Abstract
Cognitive self-efficacy (CSE), one’s belief in their ability to control their cognitive performance, is important for participation in daily activities and rehabilitation. This study aims to understand how Parkinson’s disease (PD) affects CSE. The Cognitive Self-Efficacy Questionnaire (CSEQ) was administered to 47 non-demented PD and 52 healthy comparison (HC) participants. Groups were compared on their self-reported ability to recognize (Part 1) and manage (Part 2) cognitive symptoms and to perform cognitively complex functional activities (Part 4). Relationships between CSEQ scores and individual characteristics were assessed within PD. The PD group had lower CSEQ scores than the HC group for all Parts. Within PD, Part 2 scores were lower than Parts 1 and 4, and worse depressive symptoms and higher medication dosage correlated with lower CSE. People with PD may have low CSE, which can contribute to participation restrictions and reduced engagement in treatment. Occupational therapists should consider CSE with clients with PD.
Plain Language Summary
Cognitive deficits are common in people with Parkinson’s disease (PD) and affect their quality of life. In this study, the researchers looked at cognitive self-efficacy (CSE) or the belief in one’s cognitive abilities and compared the CSE of healthy individuals with individuals with PD without dementia. The findings of the study suggest that non-demented individuals with PD have lower CSE as compared with healthy individuals. Furthermore, individuals with PD may have difficulty recognizing and managing their cognitive deficits such as memory deficits or distractibility. This may affect their ability to participate in everyday tasks that require complex cognition such as managing finances or shopping or engaging in therapy interventions focused on cognition. The study also found that greater depressive symptoms and higher dose of dopamine medications in non-demented individuals with PD lowered their CSE. This study recommends that rehabilitation professionals include assessments and interventions on CSE during treatment sessions.
Introduction
Parkinson’s disease (PD) is the second most common neurodegenerative disorder affecting older adults, with increasing incidence above the age of 60 years (Chen, 2010; Driver et al., 2009). The number of individuals with PD above the age of 50 years in most countries of the world was estimated to be 4.6 million in 2005, and this number is expected to double by 2030 (Dorsey et al., 2007). PD is associated with motor and non-motor manifestations that contribute to disability, reduced quality of life, and increased direct and indirect health care costs for people with PD, their caregivers, and society (Chen, 2010).
Cognitive impairment is one of the more common non-motor manifestations of PD (Khedr et al., 2013; Riedel et al., 2008). More than 80% of people with PD experience some cognitive decline (Khedr et al., 2013), which can range from mild cognitive deficits to dementia (Lees & Smith, 1983; Tedrus et al., 2009). Cognitive deficits in PD without dementia primarily include problems in memory, executive function, and attentional control (Aarsland et al., 2010; Rodríguez-Ferreiro et al., 2010). These deficits can impair instrumental activities of daily living (IADL), such as driving, and health management occupations, such as medication management, and are associated with carer burden, disability, and reduced quality of life (Chenoweth et al., 2008; Crizzle et al., 2012; Foster & Doty, 2021; Leroi et al., 2012; Uc et al., 2011).
Individuals with PD without dementia may have different perceptions of their cognitive capacity as compared with their actual cognitive performance or to the perceptions of others. For example, some studies have found that people with PD overestimate their executive function and cognitive IADL performance compared with their actual performance when tested (Kudlicka et al., 2013; Shulman et al., 2006). Other studies have found that people with PD underestimate their cognitive capacity compared with cognitive tests and informant reports (Kudlicka et al., 2013; Yoo et al., 2020). While overestimation may reflect decreased awareness or acknowledgment of performance challenges, underestimation may be linked to low self-efficacy or the beliefs one has about their performance capacities (Bandura, 2010, 2012). For example, a person may be aware of performance challenges (i.e., good awareness) yet at the same time believe there is little they can do to cope with or manage challenges when they occur (i.e., low self-efficacy; Toglia & Foster, 2021).
Studies in diagnoses, such as multiple sclerosis, traumatic brain injury, and dementia have found that low self-efficacy is an important factor associated with reduced quality of life (Brands et al., 2014; Schmitt et al., 2014; Tonga et al., 2020) although most studies evaluating self-efficacy have focused on physical functioning. Within the cognitive domain, low levels of self-efficacy have been noted to increase subjective cognitive complaints in clients with acquired brain injury (Brands et al., 2019). Lower self-efficacy has also been correlated to poorer attention, memory, and reaction time in individuals with multiple sclerosis (Jongen et al., 2015). Thus, there is a link between self-efficacy and cognition that warrants further investigation.
