Abstract
Background:
Cannabinoid exposures and health outcomes, including consumer safety, perceptions, and motivations, represent an emerging field of epidemiological interest. Women’s perceptions, motives, and risk for adverse health effects related to cannabinoid use may differ from those of men. Researchers aimed to assess the current body of literature on cannabinoid use specific to the historically understudied population of women.
Methods:
Researchers developed cannabinoid safety, perception, and motivation criteria and conducted searches of PubMed, Web of Science, and Embase for human epidemiological studies published between January 1, 2018, and March 31, 2023. Researchers used the Covidence review management platform to screen and extract data.
Results:
Of 270 studies identified, 80% were related to evidence of safety, 17% to motivations for use, and 10% to perceptions surrounding cannabinoid use (categories not mutually exclusive). Most articles specified cannabis/marijuana use (90%), followed by cannabidiol (CBD) (9%), and delta-9-tetrahydrocannabinol (THC) (7%). Study sample composition varied, including at least 45% women (37%), sex-specific outcomes (27%), women-only (20%), and maternal–offspring dyads (16%). Most common study designs were cross-sectional (37%), retrospective cohort (18%), and prospective cohort (16%).
Conclusions:
Research mainly covered outcomes related to cannabis/marijuana use. More longitudinal data are needed on women’s health outcomes, especially those related to cannabinoids, such as CBD and delta-8 THC. Knowledge gaps also exist in identifying motivations for using cannabinoids in pregnancy and self-treatment of women-specific conditions, and sources informing perceptions on cannabinoid use. Closing these gaps in knowledge is an important step in facilitating communication between health care providers and women.
Introduction
In 2018, Cannabis sativa L. containing ≤0.3% of delta-9 tetrahydrocannabinol (THC) by dry weight and its derivatives were defined by the Agriculture Improvement Act of 2018 (2018 Farm Bill) Public Law 115-334 as hemp and removed from control under the Controlled Substances Act. 1 Cannabinoid-containing products are now available in nearly every US state or territory, whether hemp-derived (e.g., cannabidiol [CBD], delta-8 THC) or cannabis-derived (available through state-regulated programs). Prevalence of daily cannabis use has surpassed daily alcohol use among U.S. adults, 2 and consumption has increased in the past decade among adults, including adult women. 3
Concurrently, dietary supplement usage is rising among the U.S. adult population, particularly among women (Mishra et al., 2021). 4 Some cannabinoid products contain dietary supplement ingredients (i.e., caffeine, melatonin) and are marketed for common ailments or appealing benefits (sleep, focus/energy). Research has explored safety parameters, perceptions and attitudes, and reasons for choosing cannabinoid products, but studies examining outcomes specific to women in this space and combined use of dietary supplement ingredients among women are more limited. It is important to understand the extent to which literature is applicable to women’s health in research concerning motivations, perceptions, and reasons for consuming cannabinoid products.
Although research investigating women-specific cannabis-related health outcomes is limited, 5 evidence suggests that sex differences exist in cannabis use patterns and effects 6 and that cannabis may impact women-specific health matters, such as menstrual cycle regularity. 7 Mapping the current literature and the factors it addresses related to use, such as motivations, perceptions of safety and efficacy, and health risks associated with short- and long-term use, may help inform public health efforts by identifying research gaps in this population and contributing context to future public health initiatives. As defined by Arksey and O’Malley and later refined by Daudt et al., 8 a scoping review’s purpose is to map the literature on a specific topic or research area and identify data gaps, articulate key concepts, and characterize types and sources of evidence available to potentially inform future work, policy, and research practices. Given these factors, a scoping review was conducted to map key concepts, types/sources of evidence, and incorporation of women participants within peer-reviewed literature on safety, perceptions, and motivations related to cannabinoid use and to make recommendations to help address specific data gaps.
Methods
Protocol and registration
This scoping review is registered on Open Science Framework. 9 The design follows guidance originally published by Arksey and O’Malley (2005) 10 and updated by Levac et al. (2010), 11 Daudt et al., 8 and Peters et al. (2020), 12 The reporting of this scoping review aligns with the Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) Checklist. 13
Eligibility criteria
English language human epidemiological studies published between January 1, 2018, and March 31, 2023, and based in the United States or elsewhere were considered. Randomized controlled trials and studies with randomized or nonrandomized experimental, prospective/retrospective/longitudinal cohort, crossover, case series/report, case–control, qualitative, or cross-sectional designs were eligible. Eligible studies included humans ≥17 years old 14 and ≥45% women or addressed women-specific outcomes. Studies covered evidence of safety, perceptions, and/or motivations for use of recreationally available cannabinoids (studies about prescribed medical cannabis or Food and Drug Administration [FDA]-approved pharmaceutical products were excluded). Specifically, the safety domain included evidence of risk factors for problematic use and acute/chronic health risks. Perceptions broadly related to cannabinoid use were of interest, such as consumers’ perceived safety and effectiveness of products. Motives for use were defined as self-treatment of health conditions or for adult recreational purposes. Studies that examined co-use with dietary supplements or other substances were included if they reported cannabinoid-specific outcomes. Research on offspring outcomes was included in recognition of the health effects associated with maternal use. Exclusion criteria specified no literature reviews, no meta-analyses, and no animal research studies.
Information sources and searches
Literature searches were conducted for peer-reviewed articles published between January 1, 2018, and March 31, 2023, in the Embase, PubMed, and Web of Science (WoS) databases. Boolean strings were created with guidance from an FDA institutional librarian. The WoS search string for cannabinoids included cannabis, endocannabinoid, cannabidiol, THC, CBD, marijuana, and hemp. Female WoS terms included women, female, “sex differences,” “sex dependent,” “gender differences * ,” maternal, “pregnant women,” pregnant, lactation, menopause, postmenopause, reproduction, progeny, and offspring. The WoS combined cannabinoid-dietary supplement search string included the above cannabinoid and female terms, plus dietary supplement, nutraceutical, neutraceutical, herbal, supplement, vitamin, multivitamin, melatonin, mineral, nootropics, probiotic, and catabolic. WoS terms differed slightly from PubMed and Embase terms because of operational database differences.
