Abstract
Background:
Black patients experience worse endometrial cancer outcomes compared to white patients. Endometrial thickness (ET) measures from transvaginal ultrasound (TVUS) are used in diagnostic triage to determine if further endometrial tissue sampling is needed. However, recent work suggests that TVUS may disproportionately underdiagnose Black patients and those over 60 years old, contributing to Black patients’ lower survival. Our study aimed to identify risk factors that impact ET measurement quality [visibility, missing data] from TVUS and result in nondiagnostic TVUS results.
Methods:
A retrospective analysis was conducted in a cohort of Black patients undergoing hysterectomy from 2014 to 2020. ET visibility documentation was categorized as visible or “compromised” (partially visible or nonvisible). The presence or absence of endometrial information was also assessed.
Results:
Of 2,705 patients with ultrasound information, 78% (N = 1,838) had documented ET visibility. Of those with visibility, 1,301 (71%) had complete ET visibility. Among those with compromised visibility (n = 537), 271 (50.5%) had partially visible ET, while 266 (49.5%) had nonvisible ET. Significant risk factors associated with compromised visibility included an enlarged uterus (OR: 2.89, 95% CI: 2.32–3.61) and fibroids (OR: 3.78, 95% CI: 1.94–7.39). Of 2,032 patients with ultrasound reports, 9.5% (N = 194) lacked endometrial information. Fibroids (OR: 1.81, 95% CI: 1.19–2.76) and enlarged uterus (OR: 2.61, 95% CI: 1.53–4.45) were also significantly associated with missing endometrial information.
Conclusion:
These findings suggest that a substantial proportion of TVUS examinations may not yield definitive data for diagnostic triage in Black women, potentially contributing to diagnostic delays and worse survival. Improved diagnostic approaches are needed in this population.
Introduction
Endometrial cancer (EC) is a cancer that arises from the lining of the uterus, the endometrium, and is the most common gynecological cancer in the United States. 1 The outcome disparities between Black and white patients have only widened over the past two decades. Black women face higher EC incidence and mortality after diagnosis, 2 is less likely to undergo guideline-concordant surgery, and have decreased survival relative to their white counterparts. 3 Black patients are also more likely to be diagnosed with more aggressive EC histology types. 4
Current diagnostic pathways are suspected to underdiagnose this at-risk population, leading to potentially increased mortality.5,6 The primary symptom of endometrial cancer is postmenopausal bleeding, where 90% of people with endometrial cancer will present with this symptom. 7 The American College of Obstetricians and Gynecologists guidelines for the evaluation of postmenopausal bleeding include assessment via a transvaginal ultrasound (TVUS) to triage endometrial thickness (ET) to determine endometrial biopsy (EMB), or immediate EMB, or dilation and curettage (D&C) for tissue sampling. For the TVUS pathway, an ultrasound is used to determine ET, which would lead to tissue sampling via EMB or D&C if above a certain measurement threshold. However, recent work has cast doubt on the performance of this strategy, including a retrospective diagnostic study where 9.5% of 210 EC cases among Black women were under the suggested 4 mm ET threshold to rule out cancer. 8 In addition, a cross-sectional study of over 1,000 predominantly white women presenting with postmenopausal bleeding demonstrated that the 4 mm ET threshold did not meaningfully discriminate between benign disease and EC lesions for those over 60 years old. 9 In both studies, a proportion of ultrasound results were excluded from the threshold analysis due to limited endometrium visibility or information (21% (N = 750) and 28% (N = 163), respectively).
While the accuracy of ET thresholds is under scrutiny, a more fundamental question remains unexplored: how often do ultrasound examinations fail to provide sufficient endometrium visibility for accurate measurement? This limitation reduces their usefulness in discriminating EC risk, yet risk factors for such nondiagnostic results remain unknown. Given the importance of endometrial visibility to the evaluation of EC using TVUS, work is needed to elucidate what factors are associated with ET visibility and ET reporting for symptomatic women undergoing evaluation.
This analysis aimed to identify risk factors associated with nondiagnostic TVUS results, defined as either substandard ET visibility or the absence of any ET measurement information on ultrasound reports.
Methods
Sample description
For our dataset, we used the Guidelines for TVUS in the Detection of Early Endometrial Cancer (GUIDE-EC) cohort, a merged set of abstracted electronic health record (EHR) data and secondary administrative billing information. GUIDE-EC is comprised of all patients over the age of 18 who self-identified as Black or African American who underwent a hysterectomy, identified by administrative billing codes, from 2014 to 2020 in a 10-hospital health care system in the south of the United States (N = 3,455). Briefly, clinical data was captured from the health system’s data warehouse, including the date of diagnosis and specified physician-and hospital-billed diagnostic and common procedural terminology codes. A team of professional medical record abstractors with over 20 years of experience performed the EHR data abstraction. During primary abstraction, weekly quality checking was completed via automated data checks for nonsensical entries and manual flags by abstractors of missing or uninterpretable information. In the latter case, the physician led a review of all flags to resolve data questions. 10 The abstractor team identified the first TVUS performed 24 months prior to hysterectomy, with a 30-day window before and after to indicate the symptom date. If there were no clinical notes from this time, the closest gynecological procedure (EMB, D&C) or physician note was used. Data were merged, cleaned, and de-identified, with all open-access text reviewed and recategorized until any data discrepancies were resolved. Full details on the cohort creation have been previously published. 8 For this study, our data was limited to the 2,032 (75.1%) patients of the GUIDE-EC cohort with ultrasound information. As a secondary analysis of a de-identified dataset, this current study was deemed exempt by the University of Washington IRB.
