Abstract
The aim of this review is to discuss what is known about the reflex control of the human masticatory system and to propose a method for standardized investigation. Literature regarding the current knowledge of activation of jaw muscles, receptors involved in the feedback control, and reflex pathways is discussed. The reflexes are discussed under the headings of the stimulation conditions. This was deliberately done to remind the reader that under each stimulation condition, several receptor systems are activated, and that it is not yet possible to stimulate only one afferent system in isolation in human mastication experiments. To achieve a method for uniform investigation, we need to set a method for stimulation of the afferent pathway under study with minimal simultaneous activation of other receptor systems. This stimulation should also be done in an efficient and reproducible way. To substantiate our conviction to standardize the stimulus type and parameters, we discuss the advantages and disadvantages of mechanical and electrical stimuli. For mechanical stimulus to be delivered in a reproducible way, the following precautions are suggested: The stimulus delivery system (often a probe attached to a vibrator) should be brought into secure contact with the area of stimulation. To minimize the slack between the probe, the area to be stimulated should be taken up by the application of pre-load, and the delivered force should be recorded in series. Electrical stimulus has advantages in that it can be delivered in a reproducible way, though its physiological relevance can be questioned. It is also necessary to standardize the method for recording and analyzing the responses of the motoneurons to the stimulation. For that, a new technique is introduced, and its advantages over the currently used methods are discussed. The new method can illustrate the synaptic potential that is induced in the motoneurons without the errors that are unavoidable in the current techniques. We believe that once stimulation, recording, and analysis methods are standardized, it will be possible to bring out the real "wiring diagram" that operates in conscious human subjects.
Introduction
The main function of the masticatory muscles is to break food down into pieces small enough to be swallowed. These are strong muscles that generate very large forces across very short distances and apply them via rigid teeth. Such large forces can easily damage the teeth and their supporting tissues, tongue, cheeks, and the joints unless they are controlled precisely and effectively. There is evidence that the masticatory forces are controlled very precisely and that these forces change from bite to bite, depending on the consistency of the bolus. However, we do not fully understand this control mechanism. Unless the details of the mechanism that controls the masticatory forces in health and disease are thoroughly understood, the diagnosis and treatment of masticatory-related dysfunctions (such as temporomandibular dysfunction) will remain at the present "symptomatic" state.
The aim of this review is to discuss what is known about the control of the human masticatory system and to propose a method for standardized investigation. This review is divided into six parts. The first part discusses the activation of human jaw muscle motoneurons by the central and peripheral sources. The second part discusses the receptors that are thought to contribute to the control of human mastication. In part 3, the reflexes elicited in human subjects are discussed. In part 4, simulated chewing experiments are discussed. In part 5, recording and analyzing methods are discussed, and a new method for estimating synaptic potential in human motoneurons is introduced. Finally, in part 6, three frequently asked questions are used to discuss the issues put forward in this review.
Presently, our knowledge of mastication and its control by various receptors is patchy. Most of the work in this area comes from animal species with which investigators have used widely varying reduction techniques (anesthetization, decerebration, curarization, etc.). In these preparations, when nerve pathways are the subject of the study, the nerve to be stimulated is dissected free and teased so that one or a few identified nerve fibers are stimulated. The synaptic potential that is induced by this stimulation is recorded by means of microelectrodes that are placed within the central nervous system (reviewed in Lund, 1991; Linden, 1990). It is also possible for investigators to initiate "chewing" in animal preparations, and to study the effects of various receptor systems on the development of force (Lavigne et al., 1987; Morimoto et al., 1989).
In humans, one must establish the connections of various afferents to the motoneurons that innervate the masticatory muscles under static or dynamic conditions to begin to understand the mechanism and control of mastication. The human work is challenging, since direct recording from motoneurons is not yet possible and precise stimulation of nerves is not easily controlled. Therefore, various indirect measurements have been used for the study of synaptic potential in human subjects. In most of these studies, afferent nerves are stimulated by mechanical and/or electrical means, and the responses of the motoneurons are recorded from the muscle to illustrate the basic synaptic connection between the stimulated afferent fibers and the motoneurons that innervate the muscle. However, because these techniques are indirect, they are open to numerous methodological errors in stimulation, recording, and analysis processes.
The structure and function of the human jaw muscles have been the subject of one recent comprehensive review (Hannam and McMillan, 1994). Therefore, this review will not repeat the information regarding the masticatory muscles. Instead, it will bring together what is known about the basic wiring of the human masticatory system and put forward a method for standardized investigation. While doing so, it will also remind the reader of some of the findings in animal species, since they are fundamental in the formulation of hypotheses in human experiments. We believe that once the standardized methods are used, it will be possible to bring out the real phenomenon that operates in conscious human subjects.
(1) Activation of Human Jaw Muscle Motoneurons
Jaw muscle motoneurons are activated by three sources: the motor cortex, which initiates/stops mastication and delivers pre-programmed movement patterns depending on the expectations and feedback; the central pattern generator (CPG), which provides basic rhythmic activity to jaw muscles; and the peripheral input, which may be the most powerful and yet most variable input to the motoneurons.
The motor cortex
The motor cortex sends direct and indirect input to jaw muscle motoneurons. Although the direct fibers represent only a small proportion of the total motor cortical output, investigation of this pathway is easier in human subjects with the use of various transcranial stimulation techniques (Cruccu et al., 1989). Electrical and magnetic brain stimulation has been used for the activation of this pathway. However, the reader is warned that these methods are not small-area stimulation techniques. They excite large cortical areas, and hence the limitations of these techniques need to be remembered when they are used and their results interpreted. Such experiments have shown that at least 30% of the jaw-closing motoneurons are reached by direct, fast-conducting, mainly crossed, corticobulbar fibers, and that motor-evoked potentials in jaw muscles could be elicited only during voluntary contractions (Cruccu et al., 1989; Nordstrom et al., 1999). Suprahyoid motoneurons are also reached by fast-conducting corticobulbar fibers; these connections are mainly bilateral (Cruccu et al., 1989; Gooden et al., 1999). These pathways that originate from the cortex are mainly used for the conscious initiation and termination of mastication. They can also be used when one deliberately chews, bites, or uses the jaw for performing a conscious task such as carrying an object or attacking a prey. There is also some circumstantial evidence from human work (Ottenhoff et al., 1992a,b) that the cortex may "set" the effectiveness of the synaptic input to motoneurons that innervate jaw muscles (Yang and Türker, 1999) or release a pre-programmed movement pattern (Houk, 1978; Gotlieb and Agarwal, 1980), depending upon the resistance encountered in the previous bite.
Central pattern generator (CPG)
Experiments on animals have established the existence of a rhythm generator for mastication which is produced by a group of cells known as the central pattern generator (CPG; Dellow and Lund, 1971). The CPG lies in the medial bulbar reticular formation between the motor root of the trigeminal nerve and the inferior olive. The CPG induces an inhibition in the closer motoneurons simultaneously with excitation in the openers during the opening phase of chewing. The CPG then induces excitation in the closer motoneurons during the closing phase (reviewed in Lund, 1991). Although the CPG sets the basic rhythm for mastication and alternately activates the openers and closers, control of this process is largely dependent upon the sensory feedback (Lund, 1991). In fact, the CPG-induced depolarizing potentials in masseter motoneurons are often too small to cause a discharge in fictive mastication, where peripheral feedback is not possible (Kubo et al., 1981). However, the closing phase becomes much more active in unparalyzed preparations, especially when an object is inserted between the teeth to stimulate the receptors (Goldberg and Tal, 1978). This interdependence indicates the importance of both the CPG and feedback from receptors in forming the neurological basis of mastication.
Although it has been known that rhythmic jaw movement is present in anencephalic human infants, the existence of the masticatory CPG in human subjects has not been directly shown but rather has been assumed from various pieces of circumstantial evidence, such as the existence of the sucking reflex in the infant (Finan and Barlow, 1998), the observation of phase-dependent modulation of mastication (Svensson et al., 1997), the existence of high-frequency oscillation in the jaw muscle electromyogram (Smith and Denny, 1990), and the interaction among mastication, respiration, and swallowing (McFarland and Lund, 1995).
Peripheral feedback
Forces applied to a tooth can stimulate receptors in the periodontal ligament, gingival mucosa, dentin, pulp, alveolar bone, and periosteum. These receptors that respond when a force is applied to the teeth have been termed periodontal mechanoreceptors (PMRs; Linden, 1990). If the tooth stimulation moves the mandible, in addition to these receptors, the receptors in the jaw muscles and in the temporomandibular joints are also stimulated.
The importance of peripheral feedback in controlling masticatory muscle activity has been illustrated in rabbits chewing while lightly anesthetized. The removal of sensory feedback from the periodontal receptors by disabling of the maxillary and inferior alveolar nerves greatly reduced the facilitation of the masseter muscle observed when a test strip was inserted between the teeth (Lavigne et al., 1987; Morimoto et al., 1989). When spindle cell bodies were also destroyed, the facilitation of the jaw closers disappeared almost completely (Morimoto et al., 1999). These experiments convincingly demonstrated that peripheral receptors and especially the PMRs and the muscle spindles play an important role in modulating the activity of motoneurons in "chewing" animals.
In these simulated chewing experiments on anesthetized rabbits, it was shown that the contribution from the PMRs stands out as the most important feedback source, since they generate the major part of closing muscle activity (Morimoto et al., 1989, 1999; Morimoto and Nagashima, 1989). The contributions from the muscle spindles and other receptors make up only a small portion of the muscle activity in these animals (Morimoto and Nagashima, 1989). The contribution from the cortex cannot be determined in these animals, since they were "chewing" under general anesthesia.
In humans, the importance of the peripheral receptors is illustrated in patients treated with dental bridges supported by implants. Unlike subjects with healthy teeth who display bite-to-bite variation in jaw muscle activity, these patients chew with approximately the same pattern of muscle activity during the whole masticatory sequence (Haraldson, 1983). Therefore, peripheral receptors are important for bite-to-bite changes of the masticatory muscle activity. Furthermore, reduction in the bite force in edentulous subjects, to about 20-40% of the value obtained from subjects with natural teeth (Haraldson et al., 1979; Slagter et al., 1993), may indicate the importance of the PMRs which are destroyed during the extraction process. It is also possible to illustrate the existence of the positive feedback from the PMRs by measuring maximum voluntary bite force before and during local anesthesia of the teeth. Such experiments have shown that the maximum bite force value decreased drastically unless the subject was given reassurance or visual feedback of the bite force level (Lund and Lamarre, 1973; Orchardson and MacFarland, 1980).
The contributions of the muscle spindles to the development of muscle activity in humans cannot be determined exactly but can only be speculated upon. Examining the changes in the discharge rate of human tibialis anterior motoneurons in the absence of the contribution from the muscle afferents, Macefield and colleagues (1993) have shown that the maximal discharge rate decreased by about 35% in the absence of muscle afferents. This confirmed the suggestion by Vållbo et al. (1979) that the human muscle spindles are not the main contributors to the drive of the motoneurons. The contribution of muscle spindles to the overall drive of the motoneurons has been suggested to be in the form of the α-γ co-activation, in that, as the contraction level of a muscle is increased, the muscle spindle activity also increases (via the activation of γ motoneurons that stretch the polar region of the spindles) to give additional drive to the motoneurons (reviewed in Vållbo et al., 1979).
In the jaws, when the force between the jaws increases suddenly and unexpectedly, a load compensation reflex occurs (Lamarre and Lund, 1975). The latency of this reflex suggests a monosynaptic event, and this can occur even when there is no stretch to the jaw muscles. The fact that the reflex response could be obtained without the stretch of the contracting jaw closers strongly suggested that α-γ co-activation is occurring during chewing in human subjects (Lamarre and Lund, 1975). This claim supported later findings that the spindle afferent discharges increase almost linearly with the increase in voluntary contraction in human muscles (reviewed in Vållbo et al., 1979). Such findings are difficult to explain by any mechanisms other than co-activation of the α and γ systems, where the γ system is not only compensating for the unloading effect of the muscle contraction on the spindles but also producing an overall increase in spindle discharge (Vållbo et al., 1979). It can be summarized that the contribution of the muscle afferents to total bite force developed would be a significant amount, and that the level of this contribution may increase with the intensity of bite force. This dependence of total jaw-closer muscle activity on the performance of the peripheral receptors ensures that, should resistance between the jaws suddenly yield, this positive feedback would immediately cease, hence reducing the jaw-closing muscle activity and helping to stop the jaws from forcefully coming together.
The influence of peripheral feedback on the motoneuron activation (i.e., the wiring diagram) has been studied by two different algorithms: reflex work and simulated chewing work. Before we go into the details of these methodologies and outcomes, we summarize the literature on the types of receptors that are found in the human masticatory system that may be involved in the control of mastication.
(2) Receptors of Mastication
Muscle spindles
In the human, the jaw-closing muscles, not the jaw-opening muscles, contain muscle spindles (Lennartsson, 1979; Kubota and Masegi, 1977). The temporal muscle displayed 342 (208 in the horizontal and 134 in the vertical portion), the masseter 114 (91 in the superficial and 23 in the deep portion), the medial pterygoid 59, and the lateral pterygoid muscle contained 6 muscle spindles. This is unlike the topographic representation of the spindles in animal jaw muscles, where deep portions have been found to contain almost all the spindles (Taylor, 1981). Also, unlike the spindles elsewhere in the body, spindles in the human jaw closers have been found to contain very large numbers of intrafusal fibers per spindle (up to 36). This finding reinforces the idea that the jaw-closer spindles should have a strong proprioceptive impact on the control of human mastication (Eriksson et al., 1994). Spindle discharge usually increases in animals when biting against an experimental load or hard food and hence is correlated with the tension developed by their jaw muscles (Taylor, 1990; Passatore et al., 1996). The cell bodies of the afferents that connect these receptors to the central nervous system have been found to be located in the trigeminal mesencephalic nucleus (Matthews, 1976).
Tendon organs
There is only limited evidence of the existence of tendon organs in human or animal jaw muscles (Matthews, 1975). The functional connections of these afferents, if they exist, are not known.
