Abstract
In this review, we present an overview of the signaling elements between neutrophil chemotactic receptors and the actin cytoskeleton that drives cell motility. From receptor-ligand interactions, activation of heterotrimeric G-proteins, their downstream effectors PLC and PI-3 kinase, the activation of small GTPases of the Rho family, and their regulation of particular cytoskeletal regulatory proteins, we describe pathways specific to the chemotaxing neutrophil and elements documented to be important for neutrophil function.
Introduction
Neutrophils are the key cellular elements of the innate immune system, providing protection from invading bacteria. When normal regulatory mechanisms fail, the neutrophil is also responsible for immunologically induced tissue injury (Faist and Kim, 1998). Following activation by bacterial by-products or other immune stimuli—such as lipopolysaccharide, glycolipids, and methylated DNA—neutrophils execute several specialized functions that include chemotaxis, phagocytosis, and the generation of reactive oxygen metabolites. All of these processes are required for the elimination of invading micro-organisms or cellular debris. Disturbances in these processes result in a dramatic increase in susceptibility to bacterial infection that is often reflected in the oral cavity (Ambruso et al., 2000). The oral cavity and the periodontium in particular reflect the effects of an over-exuberant neutrophil-mediated immune response (Van Dyke, 1985). Our ability to modify neutrophil activation and prevent excessive inflammatory responses is a potentially useful treatment objective for conditions associated with neutrophil-mediated tissue damage, such as periodontal diseases, sepsis, asthma, and transplant rejection (Fujishima and Aikawa, 1995). By identifying the signal transduction elements and pathways required for neutrophil recruitment to sites of infection, researchers may be able to design specific pharmacologic interventions that will allow for the dampening of overactive neutrophil responses (Benard et al., 1999b). In this review, we will highlight what is currently known about the chemotactic signaling pathways between the receptor and the cytoskeleton in neutrophils. This information can be used to identify potential therapeutic targets that will allow for the regulation of neutrophil chemotaxis, the initial recruitment phase of the innate immune response.
Chemotaxis
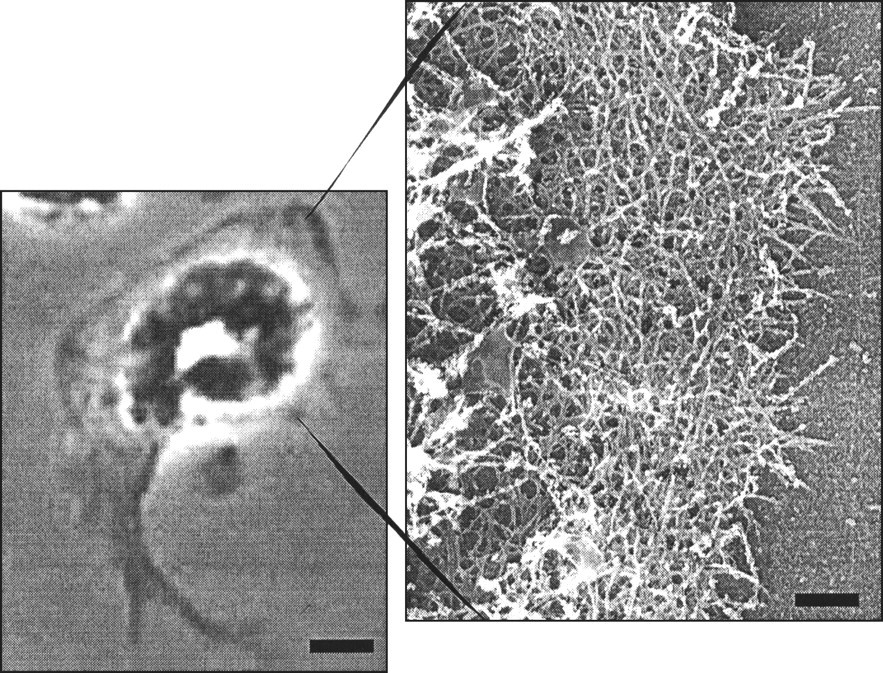
Neutrophils migrate to sites of bacterial ingress or tissue damage through the process of chemotaxis. The term chemotaxis was introduced in 1884 by Pfeffer, who described it as directional migration of leukocytes along a chemical gradient (Zachariae, 1993). Neutrophils will migrate toward the source of chemoattractants that include bacterial products such as formylated peptides, C5a, a product of the complement cascade, products of phospholipid metabolism (leukotriene B4), and chemokines such as IL-8 (Zachariae, 1993). Once a chemoattractant binds to its cell-surface receptor, a series of cytoplasmic events is triggered that results in the activation of the cytoskeletal machinery. This activation results in the neutrophil acquiring a polarized morphology that is characterized by a leading actin-rich lamella and a tail-like uropod at the rear end (Fig. 1). The leading-edge actin assembly is necessary for rapid migration of the activated cell toward the source of the chemical agent (Benard et al., 1999b).