Endometrial thickness, visibility, definition, and reporting
ET visibility was assessed in patients who underwent a TVUS and had a TVUS report that included information on the ET visibility. Reports were derived from the following: TVUS imaging reports native to the EHR, scanned TVUS reports from other health systems, TVUS results embedded in physician and other clinician notes, or any report in the EHR documenting TVUS results.
Non-native TVUS reports were classified as either summary (less than three measurements reported) or detailed (three or more measurements reported). Abstractors captured visual documentation as visible, partially visible, or nonvisible. Visible ET was defined as the presence of a numeric thickness measurement reported in millimeters or centimeters with no reported difficulties in viewing the endometrium. Partially visible was defined as measurements with notes reporting “partially visibility,” “obscured,” or any other descriptive variation indicating compromised visibility. If partially visible was selected, the abstractor documented any measurement reported and any reason that may have impacted endometrium visibility. Nonvisible ET was defined as reports with text descriptions indicating a lack of visibility, such as “unable to be measured” or “poor visibility,” and no reported ET measurement was recorded.
Endometrial information reporting
All ultrasound reports were assessed for the presence or absence of endometrial information. If the ultrasound report omitted any comment on the endometrial measurement, visibility, or any other endometrial description, it was classified as missing endometrial information. All other ultrasounds were classified as “information present.” We assessed all previously reported sources (TVUS imaging reports, scanned reports from other health systems, physician summary reports, other clinician notes, or any report in the EHR documenting ET visibility) for any mention of missing endometrial information. This included ET measurements or descriptions of the endometrium. The data were categorized into two groups: no endometrial information and endometrial information present. The information present group included all ET visibility groups (visible, partially, and nonvisible).
Risk factor selection
Potential risk factors for nondiagnostic TVUS were selected due to their relationship to EC in our previous work,5,8 from the literature,7,9,11,12 and the plausibility of the influence of TVUS quality. We considered two broad categories of risk factors: mechanical and nonmechanical. Mechanical risk factors are those that would physically cause difficulty in visualizing the ET. These include structural modifications on the uterus and endometrium: the presence of fibroids on ultrasound (US), the number of fibroids, submucosal location of fibroids, body mass index, enlarged uterus/abdomen, and ovarian cyst/pelvic mass. Nonmechanical factors were those that may cause lower concern for EC or otherwise influence the effort or perception of the need for an ET measurement. These include: endometriosis, endometrial hyperplasia, age at hysterectomy, history of fibroids, postmenopausal bleeding, any uterine bleeding, pelvic/abdominal pain, fatigue/dizziness/light-headedness, abnormal pap, urinary symptoms, menopausal symptoms, anemia, a menstrual period longer than 7 days, missed days of work, disability, employment, caregiver, smoking, blood transfusion, gender expansive identity, family history of breast cancer, family history of ovarian cancer, family history of uterine/endometrial cancer, family history of cervical cancer, depression or bipolar disorder, anxiety or posttraumatic stress disorder (PTSD), any mental health condition, number of comorbidities, and insurance. Risk factor information was obtained from the abstracted clinical and demographic data. Abstractors could select present, absent, or unknown for each risk factor. Unknown was selected when there was no clear documentation or mention of the risk factor in the EHR. Depression, bipolar disorder, anxiety, and PTSD were collapsed into a composite depression/bipolar disorder and anxiety/PTSD due to small numbers.
Data analysis
Descriptive statistics were used to summarize endometrial visibility, endometrial reporting, and potential risk factors. Risk factors were described overall by endometrial visibility and by the availability of information on the endometrium. Total numbers and percentages were used for categorical variables and means and standard deviations were used for continuous variables. Using logistic regression, each risk factor was evaluated for association with endometrial visibility outcomes excluding unknown values. Among the measures with independent associations, models were combined to evaluate their independent associations with endometrial visibility outcomes and the relative strengths of association. To avoid over-fitting, we used penalized regression using the Least Absolute Shrinkage and Selection Operator (LASSO) to select a parsimonious set of associated factors. 13 The tuning parameter was selected with 20-fold cross-validation. Through this process, we identified a common set of variables and combined them in a logistic regression model. To account for potential sampling variability, we used bootstrapping with 100 replications to generate robust estimates of the model coefficients and their standard errors.
The same process was repeated to analyze the associations between risk factors and endometrial information reporting. In this case, missing risk factor values were included as we suspected that such missing information might be associated with the absence of endometrial information available due to overall poor quality EHR reporting. We then conducted a sensitivity analysis and excluded the unknown values to determine if our selected risk factors were associated with no endometrial information reported, versus being a consequence of missing risk factor information. Additionally, we repeated all analyses stratified by age above and below 50 years old. Interactions between age and each risk factor were assessed in univariate analyses using Wald tests. p-values less than 0.05 were considered statistically significant. Analyses were conducted in Stata version 18.0 (College Station, TX: StataCorp LLC). 14
Results
Of the 3,455 patients in the GUIDE-EC sample, a total of 2,705 underwent ultrasound prior to surgery, based on EHR abstraction and billing data. Among these 2,705 patients, 673 were excluded due to having no ultrasound report present, resulting in 2,032 (75.1%) patients.