PubMed and Embase search strings for cannabinoids included cannabis, cannabidiol, cannabinoid, “cannabis use,” THC, CBD, marihuana, marijuana, “cannabis indica,” “cannabis sativa,” and hemp. Female search terms included women, female, “sex differences,” “sex differences,” “sex dependent,” “sex dependent,” “gender differences*,” maternal, pregnant, pregnancy, lactation, breastfeed, menopause, postmenopause, “reproductive health,” and offspring. The search string for combined cannabinoid-dietary supplement search included the above terms plus “dietary supplement,” nutraceutical, neutraceutical, herbal supplement, vitamin, multivitamin, melatonin, nootropics, probiotic, “catabolic supplement,” and “anabolic supplement.”
Selection of sources of evidence
Manuscripts identified from the literature search were imported into EndNote (version 20) to identify duplicates and record citations. From EndNote, titles/abstracts were exported to Covidence, 15 a digital platform for managing literature review workflows, screening titles/abstracts, reviewing full-text, and extracting data. Two reviewers independently reviewed each title/abstract according to eligibility criteria. A third reviewer served as tiebreaker if the two initial reviewers’ decisions conflicted. The same process was used for full-text review. Following full-text review, qualifying articles moved to the data extraction stage.
Data extraction and charting process
The review team created a data extraction form to gather relevant study information. Per PRISMA-ScR guidelines, 13 the form collected study characteristics (title, country, primary objectives, design, number of participants by sex); outcomes for the full sample, men-specific, and/or women-specific outcomes; cannabinoid(s) investigated; level of incorporation of women; and relevant review aim(s). One reviewer conducted each extraction, which a second reviewer checked and edited as needed. Incorporation of women was organized into the following categories: women-exclusive studies, maternal and offspring health, sex-specific outcomes, and ≥45% women but no sex-specific outcomes.
Synthesis of results
Extracted data were exported from Covidence to Microsoft Excel for cleaning and analysis. A descriptive analysis was performed to determine the frequency of articles under each research aim: articles demonstrating the safety of cannabinoid use, perceptions of safety and effectiveness, and motivations for use across healthy and vulnerable populations. Frequency of study design type, incorporation of women, and cannabinoids studied were grouped by review aim. As with all stages of the review protocol, PRISMA-ScR guidelines were followed.
Critical appraisal of individual sources of evidence
Critical appraisal of evidence is not a requirement for scoping reviews and therefore was not performed. 10
Results
Selection of sources of evidence
Of 4,276 records imported into Covidence, the WoS search returned 877 studies, PubMed returned 1377, and Embase returned 2022. Although results related to dietary supplement co-use with cannabinoids were included, all other records pertaining exclusively to dietary supplements were excluded. From the search results, 394 duplicates were removed (392 automatically by Covidence and 2 manually). Title/abstract screening deemed 660 titles/abstracts eligible for full-text review, which subsequently qualified 261 studies in the review (see Fig. 1).
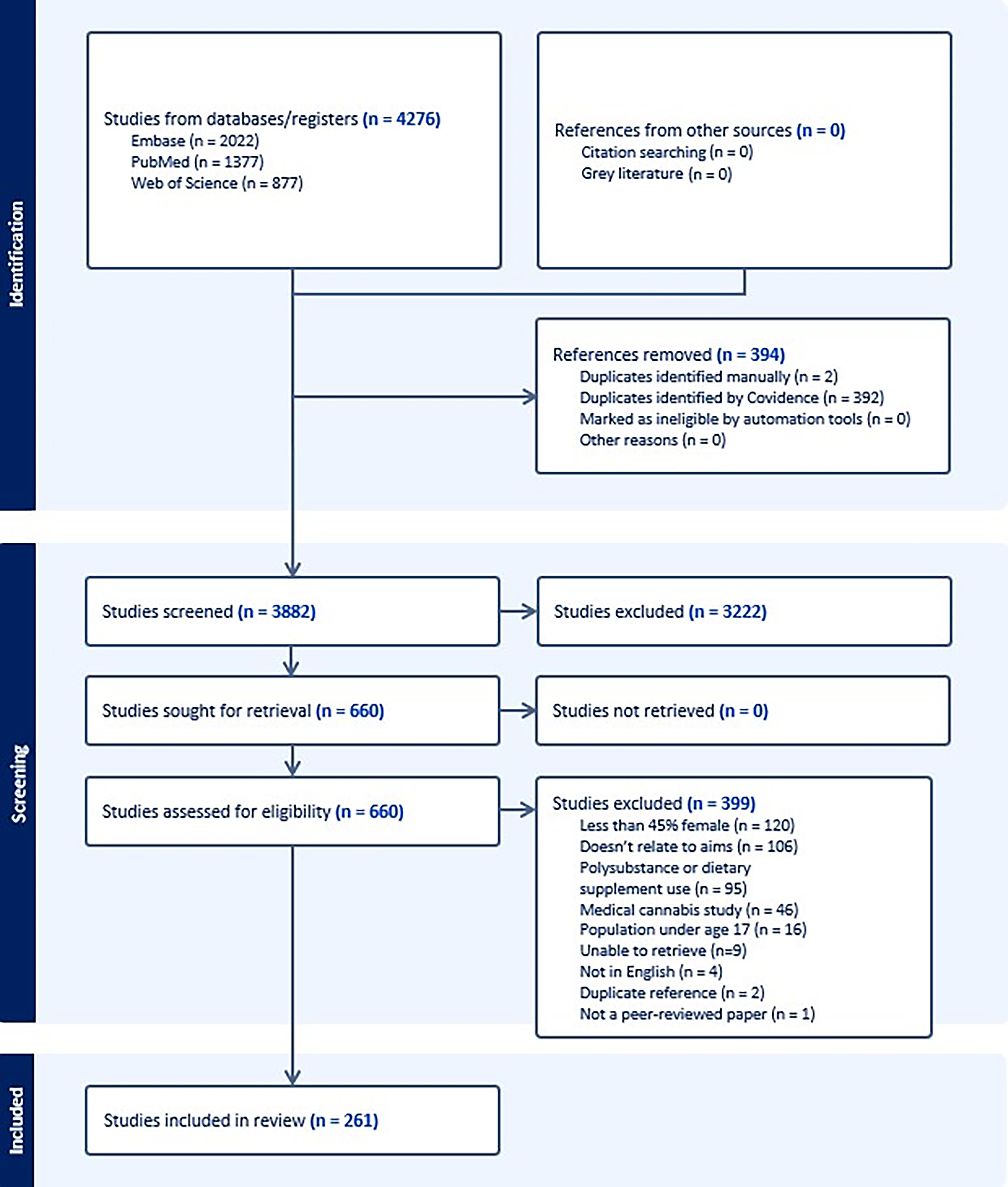
PRISMA diagram.This flowchart illustrates the selection of evidence, beginning with those identified in the search process, and proceeding through screening and full-text retrieval, ultimately resulting in 261 studies being included in the review. PRISMA, Preferred Reporting Items for Systematic reviews and Meta-Analyses.