In neutrophils, cell-surface receptor activation leads to actin cytoskeleton re-organization that drives cell motility (Glogauer et al., 2000). The most well-studied chemotactic receptor of neutrophils is the heterotrimeric G-protein-coupled, pertussis-toxin-sensitive formyl peptide (fMLP) receptor, which is now recognized to exist in high-affinity (the formyl peptide receptor) and low-affinity forms (formyl peptide receptor-like 1) (Reinhold et al., 1990; Thomas et al., 1990; De Nardin et al., 1991; Akasaki et al., 1999). These recepors recognize bacterial and mitochondrial proteins formylated at their amino terminal (De Nardin et al., 1991). The normal human neutrophil expresses about 50,000 fMLP receptors per cell, and these receptors bind formylated hydrophobic peptides with a dissociation constant of 20 nmol/L (Williams et al., 1977). The fMLP receptor is a 32-kDa glycosylated serpentine integral membrane glycoprotein containing 7 hydrophobic domains commonly interpreted as transmembrane helices. It undergoes affinity state changes modulated by a 40-kDa heterotrimeric GDP/GTP binding protein consisting of an α, β, and γ subunit (Malech et al., 1985; Jin et al., 2000). The α subunit is the GDP/GTP binding module, and in the GDP bound state it binds the βγ complex to form an inactive heterotrimer that associates with the fMLP receptor (Wieland and Chen, 1999). Ligand binding causes a conformational change in the receptor that promotes the exchange of GTP for the GDP on the alpha subunit. This induces the release of the α subunit from the βγ subunit pair (Wieland and Chen, 1999). The α subunit is then free to interact with downstream effectors, including phospholipase C (Park et al., 1993), while the βγ subunit interacts with effectors such as PI3-kinase (Clapham and Neer, 1993) and particular isoforms of phospholipase Cβ family (Park et al., 1993). It has been demonstrated in the soil amoeba, Dictyostelium discoidium, that the distribution of the βγ complex during chemotaxis is highest at the leading edge of the cell, suggesting that this subunit plays an important role in leading-edge signal transduction during chemotaxis (Jin et al., 2000).
Actin: The Cell Engine
Signal transduction elements in chemotaxis are essentially gears and regulators for the engine that drives neutrophil motility: the actin cytoskeleton. Actin is a 42-kDa protein that, under physiologic salt conditions, will assemble into filaments. A large number of proteins have been identified that modulate the assembly, disassembly, and organization of actin and actin filaments in cells. While neutrophils have a collection of actin-binding proteins similar to that found in most other cell types, they utilize their complement of actin-binding proteins to generate a motile machine capable of efficient and rapid chemotaxis.
Directional cell crawling, the intrinsic basis of chemotaxis, can be broken down into smaller processes, including extension of the cell membrane, adhesion to the surface, and contraction of the cell body in an organized and reversible way (Mitchison and Cramer, 1996). Actin-dependent protrusion of the leading edge of cells occurs through the use of two geometrically different structures. Small finger-like projections called filopodia extend out from the front of the cell (for review, see Stossel et al., 1999). In some systems, these filopodia are thought to explore the region in front of the cell, looking for permissive interactions with the substrate. In neutrophils, filopodia exist, but they are much less evident than the broad, clear lamellae at the cell front. The lamella is a sheet-like structure that is rich in actin filaments and critical for normal neutrophil motility (Fig. 1). The actin filaments within these regions are collected into highly organized structures that can behave like a solid to push the membrane forward (Stossel et al., 1999). These structures are likely to be formed with the use of different collections of actin-binding proteins, under the regulation of different components of the signal transduction cascades linking chemotactic receptors with cell movement. The processes of protrusion, adhesion, and contraction can be so well-integrated that the movement seems almost continuous.
The importance of actin and actin polymerization in chemotaxis is demonstrated by two sets of results. Cytochalasins, small molecules that bind actin filaments and prevent further addition of actin monomer (Toyama, 1984; Cooper, 1987), completely shut down motility (Mukherjee and Lynn, 1978; Dahlgren et al., 1979). Cytochalasin binds to the high-affinity end of the filament and blocks further monomer addition in vitro, and cells treated with cytochalasin fail to protrude and to assemble new actin filaments. In addition, a few patients with neutrophil dysfunction syndromes have been demonstrated to have a mutation in the gene for β actin, the actin isotype expressed in neutrophils (Nunoi et al., 1999). These results document the importance of normal actin function for neutrophil chemotaxis.
Actin filaments are polar, with each end differing in its equilibrium-binding constant for actin monomer (Wegner and Isenberg, 1983). This difference, generated by the ability of actin to bind and hydrolyze ATP (Wegner, 1977), provides a physical polarity to the filament that regulatory proteins use to drive filament dynamics with high temporal and spatial precision (Schafer and Cooper, 1995). This finding, along with the observation that cytochalasin inhibits actin assembly by binding to the high-affinity end of the actin filament, has led to the consensus that it is the regulation of the high-affinity actin filament end that is critical to normal actin polymerization and chemotaxis.
Two classes of proteins have been identified that regulate the availability of the high-affinity actin filament ends: filament-capping proteins and filament-nucleating proteins. Actin filament-capping proteins bind to the high-affinity filament end and regulate monomer addition by their presence or absence at the filament end (Schafer and Cooper, 1995; Carlier, 1998; Puius et al., 1998; Stossel et al., 1999). Actin-nucleating factors bind actin monomers under conditions otherwise unfavorable for assembly and generate a new filament with a free high-affinity end available for assembly (Pollard et al., 2000). The role of these classes of proteins in neutrophil chemotaxis, as well as the role of signaling molecules regulating these proteins, is outlined below.