Temporomandibular joint (TMJ) afferents
Innervation of the human TMJ capsule and its receptor types have been studied (Thilander, 1961; Griffin and Harris, 1975). The major innervation of the joint comes from the auriculotemporal nerve (posterior and lateral portions). The anterior portion of the capsule receives innervation from the masseter nerves (Thilander, 1961). The articulatory surfaces of the joint and the meniscus, except for its peripheral border, are not innervated (Dubner et al., 1978).
The receptor types found in the TMJ capsule include free nerve endings, Ruffini endings, Golgi organs, and Vater-Pacini corpuscles (Thilander, 1961). It has been claimed that the Ruffini endings and the Golgi organ within the capsule function as static mechanoreceptors, the Vater-Pacini endings as dynamic mechanoreceptors, and the free nerve endings as the pain receptors (Storey, 1976). The cell bodies of the afferents that connect these receptors to the central nervous system have been found to be located in the trigeminal ganglion (Lund and Matthews, 1981). Very limited information is available—and only in animals—on the connection of these afferents to masticatory motoneurons.
Skin and mucosal receptors
Human microneurography and psychophysical studies showed that, other than the rapidly adapting receptors with large receptive area (type RAII; Pacinian endings), all other receptors exist in the human facial skin and mucosa (Barlow, 1987; Johansson et al., 1988). This finding is in agreement with the findings in the rabbit (Appenteng et al., 1982). A majority of the mechanoreceptive afferent units in the skin of the human face are slowly adapting and have small and well-defined receptive fields (type SAI). Slowly adapting receptors with large receptive fields (SAII) and rapidly adapting receptors with small well-defined receptive fields (RAI) were also detected in these studies (Johansson et al., 1988).
Periodontal mechanoreceptors (PMRs)
Studies in the cat indicated that the majority of PMRs are located near the apex of the tooth root (Ness, 1954). Paring the alveolar bone overlying the root of the tooth and simultaneous mechanical and electrical stimulation were used to determine the properties of the adequate stimulus that initiated action potentials in these receptors (Linden, 1990). It was found that these receptors respond to tension but not to compression. With direct stimulation of the receptors, it has been found that the receptors with their cell bodies in the trigeminal mesencephalic nucleus are located in the middle of the fulcrum-apex, whereas the receptors whose cell bodies are situated in the trigeminal ganglion are distributed throughout the entire periodontal space. Directional sensitivity experiments have illustrated that the majority (over 70%) of the receptors with cell bodies in the trigeminal mesencephalic nucleus are located in the labial to mesial aspects of the teeth, whereas the receptors with cell bodies in the trigeminal ganglion are distributed more equally in the periodontium (reviewed in Linden, 1990).
Slowly adapting PMRs appeared to be situated in the apical third of the ligament, while the rarer, more rapidly adapting, receptors are situated below but closer to the fulcrum than to the apex. When the force was applied to the tooth, there was a graded response from the apex to the fulcrum. As far as the adaptation is concerned, the most rapidly adapting PMRs are situated near the fulcrum and the most slowly adapting ones near the apex. If a tooth is considered to rotate about a fulcrum when a force is applied to the crown, then various degrees of displacement of the root relative to the surrounding alveolar bone will occur. The displacement is graded, with the largest occurring at the apex and reducing toward the fulcrum for a given displacement of force applied to the crown. If the receptors are stimulated by the displacement of the tooth root, those receptors at the apex would receive the greatest displacement and so the greatest stimulus. Therefore, they would appear to have a lower force threshold and adapt more slowly than those closer to the fulcrum of the tooth. The observation that rapidly adapting responses can be elicited from a receptor situated at the apex by the application of a reduced force to the tooth crown further reinforces this hypothesis. The threshold also decreased closer to the apex. From these studies, it was suggested that there may be only one type of mechanoreceptor and that the rates of adaptation and threshold properties are dependent on the location of the receptor within the periodontal ligament (Cash and Linden, 1982; Linden and Millar, 1988a; reviewed in Linden, 1990).
It has also been shown that the threshold of a particular receptor depended upon the rate of application of the force to the tooth crown (Hannam, 1969; Linden and Millar, 1988b).
In humans, properties of PMRs have been studied by microneurography, and the results generally agreed with animal findings. Studies have shown that some receptors respond to slowly rising forces, while others are activated by rapidly rising components of the force stimulus (Trulsson and Johansson, 1994).
Nerve terminals in the human periodontal space have been investigated by immunohistochemistry (Maeda et al., 1990). This study has shown that the periodontal space contains both free and specialized nerve endings. There are four types of specialized nerve endings: Ruffini-like endings, that are found mainly near the root apex; coiled nerve endings, found near the mid-range of the tooth root; and spindle and expanded nerve endings, both found near the root apex. A similar description of these receptors comes from a study that used extracted teeth from humans (Lambrichts et al., 1992).
PMRs—muscle spindle interaction for calibrating the position of the jaw
Central connections of the PMRs are quite unique in that most of these receptors have their cell bodies in the trigeminal mesencephalic nucleus along with the spindle cell bodies (Linden, 1990). It has been suggested that, in the trigeminal mesencephalic nucleus, an electrical link may exist between the cell bodies of spindles and periodontal receptors (Baker and Llinas, 1971; Taylor et al., 1978).
It is also unique that the periodontal receptors and muscle spindles from jaw muscles have direct projections to the cerebellar cortex (Taylor and Elias, 1984; Donga and Dessem, 1993). It is thought that this direct connection can be used as a reliable signal of tooth contact, and this may be used to zero or recalibrate the spindle afferent discharges. Muscle spindles in the jaw-closing muscles give very finely graded information regarding mandibular movement. However, they cannot give reliable information about jaw position over a long period of time, because the spindle properties and the fusimotor activity change continuously during chewing (reviewed in Taylor, 1990). For the normal mandibular posture to be maintained, absolute positional information is needed, and that requires calibration of the muscle spindle afferent information with the exact time of tooth contact (Taylor and Elias, 1984). This calibration could be done by a comparison of the direct and reliable information received via the spindle and PMR afferents to the cerebellum (Taylor and Elias, 1984; Donga and Dessem, 1993). This comparison may allow the cerebellum to alter fusimotor activity appropriately and regulate the gain of the spindles in the jaw muscles (Prochazka, 1989).
(3) Reflex Work
In reflex work, individual pathways are stimulated, and the changes in the electrical activity of the muscle in response to the stimulus are used to estimate the profile of the synaptic connection between the stimulated afferent pathway and the motoneurons that innervate the muscle. The reflexes that are elicited in the jaw muscles can be classified according to the receptors that initiate these reflexes, such as the periodontal reflexes, spindle reflexes, and so on. These reflexes can also be classified by special nomenclature, such as periodontal-masseteric reflex, jaw-jerk reflex, jaw-opening reflex, etc. We will describe the reflexes in the jaw muscles using the method of stimulation as the discriminating factor. This is important, since it is very difficult to stimulate only one type of afferent and elicit only one reflex response in human subjects.
Stimulating one afferent type without affecting others has been a great challenge for several years in human experiments. Without the precautions stated below, stimuli are not generally reproducible, and hence results may be erroneous.
Mechanical stimulation
Many investigators have reported that the jaw-closing muscles are reflexly inhibited as a result of normal tooth contact during mastication both in animals (Thomas and Peyton, 1983) and in humans (Ahlgren, 1969). This has prompted the use of mechanical stimulation as the means of investigating jaw reflexes. There are several problems, however, with the use of mechanical stimuli in the study of jaw reflexes. First, it is difficult for a mechanical stimulus to be delivered in a reproducible manner (Bishop et al., 1984). Moreover, a mechanical stimulus which is large enough to cause a reflex response may also stimulate several receptor systems with various synaptic connections to the motoneurons. For example, a tap to the skin of the face stimulates the cutaneous receptors in the area of application (Bailey et al., 1979), the vibration-, stretch-, and position-sensitive receptors in and around the jaws (Lund et al., 1983), and also the vibration-sensitive receptors in the inner ear (Meier-Ewert et al., 1974). For this reason, it is difficult for a reflex response elicited by mechanical stimuli to be ascribed to a particular afferent system unless it is done extremely carefully.
Mechanical stimulation requires fine control of the duration, the intensity, and the exact location of the stimulus. Furthermore, the profile of the stimulus shape, including the rate of rise of the stimulus, needs to be set. If the physical relationship between the stimulating probe and the stimulated area changes, the stimulus profile and the rate of rise of the stimulus will also change. It is therefore best for the probe to be physically connected to the area to be stimulated, either by glue, or by fixing of the positions of the jaw and the probe and the application of some pre-load to take up the slack between them. These conditions will minimize the variability in the stimulus and hence make it more likely that reproducible stimulus forces will be generated. These points are illustrated in Fig. 1.
Although the exact profile of the stimulus is partly determined by the stimulating probe/area relationship as mentioned above, it is also affected by the shape of the stimulus profile as determined by the wave-generating computer (or dedicated generators). The stimulus profile may have rapidly rising and slowly rising components. We know from microneurography studies that some receptors in the trigeminal system respond to slowly rising forces, while others are activated by rapidly rising components of the force stimulus (Trulsson and Johansson, 1994). If the stimulus profile contains only slowly rising forces, then it is likely that it will activate only the slow-rate-sensitive receptors. When the stimulus profile has more fast components, the receptors that are activated will also include the fast-rate-sensitive receptors in the system. At least in the PMR system we know that the two receptor systems may generate different synaptic potentials on the masseteric motoneurons. The slow-rate-sensitive receptors seem to have an excitatory connection, while the fast-rate-sensitive receptors induce powerful inhibitory synaptic potentials (Türker et al., 1994, 1997a).
Brief, rapid stretch stimulus
In most investigations, the jaw muscles are stretched by means of a tap to the chin with a tendon hammer in a downward direction. This stimulus delivers a brief rapid stretch to jaw-closing muscles. As well as stimulating stretch-sensitive muscle spindles in jaw muscles, this stretch is also likely to stimulate the TMJ and skin receptors. This stretch stimulus induces a powerful excitatory reflex response in these muscles with monosynaptic latency (about 8 ms; range 6-10 ms; see summary of jaw-jerk latency, duration, and amplitudes in Murray and Klineberg, 1984). This stimulus is also reported to induce an inhibition in the jaw-opening muscles (Matthews, 1975). It is strongly believed that the principal receptor responsible for this reflex is the muscle spindles in the jaw closers, and hence it is suggested that the spindle afferents in the jaw closers have an excitatory connection with the jaw-closing muscles and an inhibitory connection with the antagonist muscles (Matthews, 1975).
It is argued that for this reflex to occur, actual stretch does not need to take place, as long as there is an increase in the force between the jaws (Lamarre and Lund, 1975). However, fixing the jaws at the rest position using acrylic resin, Murray and Klineberg (1984) could induce the jaw-jerk reflex in only one of the five subjects tested, indicating that vibration alone was not an adequate stimulus to evoke a jaw-closing reflex, and that stretch of the muscle was necessary to elicit a jaw-jerk reflex, at least under the relaxed conditions. In another set of experiments, small stretches applied to individual muscles in the form of taps activated the homonymous muscles much more effectively and also affected the heteronomous muscles, suggesting that the projection of muscle spindle afferent fibers to jaw closers in humans is diffuse (Smith et al., 1985).
However, in all of the above experiments, many receptor systems besides muscle spindles must have been stimulated, and hence these experiments may not indicate absolute wiring of muscle spindle afferents in the jaw muscles. To illustrate this point clearly, we delivered rapidly rising ('tap') and rapidly declining ('unload') orthogonal forces to an upper incisor tooth and examined the effects before and during local anesthetic block (Türker and Jenkins, 2000). We have noticed that whereas the tap stimulus induced a small load increase followed by a small unload on the total bite force, the unload stimulus induced only unload response on the bite force. These effects on the total bite force had very short latencies (2-6 ms) and remained during the local anesthetic block, indicating that they were mechanical artifacts of the orthogonal tooth stimulation. Although the length changes induced by the tap and unload stimuli applied to the upper incisor teeth were very small, during the local anesthetic block, they were able to generate a stretch reflex in most of the 10 subjects studied. Local anesthetic block in these experiments ensured the exclusion of the PMRs from taking part in the reflex response and hence isolated the jaw-closer muscle spindles for stimulation. Such a unique technique that does not move the jaw appreciably and yet stimulates the spindles in isolation has not been reported previously.
These experiments illustrated that even using the orthogonal stimulation of an upper incisor under the standardized conditions (as described in Fig. 1), we cannot avoid the stimulation of at least two receptor systems: PMRs and the muscle spindles (Figs. 2, 3 ).
Slow-stretch stimulus
In most studies to date, the stretch stimulus applied to the jaw muscles was in the form of a chin tap. However, it is also possible for the jaw muscles to be stretched in small steps that are controlled in their rate of force delivery (Cooker et al., 1980). Small steps of stretch induced powerful short-latency EMG responses (~ 8 ms latency) and can generate very substantial force response. No long-latency reflex was observed in these earlier experiments on jaw muscles with the slow-stretch stimulus (Lamarre and Lund, 1975; Marsden et al., 1976). However, more recently, a similar study that used controlled slow stretch of the jaw muscles reported the existence of the long-latency reflex, possibly following a transcortical path (Poliakov and Miles, 1994). This recent study also criticized the earlier work, claiming that the stretch stimuli used in the earlier investigations may have induced vibration of the bite bars, which would then have extinguished the long-latency reflex. In most studies where bite bars are used to stretch the jaw muscles, local anesthetic block of the stimulated teeth is used to confirm that both the short-latency and the long-latency reflex responses are spindles of origin. Since these reflexes were unaltered by the local anesthetic blocks, it has been claimed that the receptors responsible for the reflexes elicited by slow stretch of the jaw closers are the muscle spindles in these muscles (Lamarre and Lund, 1975; Poliakov and Miles, 1994). However, these studies did not consider the fact that TMJ receptors may also have played a role. Furthermore, it is very difficult to block the contribution of the PMRs to such stimuli completely, since physical coupling is very common between teeth, and up to 5 teeth on either side of the stimulated tooth have to be anesthetized before the subjective feeling of mechanical stimulation disappears (Türker and Jenkins, 2000).
