Abstract
Recent associations between oral health and systemic disease have led to renewed interest in the mouth and its contribution to health outcomes. Many pathways for this relationship have been postulated, among them the potential mediating role of nutrition. The link between various nutrients and systemic disease has been established, but relatively little work has been done in relating oral conditions with nutrition. We searched MEDLINE, from 1966 to July, 2001, to identify articles relating specific oral measures to nutrition outcomes. We included original articles written in English with a sample size greater than 30 that used objective oral health measures. We reviewed a total of 56 articles. Only a small proportion of these studies were methodologically sound. Although many studies were small and cross-sectional, the literature suggests that tooth loss affects dietary quality and nutrient intake in a manner that may increase the risk for several systemic diseases. The impact of tooth loss on diet may be only partially compensated for by prostheses. To date, there is little information relating periodontal disease and oral pain and nutrition. A few studies suggest poorer nutrition among individuals with xerostomia and altered taste. Further, impaired dentition may contribute to weight change, depending on age and other population characteristics. There is a paucity of well-designed studies addressing oral health and nutrition. Before we can acquire a better understanding of how nutrition and oral health interrelate, however, more studies will be required to confirm these associations—preferably longitudinal studies with larger sample sizes and better control of important confounders.
(I) Introduction
The scientific community has become increasingly interested in the possible relationship between oral conditions and systemic outcomes such as coronary heart disease, stroke, and mortality (Joshipura et al., 2000). Commonly postulated mediators of this relationship include infection, chronic inflammation, and genetic predisposition to both oral and systemic disease. Nutrition has been postulated as an alternative mediator (Joshipura et al., 1996, 1998). The impact of various oral conditions on nutritional status may relate importantly to nutrient intake and nutritional status. Several studies have established associations among nutrient intake, nutritional status, and various systemic diseases (Willett, 1994). Recent studies have clearly demonstrated the inverse association between fruit and vegetable intake and the development of cardiovascular disease (Cox et al., 2000; Joshipura et al., 2001). Increased fruit and vegetable intake has also been associated with a lower stroke risk (Joshipura et al., 1999). Multiple studies have also suggested the protective role of fruits and vegetables in cancer (Willett, 1994; Terry et al., 2001); however, studies showing no association with specific cancers have also been reported (Michels et al., 2000). Saturated fat intake has been implicated in cardiovascular disease and may play a role in increasing risk for breast and colo-rectal cancer (Lee and Lin, 2000; Zhang et al., 2000; Smith-Warner et al., 2001). Dietary anti-oxidants or fiber has been shown, in epidemiologic and intervention trials, to reduce cardiovascular disease risk; the results, however, have not been as clear-cut as those obtained for fruits and vegetables (Eastwood, 1999). With both fruits and vegetables and individual vitamins and minerals, reduction in disease outcomes occurs through several mechanisms, including protection against free radical damage, modulation of cytokine production, enhancement of endothelial function, and alteration of coagulation parameters (Grimble, 1998; Nappo et al., 1999; Nuttall et al., 1999).
This potential mediating role of nutrition in the oral-health/systemic-disease relationship has triggered our interest in reviewing the literature relating common oral conditions to nutritional outcomes. This paper will review, summarize, and evaluate original scientific articles linking specific oral health problems (tooth loss, prosthesis, periodontal disease, oral pain, xerostomia, altered taste) and their impact on nutritional outcomes (dietary/nutrient intake, body weight, body composition, blood nutrients). We will describe commonly used nutrition assessment measures used in the studies and address questions regarding how specific oral health characteristics relate to these nutrition outcomes. We will summarize the methodology, findings, and limitations of selected articles. Finally, we will provide suggestions for future research design that might provide better data and lead to better-understood conclusions.
(II) Methods
Oral health and nutrition are both defined in multiple ways and described by complex, interrelated measurements. Oral health encompasses gingival status, the well-being of the teeth and jaw, salivary quantity and quality, and sensory dimensions of taste and pain. Likewise, nutritional health includes both the quality and quantity of dietary intake and nutritional status. Dietary intake is comprised of the types of food groups ingested (such as fruits, vegetables, or dairy products) and the nutrient composition of the food eaten (micronutrients-vitamins, minerals, and macronutrients-proportion of calories consumed as protein, carbohydrates, and fats). Nutritional status includes body composition and tissue and blood levels of micronutrients. To highlight these various components of oral health and nutrition, we developed a conceptual model that describes how oral health and nutrition characteristics might relate to each other (Fig.). Characteristics noted in the shaded boxes identify patient-specific characteristics that may affect nutrition indirectly through their impact on oral health. Those characteristics, such as smoking and health behavior, may also affect nutritional status directly.
Both periodontal disease and dental caries can lead to tooth loss. Tooth loss can then result in chewing difficulty because of inadequate occlusive surfaces or the limitations of prostheses. Chewing difficulty may then lead to alteration in food selection and dietary quality, which in turn can affect both body composition and nutritional status. Oral pain can also occur as a result of caries, periodontal disease, soft-tissue lesions, and temporomandibular joint disease. Poorly fitting prostheses may also cause stomatitis and oral pain. Oral pain may lead to changes in diet and subsequent nutritional status. Xerostomia and decreased or altered taste could also, theoretically, contribute to changes in nutrient intake and nutritional status.