In the last several years, new connections among receptors, signaling molecules, and actin-binding proteins involved in the mechanics of cell motility have been established, and older connections have been strengthened. Page limitations preclude a detailed discussion of the activity of all of the actin-binding proteins found in neutrophils. However, there are many excellent general reviews on cell motility (Puius et al., 1998; Stossel et al., 1999; Pollard et al., 2000). Instead, we will focus our efforts on those proteins that current data demonstrate, or strongly suggest, are necessary for normal neutrophil chemotaxis.
Signaling molecules involved in chemotaxis
Phospholipase C
The regulation of the phosphoinositide-specific phospholipase C family of proteins is complex (Rhee, 2001). PI-specific PLC cleaves phosphatidylinositol-4,5-bisphosphate PI(4,5)P2 to generate the second messengers inositol-1,4,5-trisphosphate (IP3) and diacylglycerol (DAG). IP3 leads to intracellular calcium mobilization, whereas DAG activates some isoforms of protein kinase C. There are several isoforms of PLC in neutrophils; the β2 and β3 isoforms are activated in murine neutrophils upon stimulation with fMLP (Li et al., 2000; Wu et al., 2000). In contrast, the activation of the γ isoform of PLC in neutrophils occurs only with exposure to Mycobacterium tuberculosis (Perskvist et al., 2000). For both isoforms of PLC, activation leads to the production of bactericidal reactive oxygen intermediates. Data obtained with PLC-β-deficient mice strongly suggest that signaling of PLC-β in neutrophils is restricted to superoxide production and is not involved in chemotaxis (Li et al., 2000).
PI-3 kinases
PI-3 kinases are a family of enzymes that have been identified to be downstream of the βγ complex of G-protein-coupled receptors. PI-3 kinases phosphorylate inositol phospholipids specifically at the D3-position of the inositol ring. The main lipid products of the PI-3 kinases in higher eukaryotes are phosphatidylinositol-3-phosphate PI(3)P, phosphatidylinositol-3,4-bisphosphate PI(3,4)P2, and phosphatidylinositol-3,4,5-trisphosphate PI(3,4,5)P3. PI(3)P is present in all eukaryotes and is believed to play a role in vesicle transport and fusion (Fruman et al., 1999; Gillooly et al., 2000). PI(3,4)P2 and PI(3,4,5)P3 are implicated in a wide variety of cellular processes, including cell transformation, cell survival, cell polarization, and re-arrangements of the actin cytoskeleton (Toker and Cantley, 1997; Rameh and Cantley, 1999). There are three families of PI-3 kinases of which only the class I family has been extensively studied in mammals. All known members of this family are heterodimers consisting of a regulatory and a catalytic subunit. In one subclass (class IA), the regulatory subunit of 85 kDa (p85) forms a complex with a 110-kDa catalytic subunit (
Since the discovery of PI-3 kinase and its lipid products in 1988, PI-3 kinase signaling has been implicated in neutrophil chemotaxis. Production of PI(3,4,5)P3 in cells was first demonstrated in neutrophils stimulated with fMLP (Traynor-Kaplan et al., 1988, 1989). As early as 1990, PI-3 kinase signaling was tentatively linked to actin polymerization in these cells (Eberle et al., 1990). Many publications suggest a role for PI-3 kinase and its lipid products in neutrophil chemotaxis (for an overview, see Rickert et al., 2000; Wu et al., 2000; Wymann et al., 2000)). However, the precise role of the lipid products of PI-3 kinase in neutrophil chemotaxis is still a topic of active research.
Two known inhibitors of PI-3 kinase activity, wortmannin and LY294002, are commonly used in the study of PI3-kinase activity in a variety of cellular processes. In stimulated neutrophils, these compounds completely or partly block respiratory burst (Baggiolini et al., 1987; Thelen et al., 1994), activation of phospholipase D (Reinhold et al., 1990), increase in tyrosine phosphorylation (Naccache et al., 1993), activation of MAP/ERK-kinase 2 (MEK2) (Downey et al., 1996), chemotaxis (Thelen et al., 1995), and cell polarization (Niggli and Keller, 1997). In contrast, fMLP-mediated actin polymerization in neutrophils seems to be only partially affected by blocking PI-3 kinase activity (Arcaro and Wymann, 1993; Niggli and Keller, 1997). However, generalized inhibition of PI-3 kinase activity reveals only limited information about the precise role of this enzyme in a cellular process. For example, it does not reveal information about the spatial and temporal regulation of this enzyme that is likely to be important to its function in neutrophil chemotaxis.
In a more recent approach, investigators tried to overcome this limitation by using a fluorescent protein (usually Green Fluorescent Protein) fused to a pleckstrin homology (PH) domain. PH domains are small domains of about 120 amino acids that bind the hydrophilic head groups of polyphosphorylated phosphoinositides. A GFP-fusion protein with a PH domain specific for PI(3,4,5)P3 and PI(3,4)P2 was used to localize PI-3 kinase products in neutrophil-like cell lines upon stimulation. The experiments revealed that these cells respond to very shallow external gradients of chemoattractant (fMLP) by creating a much steeper internal gradient of PI(3,4,5)P3 that could activate components of the motile machinery in the appropriate locale. Sites of high PI-(3,4,5)P3 concentration co-localized with the leading edge of the lamella, the site of maximal actin polymerization in activated neutrophils (Servant et al., 2000). Creation of this gradient is dependent on the activity of members of the Rho subfamily of small GTPase (including Rho, Rac, and CDC-42h), suggesting a possible positive feedback between GTPase activity and PI-3 kinase activity (Servant et al., 2000).
