce cardiovascular responses. The pharynx has an array of sensory receptors, with most responding to changes in touch/pressure, water, and chemical stimuli. Sensory input from the pharynx continually streams toward the CNS to adjust the peristaltic contraction of the pharyngeal constrictor muscles during pharyngeal swallowing as well as the level of excitability to elicit successive swallows. Many of these sensory receptors have direct reflexes within the CNS to protect the lungs by controlling the laryngeal muscles. Sensory receptors of the pharyngeal region, when properly stimulated, can induce the contraction of the laryngeal adductor muscles. Sensory input from the pharynx and its portal region around the pillars of fauces protects the lungs from aspiration of liquids and food while eliciting the swallow. In addition to its functions in respiration and swallowing, the pharynx is important to the production of intelligible speech sounds and interacts with the oral region as a resonant chamber.
(B) Defining the term “reflex ”
Classic definitions of reflexes, such as those in the tradition of Charles Sherrington (Miller and Sherrington, 1916), indicate that specific sensory inputs will subconsciously induce motor responses that often have reciprocal effects on different motoneuron pools. The reflex consists of neural receptors located on sensory fibers, the afferent pathway of sensory fibers, central synaptic connections that almost always use interneurons, the efferent pathway composed of the motoneurons, or autonomic post-ganglionic neurons innervating the effector organ. The effector organ in a somatic reflex is the striated muscle innervated by the alpha motoneuron. The effector organ in an autonomic reflex involves a peripheral neuron extending its post-ganglionic axon to internal organs and blood vessels. Reflexes provide a relatively “hardwired” circuit through the CNS to control a set of often antagonistic muscles to coordinate a given motor response (e.g., jaw opening).
While reflexes can often be studied in the laboratory animal to assess the neurological system subservient to them, most reflexes function more as supporting networks of neurons assisting in the control of complex motor responses. The intact mammalian system relies on continuous reflex networks to modify motoneurons that receive synaptic input from higher centers to control or modulate speech, intra-oral transport of food, chewing, incising, and swallowing. Each of these complex motor responses is modulated by continued reflex arcs that adjust the final motor signal to the designated muscles.
Much of the information presented below has been discussed in greater detail in several valuable books and reviews (Bradley and Mistretta, 1975; Anderson and Matthews, 1976; Sessle and Hannam, 1976; Dubner et al., 1978; Kawamura and Dubner, 1981; Carpenter, 1989; Jean, 1990; Jean et al., 1994; Capra, 1995; Sant’Ambrogio et al., 1995; Shaker and Lang, 1997; Pierce and Worsnop, 1999). Many studies have emphasized the larynx and its role in the upper airway with the use of multiple reflexes to maintain patency and protect the respiratory system, but such information is beyond the scope of this review and is found in several excellent publications (Sant’Ambrogio et al., 1995).
(II) Reflexes
The oral-pharyngeal region serves multiple functions: sucking, swallowing, respiration, speech, and mastication, as well as gagging, vomiting, coughing, and snoring. These functions involve the large jaw muscles, the smaller jaw-opening muscles, and muscles of the face, lips, cheeks, tongue, palate, and pharynx. Reflexes underlie and support the interaction of these muscles.
(A) Simple oral reflexes
The simple oral reflexes are defined by a relatively short latency response to a particular type of stimulus (Table 1). Some of these reflexes are discussed below.
(1) Orienting reflex: tongue-tongue reflex
Touching the tongue of a newborn human infant with a particular tactile stimulus will induce movement of the tongue tip to the side of the stimulus (Weiffenbach, 1972). Supposedly, the ipsilateral movement brings the sensory-rich tip of the tongue into alignment with the stimulus. The response can be elicited in a variety of states, including that between feedings as well as when the infant is awake or asleep. The response can be elicited by stroking the tongue with a wax probe (1 mm in diameter) on all the surfaces. The most effective method for testing this response is to apply a single punctate stimulus to one position. Stimulation with nylon filaments at the end of a Lucite rod allows for a range of force from 68 mg to 3.6 g. The more intense the stimulus, the higher the probability of eliciting the tongue movement. The most intense stimulus elicits a positive response 95% of the time. The least intense stimulus elicits the response in less than 15% of the time. Either a medially or a laterally directed stroking along the margin can determine the direction of the tongue movement.
(2) Modulating the tongue
Several reflexes affect tongue muscles, and most are evoked by stimulation of branches of the trigeminal nerve—hence the term “trigemino-hypoglossal reflexes”.
(a) Lingual-hypoglossal reflex
The tongue has striated muscles that compose muscle mass without attachment to bone (i.e., intrinsic muscles) and muscles that attach to at least one bone (i.e., extrinsic muscles). The intrinsic muscles alter the shape of the tongue. The extrinsic muscles allow the tongue to protrude, retrude, and move laterally (Abd-El-Malek, 1955). The genioglossus muscle is the main protruding muscle, whereas the styloglossus and hyoglossus muscles retract the tongue. Early studies showed that mechanical stimuli applied to the “backward-directed filiform papillae” caused the tongue to curve the tip upward reflexively (Miller and Sherrington, 1916). Electrical stimuli applied to the lingual-chorda tympani nerve caused the tongue to become “hollowed from side to side so that its lateral edges curve upward and the back part is somewhat depressed” (Miller and Sherrington, 1916). Stimulation of either lingual nerve produced bilateral but not symmetrical movement of the tongue. This same lingual nerve stimulation also induced other reflexes, including opening of the jaws, closure of the glottis, and elevation of the palate, all of which accompanied reflex changes in the tongue.
Stimulation of the lingual nerve induces discharges in the fibers of the hypoglossal nerve, with a latency indicative of a polysynaptic reflex (Blom, 1960; Porter, 1967). Stimulation of the lingual nerve induces an early evoked potential (latency of 4 to 7 ms) followed by a later and smaller second potential (Porter, 1967). Stimulation of the contralateral lingual nerve evokes a similar response but with a slightly longer delay of 1 to 2 ms. Simultaneous bilateral stimulation of both lingual nerves induces a larger evoked response for both the early (large) and later (smaller) components. The stimulation could be paired to both bilateral lingual nerves in a conditioning-test sequence. This approach shows that the contralateral lingual sensory fibers have a polysynaptic excitatory influence on the hypoglossal axons innervating the intrinsic tongue muscles. After this period of facilitation (8 to 10 ms), the ipsilateral lingual-hypoglossal reflex is depressed, suggesting that the contralateral lingual sensory fibers also excite interneurons that eventually inhibit contralateral hypoglossal motoneurons. Mechanical stimulation of the tongue will induce the same reflex as direct stimulation of the lingual sensory nerve fibers (Porter, 1967; Yokota et al., 1974). A light probe drawn slowly from the back to the front along the dorsal tongue surface induces increased numbers of excitatory post-synaptic potentials (EPSPs) within the hypoglossal motoneurons. In contrast, pressure on other regions of the tongue or stretching of the tongue does not synaptically affect the hypoglossal motoneurons. This last finding is significant, since most investigators believe that the coordination of reflexes of tongue muscles relies heavily on exteroceptors on the tongue surface.
The effect of sensory input on hypoglossal motoneurons depends on where the sensory input is elicited in the oral-pharyngeal region and on whether the muscles cause the tongue to retrude or protrude (Morimoto et al., 1968). The medial branch of the hypoglossal nerve innervates the protrusive muscle and the intrinsic muscles (Fig. 1). The lateral division innervates the retrusive muscles. This anatomical distinction of the hypoglossal nerve has allowed for detailed analysis of the many reflexes that affect the two major divisions of the tongue muscles. Most of these reflexes, induced through the lingual nerve, use di- and trisynaptic pathways with relatively rapid responses. Some of these interneurons are located in the area ventrolateral to the hypoglossal nucleus (the perihypoglossal region) and include interneurons and premotor neurons that transmit inputs from trigeminal, hypoglossal, and vagal afferents (Ono et al., 1998). Intracellular recordings from the hypoglossal motoneurons support findings from extracellular recordings from the hypoglossal nerve branches. Whether intracellular EPSPs or short- and long-lasting inhibitory post-synaptic potentials (IPSPs) are elicited by stimulation of the lingual nerve depends upon the muscle the motoneuron is innervating (Blom, 1960; Lowe et al., 1976; Takata, 1981). Stimulation of the lingual nerve evokes mostly IPSPs in motoneurons innervating protruding muscles. Most motoneurons to protruding muscles (40%) demonstrate an IPSP or a short IPSP-long IPSP sequence (30%). A glycine antagonist (e.g., strychnine) diminishes the short IPSP, and picrotoxin diminishes the long IPSP. The remaining 30% of the protruding motoneurons first produce an EPSP, followed by an IPSP. In contrast, most of the hypoglossal motoneurons innervating the retruding tongue muscles (68%) exhibit an EPSP-IPSP sequence. A small percentage of retruding motoneurons (7%) exhibit only an EPSP. The remaining retruding motoneurons (25%) exhibit an IPSP-IPSP sequence. The overall effect of stimulating the lingual nerve is to induce tongue retraction.