However, these oral health-nutrition relationships are complicated by the fact that health habits (such as smoking), concurrent illnesses, and attitudes and beliefs about health and health behavior can affect oral health, as well as nutrition. Thus, oral health may appear to be related to nutrition even if it has no impact on nutrition. Hence, it is difficult to distinguish whether the oral characteristics or some of these other confounding characteristics (such as health behavior) are responsible for the relationships seen between oral health and nutrition.
Using this model as a basis for describing oral health and nutrition relationships, we utilized the nutritional and oral health terms noted in the Fig. to identify appropriate key terms and MeSH terms in our literature search, using MEDLINE as our primary database. We searched MEDLINE for original articles for the time period spanning from 1966 to 2000. Additional articles were generated from references cited by articles generated from our MEDLINE search. To be included in this review, studies had to have a sample size over 30, contain objective oral health measures (with the exception of oral pain assessment), and be written in English. Articles with chewing complaints or masticatory performance as the only oral health exposure were not included. Clinical trials and observational studies were included. Review articles were excluded.
(III) Results
We identified many studies that address the relationship between some aspect of oral health and nutrition. Unfortunately, many of these studies have small sample sizes and multiple comparison groups, making them underpowered for the adequate assessment of associations. To maintain brevity in the tables, we listed only those studies with sample sizes over 100.
Following the conceptual model noted in the Fig., we review the literature on the possible oral-health/nutrition relationships described.
(A) How does tooth loss affect dietary /nutrient intake ?
Using the oral health MeSH terms “tooth loss, dentition, edentulous jaw or partially edentulous jaw” and nutrition MeSH terms “eating, food habits, dietary proteins, dietary fats”, and “energy intake”, we evaluated 10 studies that address the relationship between dentition status or tooth number on dietary choices and intake (Table 1).
Hildebrandt et al. (1997) compared dietary behaviors with the number of functional units of teeth (any opposing natural or prosthetic tooth pair). The authors reported that decreased numbers of functional units are associated with the avoidance of stringy (beef/steak), crunchy (carrots), and dry solid food (bread). However, he did not adjust for other known confounders such as education or economic status, nor did he address the presence of xerostomia, which also may contribute to food avoidance.
Several studies suggest that edentulous individuals are particularly prone to inappropriate dietary intake (for example, ingesting too few nutrient-dense foods and too much calorie-rich, high fat foods) compared with dentate subjects. In a study of older adults in Sweden, Johansson et al. (1994) found that edentulous men consume fewer fruits and vegetables and have a lower intake of fiber than do dentate men. Edentulous women consumed a higher amount of fat than did dentate women. Both edentulous men and women ate more sweet snacks than their dentate counterparts, after adjustment for age and education. Norlen et al. (1993) confirmed, in his study of older women, an association between edentulousness and ingestion of a larger intake of fat. In neither study, however, was the type of fat differentiated (saturated vs. unsaturated).
In three large studies in the United States and Europe, decreasing numbers of teeth were also associated with worse dietary quality. Joshipura et al. (1996) analyzed the association between disease-related nutrients and foods and numbers of teeth. After adjusting for age, smoking, exercise, and profession, they observed that edentulous participants consumed fewer vegetables, less fiber and carotene, and more cholesterol, saturated fat, and calories than participants with 25 or more teeth. In this homogenous population of male health professionals, gender and socio-economic and behavioral factors were inherently controlled.
Krall et al. (1998) found that subjects with intact dentition or partially compromised dentition have significantly better masticatory function than subjects with removable partial dentures, compromised dentition, or full dentures. In their analyses, the calorie-adjusted nutrient intakes decreased with progressively impaired dentition status after adjustment for age, smoking status, and alcohol use. They also found that better masticatory function is associated with higher intakes of fiber, vitamin B6, folic acid, vitamins A, C, and D, carotene, thiamin, riboflavin, magnesium, phosphorus, and iron. This study, however, did not control for socio-economic status.
In a cross-sectional study of British older adults, subjects with more teeth had higher mean intakes of energy, protein, fat, carbohydrate, fiber, calcium, iron, pantothenic acid, and vitamins C and E (Sheiham et al., 2001). This was the one study that reported a higher intake of fat among individuals with more teeth. The type of fat, however, was not characterized. Smaller cross-sectional studies showed no differences in nutrient intake by dentition status (Baxter, 1984; Gordon et al., 1985). These studies had multiple comparison groups and were probably underpowered for the detection of a difference, if it existed.