These results support the earlier observation that neutrophil polarization is blocked by the PI-3 kinase inhibitor wortmannin (Niggli and Keller, 1997) (see above), and they are in agreement with data obtained from PI-3 kinase γ knockout mice. Neutrophils from these mice have a severe defect in chemotaxis and cell polarization, suggesting that PI-3 kinase activity is important in establishing cell polarity. However, neutrophils from PI-3 kinase γ -/- mice are still able to polymerize actin and to migrate upon stimulation, even though they do not produce detectable amounts of PI(3,4,5)P3 upon stimulation (Hirsch et al., 2000; Li et al., 2000; Sasaki et al., 2000). This finding and the observed increase in actin assembly in stimulated neutrophils treated with the PI-3 kinase inhibitor wortmannin suggest that there is a PI-3 kinase-independent pathway leading to actin polymerization and motility, and that the main function of PI-3 kinase activity is in the detection and signaling of a chemotactic gradient.
Guanosine exchange factors
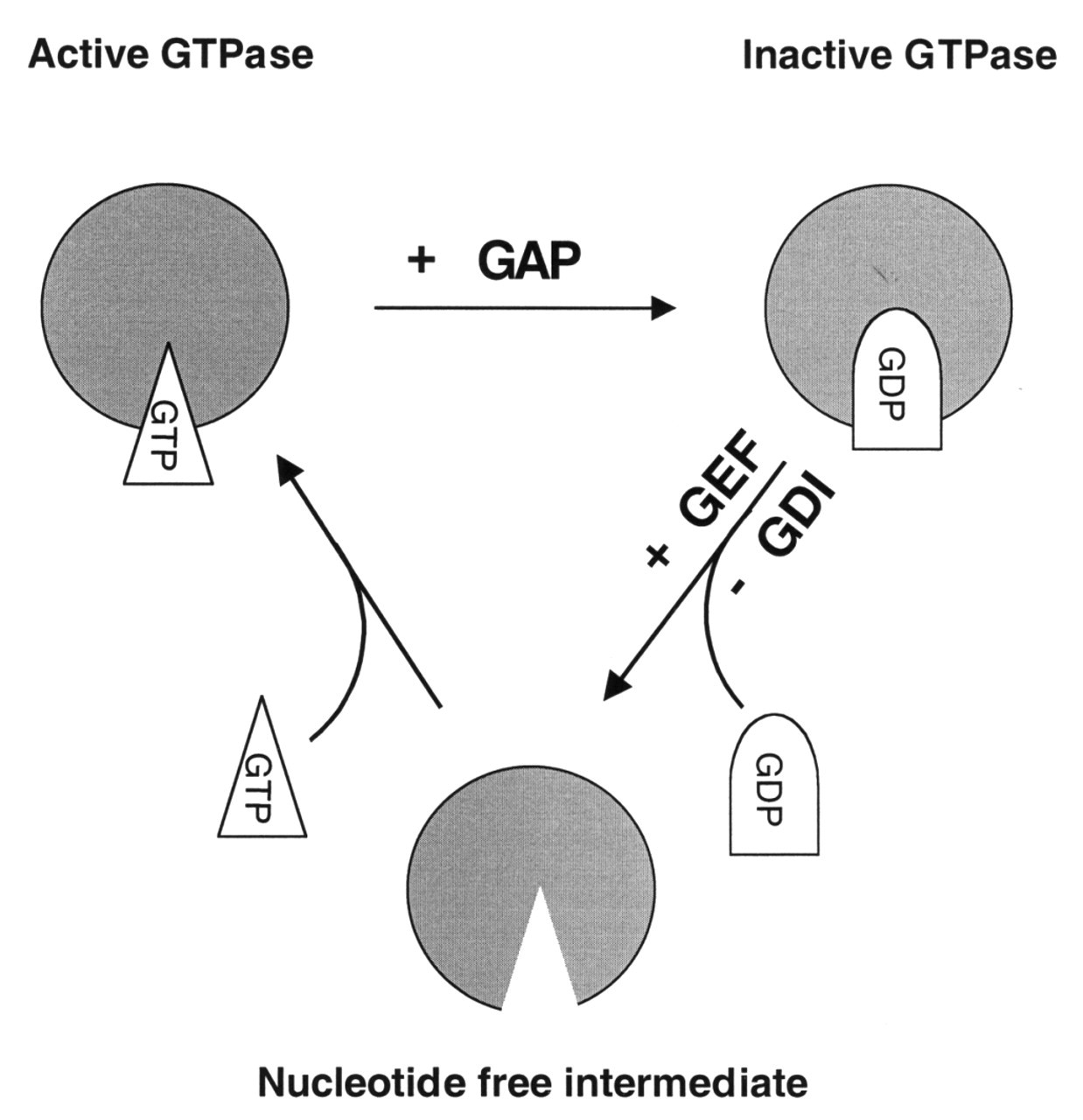
In many systems, PI-3 kinase activity has been linked to the activation of a family of small GTP-binding proteins related to Ras. GTP-binding proteins of the Ras superfamily are regulated by the phosphorylation state of the bound nucleotide. A subfamily of the Ras GTPases related to Rho and including Rho, Rac, and CDC-42 is an important regulator of motility and cell shape. These small GTPases cycle between an active GTP-bound state and an inactive GDP-bound state (Fig. 2) in a cycle that is tightly regulated by several accessory proteins, such as Guanosine Dissociation Inhibitors (GDIs), GTPase Activating Proteins (GAPs), and Guanosine Exchange Factors (GEFs). The former two promote a GDP-bound, inactive state for small GTPases either by inhibiting the dissociation of GDP from the GTPase (GDIs), or by activating the intrinsic GTPase activity of these proteins (GAPs). GEFs, on the other hand, are the only known proteins that enhance the activity of small GTPases. Upon binding to a GTPase, they lower the affinity of the GTPase for GDP, allowing for the dissociation of GDP and promoting the binding of GTP. This converts the small GTPase to its active form. GEFs are generally large (at least 90 kDa) complex proteins containing a dbl-homology domain that is thought to mediate GEF activity, one or more Src homology domains (SH2 and/or SH3 type), and at least one PH domain (Quilliam et al., 1995; Cherfils and Chardin, 1999). Most current models attribute the PI-3 kinase-sensitive activation of small GTPases of the Ras superfamily to a PI-3 kinase-dependent activity of GEFs.
A possible candidate for mediating PI-3-kinase signaling to small GTPases in neutrophils is the GEF Vav1. Vav1, which is expressed in hematopoietic cells and in the buds of developing teeth, and the more ubiquitously expressed isoforms, Vav2 and Vav3, activate the small GTPase Rac in vitro and in vivo (Han et al., 1998; Ma et al., 1998). In vitro, Vav1 was shown to bind PI(4,5)P2 as well as PI(3,4,5)P3. Interestingly, binding to PI(4,5)P2 inhibited Vav1 GEF-activity, whereas binding to PI(3,4,5)P3 activated GEF activity of Vav1 (Han et al., 1998). This suggests that Vav could connect PI-3 