Endometrial thickness visibility
Patient characteristics for endometrial thickness visibility
Of the 2,032 patients with available ultrasound reports, 1,838 (90%) were included in this endometrial visibility analysis, and 194 patients were excluded due to having no endometrial information on the ultrasound report. Among those included, 1,301 (71%) were categorized as having visible ET, and 537 (29%) had compromised visibility. Among those compromised, 271 (50.5%) had partially visible ET, and 266 (49.5%) had no visible endometrium.
Table 1 describes patient-level characteristics overall and stratified by ET visibility. There were a few notable differences between visibility groups. Regarding mechanical factors, an enlarged uterus was markedly more common in the partially/nonvisible ET group (69.1%) compared to those with fully visible ET (43.6%). For other mechanical factors, BMI, fibroid factors, and pelvic mass featured minimal (<5%) differences between visibility groups. In nonmechanical factors, the diagnosis of endometrial hyperplasia was more common in the visible ET group (9.1%) compared with the partial/nonvisible ET group (3.9%). There were also fewer uninsured patients in the fully visible group (6.7%) compared with the partially/nonvisible group (12.3%). Other nonmechanical factors that differed by minimal (<5%) differences included postmenopausal bleeding, anemia, missed days of work, and any mental health conditions.
Patient-Level Characteristics by Visibility
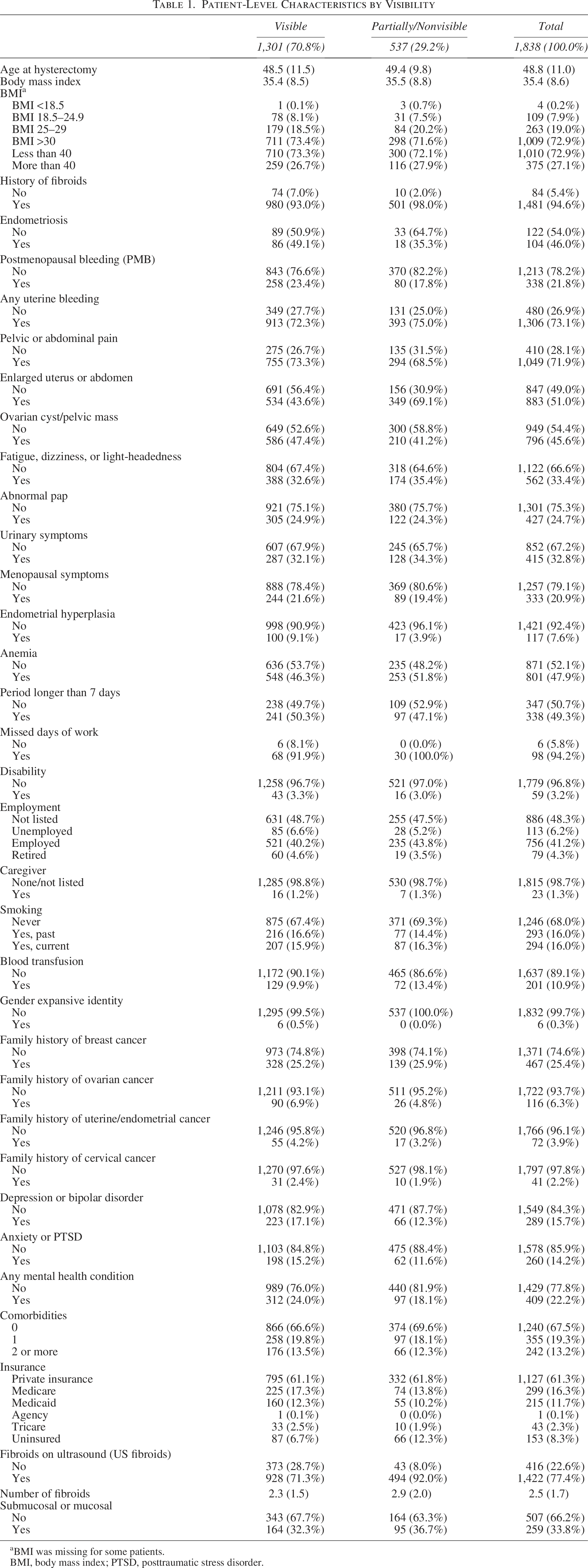
BMI was missing for some patients.
BMI, body mass index; PTSD, posttraumatic stress disorder.