Several of the larger studies noted above suggest that dentition status is associated with dietary intake. However, most of these studies fail to show whether these associations are independent of common factors (confounders) or total calorie intake. Tooth loss has been found to be associated with factors such as limited financial resources, low education status, and history of smoking that generally reflect negative health behaviors and attitudes (Kay and Blinkhorn, 1987; Burt et al., 1990; Worthington et al., 1999). Subjects concerned with their health might be more conscious about both oral health and eating behaviors, and that might explain the differences in dietary patterns among those with different dentition status. Without adjusting for these confounding effects, we cannot evaluate the independent association between oral health and dietary intakes. Adjusting nutrient intake for total caloric intake is also important because people with higher intake of calories may be expected to have higher intake of most foods and nutrients. It is difficult to interpret differences in nutrient intake when total caloric intake is not controlled (Willett, 1998). Controlling for caloric intake additionally helps reduce extraneous variation in nutrients.
In summary, most of the studies relating tooth loss and nutrition suggest that nutrient intake deteriorates in quality with fewer teeth. These changes seen in fruit, vegetable, and micronutrient intake with tooth loss may explain part of the tooth-loss/cardiovascular-disease association.
Many of the studies we identified in the literature have various approaches to the assessment of nutrition, making comparisons across studies difficult. Commonly used approaches to eliciting information on food intake in the studies reviewed included the 24-hour recall, food records, and food frequency questionnaires. The 24-hour recall elicits all the foods and beverages the subject has consumed in the preceding 24 hours. Although relatively straightforward to perform, it may not provide enough information to generalize about the individual's usual intake. In addition, it tends to underestimate the person's usual fat and calorie intake. This method is hindered by the subject's memory of what has been consumed in the day before (Gibson, 1990). The food record or diary method consists of a detailed listing of all foods consumed by a subject on one or more days. Food intake is recorded by the subject at the time the food is eaten, to minimize recall error. Portion sizes can be accurately measured; however, the process of keeping a food diary may alter the subject's behavior and not reflect usual intake. With both the 24-hour recall and the food record, multiple dates of intake are needed for usual individual intake to be estimated (Buzzard, 1998). The food frequency questionnaire consists of two components: a food list and a frequency response section for individuals to state how often each food was eaten. This approach relies less on immediate recall and more on generic recall (what usually occurs). It provides a good estimate of usual intake, but provides less detail about cooking methods. It may also not capture foods eaten by ethnic groups that are not mentioned on the questionnaire. A few studies used biochemical indicators of dietary intake. Biochemical measures are objective and thus may appear less error-prone than other nutrition assessment methods. Misclassification may still occur due to inappropriate specimen collection, storage, or analysis. In addition, biochemical measures may not completely reflect intake or tissue levels, depending on the measure's bioavailability and the subject's kinetics with respect to the particular nutrient (Hunter, 1998).
A weakness of the studies noted above and many others reviewed is that many of these studies are cross-sectional, making the direction of the oral-health/nutrition relationship difficult to ascertain, and determination regarding causality impossible. The interrelation between dental status and dietary intake is complicated. In addition to dentition status causing a change in dietary pattern, several nutrients—such as vitamins A, B6, C, and D, calcium, niacin, and thiamin—may affect oral health (Midda and König, 1994). The association between dentition status and nutrient intake observed in cross-sectional studies might be partially explained by the potential effect of dietary intake on oral health. Therefore, it is difficult to evaluate the temporal roles of dentition status and dietary intakes in cross-sectional studies.
An exception was an analysis by Joshipura et al. (1996) of the longitudinal effect of tooth loss over a four-year period on dietary change adjusted for age, smoking, exercise, and professions. Baseline intake suggested that loss of teeth might lead to detrimental changes in diet. However, there was insufficient tooth loss among the health professionals' population for significant difference to be detected (Joshipura et al., 1996). More longitudinal studies are needed to support and clarify the relationship between oral health and dietary intake, before this association can be implicated in the oral-health/systemic-disease relationship.
(B) How do prostheses affect dietary /nutrient intake ?
Using the oral health MeSH terms, “denture, partial denture, dental implant” or “dental prostheses”, and the nutrition MeSH terms, “eating, food habits, dietary proteins, dietary fats”, and “energy intake”, we evaluated 21 studies that address the relationship between dentition status or tooth number on dietary choices and intake (Table 2).
Several investigators have compared nutrient intakes among groups with prostheses. The nutrient assessment varied from crude food groups to specific nutrients derived with the use of nutrient databases and foods selected based on textures. Studies were cross-sectional or compared two groups of people before and after prosthesis, with different prostheses assigned randomly or non-randomly. Overall, prosthesis and type of prosthesis influenced patient satisfaction and chewing efficiency, and variably affected nutrient intake.