kinase signaling with downstream small GTPases and their effectors. However, little has been published on the role of Vav1 in neutrophil function. Results obtained from vav1 -/- mice suggest a critical function for Vav1 in T-cell development and for Vav1 and 2 in B-cell development (Turner et al., 1997; Cantrell, 1998; Costello et al., 1999; Tedford et al., 2001). Taken together, very little is known about the function of specific GEFs in neutrophils, even though these molecules are crucial components in signaling pathways.
Experimental evidence suggests that there may be multiple connections between signaling components and that individual signal elements may function at multiple steps within a signal pathway. Mice that are deficient in the small GTPase Rac2, the isoform predominantly expressed in hematopoietic cells, have neutrophils that display defects in chemotaxis and cell polarization (Roberts et al., 1999). Similar defects were observed in the PI-3 kinase γ deficient mice (see above). In contrast, fMLP stimulation of neutrophils from PI-3 kinase γ -/- mice leads to normal activation of the small GTPase Rac (Li et al., 2000). This suggests that there may be multiple pathways with cross-talk connections in the network of signals between the receptor and the motile machinery. To complicate matters, species differences may be important, since activation of Rac in human neutrophils with fMLP is wortmannin-sensitive and likely PI-3 kinase-dependent (Akasaki et al., 1999; Benard et al., 1999a).
The Rho family of small GTPases
In neutrophils, as in other cell types, the small GTPases of the Rho subfamily are active regulators of cell shape change and motility (Hall and Nobes, 2000; Katanaev, 2001; van Nieuw Amerongen and van Hinsbergh, 2001). Many of the results connecting these small GTPases to particular cytoskeletal proteins have been derived from studies using neutrophil extracts, since mature native neutrophils have not been successfully microinjected or transfected. The paradigm, derived from the now-classic studies of the Hall lab, is that individual small GTPases lead to specific functional responses within the actin cytoskeleton (Ridley and Hall, 1992; Ridley et al., 1992). Cdc-42h is thought to induce fine finger-like projections called filopodia, and to activate the small GTPase Rac. Rac activation induces lamellae formation, a broad, thin, actin-rich, organelle-excluding region most often found at the front of motile cells (Nobes and Hall, 1995). In cytoplasmic extracts of neutrophils, active CDC42h stimulates actin filament assembly (Zigmond et al., 1998), and Rac stimulates actin assembly in a permeabilized neutrophil model (Glogauer et al., 2000).
The small GTPase Rac is critical for normal neutrophil function. A patient with neutrophils having ineffective bactericidal and motile function was found to have a point mutation at amino acid 57 in the Rac2 gene, the Rac isoform expressed in neutrophils. This change caused the mutant protein to suppress the activity of the remaining normal Rac protein, having a dominant-negative effect on total Rac activity (Ambruso et al., 2000). Rac2-deficient mice display similar neutrophil functional defects and in vivo susceptibility to infection (Roberts et al., 1999).