Risk factors for partial/nonvisible endometrial thickness visibility
Using logistic regression, some risk factors were significantly associated with the outcome of partial/nonvisible ET (Supplementary Data S1): enlarged uterus (OR: 2.89, 95% CI: 2.32–3.61), history of fibroids (fibroids) (OR: 3.78, 95% CI: 1.94–7.39), and fibroids seen at ultrasound (US fibroids) (OR: 4.62, 95%: 3.31–6.45). Factors negatively associated with partial/nonvisible ET included hyperplasia (OR: 0.40, 95% CI: 0.24–0.68), depression or bipolar disorder (OR: 0.68, 95% CI: 0.50–0.91), and pelvic mass (OR: 0.78, 95% CI: 0.63–0.96). Other risk factors for partial/nonvisible ET with significant associations included uninsured status (OR: 1.82, 95% CI: 1.29–2.56), transfusion history (OR: 1.41, 95% CI: 1.03–1.91), and anemia history (OR: 1.25, 95%: 1.01–1.54). In patients over 50 years old, postmenopausal bleeding (PMB) (OR: 0.53, 95% CI: 0.37–0.75) was significantly associated with lower odds of partial/nonvisible ET visibility, but not for those under 50. When assessing the interaction between age groups and risk factors, those below 50 were more likely to have partial/low ET visibility if they experienced pelvic pain (p for interaction = 0.003), had an enlarged uterus (p for interaction = 0.02), and a period longer than 7 days (p for interaction < 0.001).
Figure 1 reports the relationship between risk factors and partial/nonvisible ET measurement in our LASSO model. Overall, an enlarged uterus (OR: 2.28, 95% CI: 1.74–2.97) and fibroids present on ultrasound (OR: 3.75, 95% CI:2.63–5.33) were the most significant factors for partial/nonvisible ET, followed by uninsured status (OR: 2.03, 95%: 1.43–2.89). Age stratification yielded different results. For those below age 50, fibroids on ultrasound (OR: 4.07, 95% CI: 1.37–12.1) and enlarged uterus (OR: 2.02, 95% CI: 1.40–2.92) were independently associated with a partial/nonvisible ET measurement. For those above 50, fibroids present on ultrasound (OR: 2.84, 95% CI: 1.54–5.24) and enlarged uterus (OR: 2.10, 95% CI:1.35–3.27) were independently associated, but the history of fibroids was not.
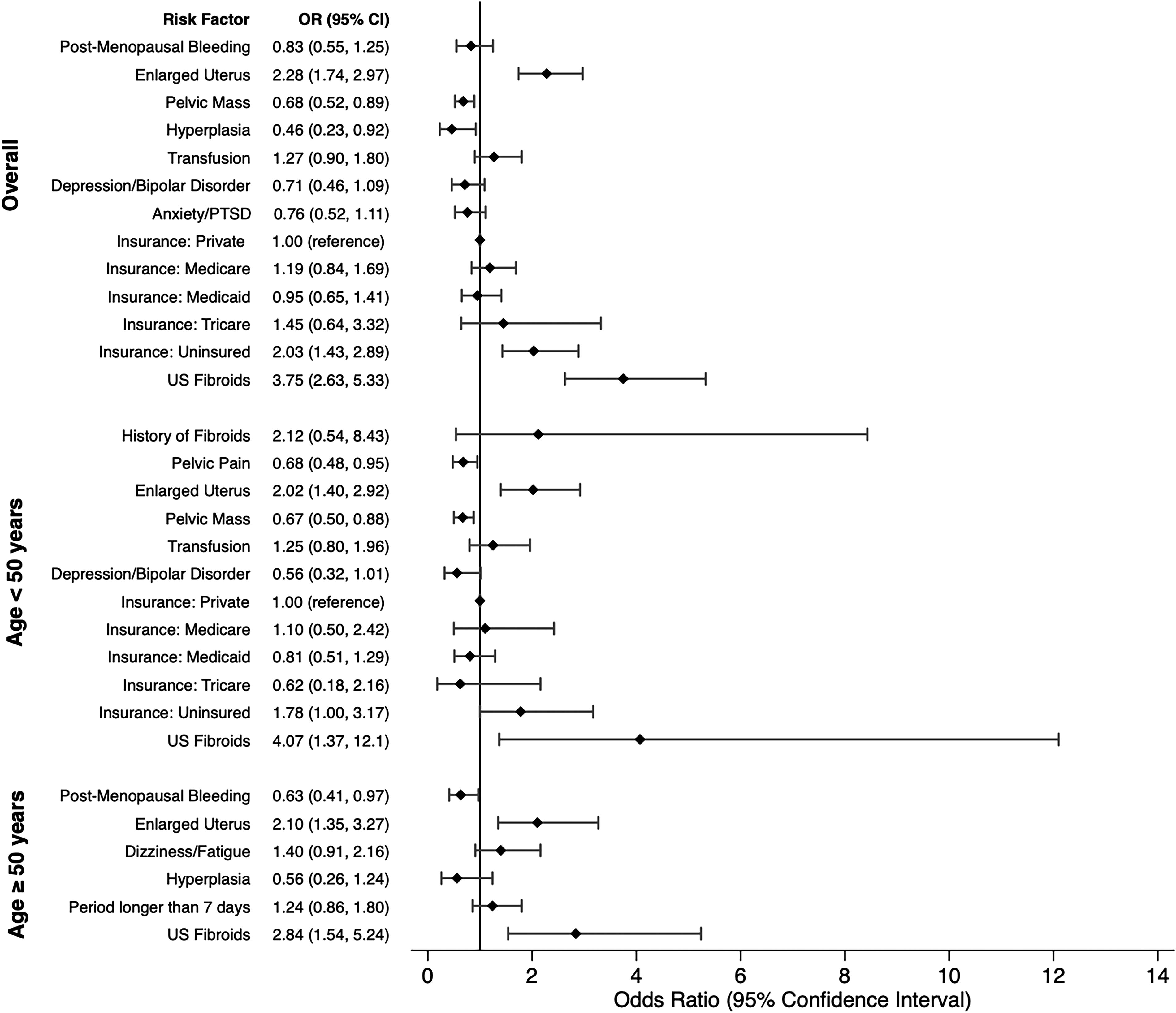
Results for partial/nonvisible compared with visible endometrium.