A group of studies compared subjects having full dentures with dentate subjects. In Krall et al.'s (1998) study of veterans, individuals with full dentures consumed fewer calories, thiamin, iron, folate, vitamin A, and carotene than did individuals with other dentitions. Papas et al. (1998a) evaluated the impact of full dentures and noted lower intake of protein and 19 other nutrients. In a separate population, Papas et al. (1998b) reported that subjects who wore dentures consumed more refined carbohydrates, sugar, and dietary cholesterol than their dentate counterparts. The above studies suggest that the presence of dentures contributes to poorer intake across multiple nutrients compared with dentate subjects. Several studies did not reveal as significant a difference in nutrient content between denture wearers and those who are dentate. Keller et al. (1997) reported decreased calorie intake among dentate men and women, but in men, poorer dietary quality among those with their own teeth. In Appollonio et al.'s (1997) study of older Italian subjects, denture wearers had the same nutrient intake as those who were dentate, with the exception of lower vitamin A intake. Greksa et al. (1995) noted lower intake of vitamins A and C and calcium, but otherwise no other differences between groups in dietary diversity. Hartsook (1974) study showed little difference in diets between patients wearing various combinations of dental prostheses; however, the study was small, and dietary intake was highly variable between the different dental groups. All of these studies are cross-sectional, with only a few controlled adequately for confounding.
Many of these studies did not address denture fit among denture wearers, which might explain some of the variability in findings regarding nutrient intake. In a study of denture wearers in Quebec, those who wore dentures that provided poor masticatory performance consumed significantly fewer fruits and vegetables than those with dentures that provided good masticatory performance (Laurin et al., 1994). Likewise, in Swedish older adults, poorly fitting upper dentures were associated with decreased intake of vitamin C (Nordstrom, 1990). In a survey of older Australians, women who reported poorly fitting dentures consumed greater amounts of sweets and dessert items (Horwath, 1990). Future longitudinal studies of the impact of prostheses on diet will need to be performed that address both prosthesis fit and confounding.
In comparisons between edentulous subjects without dentures and edentulous subjects with dentures, edentulous subjects without dentures, not surprisingly, consumed more mashed food (Lamy et al., 1999), and in a study of Swedish women, edentulous women consumed more fat (Norlen et al., 1993). Fontijn-Tekamp et al.(1996) reported that edentulous subjects without any prostheses had significantly lower carbohydrate and vitamin B6 intake. None of these studies adjusted for factors other than gender and region in the analysis.
Most of the studies of the impact of new dentures on dietary intake are small and with no control group. Garrett et al. (1997) conducted a randomized trial of no dentures, fixed partial dentures (FPD), and removable partial dentures (RPD) among partially edentulous subjects; diet was recorded by seven-day diaries. He evaluated calories and 30 nutrients and found no important differences, other than regression to the mean, among those with extreme intake. Since there was no control group, it was not clear whether the regression to the mean was beyond what might be expected in the control group. Sebring et al. (1995) studied the effect of conventional maxillary + implant-supported or conventional mandibular dentures. He evaluated both caloric intake and 27 nutrients, using three-day diet records and adjusting for body weight. In both groups, caloric intake decreased; percentage of calories from fat also decreased significantly over the subsequent three years. Lindquist (1987) evaluated the impact of prosthetic rehabilitation, using optimized complete dentures and then tissue-integrated mandibular fixed prostheses (TIP) on 64 dissatisfied complete-denture wearers. There was no change in diet after complete dentures were optimized, but there was a persistent increase in fresh fruit consumption after placement of TIP. Hamada et al. (2001) performed a similar study among diabetic patients and again showed no real change in diet except for a slight decrease in mean caloric intake. In contrast to the above studies, where there was a slight improvement in overall dietary quality, in a study by Moynihan et al. (2000), patients receiving either resin-bonded bridges or conventional removable partial dentures demonstrated a significant increase in the proportion of energy obtained from fat, 12 months after treatment in the denture group. There was no significant difference in fruit and vegetable intake between baseline and one year post-intervention in either group.
Olivier et al. (1995) conducted the only study that evaluated dietary counseling in addition to prosthetic relining for the edentulous on chewing efficiency, dietary fiber intake from various sources, and gastrointestinal esophageal and colonic symptoms. The authors used food frequency questionnaires and made pre- and post-intervention comparison using paired t test and McNemar's test, thus controlling for subject level factors and increasing efficiency. Chewing ability and fiber intake from fruits and vegetables were significantly improved. However, because there was no group that did not receive dietary counseling, it was not possible to separate the effect of relining from that of the counseling.
Sebring et al. (1995) adjusted all nutrients and total calories for body weight; however, none of the studies adjusted nutrients for total caloric intake. None of the above studies considered controlling for potential confounders, which would be an important concern for between-group comparisons, where the prostheses were not assigned randomly. Confounding is not an important concern in randomized clinical trials (RCT) (Garrett et al., 1997) where the prostheses is assigned randomly or in analyses that compare pre- and post-prosthesis (Lindquist, 1987). However, in the studies discussed above, the analyses were often only descriptive or simple between-group analyses. Other than the study by Olivier et al. (1995), studies that compared pre- and post-intervention did not use methods for paired data. Baseline differences in intake of the nutrient under consideration, energy intake, or other potential confounders were not factored into the analyses. Future intervention studies would benefit from larger comparison groups, random assignment, paired data analysis, and consistent adjustment of nutrients for total calories. Given the behavioral aspects of food intake, more studies evaluating the impact of concurrent dietary education concurrent with dental intervention would be informative.
(C) How does tooth loss or prostheses affect nutritional status ?