Cytoskeletal effectors of small GTPases
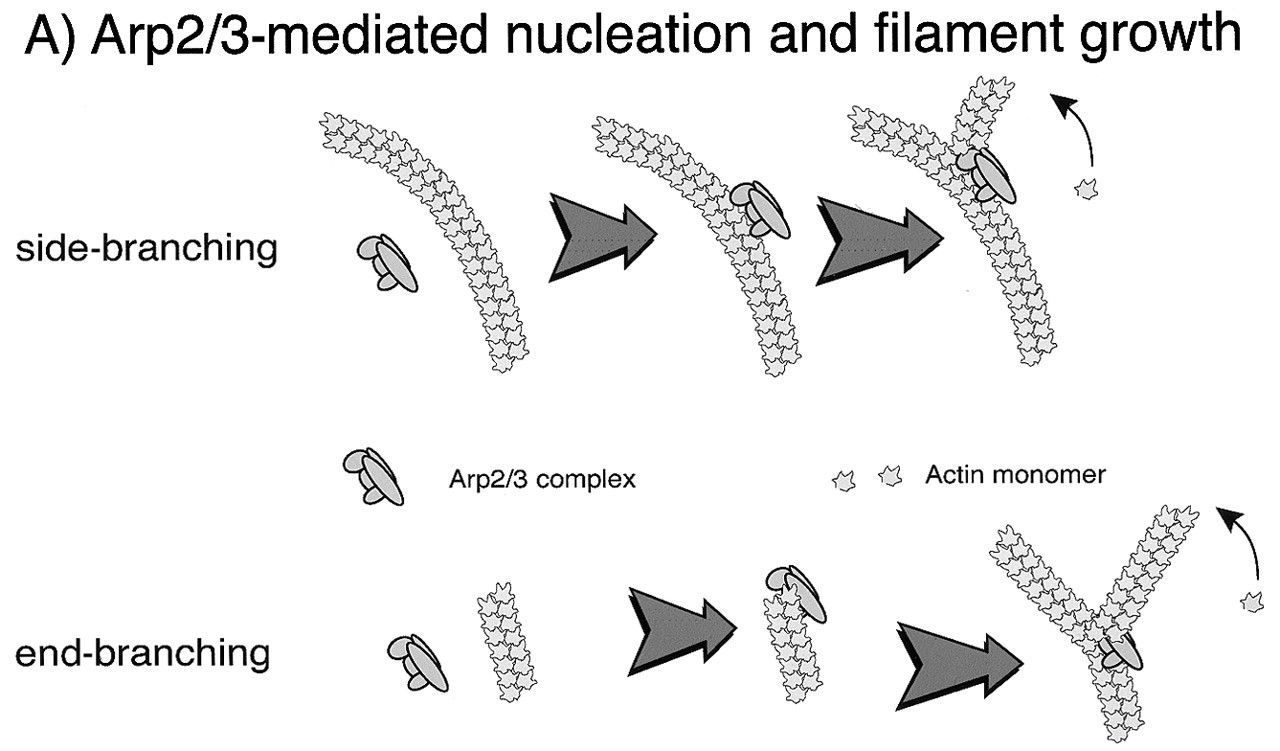
In the last several years, growing numbers of actin-related proteins have been identified. Similar in sequence to actin, but with regions of significant structural and sequence divergence, these proteins have been implicated in several important processes (Schafer and Cooper, 1995). A complex containing actin-related proteins 2 and 3 (Arp2/3) and five other proteins was identified by its ability to bind profilin (Machesky et al., 1994). Interest in this complex increased significantly when it was found to be necessary for actin filament generation in cell extracts that supported the actin-based motility of the intracellular pathogen Listeria (Welch et al., 1997). Further work demonstrates that the Arp2/3 complex is connected to multiple pathways that signal to actin assembly.
In vitro studies with purified proteins or cytoplasmic extracts derived from neutrophils demonstrate that the Arp2/3 complex is stimulated to nucleate actin polymerization upon binding a complex of GTP-bound CDC-42h, N-WASP (a member of the Wiskcott Aldrich Syndrome protein family), and the lipid PI(4,5)P2 (Zigmond et al., 1998; Higgs and Pollard, 2000). The Arp2/3 complex localizes to regions of active actin polymerization in neutrophils (Machesky et al., 1997), and its dynamics have been beautifully imaged in a tissue culture model of neutrophils as the cells move toward fMLP (Weiner et al., 1999). While these results strongly suggest that the Arp2/3 complex is an important factor downstream of CDC-42h in the regulation of chemotaxis, there are undoubtedly many other factors. In a permeabilized neutrophil model in which signaling from fMLP to actin assembly is maintained, inhibiting Arp2/3 activity with a peptide that prevents its binding to the WASP family member inhibited only half of the total actin assembly (Glogauer et al., 2000). Fig. 3 illustrates the role of the Arp2/3 complex and its mechanism of action in actin assembly during neutrophil motility.