Endometrial information reporting
Patient characteristics
Of the 2,705 with ultrasounds, there was a total of 2,032 patients with ultrasound reports included in this endometrial information analysis: 1,838 (90.5%) had endometrial information present, and 194 (9.5%) had no endometrial information reported. The 673 patients (33.1%) without an ultrasound report were excluded from the endometrial information assessment. Table 2 describes overall patient-level characteristics and is stratified by endometrial information, reporting notable differences. Regarding mechanical factors, patients with no information present (82.4%) were likely to have a BMI less than 40 compared with those with information present (72.9%). However, patients with information present (27.1%) were more likely to have a BMI over 40 than those with no information (17.6%). Other mechanical factors with minimal (<5%) differences included an enlarged uterus and fibroids on ultrasound.
Patient-Level Characteristics by Presence of Endometrial Information
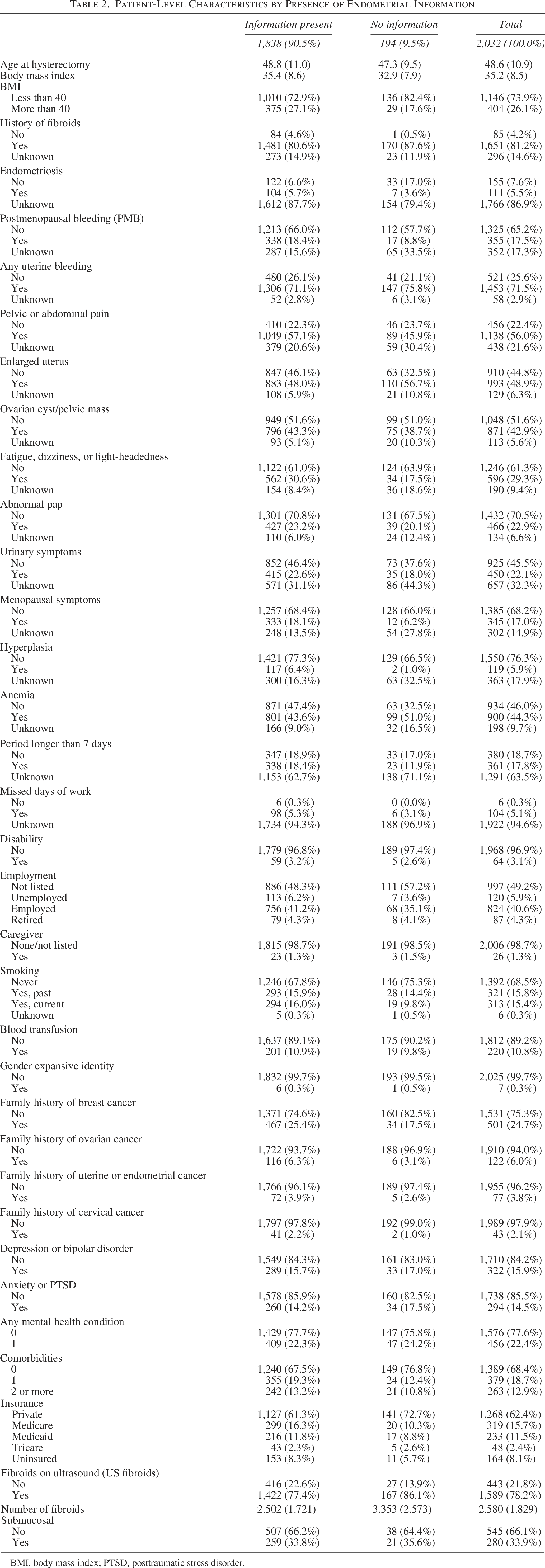
BMI, body mass index; PTSD, posttraumatic stress disorder.
When looking at nonmechanical factors, patients with endometrial information present were diagnosed with higher rates of pelvic/abdominal pain (57.1%), fatigue/dizziness or lightheadedness (30.6%), and hyperplasia (77.3%) compared to those with no information (45.9%, 17.5%, 66.5%, respectively). Lack of endometriosis diagnosis was reported to have no endometrial information (17%) compared to information available (6.6%). Other nonmechanical factors that differed by minimal (<5%) differences included history of fibroids, any uterine bleeding, urinary symptoms, smoking status, history of breast cancer, comorbidities, and insurance status.
Patients with no endometrial information had higher rates of unknown/missing variables in the following risk factors: postmenopausal bleeding, pelvic/abdominal pain, ovarian cyst/pelvic mass, fatigue/dizziness or lightheadedness, abnormal pap, urinary symptoms, menopausal symptoms, endometrial hyperplasia, anemia, and periods longer than 7 days.