Summary of study characteristics
This review included 261 primary research studies (Table 1) conducted across six continents, most of which were performed in North America (n = 199, 77%). Of the 12 study designs identified, most were cross-sectional (n = 96, 37%), prospective cohort (n = 46, 18%), and retrospective cohort (n = 42, 16%). Publication year ranged from 2018 (n = 10, 4%) to 2023 (n = 78, 30%); the number of studies increased linearly with time. Sample sizes varied from 1 to 100 (n = 62, 23%), 101 to 500 (n = 61, 24%), 501 to 1,000 (n = 25, 9%), 1,001 to 10,000 (n = 60, 23%), and >10,000 (n = 52, 20%). Studies included ≥45% women (n = 97, 37%), sex-specific results (n = 70, 27%), all-women samples (n = 51, 20%), or mother–offspring dyads (n = 43, 16%). Investigated product types included cannabis/marijuana (n = 234, 90%), CBD (n = 25, 9%), delta-9 THC (n = 20, 7%), delta-8 THC (n = 1, 0.4%), and synthetic cannabinoids (n = 2, 0.7%). Evidence of safety was the most frequently cited research aim (n = 210, 80%), followed by motivations for use (n = 43, 17%), and perceptions (n = 26, 10%). Review aim categories were not mutually exclusive, as some studies addressed more than one.
Study Characteristics (N = 261)
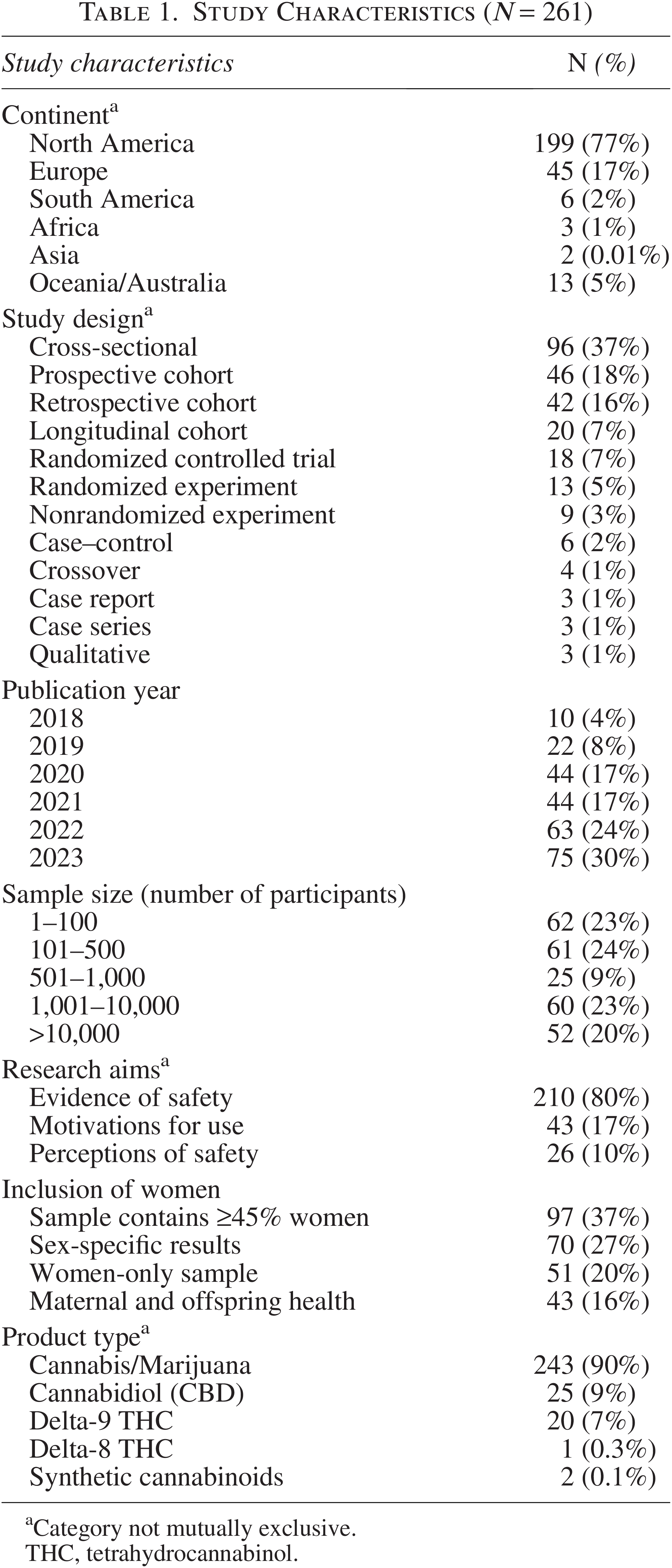
Category not mutually exclusive.
THC, tetrahydrocannabinol.