Rac is likely to have several targets within the pool of neutrophil actin-binding proteins. However, several lines of evidence suggest that, in neutrophils, gelsolin is an important effector for Rac. Gelsolin was originally identified in extracts of macrophages (Yin and Stossel, 1979), and is also highly expressed in neutrophils (Kwiatkowski, 1988). Gelsolin is activated by calcium ions to sever actin filaments and to bind tightly to the high-affinity end of the actin filament (Janmey et al., 1985). This capping of the preferred end for assembly can be disrupted only by phosphoinositides such as PI(4,5)P2 (Janmey and Stossel, 1987). Mice genetically deficient in gelsolin have been engineered, and their neutrophils have reduced motile response both to in vivo insult and in in vitro assays (Witke et al., 1995). Phagocytic activity through the Fc receptor is deficient in gelsolin null neutrophils, while complement-mediated phagocytosis is unimpaired (Serrander et al., 2000). In a possible compensation mechanism, gelsolin-deficient mice have increased expression of Rac (Azuma et al., 1998). In neutrophil extracts, the active GTP-bound form of Rac stimulates the removal of gelsolin from the end of the actin filament (Arcaro, 1998), and in the permeabilized neutrophil model, fMLP stimulates Rac-dependent actin assembly (Glogauer et al., 2000). In other cell types, some of the characteristic shape changes induced by activated Rac are deficient in gelsolin-null animals (Azuma et al., 1998). The possible role of gelsolin and other capping proteins in regulating new actin filament assembly in the neutrophil is illustrated in Fig. 3.
The role of Rho in neutrophil function is unclear. In fibroblasts, Rho induces stress fibers and cell contraction by regulating the phosphorylation status of the regulatory light chain of non-muscle myosin II, the classic myosin capable of forming bipolar filaments (Somlyo, 1999). The role of myosin motor activity in cell motility has often been questioned. Initially, generation of motile Dictyostelium amoebae that lack the classic non-muscle myosin argued against a necessary role for myosin in motility and chemotaxis (De Lozanne and Spudich, 1987). Since that original work was published, the myosin superfamily has grown to 15 or more myosin types with multiple genes, complicating our understanding of the role of myosin in cell function (Sellers, 2000). In neutrophils, recent work has nicely demonstrated that the activity of myosin-II, the classic non-muscle bipolar myosin, is critical for cell release and contraction of the rear of the cell, or uropod, toward the leading edge as the cell moves forward (Eddy et al., 2000). An increase in the level of free calcium ions, capable of activating myosin II molecules through the activity of myosin light-chain kinase, has also been detected in the rear of the cell (Marks and Maxfield, 1990; Hendey and Maxfield, 1993; Mandeville et al., 1995; Mandeville and Maxfield, 1997). The role of the Rho GTPase in modulating this process in neutrophils has not been examined. Recent work from other systems suggests that active Rho can decrease the activity of the phosphatase that inactivates the regulatory light chain as well as possibly inducing phosphorylation of the regulatory light chain itself (Somlyo, 1999). Therefore, Rho activation would promote myosin activity and contraction.
Multiple roles of phosphoinositides
The downstream connections between the small GTPases and their actin-binding protein targets have to some degree been identified. Poly-phosphorylated phosphoinositide lipids function at multiple steps within these pathways. PI(4,5)P2 is a necessary co-factor for CDC-42 to bind to the ARP2/3 complex, an actin-nucleating factor, as discussed in detail below (Higgs and Pollard, 2000). PI(4,5)P2 production is also necessary downstream of Rac, since a mutated PI-4P 5-kinase that is able to bind, but not be activated by, Rac inhibits actin assembly in several model systems and cell types (Tolias et al., 2000). Many actin-binding protein activities are also directly modulated by phosphoinositides in vitro (Janmey et al., 1999).
Other cytoskeletal proteins critical for normal neutrophil function
Lymphocyte-specific protein, also known as LSP-1, was found to be the cause of a neutrophil dysfunction syndrome (NAD 47/89) (Howard et al., 1994). Patients with this syndrome overexpress a 47-kDa protein and show reduced expression of a currently unidentified protein of 89 kDa. The 47-kDa protein was identified as lymphocyte-specific protein 1, which is now known to be leukocyte-specific (Howard et al., 1994; Li et al., 1995). LSP-1 induces bundling of actin filaments in vitro (Zhang et al., 2000), and overexpression in multiple cell types induces actin-rich hair-like structures and a reduction in motility (Howard et al., 1998). LSP-1 is also phosphorylated in response to extracellular stimuli by MAP kinase II (Huang et al., 1997), and in a hairy cell leukemia model, phosphorylation regulated LSP-1's ability to generate the unusual hairy morphology consistent with cells overexpressing LSP-1 (Miyoshi et al., 2001). An LSP-1-deficient mouse has been generated, and the neutrophils and macrophages show enhanced migration into the peritoneum in response to insult and enhanced chemotaxis to both fMLP and the C-X-C chemokine, KC, in vitro (Jongstra-Bilen et al., 2000). These results suggest that LSP-1 and its ability to bundle actin may stabilize the cytoskeleton as part of its normal function.