Risk factors for missing endometrial information on ultrasound reports
In the univariate analysis, a history of fibroids (OR: 9.64, 95% CI: 1.33–69.7) and fibroids seen on ultrasound (OR: 1.81, 95% CI: 1.19–2.76) had increased odds of having no endometrial information reported. In addition, for most characteristics, the unknown/missing variable was significantly associated with missing endometrial information reporting, such as unknown postmenopausal bleeding (OR: 2.45, 95% CI: 1.76–3.42), unknown enlarged uterus (OR: 2.61, 95% CI: 1.53–4.45), and unknown anemia (OR:2.67, 95% CI: 1.69–4.21) (Supplementary Data S2).
Table 3 shows which risk factors were associated with having no endometrial information reported. Similar to our findings on ET visibility, enlarged uterus (OR: 2.61, 95% CI: 1.53–4.45), history of fibroids (OR: 9.64, 95% CI: 1.33–69.7), and fibroids present on ultrasound (OR: 1.81, 95% CI: 1.19–2.76) were risk factors that were associated with having no endometrial information. Several unknown variables were also associated with missing endometrial information: endometriosis (OR: 0.35, 95% CI: 0.23–0.54), postmenopausal bleeding (OR: 2.45, 95% CI: 1.76–3.42), dizzy/fatigue (OR: 2.11, 95% CI: 0.23–0.54), hyperplasia (OR: 2.31, 95% CI: 1.67–4.21), and anemia (OR: 2.67, 95% CI: 1.69–4.21).
LASSO Results for No Information on Endometrium Reported Compared with Information Available (Visible, Partially, Nonvisible), with Unknown Variables Included
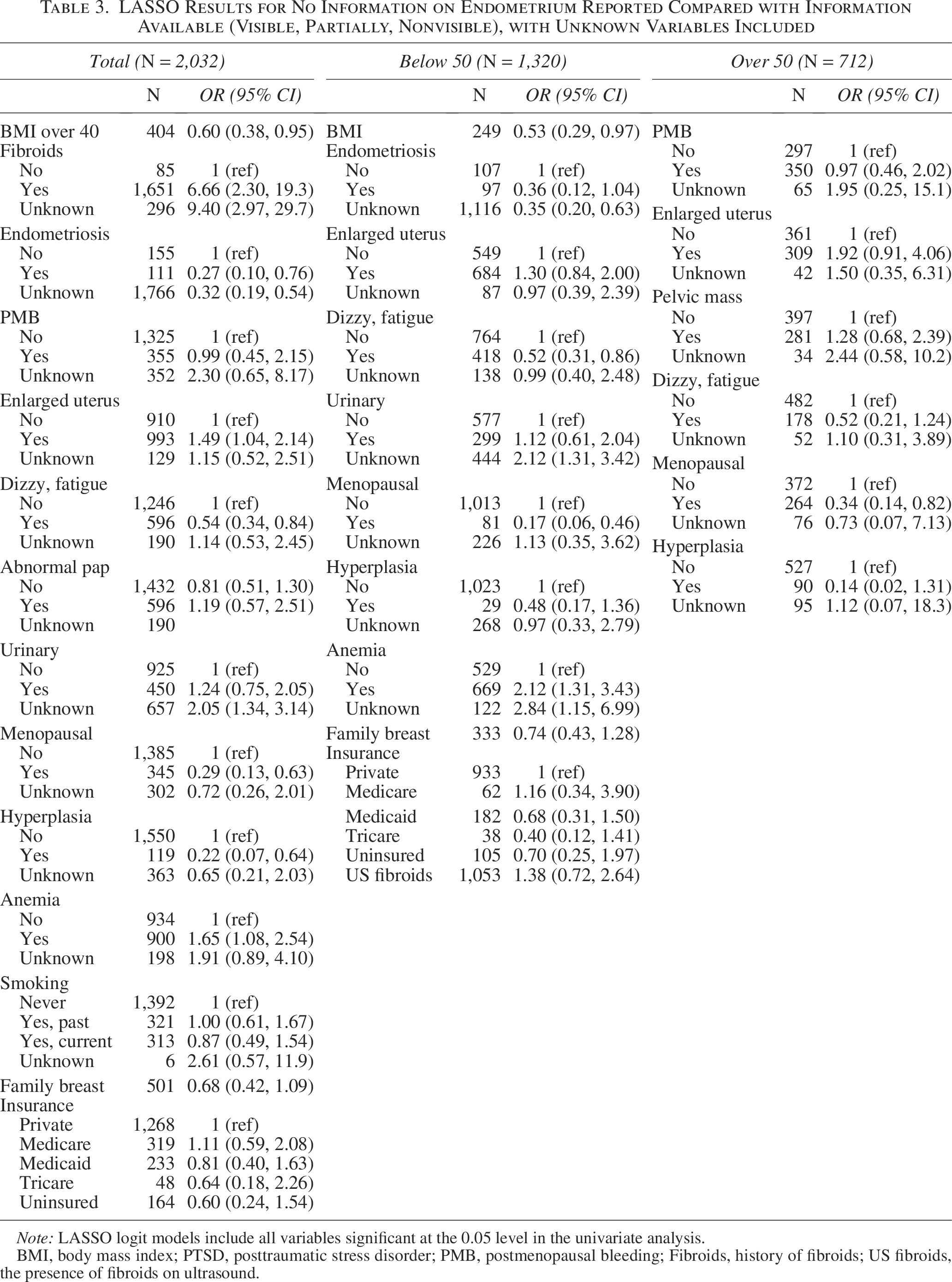
Note: LASSO logit models include all variables significant at the 0.05 level in the univariate analysis.