Topics covered under evidence of safety included health effects related to cannabinoid use, acute cannabinoid effects, and risk factors for problematic use. Motivations for use explored among healthy and clinical populations included chronic pain, mood/anxiety, and sleep disturbance (regardless of overarching condition). Among pregnant women, motivations for use tended to be pregnancy-related nausea and vomiting, or other pregnancy-related symptoms. Sixty-seven percent (67%) of motivational study designs were cross-sectional. Perceptions of cannabinoid safety was the least investigated topic overall (10%). Within this aim, the study population was often consumers, physicians, or populations with a specific health condition(s), and 65% of the studies were cross-sectional.
Synthesis of Results
Evidence of safety
Of 210 studies16–225 relating to evidence of safety, most designs were cross-sectional (n = 62, 31%), retrospective cohort (n = 41, 19%), and prospective cohort (n = 37, 18%) (Table 2). Study populations commonly included ≥45% women without women-specific outcomes (n = 70, 33%). Cannabis/marijuana was most studied (n = 186, 89%). Topic areas included cannabinoid use and associated risk for developing or worsening health parameters, effects of maternal use on child outcomes, acute cannabinoid effects, and risk factors associated with problematic cannabinoid use.
Evidence of Safety Study Characteristics (n, %) (N = 210)
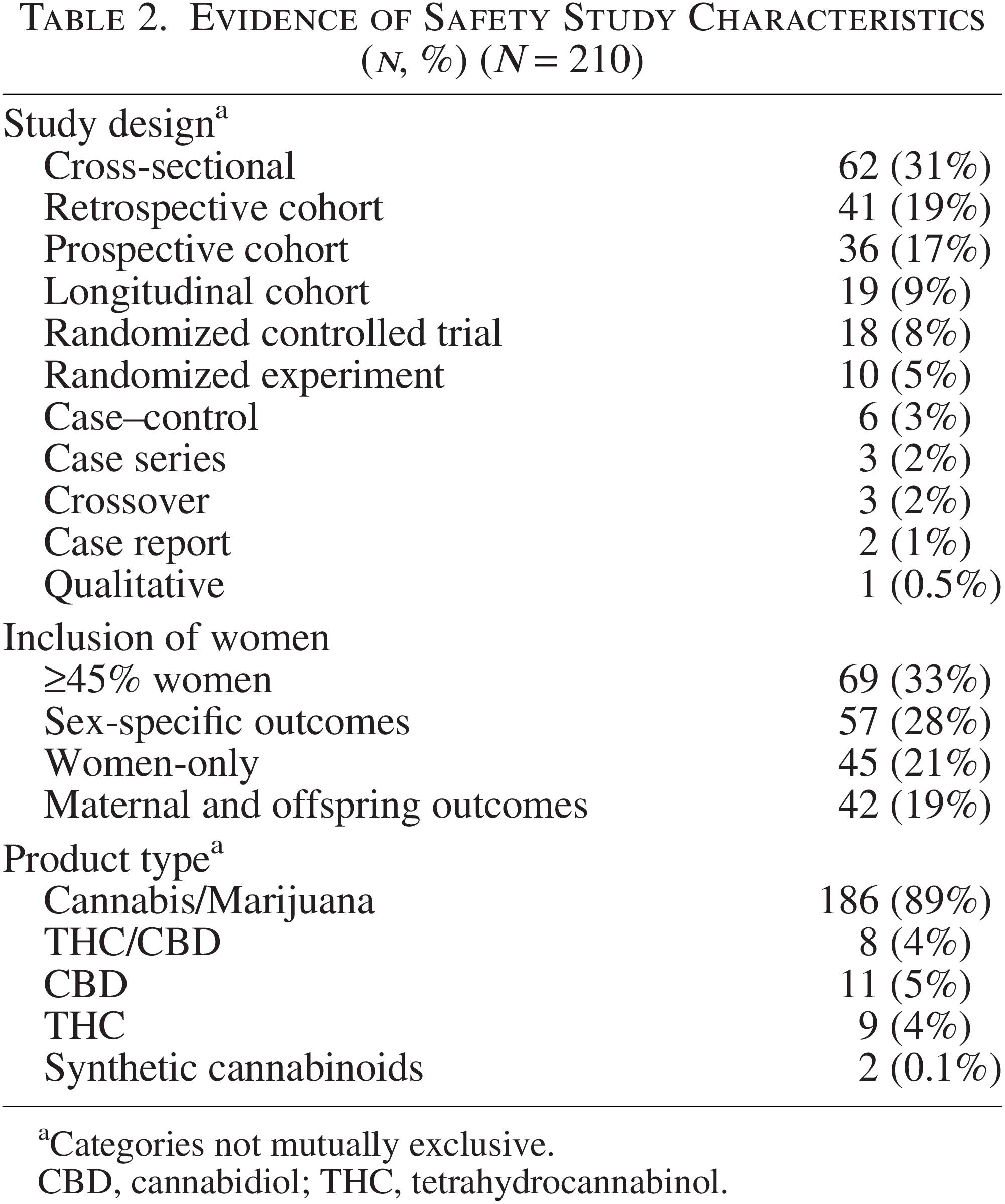
Categories not mutually exclusive.
CBD, cannabidiol; THC, tetrahydrocannabinol.
Of 74 studies16–89 addressing cannabinoid use associated with developing or worsening conditions among adults, outcomes most often covered were mental health and psychiatric disorders (n = 23),16–38 cardiovascular health (n = 12),39–50 and gastrointestinal health (n = 10)51–60 (Table 3). Among mental health or psychiatric disorder studies (n = 23), 81% reported adverse associations with cannabis use. Three studies44,61,62 examined human immunodeficiency virus-related health outcomes, finding beneficial associations between cannabis use and risk for diabetes, cardiac health biomarkers, and inflammatory biomarkers (Table 3).