Stimulating the lingual nerve will induce other reflexes, such as jaw opening, that can occur simultaneously with reflexes in the tongue. Studies in the anesthetized cat show that inducing pharyngeal swallowing by stimulation of the superior laryngeal nerve, or by water applied to the tongue surface, will suppress the lingual-hypoglossal reflex induced by stimulating the low-threshold lingual afferents (Ono et al., 1999). This finding suggests that triggering the pharyngeal swallow will suppress several reflexes, including those involving the tongue.
(b) Masseter-hypoglossal reflex
Mechanoreceptors in other areas of the oral-pharyngeal region have sensory fibers that will synaptically affect hypoglossal motoneurons but with a latency slightly longer than that in the lingual-hypoglossal reflex (Blom, 1960; Schmitt et al., 1973; Yamamoto, 1975). Stimulation of the masseter nerve innervating a jaw-closing muscle will elicit tongue reflexes (Fig. 2). This sensory input excites motoneurons innervating intrinsic muscles or the genioglossus (Sumino and Nakamura, 1974). The primary effect of stimulating the masseter nerve, however, is to inhibit hypoglossal motoneurons, particularly to retrusive tongue muscles (Mizuno and Sauerland, 1970). The close interaction of tongue movements with the jaw during feeding, chewing, and swallowing suggests that such reflexes exist between muscles of the jaw and tongue.
(c) Inferior alveolar-hypoglossal reflex
Stimulation of another branch of the trigeminal system, the inferior alveolar nerve, usually inhibits both retrusive and protrusive motoneurons to the tongue muscles. Electrical stimulation of the lower lip in the human inhibits tonic activity in the genioglossus muscle during tongue protrusion (Ishiwata et al., 1997).
(d) Glossopharyngeal-hypoglossal reflex
Stimulation of the glossopharyngeal nerve, which carries sensory fibers innervating the posterior one-third of the tongue, will induce reflex discharges in hypoglossal motor nerve fibers (Blom, 1960). Stimulation of the glossopharyngeal and superior laryngeal sensory fibers excites the intrinsic tongue muscles and the genioglossus muscle while usually inhibiting the retrusive motoneurons (Lowe et al., 1976) (Fig. 3). These findings suggest that most stimuli applied to the anterior oral region induce tongue retraction, whereas stimuli applied to the pharyngeal region induce tongue protrusion. The various sensory inputs that affect the hypoglossal motoneurons also interact to modify each other. Stimulating the infra-orbital nerve, the tooth pulp, the TMJ, the glossopharyngeal and lingual nerves, or nerves from the jaw muscles as conditioning stimuli before eliciting a hypoglossal reflex (with electrical stimulation of the lingual nerve) will often result in short- and long-term inhibitions lasting many milliseconds (150 to 400 ms).
(e) Jaw-hypoglossal reflex
Research studies in both laboratory animals and humans indicate that opening the mandible increases activity in the genioglossus muscle (Lowe and Sessle, 1973; Lowe, 1984; Ishiwata et al., 1997, 2000). Sensory receptors from the TMJ that have fibers passing through the mandibular nerve may modulate tongue reflexes (Lowe and Sessle, 1973). Opening the mandible increases the electromyographic (EMG) activity in the human genioglossus. In humans, the position of the mandible can affect the amount of tongue protrusion and may affect tongue posture (Lowe et al., 1977). The significance of such a reflex is to protect the pharyngeal passage during respiration. Protruding the tongue brings the posterior tongue forward and opens the pharyngeal passage.
(f) Temporalis-hypoglossal reflex
Depressing the mandible in the decerebrated cat induces retraction of the tongue. This reflex is called the jaw-tongue reflex and has been studied in the styloglossus muscle, one of the tongue-retracting muscles. Determining whether sensory input comes from the TMJ depends on whether the jaw-tongue reflex decreases with the application of a local anesthetic to the TMJ (Morimoto et al., 1978). Morimoto et al. indicated that this effect might be due to the anesthetic affecting the nerve branches to the temporalis and masseter muscles as these nerve branches pass through this region near the joint. Isolating the condylar process from the mandibular body without damaging the masseteric and auriculo-temporal nerves allowed for rotation of the condyle alone. Rotation of only the condyle did not evoke a reflex to the styloglossus muscle. Injecting xylocaine solution into the TMJ capsule did not stop the evoking of the jaw-tongue reflex that lowers the mandible and condyle. Sectioning the masseteric nerve did not affect the tongue reflex. In contrast, removing the insertion of the temporalis muscle to the temporal bone significantly decreased the jaw-tongue reflex for the ipsilateral tongue muscle. Isolating the coronoid process with its attached temporalis muscle and pulling it downward induced the jaw-tongue reflex. The above evidence strongly suggests that sensory input from the temporalis muscle affects the hypoglossal motoneurons innervating the tongue.
More recent studies have shown that the application of punctate light pressure to the temporalis muscle induces a depolarizing potential in hypoglossal motoneurons (Ishiwata et al., 2000). During jaw opening, electrical stimulation of the stretching temporalis muscle slightly inhibits the EMG activity in the genioglossus and styloglossus muscles. The afferent fibers from the muscle spindles in the jaw-closing muscles proceed to their cell bodies, located in the trigeminal mesencephalic nucleus, through a central tract, Probst’s tract. Lesions in this tract abolish the jaw-tongue reflex. These findings suggest that Group II muscle spindle afferents from the temporalis muscle are primarily responsible for the jaw-hypoglossal reflex.
(g) Taste-hypoglossal reflex
The stimulus of taste can elicit a tongue motor response. Placing a drop of glucose solution on one side of the tongue in the human neonate evokes a lateral tongue movement (Weiffenbach and Thach, 1973). The type of taste stimulus can induce different tongue movements in the human infant (Steiner, 1973). In decerebrate rats, the application of sucrose, NaCl, or HCl to the tongue evokes rhythmic tongue protrusions followed by lateral tongue movements (Grill and Norgren, 1978a,b). In contrast, quinine evokes gaping, or wide opening of the jaw. Recordings from hypoglossal motoneurons in the decerebrate rat indicate that the application of NaCl solution to the dorsal surface induces a rhythmic motor discharge in the hypoglossal nerve fibers (Kawamura and Dubner, 1981). Applying cold to the tongue surface induces a tonic discharge with some rhythmic pattern. Applying warmth induces a sustained tonic discharge. In the intact but lightly anesthetized rat, applying sucrose, NaCl, HCl, quinine-HCl, or different temperatures of water (5° or 43°C) can induce rhythmic bursts from the hypoglossal motoneurons. Approximately 20% of the hypoglossal motoneurons sampled exhibit tonic discharges without any sensory stimulation in the decerebrate rat and a smaller percentage (7%) in the lightly anesthetized rat. These results indicate that multiple tongue sensory inputs synaptically affect hypoglossal motoneurons.
(B) Simple pharyngeal reflexes
(1) Protecting the pharynx and respiratory system
(a) Gag reflex
The gag reflex is one of the reflexes induced by stimulation of the pharyngeal and velar regions (Roberts et al., 1989; Davies et al., 1995). It is induced by touching of the base of the tongue, soft palate, uvula, or posterior pharyngeal wall with a tongue blade, or finger. The gag reflex is considered a protective reflex supposedly intended to prevent unwanted material (i.e., foreign objects) from entering the pharynx. The gag reflex can be defined strictly as constriction of the pharynx but often is associated with more complex behavioral responses, including lowering of the mandible, forward and downward movement of the tongue, and pharyngeal and velar constriction associated with mild coughing (Leder, 1996). The more severe gagging (i.e., hypergag) involves forceful pharyngeal and velar contraction, and retching. The more severe gag actually appears to be a combination of reflex responses, with both gagging and some aspect of the emetic response.
The evoking of the gag reflex varies widely among subjects (Perlman et al., 1989; Leder, 1996). Studies in 140 normal subjects of various ages showed that the gag reflex was absent in 37% of the subjects, whereas pharyngeal sensation was absent in only one subject (Davies et al., 1995). In another study, 13% of the 69 subjects without dysphagia had no gag reflex (Leder, 1996), 84% of the subjects had a normal gag reflex, and 3% had a hypergag response. Pharyngeal sensation and the gag reflex are not as tightly interrelated as might be expected. The gag reflex has been used clinically as a potential indicator of dysphagia. However, the gag reflex is difficult to elicit in many normal subjects and does not appear to be a valid reflex for assessing patients with compromised pharyngeal swallowing (Leder, 1996). While acute stroke and dysphagia are often associated with aspiration, the presence of the gag reflex does not correlate with the potential for aspiration (Davies et al., 1995).
The most effective method for blocking the gag reflex in humans is to induce a pharyngeal plexus block (Valley et al., 1992). Applying a topical 10% lidocaine spray to the oropharyngeal mucosa is much less effective in blocking the onset of the gag reflex. Such findings suggest that the sensory input evoking the gag reflex depends upon stimulating both superficial and deep sensory receptors.