Muscle spindles are rarely present in jaw-opening muscles. Despite this fact, an excitatory reflex response similar to the stretch reflex but with longer latency (about 24-34 ms latency) has been demonstrated as a result of loading of the voluntarily contracting digastric muscle (Lamarre and Lund, 1975; Hellsing, 1987). Simultaneous inhibition in the antagonists (jaw closers) has also been shown in these studies, indicating a reciprocal organization of the pathways (Hellsing, 1987). In searching for the receptor system responsible for this reflex, Lamarre and Lund (1975) induced a local anesthetic block around the stimulated teeth to exclude the PMRs. The digastric reflex was unaffected by the local anesthetic block; hence, PMRs were not responsible. Since these muscles do not contain muscle spindles (discussed earlier), the receptor origin of this digastric stretch reflex may be the TMJ receptors (Kawamura and Majima, 1964; Lamarre and Lund, 1975).
Unloading of the jaw
The unloading mechanism in the jaws has been experimentally studied by investigators introducing a load between the jaws and suddenly and unpredictably removing the load. In response to such stimuli, unloading reflexes were recorded for both jaw-opener and -closer muscles (Lamarre and Lund, 1975; Miles and Wilkinson, 1982). The most prominent component of the jaw-unloading reflex is a short-latency (10-20 ms) reduction in the surface electromyogram (SEMG) of the masseter muscle. In addition, an increase in the SEMG of the anterior digastric muscle is observed at a slightly longer latency (approximately 25 ms) (Hannam et al., 1968; Lamarre and Lund, 1975; Miles and Wilkinson, 1982). Several receptors are affected by the fracturing of a brittle object between teeth and thus could be responsible for initiating this reflex. These receptors include muscle spindles, TMJ receptors, PMRs, and also acoustic receptors (Duncan et al., 1992). However, there has been little direct investigation into which receptors actually control the reflex.
There have been few attempts to determine whether the PMRs contribute to the unloading reflex. Blocking periodontal input by infiltration of local anesthetics around the roots of the teeth failed to alter the unloading reflex (Lamarre and Lund, 1975; Poliakov and Miles, 1994; Miles et al., 1995). Therefore, it was concluded that PMRs were of minimal importance. Instead, it was put forward that muscle spindles are the principal contributors to the unloading reflex, primarily because the latency of the onset of SEMG reduction is consistent with a monosynaptic pathway, but also because muscle spindles are thought to be responsible for the unloading reflex in the limbs (Angel et al., 1965, 1973; Lamarre and Lund, 1975; Miles and Wilkinson, 1982; Poliakov and Miles, 1994; Miles et al., 1995).
However, the masticatory system is quite unique in that it is possible not only to exclude PMRs by local anesthetics but also to destroy selectively the spindle cell bodies (in animals) that originate from the jaw muscles and to investigate the contributions of spindles to various reflexes and tasks. Goodwin and Luschei (1974) showed that destruction of the spindle cell bodies in the trigeminal mesencephalic nucleus of monkeys did not alter the jaw-unloading reflex. Therefore, reduced facilitation due to the removal of spindle input cannot be fully responsible for the jaw-unloading reflex.
We have recently introduced a new technique for inducing the unloading reflex by the withdrawal of orthogonally applied forces to teeth. This approach does not move the jaws appreciably but induces inhibition in the jaw closers which is totally abolished by the local anesthetic block of the stimulated teeth (Türker and Jenkins, 2000). This study showed that PMRs do contribute to the jaw-unloading reflex (see Figs. 2, 3 ). Details of this study are fully described under the heading "Unloading of a tooth" in this review.
Therefore, as well as the other mechanical and stiffening factors, the unloading reflex may have an important role in preventing further jaw closing from occurring after the initial slowing of the jaw when an object breaks between the teeth. If inhibition in the jaw closers did not occur following unloading, the level of contraction that was present prior to unloading would rapidly be re-established, and the jaw-closing movement would resume.
Unloading of jaw openers elicited a short-latency decrease (~ 20 ms) in EMG, followed by a short silent period and sometimes a short burst of activity in their antagonists (Lamarre and Lund, 1975). This reflex is not thought to be due to periodontal or cutaneous receptors.
Unloading of a tooth
In one of our recent studies, unloading of the tooth was achieved in the orthogonal direction so that movement of the jaws was kept to a minimum. We did this to study the contribution of the PMR system to the unloading reflex in isolation from the activity of other receptors. Rationale for the orthogonal stimulus is as follows: Using the microneurography technique, Trulsson and colleagues (1992) have shown that individual human PMRs can be activated by forces applied to the tooth in almost all directions. Most afferents are activated by static forces in two or three of the four horizontal directions and in one or both of the axial directions. These findings suggest that the location of a single receptive field within the periodontal space may not be important, and/or that each receptor may have a diffuse 3D field (Türker and Jenkins, 2000). Furthermore, since an afferent fiber originating from a single receptive field would induce a defined synaptic potential at the central synapse, synaptic potential (determined from the reflex response) induced by axial and horizontal stimulations can be similar. Since unloading in the axial direction activates many other receptor systems (TMJ receptors, muscle spindles, skin and mucosal receptors) as well as the periodontal mechanoreceptors, and since we wanted to isolate the periodontal component of the jaw-unloading reflex, we preferred to use horizontal unloading. Unloading in the horizontal direction achieved this goal quite well, since local anesthesia of the teeth abolished the inhibitory reflex response in all subjects.
This stimulus resulted in a reflex inhibition in the SEMG of both the ipsilateral and contralateral masseter muscles. Since this inhibitory response was abolished when periodontal input was blocked by local anesthetic, it can be concluded that it is of periodontal origin. Thus, in contrast to previously held views regarding the contributions of the PMRs to the unloading reflex (Lamarre and Lund, 1975; Öwall and Elmqvist, 1975; Duncan et al., 1992; Poliakov and Miles, 1994; Miles et al., 1995), our recent findings indicate that these receptors do in fact contribute to this reflex (Türker and Jenkins, 2000).
Stimulus-rate-related responses in PMRs may explain the masseteric inhibition observed with tooth unloading. First, the reduction in muscle activity may be due to the stimulation of an inhibitory pathway by the sudden unloading ('off') stimulus. A proportion of PMRs has been shown to exhibit an 'off' response, that is, they are stimulated when a force is removed from a tooth (Ness, 1954; Hannam, 1970; Lund et al., 1983; Loescher and Robinson, 1989). Receptors displaying this response most likely are the 'rapidly adapting' receptors that are highly sensitive to changes in the rate of force application (Linden, 1990; Trulsson and Johansson, 1994). Second, the masseteric inhibition may in fact be a reduced facilitation due to the removal of a tonic excitatory input. The results of several studies (Lavigne et al., 1987; Morimoto et al., 1989; Ottenhoff et al., 1992a,b) indicate that, during chewing, PMRs are predominantly responsible for eliciting the additional muscle activity that is required to overcome the resistance encountered between the teeth. If this excitatory pathway is stimulated by pressure on the teeth during biting, then the removal of this pressure, that would occur with tooth unloading, would result in the removal of the excitatory input, and hence a reduction in masseter muscle activity. Thus, the inhibition seen with periodontal unloading could be due to both an activation of the inhibitory pathway and a removal or reduction of the activity in the excitatory pathway (Fig. 4).
The onset latency of the masseteric inhibition seen with use of the orthogonal unload stimulus was approximately 20 ms, which is later than the onset of masseteric inhibition seen in jaw-unloading studies where the unloading was applied in the axial direction (approximately 10 ms; Hannam et al., 1968; Lamarre and Lund, 1975; Miles and Wilkinson, 1982). This is because of the fact that the orthogonal stimuli generate an early excitatory reflex response which delayed the expression of the inhibition (Türker and Jenkins, 2000; see also Figs. 2, 3 ). In addition, natural axial unloading is likely to be faster (~ 10,000 N/s; Nagashima et al., 1997) than the rate of unloading used in the orthogonal study (~ 400 N/s; Türker and Jenkins, 2000).
The sizes of the inhibitory responses seen in the orthogonal tooth-unloading experiments are likely to be an underestimation of the PMR-mediated inhibition that occurs under normal jaw-unloading situations. Fracturing of an object between the jaws would unload several teeth in the upper and lower jaws and stimulate many more receptors than would unloading of a single tooth in the orthogonal direction. Furthermore, the overlap of the early excitatory response would further reduce and delay the impact of the inhibitory response (Figs. 2, 3 ).
Rapid loading of a tooth
It has long been reported that a tap to the teeth induced a series of reflexes in the SEMG of the human jaw-closing muscles. The sign of the earliest reflex response to a tap stimulus has been the subject of much study and controversy. Several groups have reported that the earliest response was an inhibitory reflex with a latency of about 10 ms (Sessle and Schmitt, 1972; Van Steenberghe et al., 1981; van der Glas et al., 1984, 1985, 1988; Dessem et al., 1988; Bjornland et al., 1991; Bonte et al., 1993; Brodin et al., 1993; Sato et al., 1994). In many of the studies, the local anesthetic block of the stimulated tooth abolished the inhibitory response (Sessle and Schmitt, 1972; van der Glas et al., 1985; Brodin et al., 1993; Sato et al., 1994), indicating that the inhibition originated from the PMRs and the involvement of other receptors, such as the vibration-sensitive spindles, was rejected (van der Glas et al., 1985; Brodin et al., 1993; Sato et al., 1994).
Other groups, however, reported that the tap stimulus induced an early excitatory reflex which preceded the inhibitory reflex response (Hannam et al., 1970; Goldberg, 1971; Sessle and Schmitt, 1972; Orchardson and Sime, 1981). This reflex was termed the periodontal-masseteric reflex (Goldberg, 1971). Comparing the jaw-jerk reflex with the periodontal-masseteric reflex, Goldberg noted that the average jaw-jerk latency was 10 ms but the earliest response occurred with a latency of 8.8 ms. The periodontal-masseteric reflex, on the other hand, occurred at a shorter latency of 7-7.5 ms.
Even more confusing, in some studies this early excitatory response continued to be observed after the local anesthetic block of the stimulated tooth (Carels and Van Steenberghe, 1985, 1986). It is possible that the response originates from the periodontal space, since it disappeared in some experiments after the local anesthesia. It is also possible that this early excitatory reflex, or so-called "periodontal-masseteric reflex" (Goldberg, 1971), may originate from the PMRs that excite spindle cell bodies in the trigeminal mesencephalic nucleus, possibly by electric coupling (Baker and Llinas, 1971; Goldberg, 1971). Furthermore, it is likely that this reflex may originate from vibration-sensitive muscle spindles in the jaw closers, though the latency of this reflex appears shorter than the spindle-mediated jaw-jerk reflex (Carels and Van Steenberghe, 1985).
In another group of studies, the early response, "the P wave", was induced only in a minority of the subjects or in some of the trials (Carels and Van Steenberghe, 1985; Sato et al., 1994). In yet another group of studies, the early excitation appeared when the bite strategy was changed from maximal occlusion to edge-to-edge position (Carels and Van Steenberghe, 1986). However, we believe that it is not possible to indicate the receptor responsible for the early excitatory reflex until it is elicited reliably and the rules for eliciting it are studied carefully.
Looking at this problem in a more focused manner, Widmer and Lund (1989) illustrated that the inhibitory reflexes can "cause" "excitatory-like" artifacts in rectified averaged records. Although they used electrical stimuli to induce these reflex responses, nevertheless their findings were sufficient to cast doubt on the 'excitatory' reflexes that preceded inhibitory reflex events. Therefore, rapid loading of a tooth may generate only one reflex response, i.e., inhibition, and the 'periodontal-masseteric reflex' may be an artifact of the averaging process.
However, studies in animals clearly illustrated the existence of the neural structures for the early excitatory reflex. In decerebrate cats, Dessem (1995) has shown that tooth displacement induces short-latency depolarizations in spindle cell bodies in the mesencephalic nucleus of the trigeminal nerve and in the motoneurons of the jaw elevator muscles. Similar short-latency excitatory reflexes in the masseter have been observed following intra-oral stimulation in rats (Funakoshi and Amano, 1974). In the decerebrate cat, tooth tap stimulation activated the masseter motoneurons (both α and γ) with a latency of about 6 ms (Sessle, 1977). Therefore, the pathway for the early excitatory reflex that is induced by a tooth tap stimulus seems to exist, at least in experimental animals. We have recently studied this problem using taps and unload stimuli (Figs. 2, 3 ). We found that the unloading stimulus applied to an upper incisor tooth induces a small force reduction which may become a stimulus to the spindles in the jaw elevators. This small 'stretch' stimulus induced a significant jaw-jerk reflex in six out of 10 subjects (larger than the background variation in the surface EMG) during the local anesthetic block of the stimulated tooth (Türker and Jenkins, 2000; Figs. 2, 3 ).
Other than the early excitatory stimulus which becomes more obvious when local anesthetic block is applied to the stimulated tooth, the tooth tap stimulus induced a prominent inhibition of the jaw-closing muscles at a latency of 12 ms, presumably by stimulating the high-rate-sensitive, rapidly adapting receptors (Sessle and Schmitt, 1972; van der Glas et al., 1985; Brodin et al., 1993; Louca et al., 1996). The latency for tap-induced inhibition was shorter than the inhibition in the unload experiments. This is probably because the early excitatory reflex arrives late in the tooth tap experiments and hence does not affect the latency of the inhibitory reflex response as much as in the tooth unload experiments. In fact, the early excitatory reflex in the tooth tap experiments became apparent only after the local anesthetic block of the inhibitory reflex response (Figs. 2, 3 ).
Animal work has shown an early excitatory response in the digastric muscle in response to force applied (about 4 N) to the canine tooth after a mean latency of about 6 ms (Hannam and Matthews, 1969; Dessem et al., 1988). There is evidence that the receptors concerned are not the low-threshold PMRs that cause inhibition of the jaw-closing muscles in the cat (Dessem et al., 1988). Unlike animals, there is no evidence of activation of the digastric in humans (Goldberg, 1976).