Using the MeSH terms, “tooth loss, dentition, dentition status, edentulous jaw or partially edentulous jaw, denture, partial denture, dental implant or dental prostheses”, and the nutrition MeSH terms, “nutritional status, obesity, body weight, body composition or body mass index, weight change, weight loss”, and “vitamin levels”, we evaluated 13 articles that address the relationship among tooth loss, prostheses, and nutritional status (Table 3).
Most studies addressing nutritional status do not completely measure every aspect of nutritional status. Indeed, in the nutrition literature, much controversy exists regarding comprehensive measures of nutrition. Most nutrition indices do not fully measure nutrition and often incorporate non-nutritional measures of co-morbidity, physical function, and medication use (Lyne and Prowse, 1999). Given the lack of a well-accepted comprehensive measure of nutritional status, investigators are constrained to look at several separate clinical indicators of nutrition, including body weight and body mass index (an indirect measure of protein, fat, and carbohydrate stores), vitamin levels, indirect measures of vitamin status (hemoglobin) or protein status (visceral protein stores such as albumin), and indicators of energy imbalance (weight change).
(1) Body mass index
Six studies address weight, weight/height, and body mass index (BMI) in relation to dental status. Two studies (Gordon et al., 1985; Dormenval et al., 1995) reported an association between dental status and body composition, but these studies assessed bivariate relationships and did not control for co-morbidity, age, and sociodemographic status. In Mojon et al.'s (1999) study of nursing home residents, compromised oral functional status was associated with a BMI of less than 21 kg/m2, after adjustment for functional dependence and age. In Hirano et al.'s (1999) study of community-dwelling older adults, the authors reported a similar association between masticatory ability and body weight, after controlling for age and sex. In Johansson et al.'s (1994) cross-sectional study of older adults in Sweden, edentulous patients actually had higher BMIs, compared with dentate subjects; and in Elwood and Bates' (1972) study of older Welsh adults, there was also a trend toward higher weight among subjects with no teeth, compared with subjects with dentures, but the differences did not reach statistical significance. Some of the differences in findings in these studies may be due to the different characteristics of the populations evaluated. Mojon et al. (1999) studied nursing home residents, Dormenval et al. (1995) evaluated hospitalized patients, Johansson et al. (1994) surveyed community-dwelling older adults. It is hard to know whether oral conditions precede or follow weight change.
(2) Weight
We evaluated three articles that address the relationship between dentition status and number of teeth on nutritional status. The first study to report an association between oral health and weight loss was that by Sullivan et al. (1993). Compromised oral health was broadly defined and included multiple oral health domains. Blaum et al. (1995) evaluated risk factors for weight loss in nursing homes. The authors noted an association between chewing difficulty and subsequent weight loss, but did not demonstrate an association between specific oral health problems and weight loss. However, oral health problems were based on a standardized nursing assessment form, which might not have been reliable for oral assessment. Ritchie et al. (2000) reported on oral health risk factors associated with significant weight loss among community-dwelling older adults in the New England area. After adjustment for gender, income, age, and baseline weight, edentulousness remained an independent risk factor for significant weight loss over a one-year period.
(3) Blood nutrient levels
Several studies, in addition to evaluating nutrient intake, obtained blood for nutrient or lipid analysis. The largest study to date to evaluate blood nutrient status in relation to dental status is the British National Diet and Nutrition Survey (Sheiham et al., 2001). In their cross-sectional study of 490 free-living and institutionalized older adults, the authors reported that edentate subjects had significantly lower mean plasma levels of retinol, ascorbate, and tocopherol than dentate subjects, after adjustment for age, sex, social class, and region of residence. Among dentate subjects, mean plasma vitamin C levels were positively associated with increased numbers of occlusal pairs of teeth. Longitudinal studies supporting these relationships between vitamin status and dental status are needed to strengthen this potentially very important association. Another study of adults in Sweden (Johansson et al., 1994) reported lower serum high-density lipoprotein (HDL) levels among edentulous individuals compared with levels in those who were dentate. It is unclear in this study, however, whether other common risk factors for oral health and lipid status (such as health behavior) were adequately controlled for. In a study of older Welsh adults (Elwood and Bates, 1972), hemoglobin, vitamin B12, and folate levels were evaluated. Only hemoglobin was noted to be lower in the women with no teeth or dentures.
Some investigators have also looked at composite nutritional indices that include both risk factors for poor nutrition (such as multiple medications or depression), anthropometric measures (such as body mass index), and consumption of specific food groups (such as fruits and vegetables). The Mini Nutritional Assessment (MNA) is one such index. The MNA score is based on nutritional status, weight change, and risk factors for nutritional inadequacy (such as appetite, co-morbidity, dementia, and depression). It has been validated in several different older adult populations and correlates well with physician-nutritionists' assessments of overall nutritional status (Vellas et al., 1999).