(b) Apneic reflex
Stimulation of regions within the oral cavity, pharynx, and larynx evokes reflexes that protect the respiratory tract. The reflexes consist of apposition of the epiglottis and aryepiglottic folds, adduction of the false and true vocal cords, and inducement of respiratory cessation (i.e., apnea). Pharyngeal and laryngeal stimulation induce prolonged apnea in both laboratory animals (Storey and Johnson, 1975; Miller and Dunmire, 1976; Boggs and Bartlett, 1982; Khater-Boidin et al., 1994) and humans (Davies et al., 1988, 1989; Pickens et al., 1988; Loughlin, 1989; Thach et al., 1990).
Stimulation of the oral region in the kitten induces apneic responses that are particularly evident in the early post-natal stages of development (Khater-Boidin et al., 1994). Stimulation of the lingual nerve, which carries the sensory input from the anterior two-thirds of the tongue, induces apnea in the anesthetized animal. Applying serotonin to the floor of the IVth ventricle evokes the same type of apnea, suggesting that this region of the brain stem includes neurons that coordinate this reflex. The duration of the apneic responses decreases with age (Wallois et al., 1993). In the first 5 days, swallowing in the kitten is rarely induced with oral or pharyngeal stimulation, whereas apnea is readily induced (Miller and Dunmire, 1976). In the next 3 weeks, apnea is elicited with swallowing. By the third to fourth weeks, the adult response of respiration alternating with swallowing appears in the kitten. These results show that the coordination between breathing and swallowing matures, which suggests that the central brain stem neuronal network continues to develop the synaptic contacts post-natally. There is an early period in mammalian CNS development in which oral-pharyngeal stimulation has a much more significant impact on inducing cessation of breathing.
Studies in newborn lambs and puppies show that placing isotonic saline on the laryngeal surface of the epiglottis and the arytenoid processes evokes swallowing with little effect on breathing (Storey and Johnson, 1975; Boggs and Bartlett, 1982). In contrast, applying water to the same region induces several swallows and instant apnea. The apnea remains until the water is removed from the pharyngeal region. Applying amniotic fluid does not elicit swallowing and does not affect breathing. Recordings from single sensory fibers of the superior laryngeal nerve innervating the hypopharynx demonstrate that sensory fibers discharge to a water stimulus, which is indicative of the presence of water receptors in the hypopharynx.
Neonatal apnea is frequently associated with upper airway obstruction (Thach and Stark, 1979; Martin et al., 1994). Inducing CO2 re-breathing (i.e., hypercapnia) in an anesthetized, paralyzed, ventilated piglet increases activity in the hypoglossal nerve fibers. Discontinuation of ventilation for 60 sec to induce both hypercapnia and hypoxia markedly increases the hypoglossal nerve activity. This reflex is suppressed with cooling of the ventral medullary surface, suggesting that this central chemosensitive region is vital to this hypoglossal response. Any damage to or dysfunction of the ventral medullary site could suppress the hypoglossal reflex that maintains a patent upper airway, particularly in premature infants (Miller and Martin, 1992).
(c) Aspiration reflex
Mechanical stimulation of the nasopharyngeal mucosa causes repeated contractions of the diaphragm. This has been described as the aspiration reflex or sniff reflex (Korpas and Tomori, 1979; Widdicombe, 1986). A catheter, water, saline, or a jet of air will evoke the reflex when these stimuli are placed on the nasopharyngeal mucosa. Large myelinated sensory fibers serve as the afferent pathway (Nail et al., 1969, 1972). The fibers could be carried in both the glossopharyngeal nerve and branches of the trigeminal nerves. The main sensory innervation of the nasopharynx is from trigeminal nerve fibers. The aspiration reflex is more prevalent in some species (e.g., cats, dogs, rats, mice, pigs) than in others (e.g., rabbits, guinea pigs, humans). In humans, the insertion of a perinasal catheter rarely evokes the response.
Aspiration of material into the respiratory system (i.e., trachea, bronchi, and alveoli) does appear to relate to pharyngeal sensation. Pharyngeal sensation is rarely tested, but the one study that compared aspiration with pharyngeal sensation indicated that intact pharyngeal sensation was associated with no aspiration (Davies et al., 1995). Much more experimental and clinical research needs to be done in this area.
(2) Respiratory reflexes
Stimulation of the larynx has marked effects on the respiratory pathway, but similar reflexes are often induced by stimulation of the pharynx. Stimulation of the pharynx can induce forced expiration (Korpas and Tomori, 1979), affect the bronchial secretions (Phipps and Richardson, 1976), and induce constriction (Karlsson et al., 1988). These respiratory reflexes protect the respiratory system from invading material and complement similar reflexes induced rostrally and caudally to the oral-pharyngeal region.
(3) Cardiovascular reflexes
Laryngeal and esophageal stimulation induces cardiovascular reflexes (Korpas and Tomori, 1979; Widdicombe, 1986). Some reflexes induced by pharyngeal stimulation can affect the autonomic control of the cardiovascular system, inducing bradycardia and, less often, tachycardia (Kaplan and Schuster, 1991; Cunningham et al., 1992). Often, these cardiovascular responses are induced with swallowing (Levin and Posner, 1972; Engel et al., 1976; Palmer, 1976; Kalloor et al., 1977; Elam et al., 1989; Shirayama et al., 1989). The issue is whether a particular stimulus to the pharyngeal region (e.g., mechanical, temperature, pressure) induces the cardiovascular reflex independently of swallowing, in parallel with swallowing, or as a result of swallowing. The sensory inputs that induce the cardiovascular responses include carbonated beverages (Elam et al., 1989), hot liquids (Armstrong et al., 1985; Kunis et al., 1986), and cold liquids (Armstrong et al., 1985).
(C) Complex reflexes/responses
Some of the complex reflexes are discussed below (Table 2).
(1) Oral respiration
Some reflexes modifying the oral-pharyngeal region may depend upon alterations in sensory input from another region. Oral respiration is a term applied to chronic breathing through the mouth during normal rest without exercise. Many individuals who do this should be evaluated for narrowing of the nasal and oropharyngeal passages. Sufficient narrowing of these passages can impair the movement of adequate tidal volumes of O2 or CO2. When nasal impedance exceeds a certain level, subjects will breathe through their mouths. Sensory input from the oral-pharyngeal region might monitor airway resistance or effort. Subjects detect static changes in airway resistance of 25 to 30% and dynamic changes as high as 42% (Chiou et al., 1996). The source of this sensory perception is still unknown. Sensory receptors responding to pressure reduction exist in the pharynx and larynx. Presumably, when nasal obstruction decreases airflow sufficiently to modify the stimulation of these pressure receptors, the CNS induces oral respiration by parting the lips, flattening the tongue, raising the soft palate, and slightly lowering the mandible.
Inducing total nasal obstruction in awake lambs induces oral respiration only after a significant decrease in blood levels of O2 (hypoxanemia) (Harding, 1986). When a few breaths restore the blood gas levels, the mouth closes. This suggests that the central neural pathway inducing the complex oral-pharyngeal response for oral respiration depends primarily on changes in systemic blood gas levels for its sensory cue. Experimentally inducing nasal obstruction in the monkey produces rhythmic EMG discharges from muscles of the tongue (Miller, 1978) and pharynx (Rowe et al., 1984), as well as from mandibular and facial muscles. These findings indicate that the central brain stem respiratory pathway can synaptically control specific oral-pharyngeal muscles and their motoneurons.
Experimental studies in the rat indicate that the pattern of stimulation of the hypoglossal nerve affecting the tongue muscles can influence the route of airflow (Fuller et al., 1999). Selective stimulation of the protruding muscles may favor oral respiration. Simultaneous activity in both retracting and protruding muscles may support more nasal respiration. During intensive exercise, the genioglossus EMG activity increases significantly with the change from nasal to oronasal breathing (Williams et al., 1998).
(2) Maintaining a patent pharyngeal passage: respiratory-tongue response
The respiratory pathway extends from the nares and lips to the alveoli in the lungs. Several sites above the trachea can increase airway resistance. These sites include the external nares in the nasopharynx, the tongue in the oropharynx, the hyoid bone, and the vocal cords in the larynx (Van de Graaff et al., 1984). Muscles in these regions can significantly reduce resistance and prevent obstruction. These muscles are active in phase with respiration and will enhance their activity when there are changes in chemical stimulation of peripheral and central respiratory receptors that monitor O2 and CO2 levels.
As part of the upper airway, the pharynx is one site that can alter airway resistance. During sleep in normal subjects, the upper airway in the region of the pharynx partially collapses and increases the upper airway resistance (Pierce and Worsnop, 1999). The posture of the tongue is one of the primary factors that determine the patency of the pharynx. The activity in the tongue-protruding muscle (genioglossus) can improve upper airway flow mechanics by dilating the oropharynx and reducing flow resistance (Bartlett, 1986). Some subjects suffering from obstructive sleep apnea demonstrate a greater collapse of the upper airway that is sufficient to induce hypoxia. Such subjects demonstrate a loss of tone in the oropharynx and less inspiratory drive of the tongue muscles (Urban et al., 1996). Evaluation of pre-term infants with the clinical diagnosis of apnea, by means of a pressure catheter in the pharynx, indicates that most apneic episodes are associated with “high pharyngeal obstruction” (Mathew et al., 1982).