BMI, body mass index; PTSD, posttraumatic stress disorder; PMB, postmenopausal bleeding; Fibroids, history of fibroids; US fibroids, the presence of fibroids on ultrasound.
When we excluded the unknown variables, history of fibroids (OR: 9.64, 95% CI: 1.33–69.7) and fibroids present on ultrasound (OR: 1.81, 95% CI: 1.19–2.76) were still associated with missing endometrial information on ultrasound reports. However, with our LASSO model, only fibroids on ultrasound in those below 50 years old (OR: 2.07, 95% CI: 1.21–3.56) were associated with missing endometrial information (Table 4).
LASSO Results for No Information Compared with Information Available (Visible, Partially, Nonvisible) without Unknown
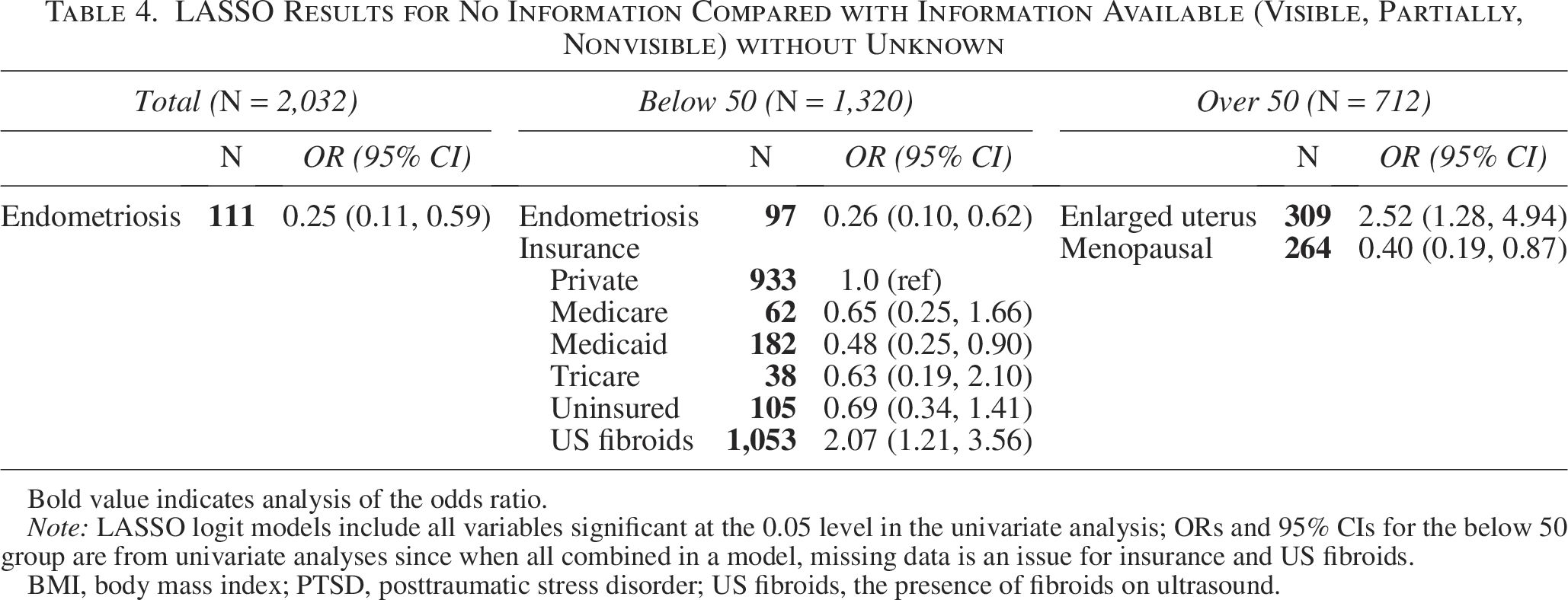
Bold value indicates analysis of the odds ratio.
Note: LASSO logit models include all variables significant at the 0.05 level in the univariate analysis; ORs and 95% CIs for the below 50 group are from univariate analyses since when all combined in a model, missing data is an issue for insurance and US fibroids.
BMI, body mass index; PTSD, posttraumatic stress disorder; US fibroids, the presence of fibroids on ultrasound.
Discussion
We were able to analyze health care information across 10 hospitals and associated health care systems on Black patients who underwent pelvic ultrasound prior to undergoing a hysterectomy for a variety of indications. We assessed the quality of endometrial visibility and endometrial information reporting from these ultrasounds to identify any risk factors that may be associated with a nondiagnostic result with respect to endometrial cancer assessment needs.