Slow loading of a tooth
The studies that used rapidly loading stimuli suggested that the reflex responses from these receptors are principally inhibitory (Sessle and Schmitt, 1972; van der Glas et al., 1985; Dessem et al., 1988; Bonte et al., 1993; Louca et al., 1996). In contrast, other researchers have demonstrated evidence for an excitatory connection between the PMRs and the motoneurons that innervate the jaw-closing muscles (Lund and Lamarre, 1973; Amano and Yoneda, 1980; Funakoshi, 1981; Lavigne et al., 1987). Recently, we have stressed the importance of the rate of rise of the stimulus force in eliciting excitatory or inhibitory responses from the masseter. The slowly rising stimulus induced mainly an excitatory reflex, while the rapidly rising stimulus usually induced inhibition (Brodin et al., 1993; Türker et al., 1994). There were also other variables that affected the success of the stimulus in inducing a certain type of reflex response, such as the presence of a pre-load and the exact stimulus force profile (Türker et al., 1997a).
A slowly rising mechanical stimulus is likely to stimulate only the PMRs, since the reflex response to a push stimulus of up to 3 N disappeared when local anesthetic was infiltrated around the stimulated tooth (Brodin et al., 1993).
Reflex work where slowly rising mechanical stimuli were applied to the incisor teeth in the orthogonal direction suggested that, at low levels of background activity, the stimulus generates a net closing force, whereas at high levels of background activity, it induces a net reduction of the closing force (Yang and Türker, 1999). This may be due to the differential activation of motor units by PMRs, where small motor units receive excitatory input and large motor units receive inhibitory input (Yamamura and Shimada, 1992). Therefore, at low bite-force levels, reflex activation of small-sized motor units would be relatively intense, which will help keep the food between the teeth (Trulsson and Johansson, 1995). However, when the background bite force is already high, the same periodontal input may inhibit the larger motor units and hence limit further increase in bite force which would have damaged the teeth and supporting structures.
Acoustic stimulation
During mastication, several receptor types—such as the spindles, periodontal mechanoreceptors (PMRs), and temporomandibular joint (TMJ) receptors—are activated. Furthermore, since mechanical stimulation from the food on teeth evokes bone-conducted vibrations in the head, acoustic receptors are also stimulated (van der Glas et al., 1988). To study the influence of acoustic receptors on jaw muscle activity, van der Glas and colleagues (1988) delivered tap stimuli to an upper central incisor tooth and induced well-known inhibitory and excitatory influences in masseter muscles in response to such stimuli (Van Steenberghe, 1979; van der Glas et al., 1984, 1985). In these studies, when local anesthetic block was applied around the stimulated tooth, the reflex response was reduced by about 89%, indicating the level of contribution by the PMRs to the reflex response. In another set of studies, when the sound of the tooth tap was suppressed by white noise applied to the ears by head phones, the reflex response was found to be reduced by about 33% (Van Steenberghe et al., 1981). Therefore, it was claimed that the influences of these receptor systems are not additive but rather interactive, since the sum of the PMR and acoustic contributions is larger than 100% (van der Glas et al., 1985, 1988).
However, the influence of the acoustic receptors may have been overestimated in these studies, since it was assumed that no receptor systems were stimulated with this type of tooth stimulus other than the PMRs and the acoustic receptors. Yet, we have recently shown that even the tiniest taps or unloads applied to the upper incisor teeth can cause a mechanical artifact which induces a muscle spindle response (Figs. 2, 3 ). This response became clearer during the local anesthetic block of the stimulated tooth. It is therefore tempting to conclude that no matter how carefully teeth are stimulated, three types of receptor systems will always be stimulated: PMRs, muscle spindles, and the acoustic receptors. Researchers should take this into consideration when investigating human reflexes using mechanical tooth stimulation.
Electrical stimulation
Although it is not a natural stimulus such as the pressure on the teeth or stretch of the muscles that occurs normally during mastication, electrical stimulation offers the following advantages over mechanical and acoustic stimulation in the study of jaw reflexes: The stimulus parameters (intensity, duration, and frequency) can be readily set and kept constant. The electrical stimuli do not cause vibration and hence do not stimulate the vibration-sensitive receptors in muscles and in the inner ear. The electrical stimuli do not change the jaw position (as mechanical stimulation does) and hence do not activate the position-sensitive receptors that are situated in the jaw muscles and in the TMJ.
Mucosa and skin stimulation
Electrical stimuli can be applied with ease to the mucosa and to the skin for study of the synaptic connection of these afferents to motoneurons. Usually, the sensory perception threshold (T) of the subject is used to standardize the stimulus intensity rather than the actual voltage or the current delivered (Türker, 1988). Intensities of 1-3 T are used to stimulate low-threshold afferents which convey information about touch sensation (Türker and Miles, 1985; Brodin and Türker, 1994). When the stimulus intensity is increased, however, as well as stimulating the low-threshold afferent fibers, the stimulus current begins to activate the higher-threshold afferents that convey information about squeeze, pin prick, and pain to the central nervous system. Yemm (1972a,b) observed that mildly noxious electrical stimulation of the oral mucous membrane inhibits the activity of jaw closers. Using this approach, several investigators have studied mucosa- and skin-induced jaw reflexes both in neurologically normal individuals (Godaux and Desmedt, 1975a) and in dysfunctional patients (Hussein and McCall, 1983).
However, the problem of stimulating only one type of afferent fiber has always limited the usage of electrical stimulation techniques. Methods such as pressure application and local anesthetics can be used to reduce the number of afferents stimulated; however, such experiments are difficult to perform, and the results are hard to interpret (Godaux and Desmedt, 1975b).
Stimulation of the peri-oral skin or mucosa in humans can result in excitation or inhibition of the jaw closers, depending on the stimulus intensity. Stimuli that were just above the perception threshold induced excitation, and stronger stimuli induced inhibition (Brodin and Türker, 1994). The inhibitory stimulus does not have to be painful, since the range of subject responses to such 'adequate' stimuli varied from 'slightly painful' to 'painless' (reviewed in Lund et al., 1983). These adequate stimuli do not, however, initiate activation of the digastric in the human (Yemm, 1972a,b).
In the animal, strong stimuli to the mucosa or the skin initiate the jaw-opening reflex, which involves a reduction in the ongoing activity of the jaw closers and an increase in the activity of the jaw openers. Stimulation with light mechanical or electrical means, however, induces a jaw-closing reflex with an increase in the jaw-closer activity (Thexton, 1974, 1975).
Nerve stimulation
Direct electrical stimulation of nerves is very difficult in the jaws region, since the nerves are located deep in the face and close to numerous blood vessels (Scutter et al., 1997). For example, insertion of one or two needles to a depth of about 2 cm has been used to stimulate the masseteric nerve (Godaux and Desmedt, 1975b; Macaluso and De Laat, 1995) in an area in the vicinity of the pterygoid plexus. In addition, in these experiments, distortion of the needle electrodes during movements of the jaw is a distinct possibility. Once the electrodes are positioned, fine lateral movements in relation to the masseteric nerve are not possible, which may necessitate repeated insertions for satisfactory results to be achieved.
We have recently developed a stimulating technique which is non-invasive, flexible, and effective for producing H-reflexes in the masseter (Scutter et al., 1997; Scutter and Türker, 1999). This 'transmuscular' stimulation technique incorporates an anode and a cathode into a U-shaped stainless steel frame. The frame is held in position by a bite plate constructed of dental impression material which is moulded to the shape of the subject's teeth and attached to a flat stainless steel plate near the anode. The cathode can be finely adjusted to locate the best stimulating position. The bite plate maintains the bite position. When the jaw-closing muscles are relaxed, the bite plate rests comfortably between the teeth. Although this stimulation technique is non-specific to the masseteric nerve alone, it is preferred, since it is non-invasive and simpler to apply.
(4) Simulated Chewing
The information regarding the role of peripheral receptors within the feedback system has been confusing in many instances. For example, although most of the PMRs reach their saturation point (maximum discharge rate) at low force levels (Trulsson and Johansson, 1995), and stimulation of the teeth usually initiates inhibition (Louca et al., 1996), the feedback from these receptors during chewing makes a large contribution to the closing force (Morimoto et al., 1989; Ottenhoff et al., 1992a,b). How can this work?
It is possible that the effectiveness of the synaptic input from the primary afferents on jaw-muscle motoneurons may change during mastication, when many receptors are stimulated in concert. There is a large body of animal evidence suggesting that such a modulation of synaptic inputs is a rule rather than an exception in the masticatory system (Lund, 1991). For example, it has been suggested that the inhibitory circuits from oral mechanoreceptors to jaw closers and the excitatory pathway to the jaw openers are shut down during mastication, allowing for vigorous elevator muscle activity (Appenteng et al., 1982). However, in human subjects, such direct experiments cannot be done. Instead, a clever approach of 'simulated chewing' experiments has been devised by a Dutch group (Ottenhoff et al., 1992a,b; van der Bilt et al., 1995, 1997). These experiments are now reporting new findings on the feedback control of mastication.
The simulated chewing experiments are complex in the set-up. In these experiments, the subject is asked to 'chew' (open and close the jaws) regularly once per second on freely moving bite bars. While the subject chews, a resistance is suddenly introduced when the bite bars are stiffened ('force on' experiments that simulated food insertion into the mouth; Ottenhoff et al., 1992a,b). In the very first cycle with resistance, only a late excitatory response to this sudden introduction of force was observed. The latency of this response was very close to the reaction time of the subject (129 ms and 150 ms, respectively). In the next cycles, however, two additional responses were observed: an anticipatory response and a short-latency reflex response. The anticipatory response started about 70 ms before the onset of force, and the short-latency reflex response started about 23 ms after the onset of force. In these cycles, the long-latency excitatory response—which was the only response in the first cycle—disappeared.
These 'force on' experiments suggested that the reflex mechanism does not play an important role in the very first cycle and comes to be used only from the second cycle on. This may mean that the trigeminal system does not allow for reflex increase in muscle activity to occur without the subject's conscious realization regarding the meaning of the resistance and whether to allow high forces to be used in the next cycle to overcome the resistance or simply to stop chewing and spit the resistance-generating matter (often a small stone in the food) out.
In these simulated chewing experiments, it was also possible to withdraw the resistance between the jaws and observe the effect of a sudden yield of resistance ('force off' experiments; Ottenhoff et al., 1992a,b). In these experiments, the anticipatory response still occurred at the latency of about 70 ms before the onset of the force removal. However, most of the additional muscle activity disappeared about 27 ms after the removal of the force. These 'force off' experiments suggested that the additional muscle activity that is generated to overcome the resistance between the jaws is under reflex control, with a latency of about 25 ms (Ottenhoff et al., 1992a,b).
The insertion of force in these experiments was done by means of magnetic fields that affected all teeth simultaneously. This is of course unlike a real-life situation, where normal mastication of food occurs unilaterally and involves only a small number of teeth. Therefore, the reader is reminded about the limitations of this study and warned regarding the interpretation of these results.
From the available data, one can speculate that both the reflex mechanism and the control of the synaptic efficacy of primary afferent inputs by the central nervous system may help protect teeth from damaging forces. They may, at the same time, allow large chewing forces to develop, as long as the central nervous system is aware of the resistance between the teeth. This system would work quite well, in that, each time a large force is required to overcome an unexpected resistance, the force increase would involve the higher centers which may control its damaging effects (Ottenhoff et al., 1992a,b; Yang and Türker, 1999). Conscious interference in bite force can also increase the flow of information from the receptors to the cortex, which is reduced during normal chewing (Olsson et al., 1986; Lund, 1991), giving the cortex precise information about the resistance-inducing matter.
The receptors responsible for the reflex portion of the additional muscle activity can be the PMRs and/or the muscle spindles. The involvement of muscle spindles was rejected by the Dutch group (Ottenhoff et al., 1992a,b), since the reflex latency was too long (23 ms) for the monosynaptic muscle spindle reflex (usually around 6-10 ms). Furthermore, involvement of the muscle spindle long-latency reflex response was rejected, since no such reflex was shown at the time of that study. However, since then, the existence of a spindle-mediated long-latency reflex in the jaw closers has been shown (Poliakov and Miles, 1994; Miles et al., 1995). Therefore, although the PMRs were claimed to be the receptor responsible for the reflex responses induced during the 'force on' experiments, it is also possible that the muscle spindle system may contribute to it appreciably.
Recent studies using the same simulated chewing methodology also illustrate that the stretch reflex sensitivity is modulated during various phases of rhythmic open-close movement (van der Bilt et al., 1997). Zero or a very weak stretch reflex could be induced during jaw opening, whereas strong reflexes could be elicited during jaw closing. The importance of these modulations on the normal function of jaw movements is discussed in the section under "How do we open our jaws?".
(5) Recording and Analyzing the Results
Since it is not possible to record directly from human motoneurons, indirect methods have been developed to record the activity of muscles and muscle fibers that are innervated by the motoneurons. Surface electromyogram (SEMG) is the simplest form of recording the effect of a stimulus on the motoneuron pool of a muscle. Intramuscular EMG that involves the insertion of thin needles or wires into the muscle can bring out the action potentials of several muscle fibers. The changes in the activity of these action potentials can reflect the sign of the synaptic potential that develops as a result of stimulus delivered. Finally, single-motor-unit action potentials (MUAPs) can be recorded by means of tip-active wire or needle electrodes that give the activity of only one motoneuron. This is of the utmost importance, since the main aim of the reflex studies is to investigate the synaptic potential that develops in one motoneuron to find the distribution of the afferent inputs to cells with different input impedances. These methods will now be examined in some detail.