Studies on the effects of increased CO2 blood levels (hypercapnia) and decreased O2 blood levels (hypoxia) on hypoglossal motoneurons in the decerebrate cat indicate a strong control of hypoglossal motoneurons by the respiratory pathway (Mitra et al., 1986; Bartlett and St. John, 1988; Withington-Wray et al., 1988; Nunez-Abades et al., 1993; Martin et al., 1994; Hayashi and McCrimmon, 1996; Eastwood et al., 1998; Fenik et al., 1998). In the sleeping cat, induction of hyperoxic hypercapnia increases the phasic EMG activity from the genioglossus muscle (Haxhiu et al., 1987). This EMG activity increases more at higher levels of CO2 (3.4 to 7.4%) than at lower levels (0 to 3.4%).
Much of this research on oral respiration has emphasized the effect on the genioglossus muscle. However, studies in the anesthetized rat indicate that stimulation of chemoreceptors can induce activity in both tongue-protruding and -retruding muscles (Fregosi and Fuller, 1997; Fuller et al., 1998, 1999). Airway occlusion in an anesthetized, tracheotomized rat (i.e., one in which both hypercapnia and hypoxia have been induced) increases EMG activity in both retractor and protractor tongue muscles, and the net effect is a greater retraction of the tongue (Fig. 4). Inducing hyperoxic hypercapnia and poikilocapnic hypoxia also increases activity in both retractor and protractor muscles. The combined effect on both groups of muscles alters the tongue retraction force. Co-activation of both retracting and protruding tongue muscles does not dilate the pharyngeal airway but does decrease pharyngeal collapsibility (Fuller et al., 1999). In contrast, activation of only the tongue-protruding muscles dilates the oropharynx but has little effect on pharyngeal airway collapsibility.
A second sensory input of pressure can modify the tongue muscles. In anesthetized rabbits, a change in airway breathing from normal pharyngeal passage breathing to tracheal breathing affects pharyngeal closure. In the animal breathing through the trachea, pharyngeal closure is restored either by blocking the tracheal tube to re-institute pharyngeal breathing or by creating cyclic pressure changes in the nose and pharynx (Abu-Osba et al., 1981). Applying a topical anesthetic to the upper airway mucous membrane (nose, pharynx, and larynx) eliminates this sensory input from pressure and results in an obstructed pharyngeal airway and death. This reflex effect is shown only in live animals. Dead animals have a patent airway resistant to collapse. This finding suggests that the airway-restricting muscles actively close the pharynx unless sensory input from cyclic pressures of flowing air is present. Applying pressure oscillations to the isolated upper airway in the tracheotomized, unanesthetized dog tonically activates the EMG activity of the genioglossus muscle. These low-amplitude, high-frequency pressure oscillations simulate snoring and suggest that the upper airway has mucosal mechanoreceptors that induce activity in the genioglossus muscle to maintain a patent airway (Eastwood et al., 1999).
The hyoid arch is a third region that can affect the patent pharynx (Brouillette and Thach, 1980). The hyoid arch partially surrounds the hypopharynx above the larynx and below the tongue (Van de Graaff et al., 1984). More than 12 muscles insert into the hyoid arch. Inducing hypercapnia in anesthetized dogs increases the EMG activity in the geniohyoid, thyrohyoid, and sternohyoid muscles. Increasing the negative pressure in the upper airway enhances the duration and amplitude of the EMG activity in these muscles. The muscles attached to the hyoid arch can affect patency and resistance of the upper airway, and are reflexively controlled by chemical and pressure changes.
(3) Transport and protection: pharynx
(a) Pharyngeal swallowing
The most complex all-or-none reflex elicited from the pharyngeal region is pharyngeal swallowing. While swallowing can be elicited without previous oral preparation, the normal functional sequence involves initial food intake, such as incising hard food or sipping liquid, masticating or chewing food to a smaller size or bolus, and then orally transporting the bolus toward the pillars of fauces (the anterior portals of the pharynx). Pharyngeal swallowing involves muscles of the soft palate, tongue, hyoid bone, pharynx, and larynx (Fig. 5). The soft palate elevates, the pharyngeal walls appose, the hyoid and larynx elevate, the laryngeal vestibule closes, the epiglottis tilts downward, and the upper esophageal sphincter relaxes and opens (Donner et al., 1985). The extrinsic tongue muscles include the genioglossus. The intrinsic muscles provide the ability to change the shape of the tongue, as in flattening or developing grooves. The tongue changes position by movement of the hyoid bone that involves the supra- and infrahyoid muscles. The pharynx is composed of the superior, middle, and inferior constrictor muscles that overlap to form a continuous sheath of muscles extending from the cranium to the esophagus. The muscles are thin, and their fibers are oriented obliquely and insert into a posterior median raphe (Bosma et al., 1986). The posterior pharyngeal wall includes a thin layer of elastic tissue that allows the pharyngeal wall to expand and contract without folding. During pharyngeal swallowing, the lateral and posterior walls of the pharynx move medially and anteriorly, reducing the pharyngeal area during the development of a caudally directed wavelike motion. Three other muscles elevate the pharynx—the stylopharyngeus, salpingopharyngeus, and palatopharyngeus. The muscles involved in pharyngeal swallowing are innervated by several motoneurons in several cranial nerves, including the trigeminal (V), facial (VII), glossopharyngeal (IX), vagal (X), accessory (XI), and hypoglossal nerves (XII).
The sensory regions that evoke pharyngeal swallowing with application of pressure or various liquids in both humans and laboratory animals include the soft palate, uvula, dorsal tongue surface, faucial pillars, dorsal pharyngeal wall, pharyngeal surface of the epiglottis, and glosso-epiglottidinal sinus (Miller and Sherrington, 1916; Pommerenke, 1928; Storey, 1968a,b; Sinclair, 1970, 1971). The effectiveness of eliciting pharyngeal swallowing in humans with touch/pressure depends upon the site and the intensity of the stimulation (Pommerenke, 1928). Superficial touch/pressure with a blunt rod is most effective in eliciting pharyngeal swallowing when applied to the pillars of fauces and least effective on the uvula and soft palate. The application of greater pressure, presumably recruiting both superficial and deep mechanoreceptors, will evoke swallowing most readily from the posterior pharyngeal wall. Subjects differ as to their most sensitive regions for evoking swallowing with touch/pressure. No studies have defined the factors that determine the distribution of the sensory fibers in the pharynx and the relevance of early conditioning stimuli to this distribution.
In anesthetized animals, the pre-epiglottic sinus in front of the base of the epiglottis, the glottis, and the base of the epiglottis are the most sensitive sites at which to evoke swallowing with touch/pressure and liquids (Storey, 1968a,b; Sinclair, 1970). When normal subjects swallow, the bulk of the bolus is held on the epiglottis before the forward and upward movement of the larynx (Ardran and Kemp, 1952). Detailed videofluoroscopic analysis of humans swallowing 5-mL sample substances of different consistencies shows that most pharyngeal swallows occur after the head of the bolus passes the radiopaque marker on the anterior faucial pillars (Linden et al., 1989). Not one of the 30 pharyngeal swallows tested in the 12 subjects began with arrival of the bolus to the anterior pillars. It appears that the pharyngeal area needs to be stimulated in the normal subject to elicit pharyngeal swallowing.
A well-used clinical approach to facilitate the evoking of swallowing in dysphagic patients is to apply a cold probe to the anterior faucial arches (Chi-Fishman et al., 1994). One study analyzed 25 neurologically impaired patients who presented with delays in triggering of the pharyngeal swallow (Lazzara de Lama et al., 1986). Liquid barium or barium paste was offered within 5 sec before and after stimulation of the anterior faucial arches. The time of oral and pharyngeal passage of the bolus was calculated. Pharyngeal transit times for liquids were reduced from 4.2 to 2.4 sec and for solids from 4.1 to 1.6 sec when paired with conditioned stimuli. The cold/touch/pressure on the anterior faucial arches improved the evoking of the swallow that immediately followed within 5 sec and 1 or 2 additional swallows. The improvement occurred after the first application of the probe, suggesting an immediate effect of altering oral sensation on the excitability of the central swallowing pathway. This study was careful not to use large boluses in this impaired group of patients, because larger boluses could have been aspired.
An alternative approach to ascertaining the interaction of sensory input from the oral-pharyngeal region affecting pharyngeal swallowing arose from studies that removed or temporally blocked sensory input. Studies in the early part of the 20th century showed that applying a local cocaine anesthetic to the anterior pillars of normal human subjects impaired their ability to swallow (Pommerenke, 1928). More recent work has shown that applying xylocaine to limited regions of mucosa of the oral-pharyngeal region does not seem to affect the eliciting of pharyngeal swallowing in a normal subject. However, applying a local topical anesthetic to much of the oral and pharyngeal region impairs a subject’s initiation of swallowing (Mansson and Sandberg, 1974, 1975). When a subject receives a topical anesthetic with a spray or gargle, intervals between swallowing become longer. Such results indicate that stimuli are needed to excite receptors within the oral-pharyngeal mucosa to repeat pharyngeal swallowing most effectively.