Lamy et al. (1999) and Griep et al. (2000) used the MNA score to measure nutritional status. Lamy et al. (1999) evaluated nursing home residents and found that edentulous subjects without or with only one denture had significantly lower MNA scores, reflecting poorer nutritional status. Griep et al. (2000) noted a relationship between number of teeth and MNA score, in the univariate analysis, but not after adjustment for co-morbidity, medication, and gender. Further combined nutrition scores are hard to interpret and strongly influenced by the patient's severity of illness. All of these studies were cross-sectional, so temporality could not be established.
(D) How does periodontal status /dental caries affect dietary /nutrient intake or nutritional status ?
Using the MeSH terms “periodontal disease, gingivitis, dental caries” and the nutrition terms “eating, food habits, dietary proteins, dietary fats”, and “energy intake, nutritional status, obesity, body weight, body composition”, and “body mass index (BMI)”, we were not able to identify any articles for dental caries, and we were able to identify only three articles that address the potential effect of periodontal disease on nutrition outcomes.
(1) How does periodontal status affect dietary/nutrient intake?
Osborn et al. (1977) performed a cross-sectional study of dietary intake among patients with periodontal disease. The authors found no difference in nutrient intake compared with that of the general population.
(2) How does periodontal status/dental affect nutritional status?
In a cross-sectional study, 241 healthy dentate Japanese adults were examined, and their periodontal status was estimated according to the Community Periodontal Index of Treatment Needs (CPITN) (Saito et al., 1998). The authors reported an increased prevalence of periodontitis in larger BMI groups, after adjusting for age, gender, oral-hygiene status, and smoking history. When fitness (VO2 max) was added to the logistic regression model, periodontitis was no longer significantly associated with larger BMI, suggesting that fitness or lean body mass may be more important in the association between periodontal disease and nutritional status than BMI per se (Saito et al., 1999). To date, no longitudinal studies have evaluated the relationship between periodontal disease and BMI. Given the possible role of body fat in host inflammatory response (Mohamed-Ali et al., 1998), longitudinal studies assessing periodontal status and fat and fat-free mass are warranted if we are to attain a better understanding of the direction of these relationships.
(E) How does xerostomia or oral pain affect dietary /nutrient intake or nutritional status ?
Using the MeSH terms “xerostomia, Sjögren's, facial pain, toothache, mouth diseases”, and “pain”, and the nutrition MeSH terms “eating, food habits, dietary proteins, dietary fats”, and “energy intake, nutritional status, obesity, body weight, body composition”, “body mass index (BMI)”, and “nutrient levels”, we evaluated 7 articles that address the relationship among xerostomia, oral pain, and either dietary/nutrient intake or nutritional status.
(1) How does xerostomia or oral pain affect dietary/nutrient intake?
Remarkably few studies have evaluated the relationship between oral pain and nutritional outcomes except in the case of head and neck cancer, where usually the oropharynx is bypassed and gastrostomy tubes are placed to ensure that the patient receives nutrition (Newman et al., 1998). Studies of other patients with chronic oral pain have not focused on nutritional outcomes. The only study of temporomandibular disorder pain dysfunction syndrome (TDPDS) is limited in size and scope, and suggests that dietary choices are negatively affected by TDPDS symptoms (Irving et al., 1999). In a broader study of orafacial discomfort, no relationship was identified between multiple orofacial complaints and dietary intake (Yontchev et al., 1989).
Three studies of xerostomia have found that diet/nutrition and the quality of saliva were affected by exposure to Sjögren's syndrome and xerogenic medications. Loesche et al. (1995) reported that individuals with complaints of xerostomia were more likely to avoid crunchy vegetables (carrots), dry foods (bread), and sticky foods (peanut butter). Rhodus (1988) studied 28 patients with Sjögren's syndrome and compared them with a group of controls matched for diabetes, depression, cardiovascular disease, arthritis, age, gender, and dental health. Caloric and micronutrient intakes were significantly lower among xerostomic patients. Rhodus and Brown (1990) also evaluated 84 older residents of an extended care facility. Energy, protein, fiber, vitamins A, C, and B6, thiamin, riboflavin, calcium, and iron were significantly lower in the patients with xerostomia than in those without. These studies suggest that xerostomia impairs optimal nutrient intake but are again hampered by their small size, cross-sectional design, and inadequate control for confounding factors.
(2) How does xerostomia or oral pain affect nutritional status?
The only study we were able to identify that addressed the impact of chronic oral pain on nutritional status was our study of New England elders, where self-reported oral pain was not associated with subsequent one-year weight loss (Ritchie et al., 2000).
Rhodus (1988) noted that the body mass index for the xerostomic individuals with Sjögren's syndrome was significantly lower than that for the control group. In their study of older extended-care-facility residents, they also noted a significantly lower body mass index among the xerostomic subjects (Rhodus and Brown, 1990). Dormenval et al. (1995) evaluated hospitalized older adults and noted that low unstimulated salivary flow rates were associated with low body mass index, triceps skin-fold thickness, and arm circumference. In the arena of both xerostomia and oral pain, much more research is needed before we will understand the extent to which these oral conditions affect nutrient intake and nutritional status.
(F) How does altered taste affect dietary /nutrient intake or nutritional status ?