Associations Between Cannabinoid Use and Risk for Developing or Worsening Health Effects (N = 74)
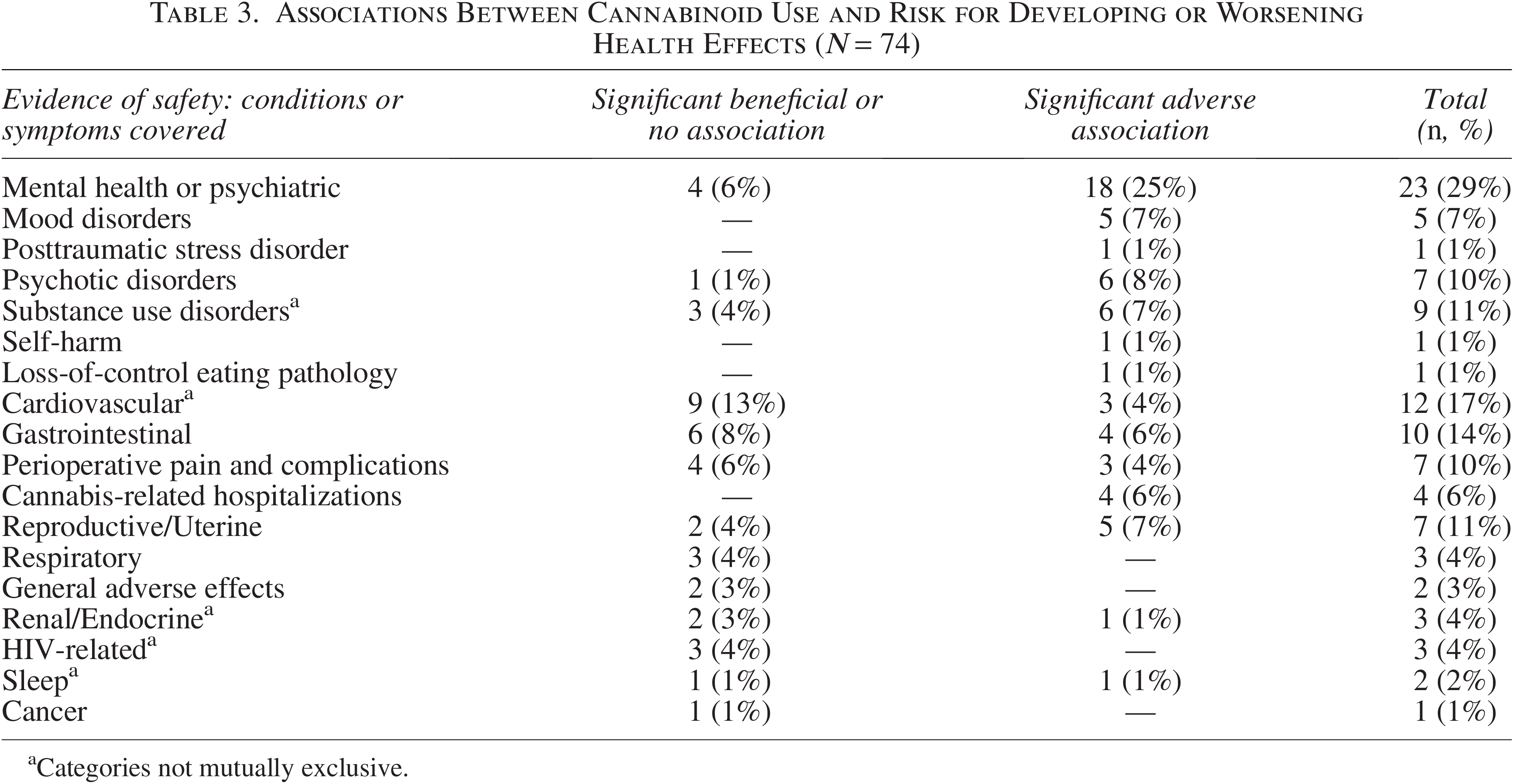
Categories not mutually exclusive.
Four studies examined the association between cannabis use and health parameters of pregnant women90–93 ; and of 42 studies focusing on offspring health,92,94–134 39 reported at least one significant association between maternal cannabis use and increased risk for one of the outcomes investigated. Thirty-one studies92,94–123 explored birth and infant health outcomes within 1 year of life; 11 included children aged 12 months to 10 years old.124–134 All articles investigating low birth weight (n = 17)94,100–106,108,109,112,114,118,120,121,123,126 found a statistically significant increased risk among women who did versus those who did not use cannabis during gestation. Preterm birth (n = 11),94,99,105,107,109–111,114,119–121 small-for-gestational-age (n = 9),99,103,108–110,112,119,121,123 and neonatal intensive care unit admission (n = 5)94,99,105,108,120 were also determined as higher risk for cannabis-exposure in-utero (see Supplementary Table S1 for other outcomes).
Studies of acute effects (n = 25)135–160 examined cannabis/marijuana (n = 8),135–139,141–143 ratios of THC:CBD within cannabis/marijuana (n = 5),144–148 delta-9 THC alone (n = 5),140,149–153 and CBD alone (n = 6).154–160 Studies included ≥45% women without sex-specific outcomes (n = 14),135–137,143–146,148,149,151,152,157,159,160 sex-specific outcomes (n = 8),135,138,139,141,142,147,150,155,158 and women-only (n = 3).140,153,156 Studies explored acute subjective or psychological (n = 19),135,136,138–144,146–148,150,151,153,155,157–160 physiological (n = 10),135,138–141,147,150,154,156 psychomotor (n = 7),135–137,142–145,148 and cognitive effects (n = 9).95,135,136,139,141,142,147,150,152,160 Two studies tested for but did not find statistically significant moderation of variables by sex155,158; however, differences by sex were observed in six studies, including the amount of smoked cannabis needed to achieve a perceived “high” (n = 1), 139 driving impairment (n = 1), 142 subjective effects (n = 4),138,141,147,150 and physiological effects (n = 1).141,147 Some studies (n = 9)191–199 considered neurological and behavioral effects from long-term, and sometimes heavy, cannabis use. Of those, five studies191,192,196–198 reported significant differences in outcomes by sex; four191,194–199 described significant adverse associations, including in brain structure and functioning.