Several lines of evidence indicate that saliva is a normal stimulus in modifying the threshold to elicit repeated pharyngeal swallowing (Mansson and Sandberg, 1975). When a subject attempted to repeat 11 pharyngeal swallows without a bolus and only with normal saliva, the intervals increased between each swallow, since each was more difficult to induce. Administering methylscopolamine to inhibit salivation increased the intervals between swallowing. When a subject sucked an acid lozenge or a neutral lozenge, the intervals between successive swallows were decreased. The acid lozenge was more effective. Fluid must modify pharyngeal mucosal receptors, but the possibility that hydrogen receptors alter the threshold of inducing swallowing also exists. These different experimental approaches demonstrate that the threshold to elicit swallowing normally depends upon mucosal sensory stimulation, and the primary stimulus can be saliva, fluid, or a dense bolus.
Comparison of the application of water, blunt probes, or ox-hair brushes to different regions of the pharyngeal mucosa in an anesthetized or decerebrate cat has shown that water is a more effective stimulus than pressure in inducing pharyngeal swallowing (Storey, 1968a,b). The properties of water were tested to see if its osmotic or mechanical characteristics are important submodalities that might enhance the evoking of pharyngeal swallowing. There was no distinction among the subsets of properties of water. Studies in the rabbit showed that several chemical solutions—sucrose, quinine-HCl, acetic acid, and ethanol—are more effective than water in evoking swallowing when applied to the pharyngeal or laryngeal mucosa (Shinghai and Shimada, 1976).
Studies in sheep have shown that the fetus swallows randomly in episodes of 1 to 9 minutes’ duration (from 2 to 7 episodes per day) (Mistretta and Bradley, 1977). The fetus swallows large amounts of amniotic fluid in the last third of gestation. The volume swallowed per episode and the total amount swallowed per day vary for each fetus. The peripheral taste sensory system is functional in both lamb and human fetuses. Previous observations of the fetus showed that injecting saccharin into the amnion caused the fetus to increase its normal swallowing rate. This suggests that taste, either reflexively or by a more complex behavioral effect, is an effective sensory stimulus for swallowing before birth (Bradley, 1972; Mistretta, 1972). Based on post-parturition studies of animals (e.g., sheep), the early post-natally developing pharyngeal swallow also depends upon continuous sensory feedback for strength of muscle recruitment and for repeated swallowing. Oral-pharyngeal sensory inputs may be fully functional by the last third of the gestational stage in both the lamb and the human. Sensory input from the pharyngeal mucosa is important to the motor discharge during pharyngeal swallowing (Sumi, 1963-1964). The infant animal is unable to discharge its motor fibers to the muscles active in pharyngeal swallowing with a high, short frequency but elicits a slow, prolonged discharge (Sumi, 1975). Anesthetizing the mucosa of the infant animal prevents the normal motor discharge, indicating that, in early post-natal development, sensory feedback is important for each muscle to contract in the swallow and for the pharyngeal swallow to be completed.
How mechanical stimulation is applied to the oral-pharyngeal region determines the type of reflex. Mechanical stimulation with a probe or brush to the pharyngeal mucosa evokes swallowing or glottic tension. Evoking swallowing with this type of mechanical stimulation is more effective when the stimulus is applied in stroking movements than as a stationary stimulus. Impeding the movement of a bolus of meat through the oropharyngeal region by tying a string around the bolus induces a gag reflex. Such observations suggest that a moving stimulus applied across multiple receptive fields within the oral-pharyngeal region will evoke a particular reflex based on how that stimulus is applied. The stationary or moving application of the stimulus and the direction of its movement across many receptive fields presumably establish different patterns of sensory input over populations of sensory fibers innervating the oral-pharyngeal region (i.e., sensory sheets; Capra, 1995). Which brain stem interneurons are affected depends partly on these patterns of sensory input. This means that a given motoneuron will respond in more than one type of reflex (e.g., coughing, sneezing) but that its recruitment is established by pre-motor interneurons that are triggered from specific sensory inputs (Oku et al., 1994; Shiba et al., 1999).
Electrical stimulation applied to specific branches of the pharyngeal plexus (19a and 19b) of the glossopharyngeal nerve in the anesthetized cat evokes pharyngeal swallowing (Sinclair, 1971). The pattern of stimulation to these branches determines the threshold to evoke pharyngeal swallowing. When nerve fibers are excited artificially by electrical stimulation in frequencies of 30 to 50/sec, pharyngeal swallowing is more easily evoked (Fig. 6). The stimulus pattern is critical to triggering the brain stem network of neurons that elicit pharyngeal swallowing (Doty, 1951; Doty et al., 1968; Miller, 1972a,b). Parameters of this stimulus pattern involving the superior laryngeal nerve and the glossopharyngeal nerve have been studied in considerable detail. These parameters indicate that a minimum number of pulses must be transmitted by the sensory neurons to the brain stem. Once the minimum number of pulses has triggered the brain stem network to elicit the all-or-none pharyngeal swallow, the network maintains its excitability to repeat the pharyngeal swallow without sensory input for at least 700 ms. The patterned sensory input can be too fast and not effectively excite the brain stem interneurons controlling the pharyngeal phase.
Sensory input from the oral-pharyngeal region not only affects the threshold to elicit swallowing but also alters the contraction of muscles involved in the pharyngeal swallow. EMG recordings from the human genioglossus muscle and from the geniohyoid (suprahyoid muscle) during the oral swallowing phase (i.e., oral transport) show that the duration of the EMG activity increases as the consistency and volume of the bolus increase (Hrychshyn and Basmajian, 1972). EMG recordings from the mylohyoid muscle (suprahyoid muscle) during the pharyngeal phase of swallowing indicate that the duration of EMG discharge is shorter in the anesthetized animal (200 ms) than in a fully awake animal (Doty and Bosma, 1956). The duration of excitation to each motoneuron pool for muscles active in pharyngeal swallowing can change with the general excitatory level of the central interneurons. The sequence of activated muscles does not change or changes only minimally (Lowe and Sessle, 1973).
The larger the bolus volume, the higher the velocity of the front face of the bolus as it enters the esophagus (Fisher et al., 1978; Hamlet, 1989). This may be due not to more pharyngeal constrictor muscle contraction but rather to more hyoid bone movement and more and longer opening of the upper esophageal sphincter (Dodds et al., 1988). However, it does suggest an association between the size of the bolus and the strength of the muscle contraction.
(b) Emesis
Emesis (vomiting) is a common medical and clinical problem. The emetic response has three components: nausea, retching, and expulsion. These three components occur as a single process in the human and dog, but their linkage in other animals varies (Wang, 1980; Barnes, 1984; Carpenter, 1989). The emetic response is another complex reflex that involves the sequential contraction of many different muscles, including those of the oral-pharyngeal region. Unlike pharyngeal swallowing, the emetic reflex primarily involves somatic muscles, with secondary contributions from the oral-pharyngeal muscles. The oral, pharyngeal, and esophageal muscles respond in a coordinated reflex that changes intestinal motility, inhibits gastric tone, and opens both the upper and lower esophageal sphincters.
Emesis is often studied by the intravenous injection of apomorphine. This response is lost if the area postrema, a region on the dorsal surface of the brain stem in the floor of the fourth ventricle, is ablated. The area postrema has been referred to as the “chemoceptive trigger zone” for emesis. It lies outside the blood-brain barrier and has a high number of blood vessels. Removal of the area postrema in five human subjects with intractable nausea and vomiting eliminated both responses. The area postrema will induce vomiting when electrically or chemically stimulated (Strominger et al., 1994). It has been postulated that direct pressure applied to the area postrema, as occurs with certain tumors or hydrocephalus, may also excite this region. Emesis can be induced by chemicals applied directly to the gastrointestinal tract (e.g., CuSO4). Electrical stimulation of the abdominal vagus will induce vomiting (Watson et al., 1995). Motion sickness induced in laboratory animals can be eliminated by bilateral labyrinthectomy or by removal of the semicircular canals (Money and Friedberg, 1964), as well as by removal of the area postrema (Brizzee et al., 1980) and the vestibular cerebellum.
(III) Conclusions
The wealth of reflexes that emanate from the oral and pharyngeal regions provides a mechanism for the protection of the anterior portal of the gastrointestinal tract, facilitation of the oral and pharyngeal regions to transport food and liquids, and protection of the same regions for respiratory responses that can include recruiting the oral cavity as part of the upper respiratory tract. Much of this control centers on the tongue and the complex array of muscles that alter its shape and position in the oral and pharyngeal regions. The reflexes provide the underlying framework of neural control upon which more complex motor responses can build and recruit in a functional framework. The sources and types of sensory stimuli determine the potential reflex. Some of the reflexes reflect a simple response, such as retraction of the tongue with stimulation of sensory nerves innervating the mandible or jaw-closing muscles. Other, more complex, reflexes, such as pharyngeal swallowing, require a finely controlled recruitment of multiple cranial motor nuclei that involves the intercession of interneuronal networks that command the sequence of activity. Ultimately, the respiratory central pathway, that can induce oral respiration and maintain a patent airway, will dominate the reflex control of the oral and pharyngeal regions, superseding almost all the reflexes. Future studies will need to focus on the pharyngeal and esophageal interaction and the multiple reflexes that link these two regions, similar to the interaction between the oral and pharyngeal areas.