Surface electromyogram (SEMG)
SEMG is easy to use and provides thought-provoking and exciting results. Unfortunately, single traces of SEMG are not reliable for reflex measurements. Instead, several manipulations have been suggested to convert SEMG records into quantifiable forms. The most common technique is full-wave rectification and averaging of the SEMG around the time of stimulation. This approach has the capacity to illustrate the direction and the strength of the reflex response (Goldberg, 1971). Several manipulations have been performed to convert the rectified averaged SEMG record for this purpose. For example, Goldberg (1971) used a planimeter to measure the area of the rectified averaged SEMG record; van der Glas and colleagues (1984) used the sum of the absolute surfaces outside the confidence interval (total normalized surface, TNS); Evans and colleagues (Evans et al., 1989) used the percentage modulation of the mean SEMG level as the calibration bar; and Cadden and Newton (1994) calculated the area to indicate the percentage change. Quantification of the synaptic potentials from the SEMG data can be criticized for two main reasons:
The synchronization type error
This error is due to phase-advancing or delaying of single MUAPs in bulk that underlie SEMG. An excitatory post-synaptic potential (EPSP) causes a phase advancement of the occurrence of the MUAPs. The phase-advanced MUAPs induce a peak in the SEMG record at the reflex latency. This peak can then induce several peaks and troughs separated from each other by about one interspike interval, as these MUAPs fire again and again in synchrony (Türker and Cheng, 1994). Therefore, a decrease in the SEMG level, following an excitatory reflex, may not always indicate an inhibitory synaptic connection but may be caused by the phase advancement of the MUAPs (Türker et al., 1997b).
Also, the synchronous activation of the phase-delayed MUAPs by an inhibitory post-synaptic potential (IPSP) induces a peak in the SEMG. Hence, an increase in the SEMG level, following an inhibitory reflex, may not always indicate an excitatory synaptic connection but may be caused by the synchronous re-occurrence of the MUAPs (Miles and Türker, 1987; Miles et al., 1987).
The count-related error
The decreased activity in the SEMG immediately after an excitatory reflex may be due to the phase-advanced spikes rather than to a genuine inhibitory reflex (Türker et al., 1997b). During this time of relative 'silence', many of the MUAPs cannot fire, since the EPSP caused them to fire only a few milliseconds earlier.
For the above reasons, it is therefore possible to consider only the very first response in the SEMG as the true representative of the post-synaptic potential in the motoneuron. The peaks and troughs that follow the very first response in the averaged SEMG records should therefore be interpreted with extreme caution. These problems are highlighted in Figs. 5-8 by means of slice preparations and single MUAP analyses.
Intramuscular EMG
The use of intramuscular electromyography (IM-EMG) for reflex studies is preferred if the muscle of interest is small, situated deep in the limb, or when defined parts of the muscle are to be studied. The major advantage of IM-EMG is that it is specific to the area of recording and is not subject to as much artifact (such as cross-talk, stimulus, and movement artifact) as the SEMG (reviewed in Türker, 1993). However, intramuscular recording is subject to the same synchronization and count-related errors as the SEMG, since both of these analyses depend upon the probability of spike occurrences. Therefore, again, other than the very first response in the averaged record, one cannot take the late responses to represent the stimulus-evoked synaptic potential in the motoneuron pool.
Single-motor-unit action potentials (single MUAPs)
Recording single MUAPs from muscles has the distinct advantage of allowing for the observation of individual motoneuron output, since there is a one-to-one relationship between the two events. Furthermore, it is virtually artifact-free, since the action potentials are all-or-nothing events, and hence records are very reliable. Since the ultimate aim of reflex experiments in humans is to estimate the synaptic events in the motoneurons themselves, single-MUAP techniques come closest to allowing for the estimation of synaptic events in motoneurons, since they directly represent the output of individual motoneurons.
Although the recording of single-motor-unit activity from human muscles is an easy process, the quantification of reflex responses has always been a challenge and has led to the development of numerous techniques (Garnett and Stephens, 1980; Awiszus et al., 1991; Türker et al., 1994). To quantify the reflex responses, most investigators have used the firing probability of the single motor unit following a stimulus (Garnett and Stephens, 1980; Kudina, 1980). A peristimulus time histogram (PSTH) is constructed from the firing of one motor unit over many trials, for assessment of the effect of the stimulus on the firing probability. In some other studies, in addition to the PSTH, the interspike interval of the firing of the motor units, in the form of raster dots, has been used for the same purpose (Kudina, 1980; Türker and Miles, 1989; Türker et al., 1989).
In general, the counts in the PSTH and the changes in the duration of the ISI have been used in a large number of calculating algorithms in an attempt to put some value on the synaptic potential developed in the motoneuron. In the PSTH approach, an excitation of a motor unit as a result of a stimulation elicits a peak, whereas an inhibition induces a trough in the PSTH. Just like the limitations of the surface EMG and the IM-EMG mentioned earlier in the PSTH approach, associating peaks with excitation and troughs with inhibition is not always correct, since the counts in the PSTH are also contaminated by the synchronization and count-related errors.
For example, a strong excitatory stimulus like an H-reflex shortens the ISIs (for the definition of excitation, see Kudina, 1980) of the firing of the motor unit. However, because of the large numbers of counts that occur in a very short time period at the reflex latency, the firing of the motor unit is now 'synchronized' to the stimulus. As in the case of the SEMG and IM-EMG, this synchronized firing will induce a series of peaks and troughs in the PSTH, and their size will gradually diminish (Türker and Cheng, 1994). Therefore, it has been proposed that: "...besides neuronal excitation as the 'usual' reason for peak, a PSTH peak can also be due to another PSTH peak occurring before" (Awiszus et al., 1991). Fig. 6 illustrates this point on a soleus motor unit.
A strong inhibitory stimulus can also induce a synchronizing effect on the firing of motor units by lengthening the interspike intervals (for the definition of inhibition, see Kudina, 1980) and by setting the action potentials to occur at a certain time after the stimulus. The timing of occurrence of the action potentials after the IPSP is fixed in relation to the stimulus and thus induces several peaks and troughs in the PSTH. Like excitatory reflexes, these peaks and troughs in the PSTH may not represent a series of excitatory and inhibitory synaptic potentials, since they could have been caused by the synchronous firing pattern of the unit that is set by the inhibitory stimulus (Miles and Türker, 1987; Türker and Cheng, 1994).
Since all techniques used for the estimation of synaptic potentials are indirect, the serious limitations and contaminations they present should always be taken into account when the significance of such findings is assessed. There is an important factor that has to be controlled before such indirect techniques can be made useful and comparisons of such results can be made.
Elimination of contaminations from the analyses
We have recently claimed that instantaneous frequency plots (peristimulus frequencygram, PSF) from single MUAPs may indicate the time course of the synaptic potential to a motoneuron more accurately than the PSTH (Türker and Cheng, 1994). We reasoned that the PSF record examines each of the discharges of the motoneuron individually, without adding the previously occurring event, and hence does not contain the synchronization and the number-related errors that are associated with all probability-based analysis methods, such as SEMG, IM-EMG, and PSTH.
Since the discharge frequency of a motoneuron reflects the net current reaching the soma (Kernell, 1965; Redman, 1976; Schwindt and Calvin, 1973a,b; Baldissera et al., 1982; Powers et al., 1992), any significant change in the post-stimulus discharge frequency should indicate the sign and the profile of the net input (Türker and Cheng, 1994; Türker et al., 1997b). Furthermore, since discharge frequency values are individual events and plotted in the PSF as independent and single values (without being altered by the previous activity at that particular time), they should avoid any number- or synchronization-related errors (Figs. 5, 6 ).
However, PSF records had never been directly compared with the characteristics of synaptic potentials measured in the same cells. Therefore, to test the hypothesis that the PSF is a better representation of the underlying synaptic potential than the PSTH, we elicited repetitive discharge in hypoglossal motoneurons recorded in slices of rat brainstem, using a combination of a suprathreshold injected current step with superimposed noise to mimic the synaptic drive likely to occur during physiological activation. The effects of repetitive en masse stimulation of afferent nerves were simulated by the further addition of trains of injected current transients of various shapes and sizes. The effects of a given current transient on motoneuron discharge timing and discharge rate were measured by the calculation of a PSTH and a PSF. We calculated the amplitude and time course of the simulated post-synaptic potentials (PSPs) produced by the current transients by convolving the current transient with an estimate of the passive impulse response of the motoneuron. We then compared the shape of the injected current transient and the simulated PSP with the profiles of the PSTH and PSF records.
The PSTHs produced by excitatory PSPs (EPSPs) were characterized by a large, short-latency increase in firing probability that lasted slightly longer than the rising phase of the EPSP, followed by a reduced discharge probability during the falling phase of the EPSP (Fig. 5). In contrast, the PSF analysis revealed a proportionate increase in discharge rate over the entire profile of the EPSP, even though relatively few spikes occurred during the falling phase. The PSTHs associated with IPSPs indicated a reduction in discharge probability during the initial, hyperpolarizing phase of the IPSP, followed by an increase in the discharge probability during its subsequent repolarizing phase. Based on the PSF analysis, the initial phase of the IPSP appeared as a large gap in the record where very few or no discharges occurred. The subsequent phase of the IPSP was associated with frequency values that were lower than the background values (Figs. 7, 8 ). By either the EPSP or IPSP, the PSTHs contained secondary peaks and troughs that were not directly related to the underlying PSP, but instead reflected the regular recurrence of spikes following those affected by the PSPs (the source of synchronization- and number-related errors). The PSF analysis is more useful for indicating the total duration and the profile of the underlying PSPs, since the discharge frequency of the spikes which followed the PSPs very closely did not contain synchronization- and count-related errors.
Figs. 6 and 8 show that the properties of the reflex response of a single motor unit depend upon the technique used for data analysis. For example, in Fig. 6, while the classic PSTH technique illustrated three separate reflex responses on the bases of significant changes in the number of counts following the stimulus, the PSF technique illustrated only one reflex response: a single long-lasting excitatory reflex response. An examination of Figs. 5 and 7 clearly shows that the PSTH is not reliable for secondary and tertiary 'reflexes', and hence the PSF record must be used if we suspect any long-latency events.
We have demonstrated that the PSF analyses illustrate an estimate of the net changes in the membrane potential of a motoneuron and hence can indicate the sign and shape of the post-synaptic potential developed by the stimulus. This is of the utmost importance for investigators attempting to hypothesize the connections and strength of connections in the central nervous system. Without this knowledge, the postulated connections may prove groundless. We therefore suggest that in addition to the classic SEMG or IM-EMG or PSTH techniques, researchers should seriously consider the usage of PSF in their investigation of synaptic input to human or animal motoneurons.
(6) Questions for Problem-based-learning Exercises
(a ) If stretch of jaw muscles induces a powerful stretch reflex, how do I open my mouth without inducing this reflex ?
It is well-established that the inputs from the primary afferents to motoneurons are modulated by pre- and post-synaptic influences (Rudomin, 1990). The synaptic effects of the primary afferents from the jaw region as well are subject to such influences (reviewed in Lund, 1991). Lip and mucosal mechanoreceptors, muscle spindle afferents, and PMRs become active immediately and synchronously as soon as the teeth encounter the bolus and continue to be active throughout the slow-closure ('power stroke') phase of chewing (Lund and Olsson, 1983). The consecutive activation of these receptor systems may allow for interaction. For example, it has been shown that cutaneous stimulation exerts pre-synaptic inhibition on the spindle inputs to motoneurons in the trigeminal system of the decerebrate cat (Goldberg and Nakamura, 1977). Furthermore, it has also been established that the central pattern generator can generate de- or hyperpolarization in the motoneurons that innervate jaw-closing muscles during closing and opening (respectively) (Goldberg et al., 1982; reviewed in Lund, 1991). In the conscious cat, masseteric H-reflex was facilitated during jaw closing and decreased during jaw opening (Chase and McGinty, 1970).
Therefore, animal work shows that the stretch of spindles during opening has little effect on the excitation of jaw-closer motoneurons, since the jaw-closer motoneurons are hyperpolarized by the activity of the CPG, and that the gammas are relatively inactive during jaw opening (Lund et al., 1979). On the other hand, during closing, feedback from muscle spindles provides servo-assistance to the motoneurons that are already excited by the activation of the CPG (Lund, 1991). This servo-assistance occurs by the increase in the activity of the gamma system during the closing (biting) phase. Gamma activity during biting prevents spindle unloading, though it cannot prevent unloading during faster movements (Goodwin and Luschei, 1975; Lund et al., 1979; reviewed in Taylor, 1990).
In the human, recent studies illustrated that stretch reflex sensitivity is modulated during various phases of rhythmic open-close movements (van der Bilt et al., 1997). Zero or a very weak stretch reflex was induced during jaw opening, whereas strong stretch reflexes could be elicited during jaw closing. This modulation is probably due to three main reasons. First, similar to the actively biting monkey (Lund et al., 1979), the gamma activity may decrease during opening. Second, the excitability of the motoneurons can affect the response to the stretch stimulus, since it is known that, during opening, a very small amount of activity remains in the jaw-closing muscles (Yemm, 1976). This reduction in the activity of the jaw-closing muscles during opening can be partly due to the CPG that hyperpolarizes the jaw-closer motoneurons during opening and partly due to post-synaptic effects of the proprioceptors, including TMJ receptors, that monitor jaw movement. And finally, the reflex amplitude may be influenced by the pre-motoneuronal adjustment of the spindle input to motoneurons (van der Bilt et al., 1997). This pre-motoneuronal adjustment may involve all proprioceptors in the jaw region that are activated by the jaw movement. This could allow for modulation of the reflex responses elicited by muscle stretch so that jaw opening can be done effortlessly.
(b ) Why does mastication in edentulous subjects become less efficient, and why do the masticatory forces in these individuals (even with the best-fitting dentures ) immediately fall to about 20-40% of the dentate value ?
It is probably because the jaw muscles in these individuals get less “positive feedback” from the PMRs. Although in most animal and human studies, stimulation of teeth has been shown to induce inhibition in the jaw-closing muscles, there are some reports indicating that excitation can also be induced by mechanical tooth stimulation (Sessle and Schmitt, 1972; Lund and Lamarre, 1973; Amano and Yoneda, 1980; Funakoshi, 1981; van der Glas et al., 1985; Lavigne et al., 1987; Dessem et al., 1988; Bonte et al., 1993; Louca et al., 1996). Recently, we have stressed the importance of the rate of rise of the stimulus force in eliciting excitatory or inhibitory responses from the masseter. The slowly rising stimulus mainly induced an excitatory reflex, while the rapidly rising stimulus usually induced inhibition (Brodin et al., 1993; Yamamura et al., 1993; Türker et al., 1994).