We found that studies addressed risks from using cannabis (no other cannabinoids were indicated) or identified factors predisposing population subgroups to experience cannabis-related problems. Thirty-five articles28,71,161–176,178–190,200–203 examined populations at risk of problematic cannabis use, reporting relationships with psychosocial factors (n = 13)166,168,173,174,176,179,182,185,187–189,200,202 and racial/ethnic status (n = 2)178,200 (categories not mutually exclusive). Eight studies70,177,186,204–208 examined risk for consequences among cannabis consumers compared with nonconsumers, including other substance use (n = 3)177,204,207 and accidents among older adult cannabis consumers (n = 2).70,206
Findings from 18 studies92,101,209–225 exploring risk factors for perinatal cannabis use reported significant associations with younger age (n = 5),92,212–214,218,223 mood or anxiety disorders (n = 4),101,211,212,222 lower socioeconomic status (n = 2),212,214 living in a recreationally legal state (n = 2),215,217 and inadequate prenatal care (n = 2).215,216 Studies used inconsistent categorizations and reported mixed results related to racial/ethnic differences, with perinatal cannabis consumers found more often identifying as non-Hispanic White; non-Hispanic Black; or multiracial/other (n = 1), 92 Black (n = 2),212,225 White or Black (n = 1), 213 Hispanic compared with White (n = 1), 214 or any racial group (n = 1). 216
Motivation for use
Forty-three studies58,77,82,143,162,217,226–261 explored motivations for cannabinoid use, with most employing a cross-sectional (n = 30)58,82,143,162,217,226–239,246,252–261 study design (Table 4). Forty-two studies58,82,143,162,226–230,232–240,247,249,250,252–261 addressed cannabis/marijuana use, followed by THC/CBD (n = 4),82,233,252 CBD (n = 1), 248 and delta-8 THC (n = 1) 231 (categories not mutually exclusive). Incorporation of women ranged from at least 45% of women in the sample (n = 21),82,143,226–239 assessing sex-specific outcomes (n = 11),58,162,252–258 and having women-only samples (n = 11).259–261 Motives for use unrelated to health conditions included recreational purposes,143,233,237,239 coping,236,255 sexual function,257,261 and social factors.226,240,249,251,256,258,262 Studies cited under sexual function did not refer to a specific diagnosable condition and thereby were included with motives unrelated to managing a health condition. Dietary supplement and cannabinoid co-use were covered in three studies.227,244,248
Motivations for Use: Study Characteristics and Motives Covered (N = 43)
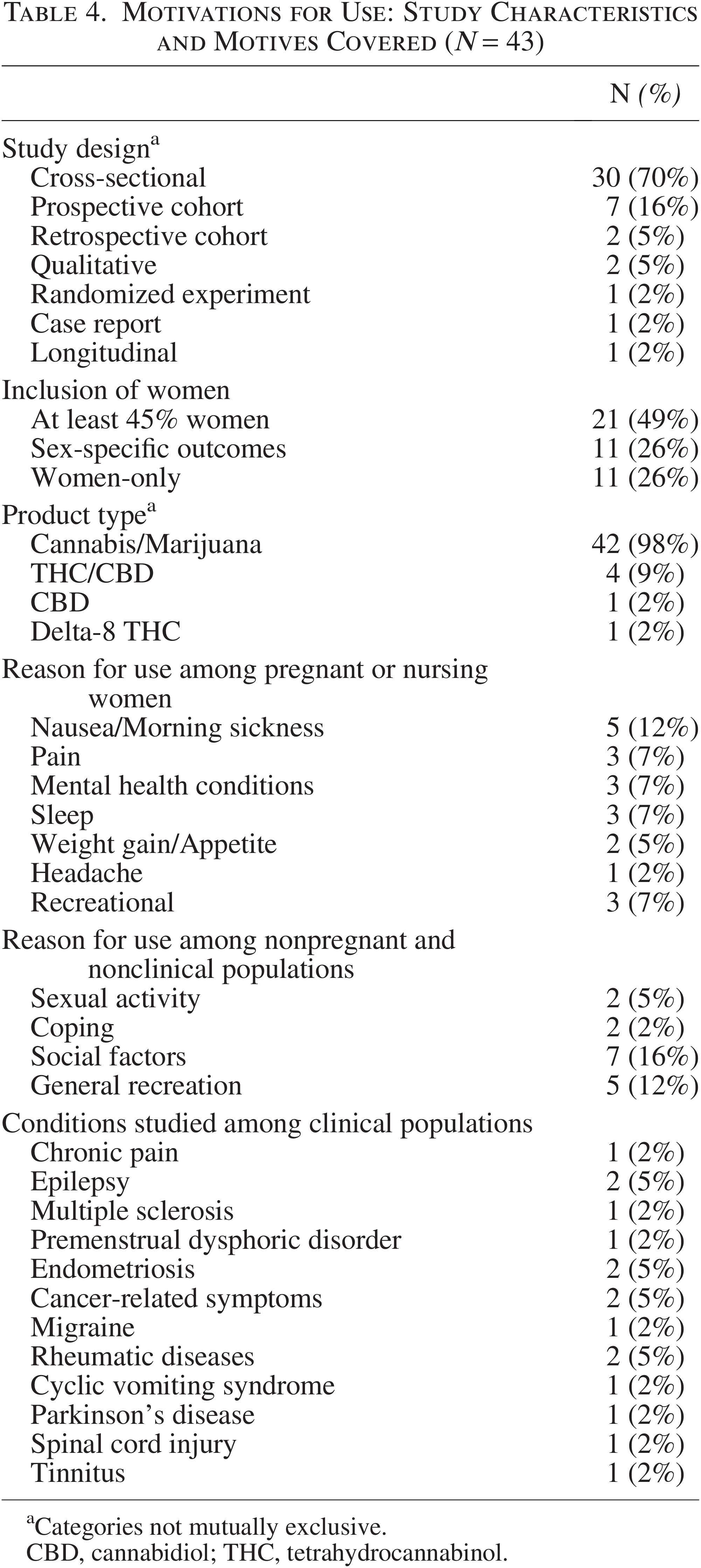
Categories not mutually exclusive.
CBD, cannabidiol; THC, tetrahydrocannabinol.