Endometrial cancer assessment pathways that utilize TVUS rely on accurate and high-quality ET measurements. Overall, we found that a substantial proportion of ultrasounds had compromised ET measurements due to poor visibility or a lack of endometrial information reporting at all. These represent two distinct but important potential gaps in care in evaluating EC risk.
Nearly one-third of ultrasounds with endometrial information reported partial or nonvisible ET measurements. Current guidelines allow a provider to bypass TVUS and perform tissue sampling, yet over half of providers (54%) preferred TVUS as the next step in the diagnostic workup.15,16 This creates a potential pathway to increased false negative results and missed cancers in Black patients, especially those with uterine fibroids.5,8,17 History of fibroids and fibroids seen in ultrasound predicted compromised visibility, consistent with their known ability to obstruct the view of the endometrium through shadowing caused by their mass and calcifications that can cause brightness that distorts the endometrial echo. 18 An enlarged uterus, independent of fibroid status, was another strong risk factor for poor visibility. Prior studies have investigated the effectiveness of TVUS in assessing the endometrium across pre- and post-menopausal individuals.19–24 The presence of fibroids was most commonly seen in post-menopausal patients, negatively impacting endometrial visualization. Other factors, such as a distorted uterine cavity and/or prior uterine surgeries (such as a D&C), were seen to impact both populations. Though we did not assess prior surgery, the data on fibroids and the uterine cavity are consistent with our findings. We noted that in addition, pelvic mass and anemia were also associated with compromised ET visibility and are common fibroid symptomology 25 Thus, factors suspicious for both fibroids and EC can make the TVUS EC diagnostic process, which relies on accurate ET measurement, less reliable.
Endometrial information is a standard part of pelvic ultrasound reporting, yet we found a small but important proportion of cases missing this vital information entirely. Given the overlap of symptoms with EC and other benign conditions, an unexpected, thickened endometrium may be the first sign of EC concern. Exclusion or omission of this information from ultrasound reports could thus obscure signs that would prompt an EC evaluation. In this data, factors associated with missing endometrial information on ultrasound reports were fibroids and an enlarged uterus, underlying the hesitation that may be needed in employing the TVUS diagnostic pathway in these individuals. Outside of our analyses, our results also demonstrate the commonality in which complete endometrial information is missing from pelvic ultrasound in symptomatic individuals.
There are important limitations to consider when interpreting our study results. For endometrium assessment, we utilized a conservative definition of partial visibility—only counting it when it was expressly reported—which may very well undercount the phenomenon. This was a deliberate choice to give physicians the benefit of the doubt, but it may mean the challenge of ET capture and reporting is underestimated in this study and may be larger in this health care setting. In terms of symptomatology, our population is likely to have many more gynecological symptoms compared to the general population, as all the patients ultimately underwent a hysterectomy. Patients who are less symptomatic may have a different profile of ultrasound reporting quality, though we would imagine that greater severity of symptoms should lead to more, not less, comprehensive reporting. Thus, in this context, the frequency of missing endometrial information we found is especially concerning.
While we identified meaningful associations between risk factors and compromised or nondiagnostic TVUS in terms of EC risk evaluation, these findings may not be generalizable to all potentially symptomatic individuals, especially those who do not go on to require hysterectomy. A quarter of the GUIDE-EC cohort was excluded from the analysis due to a lack of any available ultrasound information in the record, despite having received an ultrasound. Given our broad inclusion category, this often represented an ultrasound received outside the care system with poor coordination and transfer of records. Though we cannot assess the quality of these scans, such gaps highlight the challenges of relying on TVUS as a central component of EC risk evaluation. Additionally, limitations in EHR data capture contributed to incomplete information for some factors, leading to smaller sample sizes, less precise estimates, and wider confidence intervals. EHR data often follows a missing not at random (MNAR) pattern, particularly for symptoms and risk factors that may only be documented when clinically relevant. Thus, such missingness is not amenable to multiple imputations. In addition, it may represent a selection bias where those with the most comprehensively documented risk factor profiles differ meaningfully from those with high rates of missing factor documentation in unmeasured ways. Finally, unmeasured risk factors in this population may be associated with this lack of EHR reporting, potentially influencing our findings.
Conclusion
For patients or clinicians considering TVUS, our findings help identify characteristics associated with a higher likelihood of a nondiagnostic result, supporting a “risk-based” approach. We advocate for incorporating risk factors, such as those that increase the likelihood of compromised endometrial measurements, into the endometrial cancer evaluation process.
Authors’ Contributions
M.A.O.: Writing—original draft; and writing—review and editing; M.P.: Data curation; formal analysis; visualization; and writing—review and editing; R.K.: Conceptualization; formal analysis; and writing—review and editing; W.R.: Conceptualization; investigation; and writing—review and editing; K.M.D.: Conceptualization; writing—review and editing; supervision; project administration; and funding acquisition.
Footnotes
Acknowledgments
The authors would like to thank Julianna Alson and Patrice Williams for assisting with medical abstraction.
Author Disclosure Statement
The authors have no conflicts of interest.
Funding Information
This project was funded by the 2023 Victoria’s Secret Global Fund for Women’s Cancers Rising Innovator Research Grant in Partnership with Pelotonia & AACR.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