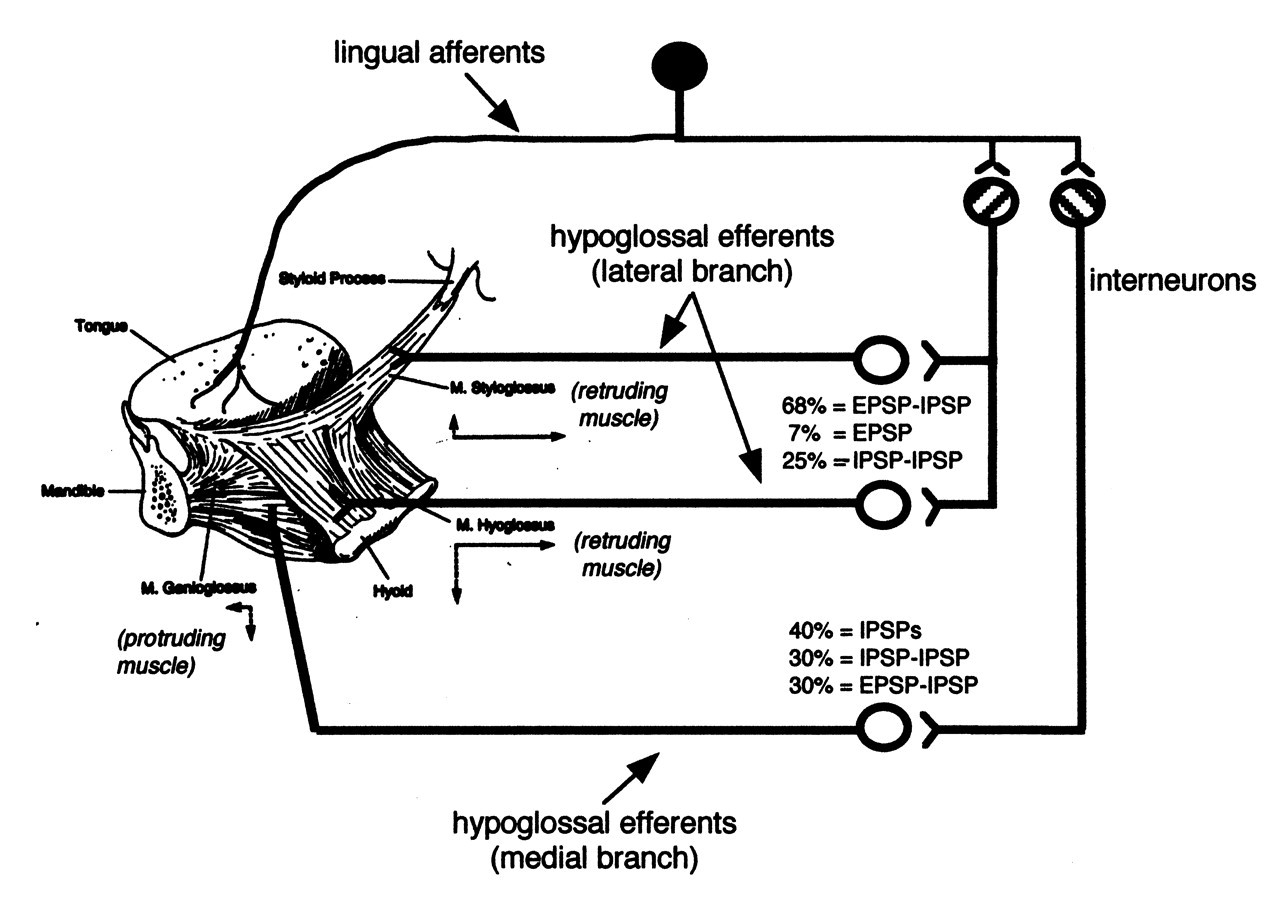
The lingual-hypoglossal reflex. This reflex has been directly analyzed by electrical stimulation of the nerve and extracellular recording from hypoglossal nerve branches. In a second approach, intracellular recordings from hypoglossal motoneurons determining excitatory (EPSP) and inhibitory (IPSP) synaptic potentials are obtained. Mechanical stimulation of the tongue will also evoke these synaptic potentials. An example of the reflex effects is given in one study in the anesthetized cat (Takata, 1981), in which a polysynaptic reflex had different proportions of excitatory and inhibitory effects, depending upon whether the motoneurons innervated the tongue-protruding or -retruding muscles. (The drawing of the dissection of the tongue muscles is adapted with permission from Fregosi and Fuller [1997].)
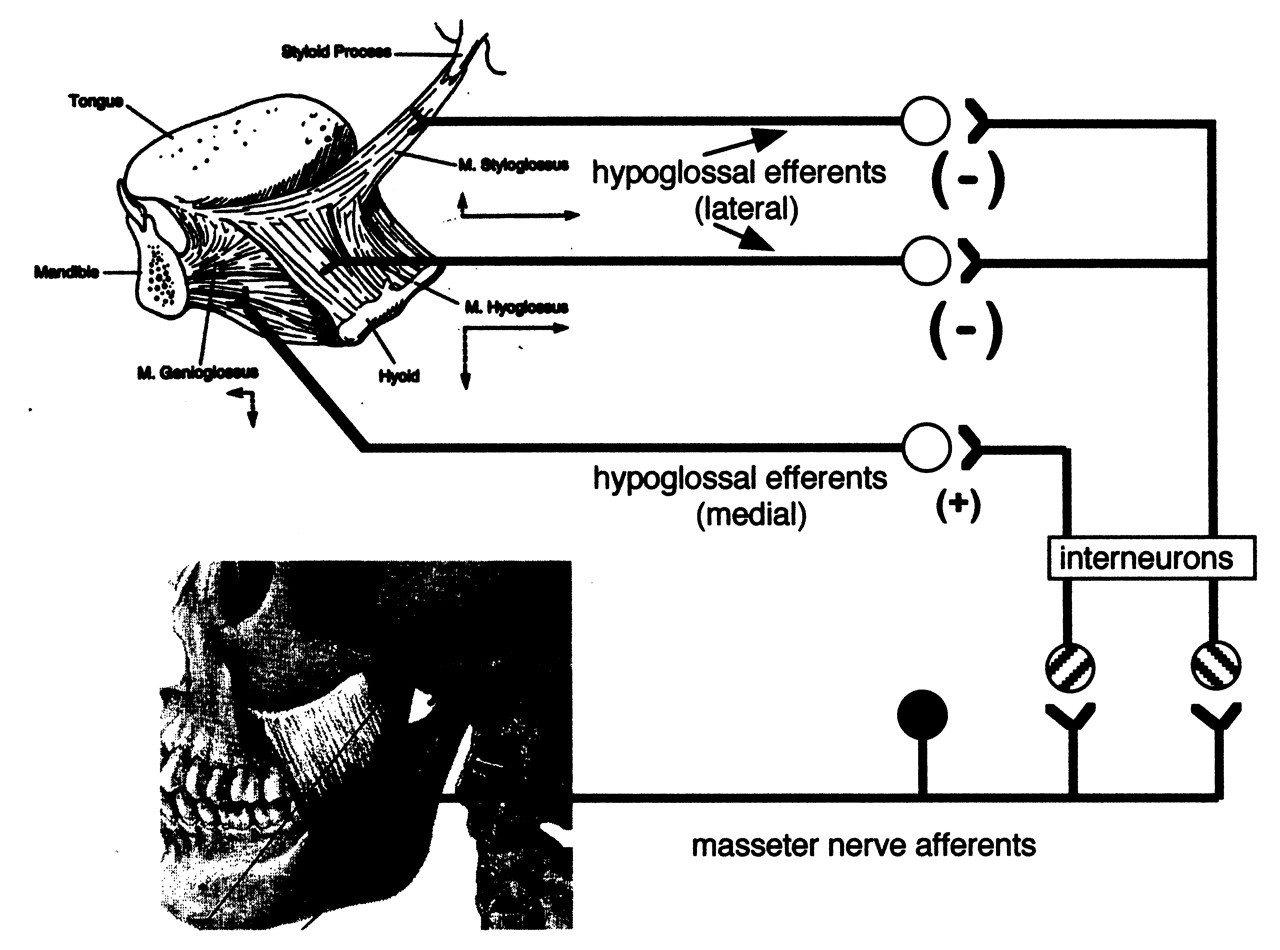
The masseter-hypoglossal reflex. Electrical stimulation of the masseter nerve induces polysynaptic reflexes in hypoglossal motoneurons. The primary effect is inhibitory on most hypoglossal motoneurons, with some excitatory inputs on motoneurons innervating the genioglossus and intrinsic muscles of the tongue.