Using the MeSH terms “taste and taste disorders” and the nutrition MeSH terms “eating, food habits, dietary proteins, dietary fats”, and “energy intake, nutritional status, obesity, body weight, body composition” and “body mass index (BMI)”, and “nutrient levels”, we evaluated 7 articles that address the relationship between altered taste and either dietary/nutrient intake or nutritional status.
(1) How does altered taste affect dietary/nutrient intake?
Very few studies have adequately evaluated changes in nutrient intake or nutritional status among patients with decreased or altered taste. The largest study is that by Mattes et al. (1990), who evaluated 118 patients with taste distortion and taste loss. No real differences were noted in nutrient intakes between groups. Patients with taste distortion were more likely to report altering their intake (either increase or decrease) than control subjects or patients with taste loss. Mattes-Kulig and Henkin (1985) studied 65 patients with dysgeusia. In their study, nutrient intake decreased progressively as dysgeusia severity increased, especially for vitamins A and C and calcium. Both of these studies were cross-sectional and were limited by their relatively small sample size. Mathey et al. (2001) evaluated the impact of flavor-enhancers in a nursing home population. Energy intake declined in the control group but remained stable in the intervention group. No difference was noted in intake of vitamins and minerals.
(2) How does altered taste affect nutritional status?
In Mathey et al., (2001) study to evaluate the impact of flavor-enhancers in a nursing home population, those in the intervention were more likely to gain weight, and those in the control group were more likely to lose weight during the follow-up period. In the studies by Mattes et al. (1990) and Mattes-Kulig and Henkin (1985), no clear differences in body weight were noted between dysgeusia patients and the controls. Clearly, many opportunities remain to explore the relationship between altered taste and nutrition outcomes.
(IV) Discussion
In summary, although many studies to date have been small and methodologically flawed, recent larger studies suggest the following regarding the questions raised in this paper:
Does tooth loss affect dietary intake? Studies suggest that tooth loss affects dietary quality and nutrient intake in a manner that may increase risk for several systemic diseases. Further, impaired dentition may contribute to weight change, depending on age and other population characteristics. Longitudinal studies in large populations with standardized nutrition and oral assessment measures will be required to confirm these findings. Do prostheses affect dietary intake? The studies to date, because of the way they were conducted, provide unclear results. Dentures appear to improve dietary quality somewhat but do not really compensate for loss of teeth. Future studies evaluating prostheses will need to control more carefully for number of teeth in evaluating two groups with and without dentures but with the same number of teeth. Intervention studies with and without dietary counseling, and with and without new prostheses, will be needed to clarify whether the lack of changes in diet in many studies is due to behavioral issues, or whether the changes reflect the inadequacy of the prostheses to optimize nutrient intake. How does tooth loss or the use of prostheses affect nutritional status? Several studies show an increased risk among individuals with oral health problems for low body mass index and weight loss, but predominantly among vulnerable populations (hospitalized patients or nursing home residents). The only large study of diet and blood nutrient status demonstrated lower vitamin levels among edentate subjects and an inverse relationship between number of occlusal pairs and vitamin C levels. Longitudinal and intervention studies are needed to confirm these observations. How does periodontal status affect nutrient intake or nutritional status? The one study evaluating this relationship showed no relationship between periodontal disease and nutrient intake. The possible relationship between obesity and periodontal disease warrants further epidemiologic evaluation to clarify the direction of the relationship and provide further data regarding strength of the association after adjustment for confounding. How does xerostomia and/or oral pain affect nutrient intake or nutritional status? The answer to this question is hampered by a lack of studies. The studies to date, however, suggest that xerostomia increases risk for both inadequate intake and lower weight. Larger longitudinal studies that control for co-morbidity will be necessary to confirm these findings. The few studies of chronic oral pain provide conflicting findings regarding the impact of oral pain on nutrition. How does altered taste affect dietary intake or nutritional status? Studies suggest an alteration in nutrient intake that varies with the type and severity of dysgeusia. Studies using standardized taste threshold measures and dietary assessment measures may provide greater detail regarding the importance of taste to nutrition.
As stated earlier, health behaviors, co-morbidity, and socio-economic status may confound the nutrition and oral health association. Many of the studies relating nutrition and oral health thus far have been cross-sectional studies with relatively small sample sizes. Nutritional measures were often not standardized, nor were nutrients adjusted for caloric intake.
Before we can achieve a better understanding of how nutrition and oral health interrelate, more research is needed to confirm these associations—studies with larger sample sizes and better control of important confounders. These studies will also require inclusion of comprehensive nutrition and oral assessment data. Many of the dietary intake assessment methods used in the studies described are acceptable. Food frequency questionnaires provide optimal dietary information. For ethnically diverse and older adult populations, dietary recalls may be preferable. Dietary recalls, however, estimate usual intake only when multiple days are assessed, so future studies should avoid one 24-hour recall. With both these assessment measures, large sample sizes are required to minimize error. All nutrients reported should be adjusted for calorie intake. Nutritional status assessment measures should include not only height and weight, but preferably indicators of fat and fat-free mass as well. More longitudinal studies would benefit from biochemical indicators of dietary intake. Only a few studies have assessed the nutritional impact of oral pain, xerostomia, and altered taste on nutrition outcomes. In particular, nutrition outcomes of temporomandibular disorders justify further study. Larger dental intervention trials will shed light on how remediable oral-health-related nutritional problems are.