Conclusion
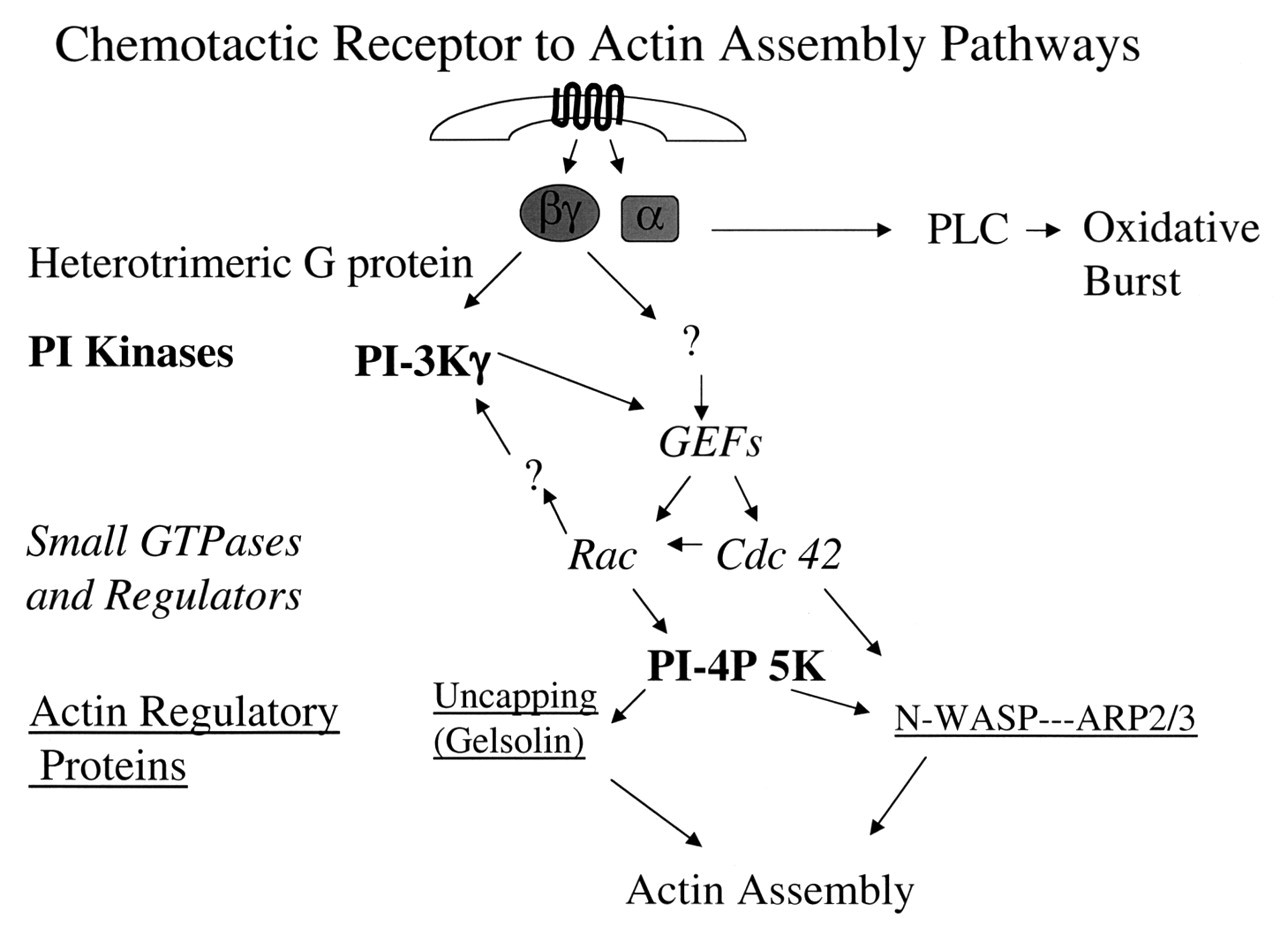
In this review, we have attempted to present an overview of the signaling elements between neutrophil chemotactic receptors and the actin cytoskeleton that drives cell motility (Fig. 4). From receptor-ligand interactions, activation of heterotrimeric G-proteins, their downstream effectors PLC and PI-3 kinase, the activation of the small GTPase of the Rho family, and their regulation of particular cytoskeletal regulatory proteins, we have illustrated pathways specific to the chemotaxing neutrophil and elements documented to be important for neutrophil function. In places, these pathways do not seem simple or to follow simple linear pathways. This may be due to complexities of the system not yet worked out, as well as to the reality that life and its biochemistry do not necessarily function in straight lines. However, the growing number of mice deficient in particular proteins, along with the continued study of patients with altered neutrophil function, will aid in identifying unexpected players in these pathways and help both the clinician and the researcher find ways to modulate neutrophil function to the benefit of us all.
Neutrophil morphology. Phase micrograph of an fMLP-activated neutrophil (bar = 5 μm) with an electron micrograph of leading lamella of an fMLP-activated neutrophil demonstrating the highly branched actin filament network (bar = 500 nm). Activation cycle of small GTPases of the Ras superfamily. Small GTPases of the Ras superfamily cycle between a GTP-bound active and a GDP-bound inactive state. GTPase Activating Proteins (GAPs) enhance the slow intrinsic GTPase activity of small GTPases. Guanosine Dissociation Inhibitors (GDIs) inhibit dissociation of GDP from the GDP-bound form of these proteins, whereas Guanosine Exchange Factors (GEFs) enhance this process. Regulation of actin filament elongation in migrating neutrophils. Summary. A schematic outline of the known signaling elements that link chemotactic receptors and actin assembly in neutrophils. It is recognized that each arrow could represent multiple elements in the signaling path.