Therefore, it is possible that when one bites on food, the PMRs can help develop excitation on the jaw-closer motoneurons as long as the bite force and the resistance increase slowly between the teeth. If the resistance between the teeth increases suddenly and unexpectedly, the periodontal receptors can then inhibit the activity of the jaw-closer motoneurons and hence reduce bite force (Türker et al., 1994, 1997a). If, however, the resistance between the teeth suddenly yields, the periodontal input can stop sending further excitation to jaw-closer motoneurons, and hence jaw movement slows down and stops (Türker and Jenkins, 2000).
Extraction of teeth destroys the PMRs and hence they can no longer provide the excitatory influence to the jaw-closer motoneurons (Linden and Scott, 1989). Importance of the PMRs in the level of activation of the motoneurons can be assessed by determination of the maximal bite force before and during local anesthetic block of the teeth (Lund and Lamarre, 1973; Orchardson and MacFarland, 1980). Such experiments in which contributions of the PMRs are temporarily blocked showed that the maximal bite force decreased drastically unless the subject was reassured about his/her bite force performance. Therefore, PMRs not only provide extra excitation to the jaw-closer motoneurons but also convey information to the central nervous system that the bite action is not damaging the teeth and support structures. Furthermore, since the PMRs do contribute to the stopping mechanism of the jaws in the event of a sudden yield of resistance, their absence would weaken the braking mechanism of the jaws.
Therefore, in the absence of these important contributions from the PMRs, the central nervous system may automatically reduce the level of achievable bite force to avoid damaging forces on teeth and their supporting structures. When we provide the feedback in the form of vision or suggestion, the original maximum bite force level can then be achieved. So, why can we not increase the level of excitation of motoneurons consciously when periodontal input is abolished in the edentulous situation? This may be due to the fact that we need this feedback/reassurance if we are to bite harder. In the absence of this valuable feedback, the central nervous system may reduce the maximal bite force to such a level that it no longer needs this feedback/reassurance for biting tasks and avoids developing damaging forces on the teeth should an object break between the jaws unexpectedly.
(c ) What stops my jaws from forcefully coming together when an object suddenly yields ?
Unloading of the jaws and teeth occurs when a brittle object is fractured between the teeth. In this event, the teeth do not crash together, despite the fact that high bite forces may be developed before the object breaks. There are several mechanical and neurological factors that are thought to prevent the teeth from crashing together. The first involves the inherent mechanical properties of the jaw musculature, such as elasticity (Yemm, 1976) and the length-tension (Mackenna and Türker, 1978) or velocity-tension relationships (Van Willigen et al., 1997) of the jaw muscles and tendons that can help reduce forces as the jaws are brought together. Second, there is an active stiffening of the openers during a biting task, especially when the resistance is expected to yield (Miles and Wilkinson, 1982; Miles and Madigan, 1983). A third mechanism is a series of neurological events termed the 'unloading reflex' (Lamarre and Lund, 1975; Miles and Wilkinson, 1982).
It has been suggested that the unloading reflex is not important in stopping the jaw, since the impact of the reflex on force development in the masseter muscles is too slow (Miles and Wilkinson, 1982; Van Willigen et al., 1997). Instead, mechanical factors such as the stiffening of the antagonist muscles (Miles and Wilkinson, 1982; Miles and Madigan, 1983) and the force-velocity properties of the jaw muscles (Van Willigen et al., 1997) have been proposed as the principal factors responsible for halting jaw closure.
The basis for these conclusions is that the delay between the decrease in EMG activity and the decrease in force is about 20 ms (Miles and Wilkinson, 1982; Miles and Madigan, 1983). However, this is the electromechanical delay reported for the leg muscles (Grillner, 1972) and as such is not accurate for the muscles of the jaw. In our recent study, we found a delay of only 8-9 ms between the EMG inhibitory period and the reduction of force (Türker and Jenkins, 2000). A similar delay has been reported between the onset of the masseteric silent period elicited by electrical stimulation of the palatal mucosa and the onset of the jaw-opening movement (Yemm, 1972a,b; Türker and Miles, 1985; Widmer and Lund, 1989). Following the unloading of the jaw, rapid jaw closure is halted within 30-40 ms (Hannam et al., 1968; Miles and Wilkinson, 1982). Thus, unloading-induced jaw-closure inhibition, which causes a decrease in bite force, may contribute to the final period of jaw stoppage.
Therefore, in addition to the other mechanical and stiffening factors, the unloading reflex may play an important role in preventing further jaw closing from occurring after the initial slowing of the jaw when an object breaks between the teeth. If masseteric inhibition did not occur following unloading, the level of contraction that was present prior to it would rapidly be re-established, and the jaw-closing movement would resume.