The pathophysiologic relationship among oral health status, diet, and systemic health outcomes such as CVD and cancer also warrant further exploration. Nutritional factors, especially anti-oxidants that may be affected by tooth loss, may modulate both periodontal disease and systemic disease by interfering with the inflammatory cascade, and preventing carcinogenesis. Likewise, tooth loss may contribute to the intake of calorie-dense, nutrient-poor diets, leading to decreased intake of anti-oxidants and increased intake of foods that foster obesity. Future studies will require careful assessment of nutritional, inflammatory, and cancer biomarkers to better understand these interrelationships.
This review highlights associations between oral health and nutrition that suggest an important potential mediating role in the oral-health/systemic-disease relationship. Ongoing aggressive evaluations of these relationships are needed to improve our understanding of this role and to clarify the exact pathways that relate oral health to cardiovascular and other health outcomes.
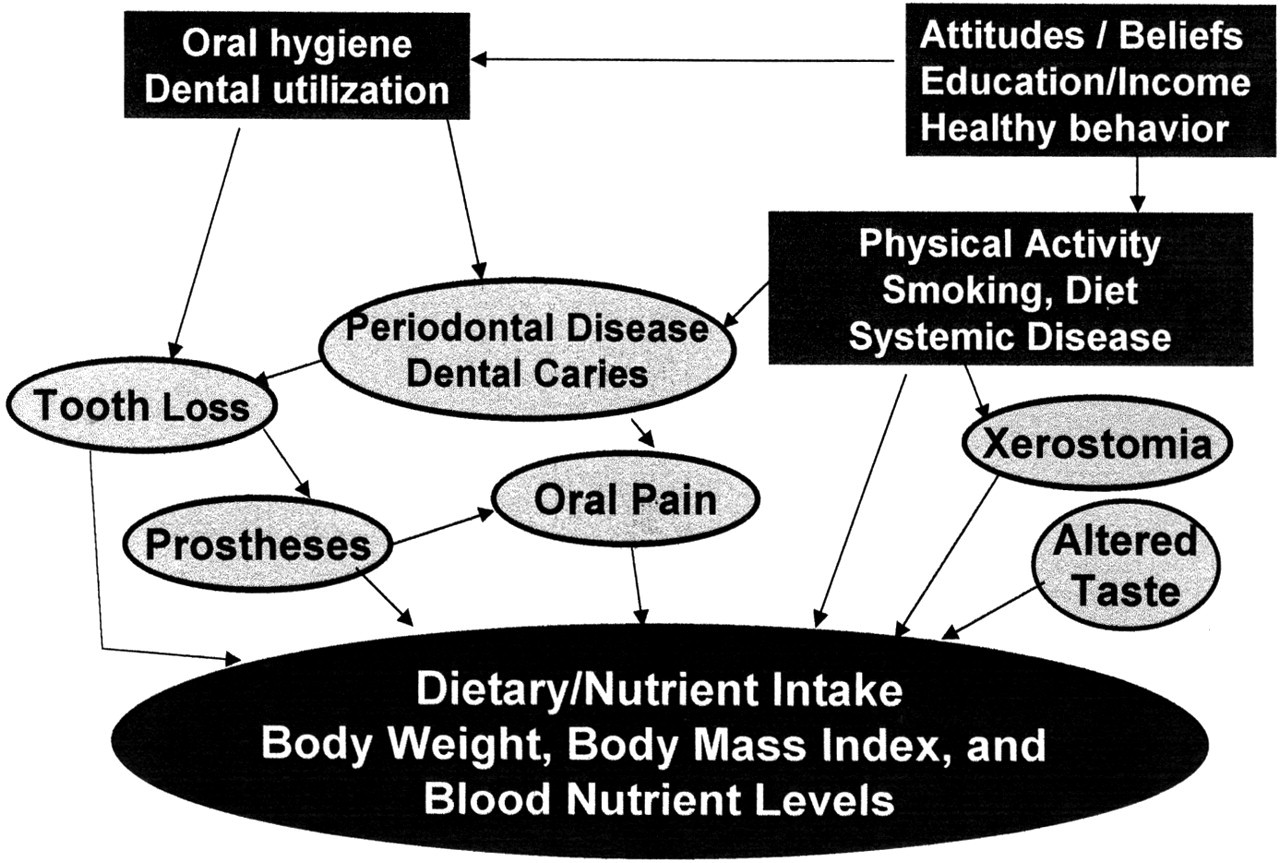
Pathways for the oral-nutrition relationship.
Footnotes
Acknowledgements
The authors thank Tom Barton for assistance with manuscript preparation and review. This work was supported in part by grant K01 AG00691 from the National Institutes of Health.