Eighteen studies58,77,82,227,228,230,232,234,235,238,241,242,247,248,252–254,260 examined reasons for self-treating conditions or adjacent symptoms with cannabinoids (see Table 4 for list of conditions). Endometriosis77,260 and premenstrual dysphoric disorder (PMDD) 242 were the only women-specific conditions investigated. Across clinical and nonclinical populations, cannabinoids were commonly used to address trouble sleeping (n = 7),82,232,233,235,246,254 mood/anxiety (n = 14),77,82,231–233,235,238,241,242,246,255,256 appetite (n = 3),82,233,246 gastrointestinal distress (n = 6),58,77,82,233,246,254,259 and pain (n = 12).77,82,209,228,230–233,235,247,252,260
Six studies examined perinatal cannabis use motives.217,243,244,246,254,259 Pregnant or nursing women more often reported using cannabis solely for therapeutic reasons (n = 3)243,254,259 than recreational and therapeutic (n = 3).217,244,246 Specific reasons for use among otherwise healthy pregnant women varied (Table 5).
Perceptions of Safety: Study Characteristics (N = 25)

Categories not mutually exclusive.
CBD, cannabidiol; THC, tetrahydrocannabinol.
Perceptions about cannabinoid products
Of 25 articles82,143,187,231,235,236,243,248,250,254,256,263–274 addressing perceptions of cannabinoid products (Table 5), 24 specified cannabis/marijuana,82,143,235,236,243,248,250,254,256,263–269,271–274 followed by THC/CBD (n = 3),82,266 CBD (n = 3),248,270,275 and delta-8 THC (n = 1) 231 (categories not mutually exclusive). Fifteen studies included ≥45% women,82,143,231,235,236,239,248,250,263–267,270 5 addressed sex-specific outcomes,254,256,268,269,275 4 had women-only samples,243,272–274 and 1 related to maternal and offspring health. 271 Researchers surveyed clinical82,248 (n = 8, 32%) and nonclinical participants231,263,264,268,269 (n = 14, 56%), pregnant women243,254 (n = 4, 16%), and physicians248,254 (n = 2, 8%) (categories not mutually exclusive).
Summary of Results
Cannabinoid products became more available following the 2018 Farm Bill, which may have prompted increased research into cannabinoid use, effects, and accessibility. This scoping review addresses the broad topic of research on cannabinoid use among adult women, including perceptions, evidence of safety, and motivations for use. We analyzed studies published from January 1, 2018, to March 31, 2023, to focus on the most recent findings. Most studies included findings related to safety and specified cannabis/marijuana. Among the 261 studies reviewed, only 70 (27%) analyzed outcomes by sex, and 51 (20%) comprised samples exclusively of women.
Of only seven articles74–78,89–91 exploring cannabis use and health conditions exclusive to women, all specified cannabis/marijuana, suggesting a lack of research into women-specific health associations with other commonly used cannabinoid products (e.g., CBD, delta-8 THC). Cannabis use was associated with higher odds of having recurrent bacterial vaginosis among adult women. 78 No association was detected between cannabis use and elevated risk for adenomyosis. 74 Pertaining to the menstrual cycle, circulating estrogen levels altered responses to delta-9 THC administration in one randomized experiment. 89 Among women who co-used cannabis and tobacco, cannabis consumers had a significantly shorter luteal phase than tobacco-only consumers, suggesting a cannabis-specific effect. 75 Thirteen women-only studies20,22,24,30,44,46,49,57,73,140,153,156,208 considered cannabinoid use and effects nonexclusive to women, such as CBD for postexercise inflammation 156 or cardiovascular health. 49
All health effects and risk factors explored among pregnant women were related to cannabis/marijuana use. Studies investigating harm to the mother found mixed results. One cohort study found that a greater proportion of cannabis-consuming pregnant women with hypertension developed cardiomyopathy compared with nonconsumers; however, 92 cannabis-consuming women were less likely to be diagnosed with COVID-19 than current nonconsuming pregnant women. 93 Eighteen studies92,101,209–223,225 examined risk factors for perinatal cannabis use, including younger age,92,212–214,218 psychiatric disorders,101,211–213,222 lower socioeconomic status,212,214 and residence in a medically or recreationally legal state.215,217 Inadequate access to prenatal care was also identified as a risk factor for maternal cannabis use,215,216 highlighting challenges in the current health care system. It is considered good clinical practice to screen at-risk patients on cannabis consumption. 276
Motivations for using cannabinoids were categorized by population: healthy recreational consumers, consumers self-treating health conditions, and pregnant or postpartum women. Those with and without reported health conditions tended to use cannabinoids for alleviating emotional or physical discomfort, consistent with findings from analysis of Brightfield Group’s nationally representative survey data, indicating 57% of women cited emotional relief (53%) and physical relief (51%) as primary reasons for using cannabis. 277
Most studies that inquired about reasons for use among pregnant or breastfeeding women found that cannabis was commonly used to improve symptoms such as nausea or sleep disturbance. Among women diagnosed with hyperemesis gravidarum (HG), 53% consumed cannabis daily and typically reported doing so because their antiemetics did not work. 259 Pregnant women with and without HG were also hesitant to discuss cannabis usage with their physician and preferred to gather information from internet and peer sources. 259 In one study reporting on frequency of use during breastfeeding, all women in the sample reported using cannabis daily or almost daily,244,259 demonstrating a need for open dialogue between women and their physicians to enable informed decision making and alternatives to ineffective medications.