Conclusions
As stated above, the aim of this review was to discuss what is known about the reflex control of the human masticatory system and to propose a method for standardized investigation. We have considered the literature regarding the current knowledge of reflex pathways. This was done to emphasize to the reader the current methods used and the advantages and disadvantages of using mechanical and electrical stimuli
We have put forth suggestions to improve the method of investigation in both systems, and proposed a new technique that offers the most accurate way of delivering the stimulus. Furthermore, a new analysis method was introduced, and its advantages over the currently used methods are discussed. The new method is able to illustrate the synaptic potential that is induced in the motoneurons without the errors embedded in the current techniques.
Once methods for stimulating, recording, and analyzing are standardized, it will be possible to bring out the real wiring diagram that operates in conscious human subjects.
Methodology for a reproducible tooth, mucosa, and skin stimulation under static conditions. The subject sits comfortably with the upper teeth held in fixed relation to a probe mounted on the moving coil of a vibrator and bites into an impression of the upper and lower teeth on a metal bite bar. The impression material on the top part of the bite bar is cut away from around the area of stimulation (in this Fig., it is the upper central incisor tooth) so that the probe can 'move' the tooth. The shape, amplitude, and frequency of the stimulus wave are determined by means of a wave-generating computer program. The computer program is set to initiate random stimuli, with the interstimulus interval varying between 1 and 5 sec. Electrodes are placed to record the surface EMG from the ipsi- and contralateral masseter and digastric muscles. Force applied to the tooth is measured with strain gauges mounted in series with the stimulating probe. For feedback, the ipsilateral EMG signal is rectified and low-pass-filtered at 0.1 Hz, and displayed on an oscilloscope screen. The subject is asked to bite in such a way as to keep the level of activity of the ipsilateral masseter muscle at a pre-determined level (10-20% of the maximal voluntary contraction). In all experiments, the sound of the mechanical stimulus is masked by white noise played into earphones at 80-90 dB. Cumulative sum (CUSUM; Ellaway, 1978) of the ipsilateral masseter surface electromyography (SEMG) and bite force responses to mechanical stimulation of an upper incisor tooth. Calibration was in 'k' (average pre-stimulus background activity) for the CUSUM and in Newtons (the calibration bar for the force data indicates 1 N) for the bite force. Thick lines are the pre-local-anesthetic records, and thin lines are the records taken during the local anesthetic block. Stimulus delivery was at 0 ms. The response to unloading stimulus (2 N load taken off within 5 ms; 400 N/s) is shown at the top; the response to the tap stimulus (2 N load is delivered within 5 ms; 400 N/s) is shown at the bottom. The vertical dotted lines indicate the minimum reaction time for the subject. The relationship between the change in the load and the reflex response is illustrated in an expanded time scale in Fig. 3. Note that the inhibitory reflex disappears but the excitatory reflex stays the same or increases in size during the local anesthetic block of the stimulated tooth. Therefore, both unload and tap stimuli activated not only the PMRs but the spindles as well. Spindle response became obvious once the PMRs were blocked by the local anesthesia. See text for details. The relationship between the 'stretch' stimulus and the first excitatory response. All records are from trials during the local anesthetic block of the stimulated tooth. Thick lines represent the CUSUM of the ipsilateral masseter SEMG; thin lines represent the bite force. Note that the unload stimulus induces a reduction in the force record, and the tap stimulus induces an increase, followed by a decrease in the force record. Since there was no change in the electrical activity of the jaw muscles preceding these changes in the bite force, they are labeled as mechanical artifacts of the stimuli. This subtle yet consistent mechanical artifact is used for eliciting and studying the muscle spindle stretch reflex responses. Note also that the latency of the early excitatory reflex response is shorter in the trials with the unload stimuli, since the 'stretch' arrives earlier with the unload stimulus. Wiring diagram of human masticatory system. Current knowledge on the wiring diagram of the human masticatory muscles estimated under static conditions only. Note that the positions of the cell bodies of the neurons and the motoneurons are all deduced from reduced animal experiments. Peristimulus time histogram (PSTH) vs. peristimulus frequencygram (PSF) in representing the underlying synaptic potential. The PSTH (upper trace) and PSF (lower trace) produced by an EPSP. The dashed line in both traces is the estimated EPSP produced by the current transient injected into