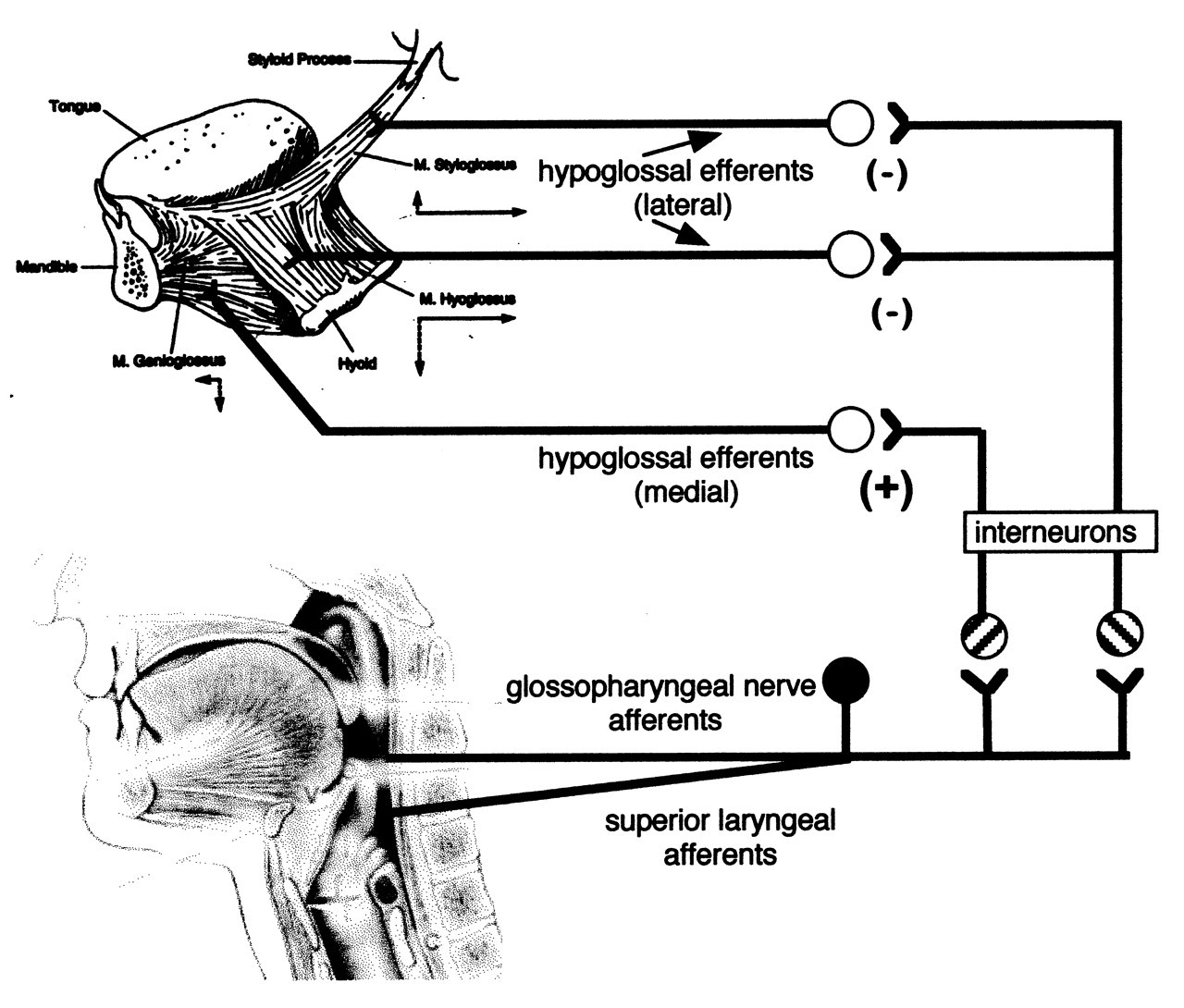
The glossopharyngeal-hypoglossal reflex. Electrical stimulation of the glossopharyngeal nerve innervating the posterior tongue and pharynx induces polysynaptic reflexes in hypoglossal motoneurons. A similar effect occurs with stimulation of the superior laryngeal nerve that innervates the hypopharynx and larynx. The primary effect is excitation of motoneurons innervating the genioglossus and tongue intrinsic muscles.
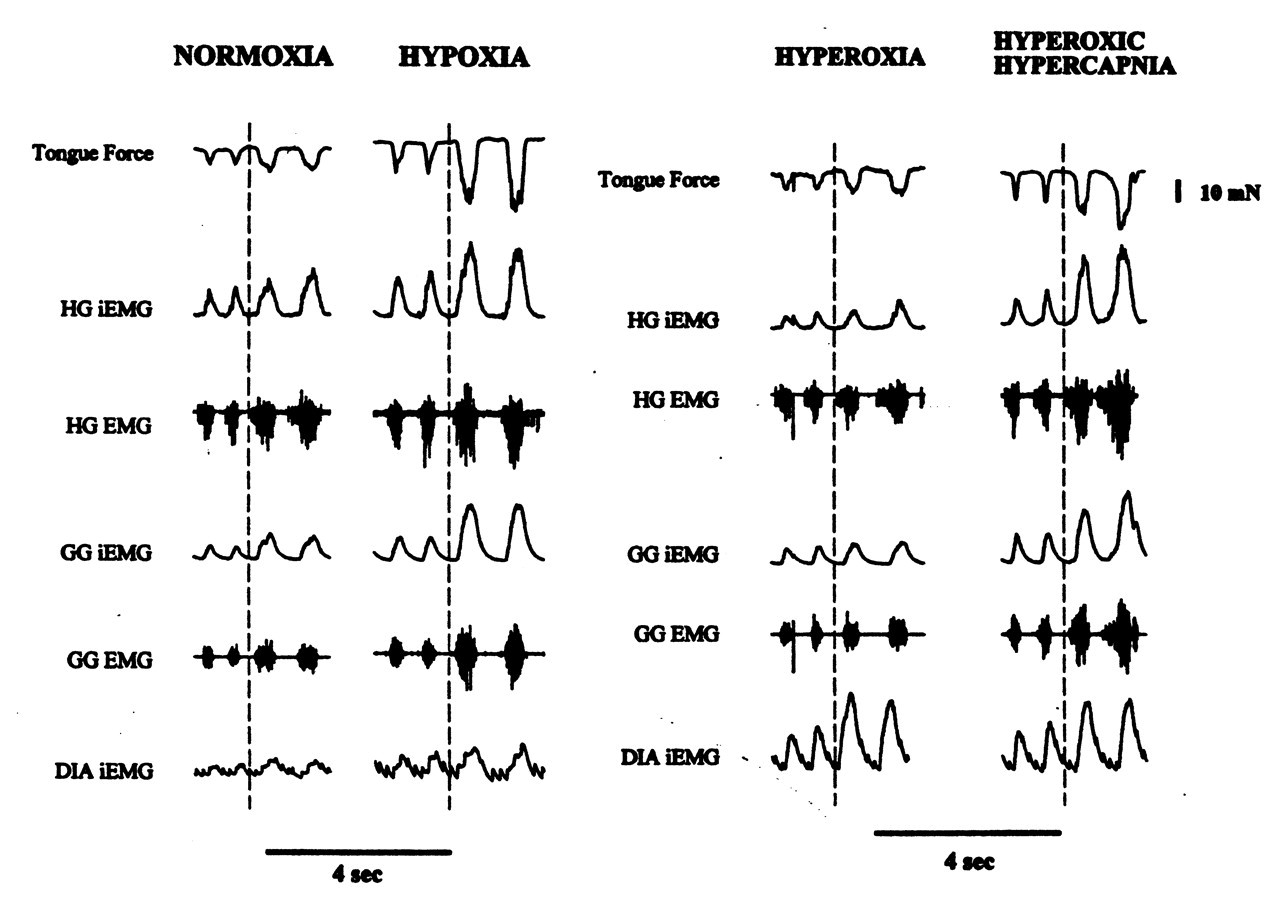
Effect of altering the mixture of O2 and CO2 on the inspired gas levels of an anesthetized rat and on the EMG activity recorded from specific tongue muscles and the diaphragm. Tongue force (upper first trace) is measured with a transducer, and retraction is denoted by a downward direction. EMG activity is shown (lower trace) as both the original signal (HG EMG, GG EMG) and the integrated level (HG iEMG, GG iEMG). The rat breathing room air (NORMOXIA) demonstrates rhythmic EMG activity from the hyoglossus retruding muscle (HG), the genioglossus protruding muscle (GG), and the primary respiratory muscle, the diaphragm (DIA iEMG). Modifying the inspired gas mixture to 10% O2 in 90% N2 (HYPOXIA) induces hypoxia and increases rhythmic EMG activity in both tongue muscles. Tracheal occlusion is induced (vertical dotted line) to prevent further respiration. Breathing only O2 (HYPEROXIA) to induce hyperoxia has minimal effect on the tongue muscles even after tracheal occlusion. Breathing O2 enriched with CO2 (HYPEROXIC HYPERCAPNIA) increases the EMG activity in both tongue muscles and induces more tongue retraction. Tracheal occlusion enhances this hypercapnic response. (Adapted with permission from Fregosi and Fuller [1997])