Of the three studies characterizing dietary supplement and cannabinoid co-use,227,244,248 one prospective cohort study surveyed breastfeeding mothers on postpartum cannabinoid and dietary supplement use, with 57% of participants reporting concurrent intake of over-the-counter medications and dietary supplements, including vitamins (nine), iron (two), calcium carbonate (one), fish oil (one), and probiotics (one). 244
Perceptions related to cannabinoid use were the least investigated topic overall. Current cannabis consumers held greater positive health perceptions toward cannabis than nonconsumers.239,266,267 Findings from surveys addressing physician competency suggested low confidence in knowledge of cannabis product safety 248 and knowledge pertaining to recreational use, 254 in alignment with reported patient perceptions of their physicians’ cannabis knowledge competency.82,250 Similarly, 70% of surveyed delta-8 THC consumers did not believe their physicians possessed in-depth knowledge about cannabinoids, and 78% did not discuss delta-8 THC use with them. 231 Surveyed participants often reported obtaining cannabis information from friends or internet searches instead of physicians or other scientific sources.82,243
Recreational dispensary store signage and product packaging influenced consumers’ perceptions about cannabis: front-of-store signage touting potential health benefits increased the odds that consumers perceived less risk from use. 264 Young adult consumers preferred products without health warning labels, but their self-reported cannabis health effect knowledge increased after exposure to such labels. 265 Among pregnant or breastfeeding women, most reported concern over long-term effects but remained comfortable using cannabis. 243
Strengths and limitations
Strengths of this scoping review include following review protocols established by Arksey and O’Malley (2005) 10 and guidelines from the Joanna Briggs Institute for PRISMA reporting. 13 A wide variety of study designs (n = 12) and multiple categories of safety evidence (associations with health parameters, acute drug effects, and risk factors) were captured. Population subgroups were analyzed as part of the evaluation of risk factors for problematic use, including in cannabis use disorder and the perinatal period. To the authors’ knowledge, although other scoping reviews have considered motivations and perceptions among certain groups of women cannabinoid consumers, 278 no other scoping review has characterized recent scientific literature involving evidence of safety, motivations, and perceptions related to cannabinoid use and evaluated the degree of participation of women.
Among this scoping review’s limitations, the three databases used and the selection criteria applied may have resulted in missing eligible studies. Although this review reported on several motives for cannabinoid use, articles focusing on medical/pharmaceutical cannabis usage may have yielded relevant information. Based on available data, study design, and bias, researchers were unable to ascertain directionality of a detected significant relationship between cannabinoid use and health effects. For example, one study found that cannabis consumers were less likely to report sinonasal symptoms compared with never-consumers. However, it is possible that those experiencing sinonasal symptoms were unlikely to try cannabis in the first place—not that cannabis is protective against sinonasal distress. 79
Additionally, regulatory requirements needed to conduct marijuana research and the limited availability of longitudinal cohort data on marijuana use may affect the heterogeneity of data available. Much of the currently available cohort data also contain non-population-representative study samples, yielding findings that suggest men tend to use cannabis more than women.40,42,50,52,53,58,69,79,184,202,247 A concerted effort is needed to incorporate population-representative women-specific studies and sex-specific analyses in cannabinoid research.
Data gaps
No studies captured in this review documented prenatal CBD exposure effects. Limited literature exists on the prevalence of women using CBD during pregnancy; however, survey results presented at the 2019 Anesthesiology Annual Meeting indicated that 29% of pregnant women respondents believed topical CBD was safe to apply during the prenatal period, along with 18% of physician anesthesiologists and 70% of doulas. 279 Other data suggest women may believe THC-containing products are a safe therapy for pregnancy-related nausea and vomiting, 280 and this perception may readily translate to CBD-dominant or CBD-isolate products. Animal model studies suggest adverse outcomes from prenatal CBD exposure, dependent on dose and sex of the offspring.281–283
Furthermore, a small number of studies addressed cannabinoid patterns of co-use with dietary supplements,227,244,248 suggesting the need for research on potential interactions of dietary supplement ingredients with cannabinoids, including identification of synergistic/adverse effects and consumer/health care provider education gaps. More work is needed on evidence of safety, perceptions of safety, and motivations for co-use of cannabinoids and dietary supplements based on data from U.S. population-representative adult women study participants to address gaps for potential reproductive health risks and risks during other life stages from co-exposures.
Safety of novel cannabinoids (i.e., delta-8 THC), motivations for using cannabinoids, and perceptions surrounding cannabinoids were lacking in this body of literature. Although these compounds are widely used among residents of states without access to medical and/or adult-use cannabis, 284 published research on motivations, perceptions, and safety parameters among women is sparse. It is possible that these products are covered in studies that did not adequately involve women, so were excluded from this effort, or were not available via the search engines used in this review.
Conclusion and considerations for future work
This review captured 261 studies covering themes of cannabinoid use health risks, problematic use risk factors, acute and long-term health effects, reasons for use, and perceptions related to safety and product knowledge among consumers and physicians, while assessing incorporation of sex-specific analyses. The majority of included research explored safety and cannabis/marijuana outcomes, used cross-sectional data, and contained samples with at least 45% women (without exploring sex-specific outcomes). Based on identified data gaps, more longitudinal data sources are needed to better understand how acute and chronic cannabinoid use among adult women of differing populations (by race/ethnicities, age group) and routes of administration (i.e., inhalation, ingestion) impacts individual health and how prenatal use affects maternal and offspring outcomes. Future research should also investigate reported motivations, behaviors, attitudes, and perceptions of safety and effectiveness pertaining to cannabinoid self-treatment of women-centric conditions, including endometriosis, PMDD, and menopause (insomnia, pain, mood swings) symptoms. With National Survey on Drug Use and Health data indicating increasing use among adult women, guidance from health organizations, and preliminary data on sex differences in use patterns and acute effects, advancement of research in this space and concerted efforts to include women in research samples are vital to public health efforts.
Authors’ Contributions
C.T.: Conceptualization (equal), investigation (lead), methodology (equal), project administration (lead), writing—original draft (lead), and writing—reviewing and editing (lead). S.P., S.R., and K.S.: Investigation (equal) and writing—original draft (equal). A.K.: Conceptualization (equal), investigation (equal), and methodology (equal). M.S.-R., C.P., K.G., and A.M.: Investigation (equal). E.M.S. and K.V.: Conceptualization (equal) and supervision (lead). S.B.-M.: Conceptualization (equal), supervision (lead), and writing—review and editing (equal). B.J.W.: Conceptualization (lead), methodology (equal), and writing—review and editing (equal).
Footnotes
Author Disclosure Statement
The authors have no conflicts of interest to disclose.
Funding Information
This work was funded by the FDA Office of Women’s Health.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