a rat hypoglossal motoneuron in a slice preparation. The PSTH exhibited a peak during the rising phase of the EPSP, followed by a trough. Note that following the increase in firing probability during the rising phase of the EPSP, two more peaks appeared in the PSTH, denoted by two pairs of vertical lines to the right of time zero. Looking at the PSTH alone, one would have to say that there was a series of excitations followed by inhibitions. On the other hand, the increase in discharge frequency in the PSF followed the time course of the EPSP correctly, indicating that there is only one long-lasting excitatory event occurring in the motoneuron. The three horizontal lines in the PSF record represent the mean background discharge rate calculated over negative time lags (solid line) ± 2 SD (dotted lines). There were 198 stimuli. Adapted from Türker and Powers (1999) with permission from the American Physiological Society. Reflex response of a single human soleus motor unit to the Achilles tendon tap. This Fig. shows two different approaches to estimating the synaptic potential in humans. The reflex response of a soleus motor unit to 838 tap stimuli is expressed by PSTH and PSF. PSTH records confirmed the classic belief that a tap to a tendon generates three different reflexes: tendon jerk reflex, inhibitory (refractory, silent) period, and long-latency excitation. The tendon jerk reflex is signified by a large number of counts in the PSTH at a latency of about 44 ms. This reflex response lasted for 5 ms and was followed by a silent period of about 60 ms. The silent period is signified by a small number of counts in the PSTH. There was also a 'long-latency' reflex starting around 120 ms after the stimulus. Alternatively, the PSF record showed only one reflex response, a single long-lasting excitation. There was a rapid increase in the discharge frequency coincident with the tendon jerk reflex latency. This increase lasted for about 65 ms, after which the discharge rate returned to the pre-stimulus level. Time zero (arrows) indicates the timing of the tendon tap. Adapted from Türker et al. (1997b) with permission from Springer-Verlag. Effects of an IPSP on motoneuron discharge probability and rate. The PSTH (upper trace) and PSF (lower trace) produced by an IPSP (dashed trace). Note that following the decrease in firing probability during the falling phase of the IPSP, three peaks appeared in the PSTH, denoted by three pairs of vertical lines to the right of time zero. The PSF produced by the same IPSP showed that the deepest part of the IPSP induced a gap in the PSF which was followed by a lower-frequency discharge during the rising phase of the IPSP. Looking at the PSTH alone, one would have to say that there was a series of inhibitions followed by excitations. The discharge rate of the PSF, however, did not change much after the initial reduction, indicating that there was only one inhibitory event. There were 198 stimuli. Adapted from Türker and Powers (1999) with permission from the American Physiological Society. Reflex response of one masseter motor unit to near-painful electrical stimulation of the lip. This Fig. shows two different approaches to estimation of the synaptic potential in humans. The reflex response of one human masseter motor unit to 500 stimuli is expressed in PSTH and PSF. This stimulus classically produces two inhibitory responses followed by an excitatory reflex response. The number of counts in the PSTH illustrates this classic belief very precisely. According to the PSTH analysis, the inhibitory period finished at around 70 ms after the stimulus, when the excitatory reflex response started. The PSF record also showed that the inhibitory responses (gaps in the frequency record) do occur. However, according to the PSF, the inhibitory period lasts much longer and terminates at around 120 ms post-stimulus. Note that the reduction in the discharge frequency between 70 and 120 ms post-stimulus signifies the rising phase of the IPSP (from Fig. 7). Therefore, the large peak in the PSTH signifies not an excitatory reflex but rather the occurrence of delayed spikes during the rising phase of the IPSP (hence the continuation of the inhibitory reflex). The discharge frequency then increases, reaching its peak around 140 ms. Therefore, the PSF analysis shows that there were two periods of inhibitory responses that lasted to about 120 ms which were then followed by an excitatory response. The vertical line indicates the timing of the stimulus (time zero). Adapted from Türker and Cheng (1994) with permission from Elsevier Science.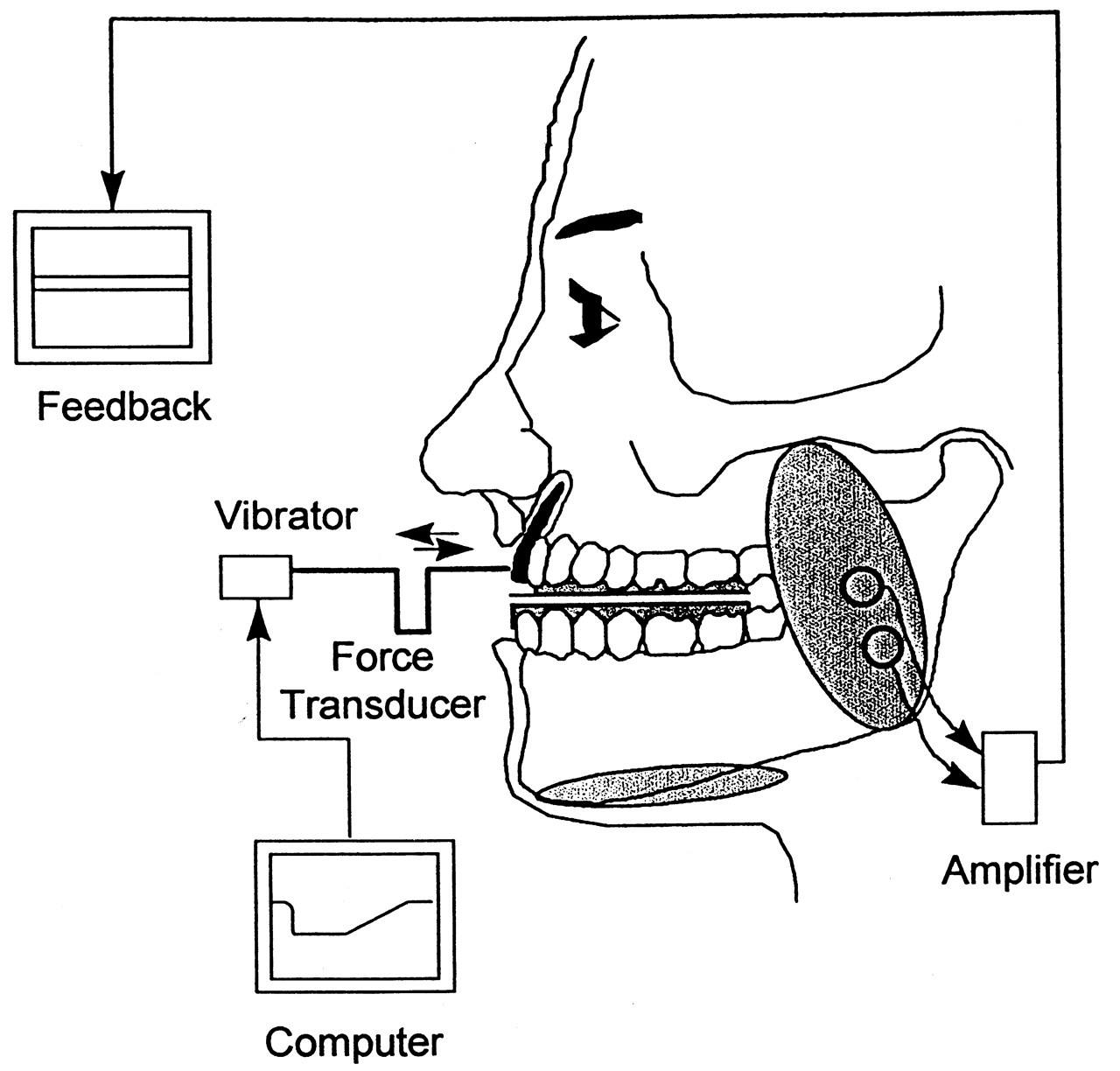
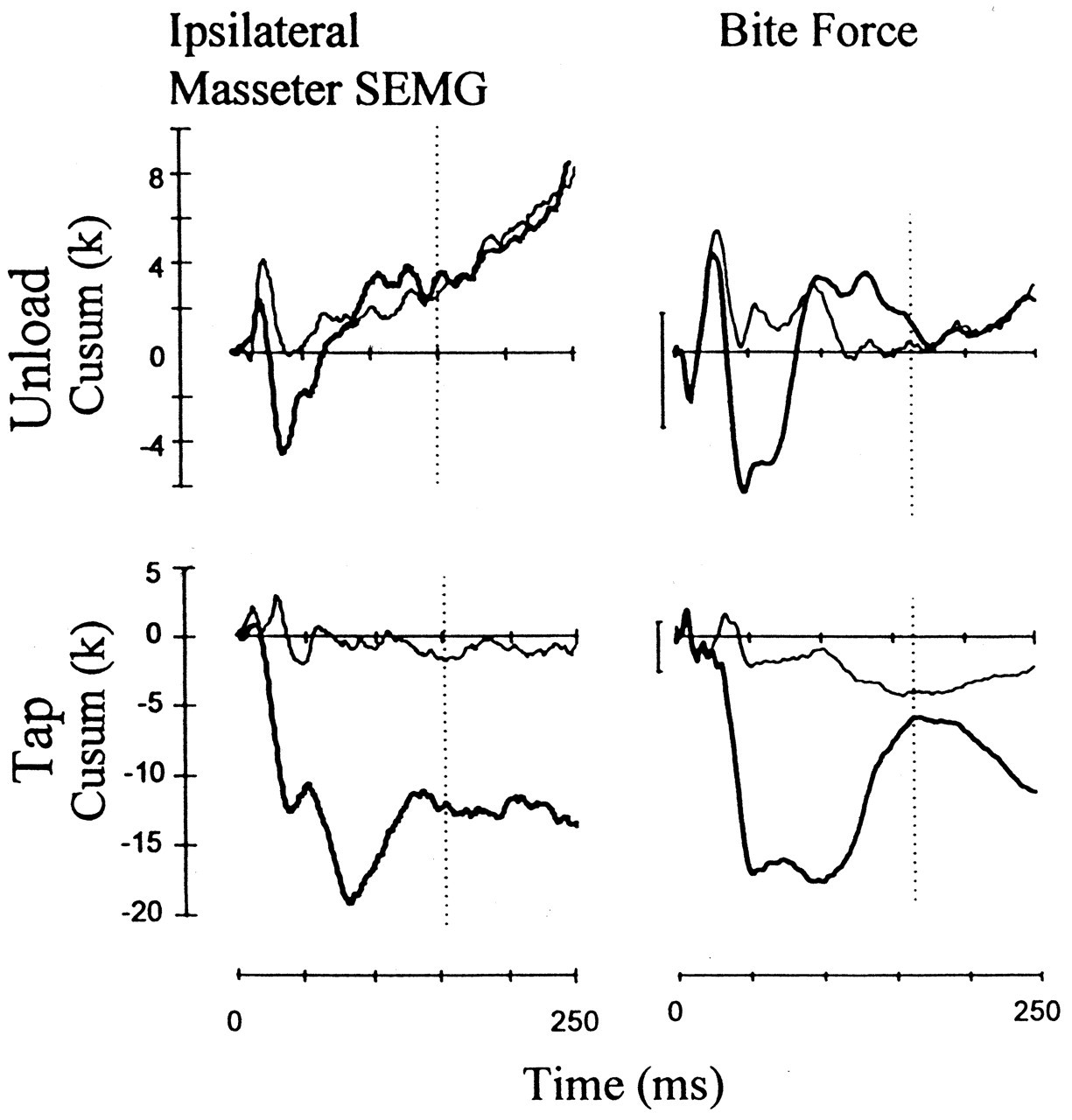
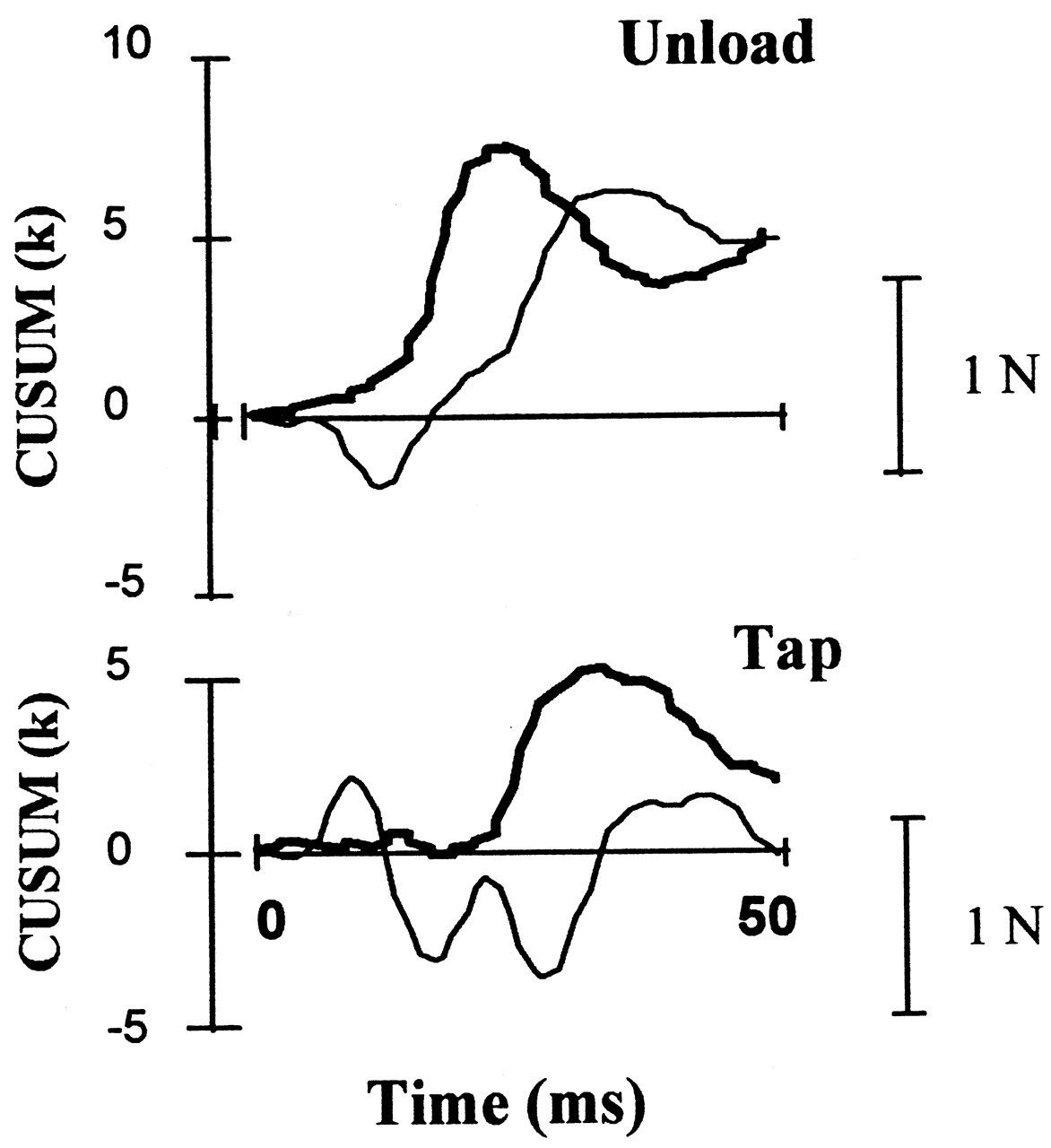
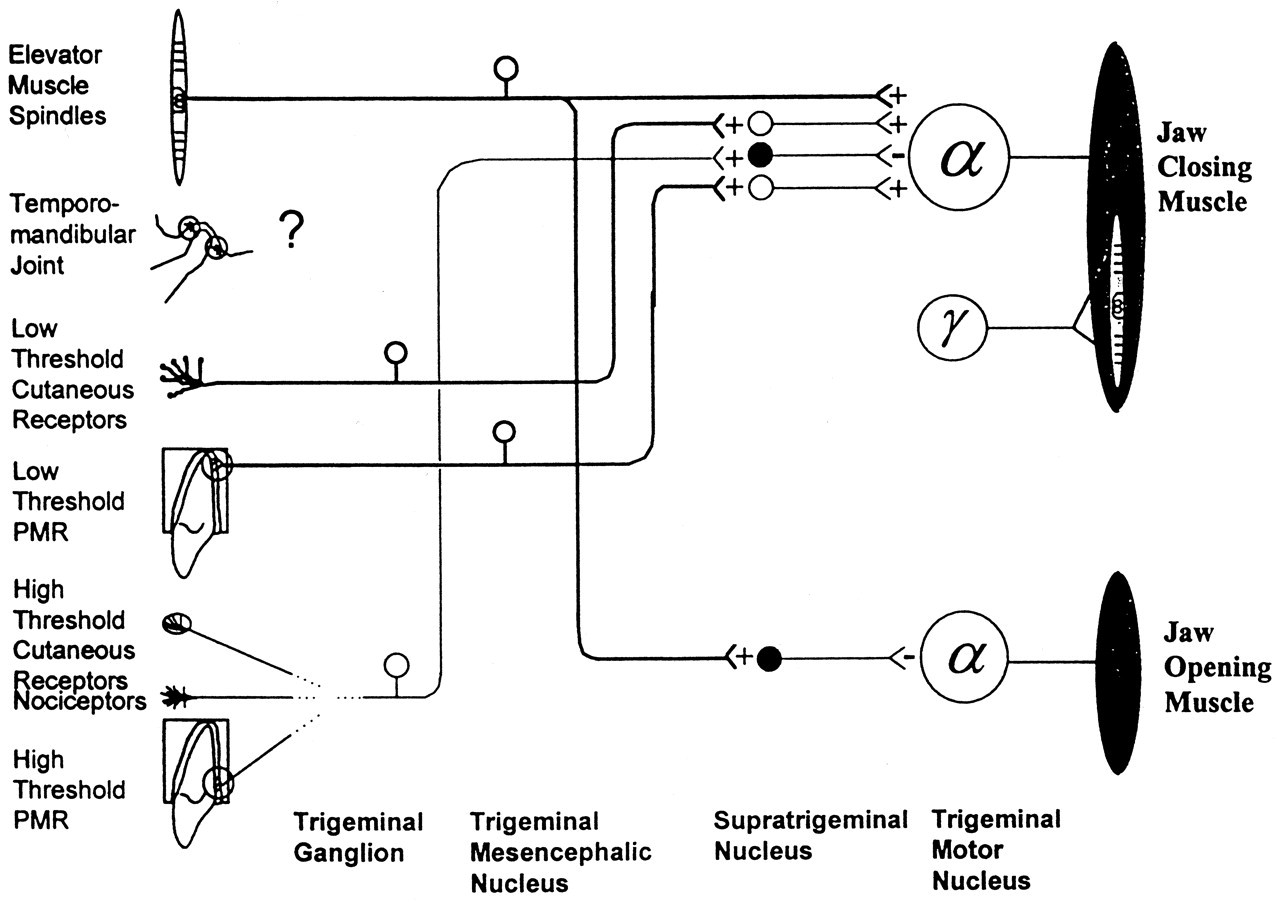
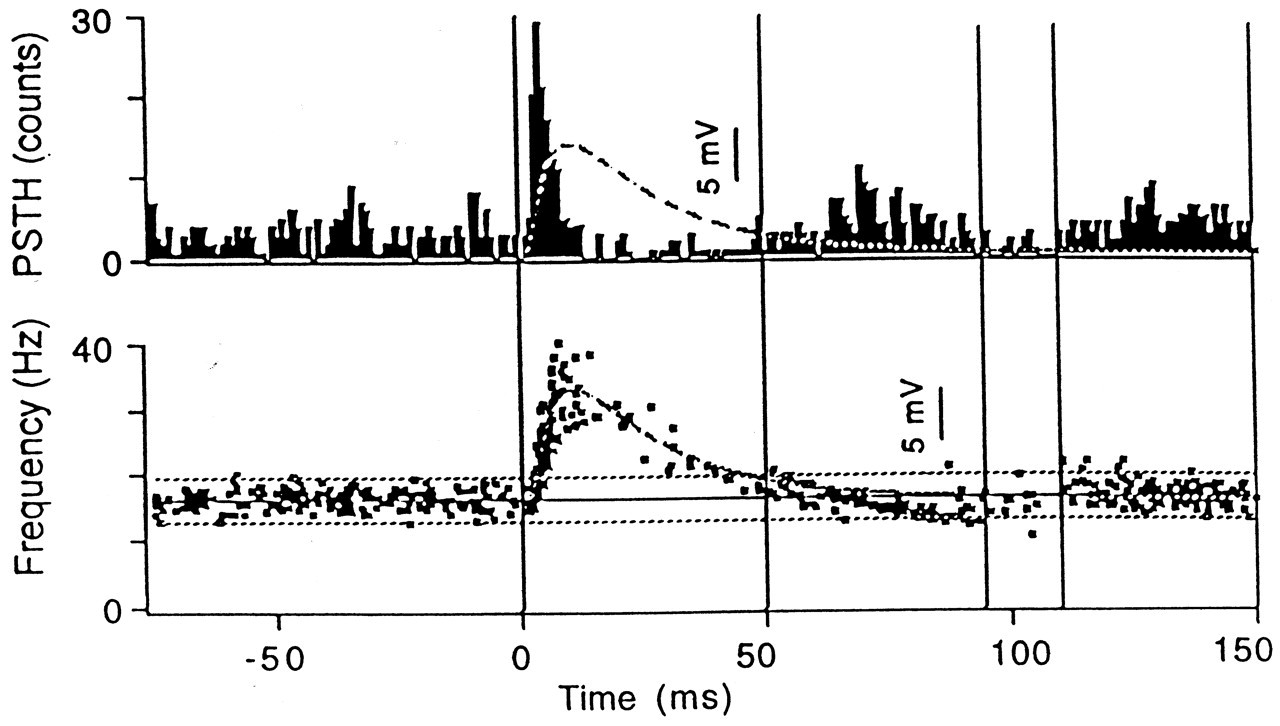
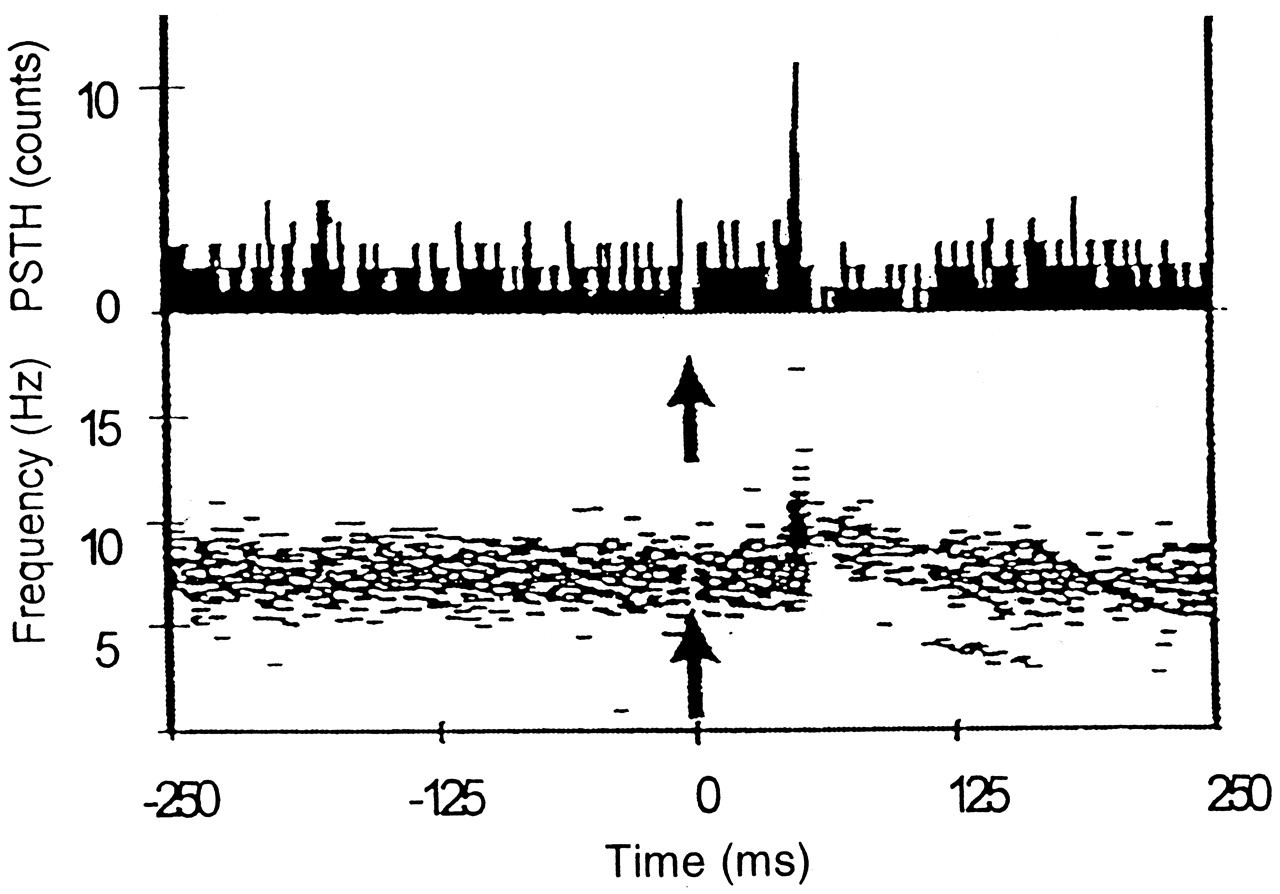
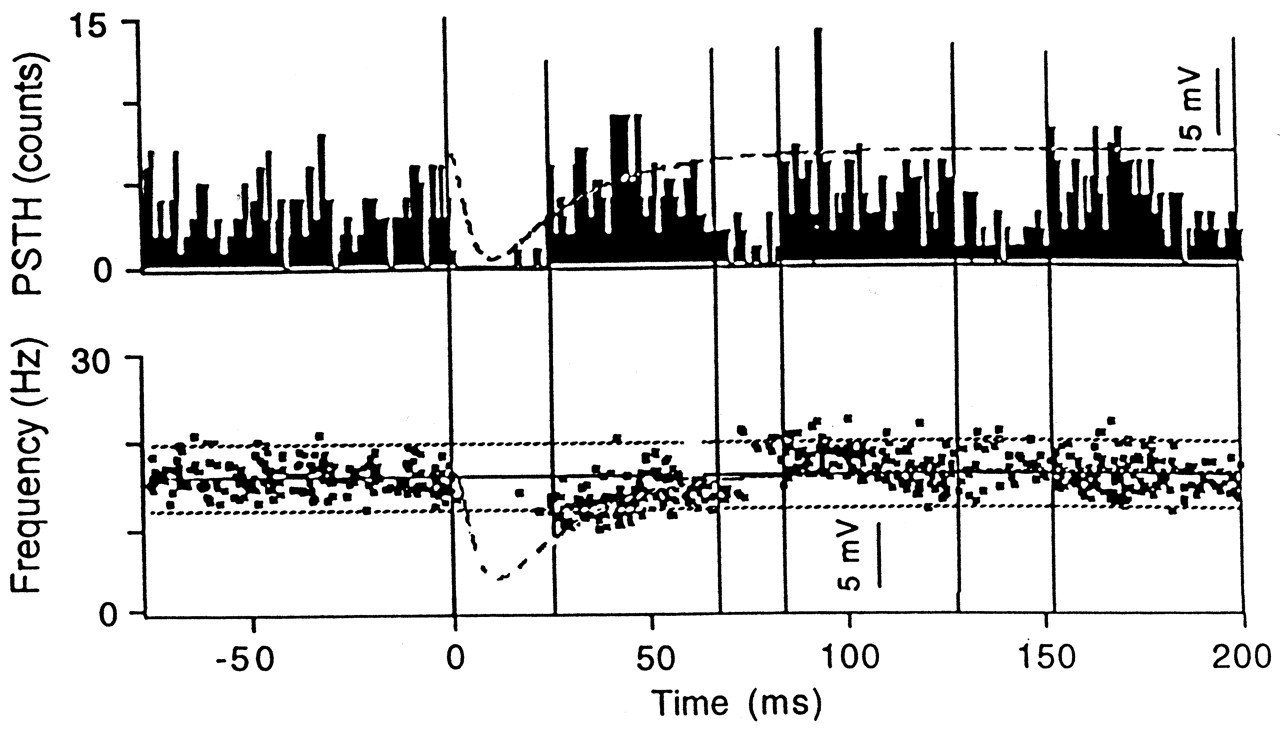
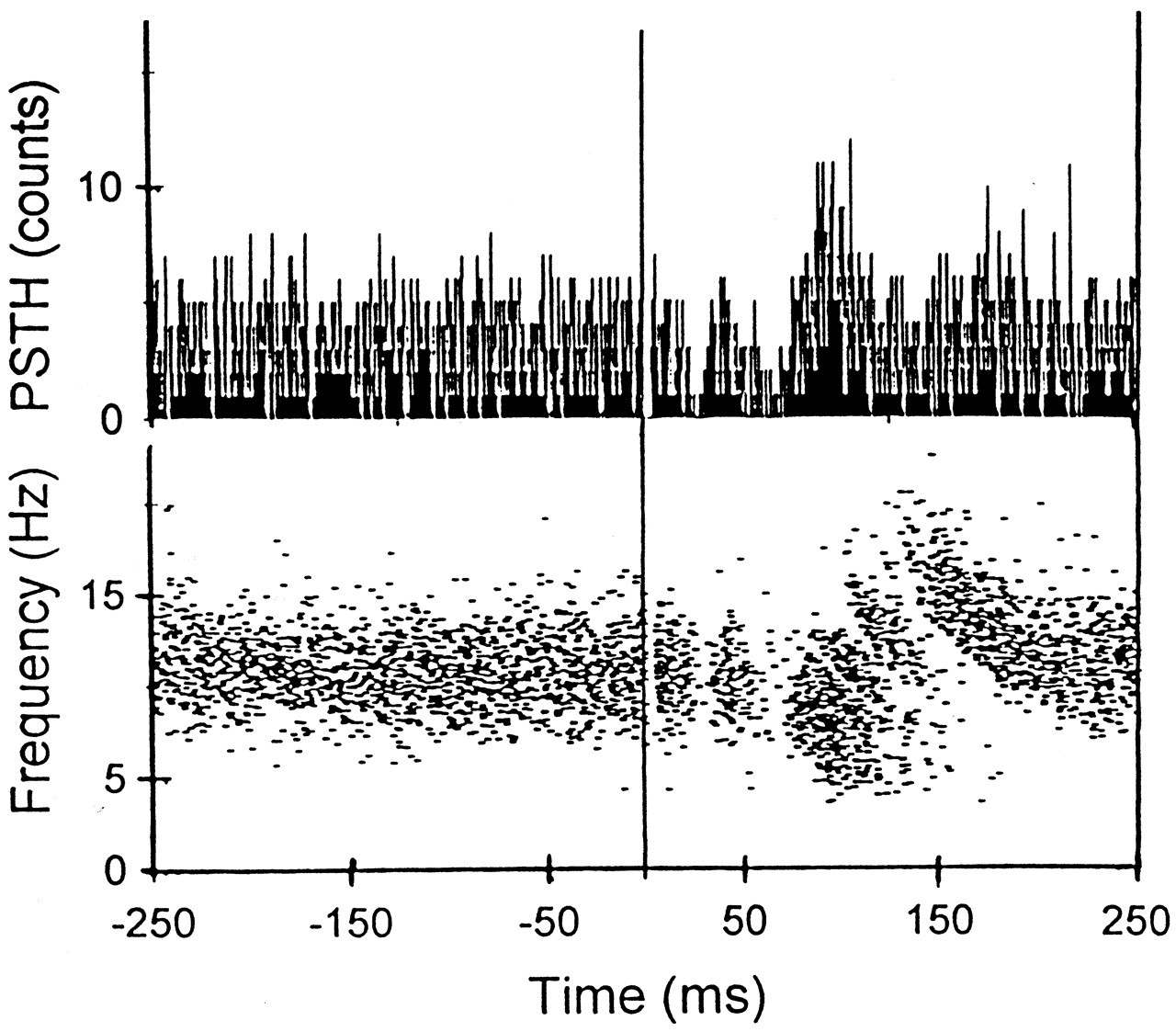
Footnotes
Acknowledgements
This laboratory is supported by the National Health and Medical Research Council (NH&MRC) of Australia. I thank Peter Abolfathi, Pål Brodin, Hong Bing Cheng, Melissa Jenkins, Nick Kerr, Tu Hoanh Le, Ian Linke, Tim Miles, Randy Powers, Sheila Scutter, Annie Schmied, Kylie Tucker, Balamir Uragun, and Jun Yang for their valuable contributions to various experiments.