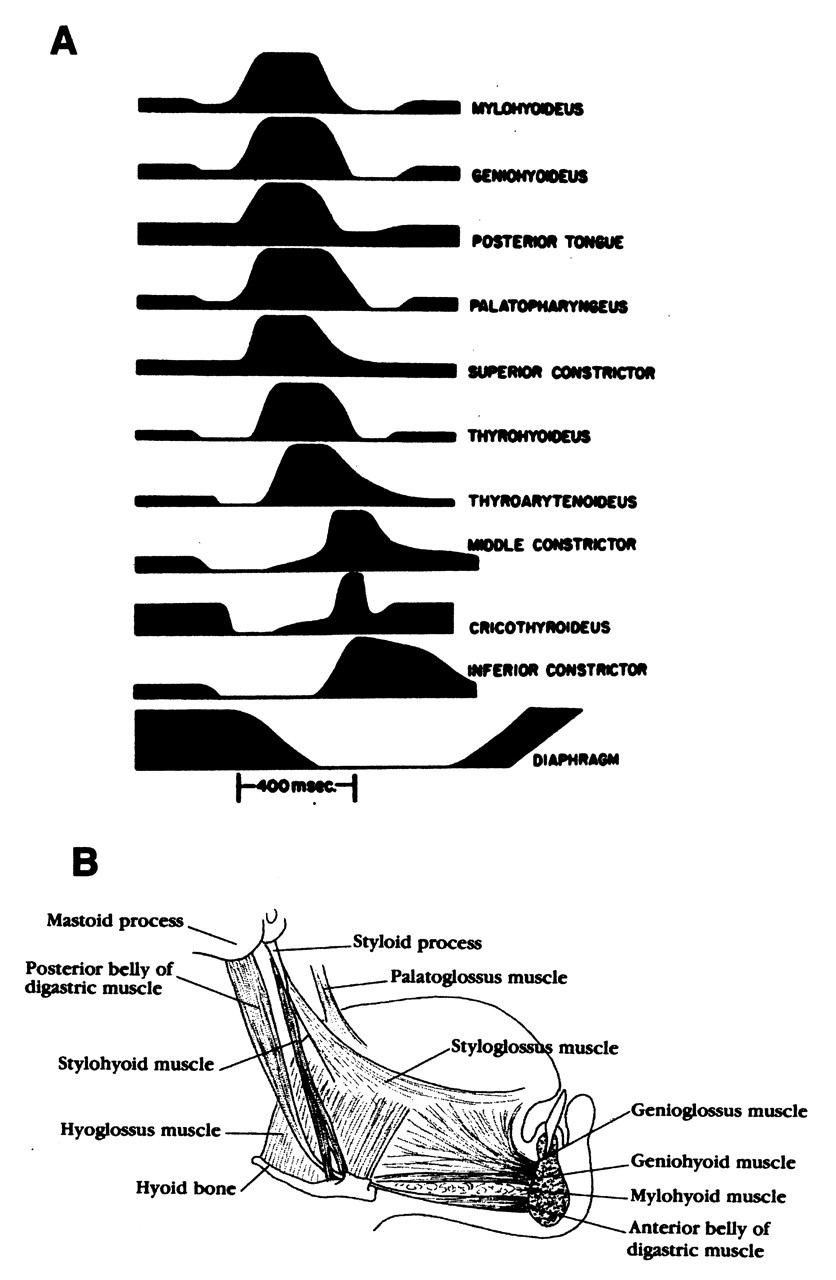
The EMG activity of muscles involved in one pharyngeal swallow as recorded from the anesthetized animal (e.g., cat, dog, monkey). (A) The level of EMG activity can decrease below its background level, suggesting inhibition of that muscle, and then recruit with much activity followed by further inhibition. The increased EMG activity precedes the contraction of the muscle. The diaphragm ceases all activity during a pharyngeal swallow (adapted with permission from Doty and Bosma JF [1956]). (B) Location of some of the muscles involved in the earliest onset of the pharyngeal swallow (adapted with permission from Dickson and Dickson [1982]).
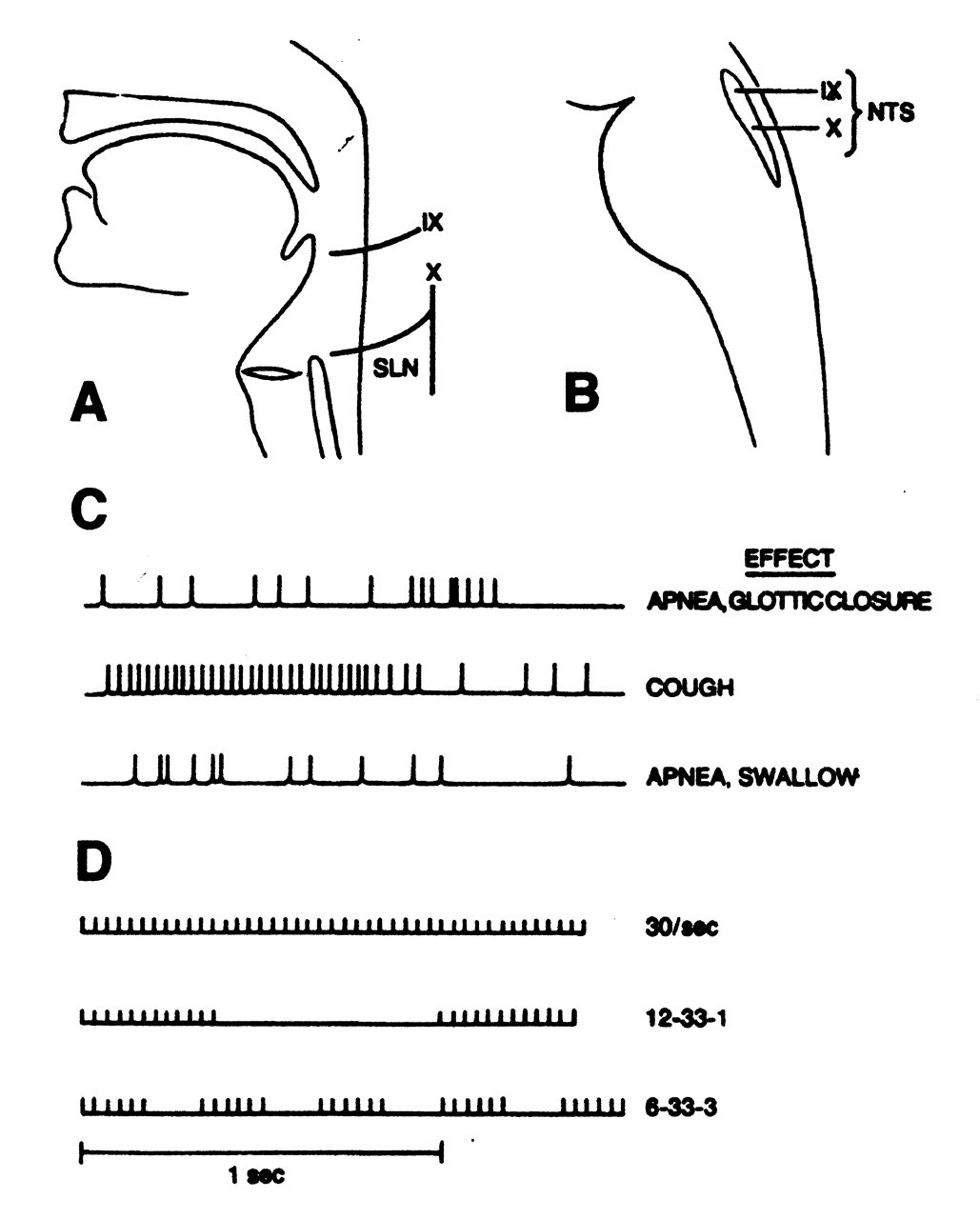
Example of discharges recorded from fibers of the two primary sensory nerves that innervate the pharynx and larynx. (A) The distribution of sensory afferent fibers for the glossopharyngeal nerve (IX) and the superior laryngeal nerve (SLN), which is a branch of the vagal nerve (X). (B) The sensory fibers for both the glossopharyngeal and superior laryngeal nerves synapse in the nucleus tractus solitarius (NTS). (C) Different discharge patterns of sensory fibers from the superior laryngeal nerve recorded from laboratory animals during the inducement of particular reflexes (e.g., apnea, cough) (adapted with permission from Storey [1968b]). (D) Different patterns of electrical stimulation applied to the superior laryngeal nerve of laboratory animals to induce pharyngeal swallowing (adapted with permission from Doty [1951]).