Cognitive self-efficacy (CSE) is one’s belief in their ability to use cognitive skills, such as attention, memory, organization, and problem-solving effectively in various situations (Toglia & Kirk, 2000). CSE comprises one’s confidence in their capacity to produce a desired cognitive performance-related outcome. CSE is relevant to everyday cognitive function and cognitive intervention among people with PD because it affects the choice, engagement, and sustainment of participation in cognitively challenging activities (Berry & West, 1993). These activities can include health management, social participation, driving, and work, all of which are important for everyday function among people with PD. Individuals with higher CSE may put forth more cognitive effort in tasks, thereby improving their performance (Berry & West, 1993; Toglia & Kirk, 2000; West et al., 2008). In contrast, those with lower CSE may avoid or give up on tasks they perceive to be above their cognitive performance capacity (Bandura, 1994), which may lead to reduced performance and participation in daily activities. In individuals with mild cognitive impairment (MCI), CSE has also been shown to affect a person’s ability to cope with the disease process after being diagnosed (Tonga et al., 2020). Relevant for occupational therapy treatment, low CSE can limit the person’s ability to engage in and benefit from interventions aimed at improving their cognitive performance. If a person does not believe that they have the capacity to control their cognitive performance problems (i.e., low CSE), they may not fully engage in treatments designed to do so (Toglia & Foster, 2021).
Despite its potential importance for daily function and clinical practice, CSE has rarely been studied. Memory self-efficacy specifically has been investigated in the context of healthy aging and MCI and found to be positively associated with memory performance (Kurasz et al., 2021; Stolder, 2012). However, studies of self-efficacy for executive function, other cognitive domains, functional cognition, or the management of cognitive symptoms are lacking, especially in clinical populations. This may be due to a lack of assessments to measure CSE. One study that investigated CSE in people with traumatic brain injury found that self-efficacy for the management of cognitive symptoms was the greatest single contributor to life satisfaction, after accounting for a number of demographic and injury-related variables, community functioning, and activity-related satisfaction (Cicerone & Azulay, 2007). These findings highlight the need for a more comprehensive understanding of CSE and its implications for rehabilitation treatment and outcomes in clinical populations at risk of cognitive dysfunction such as PD.
The purpose of this study was to investigate CSE among people with PD. We administered the Cognitive Self-Efficacy Questionnaire (CSEQ; Toglia & Johnston, 2017), which assesses beliefs regarding the ability to recognize and manage cognitive symptoms and perform cognitively complex activities, to non-demented individuals with PD and healthy older adult participants. In addition, we investigated the relationships between demographic and PD-related characteristics and CSE among people with PD.
Method
Participants
Participants were volunteers aged 50 to 75 years with PD without dementia recruited from a university movement disorders center in the Midwestern United States and age-matched healthy comparison (HC) participants recruited from the Greater New York City area and suburbs.
The PD participants met criteria for typical idiopathic PD, within Hoehn and Yahr Stages I to III (Hoehn & Yahr, 1967), and were being treated with and experienced clear motor benefit from levodopa–carbidopa. Exclusion criteria comprised suspected dementia or global cognitive impairment, as determined by physician, caregiver report, a score <27 on the Mini-Mental Status Examination (MMSE; Folstein et al., 1975), other neurological conditions (e.g., stroke, brain injury), brain surgery, history of or current psychotic disorder, significant current psychiatric symptoms (e.g., severe depressive symptoms, apathy, and sudden changes in emotions or mood), inability to understand or speak English, or any condition that would practically interfere with testing (e.g., low vision, severe dyskinesias).
The HC participants were community-dwelling and independently functioning adults. Exclusion criteria comprised suspected dementia or global cognitive decline (determined by a score >4 on the Short Blessed Test (Katzman et al., 1983), diagnosed neurological or psychiatric disorders, visual or auditory perceptual deficits that would interfere with participation, and inability to understand written or spoken English.
Research Design and Procedure
This was a cross-sectional analysis of data derived from two existing studies. The research was approved by the institutional review board of each university and all participants provided informed consent prior to participation. Demographic characteristics (e.g., age, sex, and gender) for all participants were obtained by interview. The PD participants’ disease-related characteristics (e.g., Hoehn & Yahr stage, Unified Parkinson’s Disease Rating Scale [UPDRS] motor subscale score [Fahn et al., 1987], medications, and disease duration) were obtained by interview and clinical records, and levodopa equivalent daily dose (LEDD) was calculated for each PD participant. All participants completed the CSEQ (described in the following), and the PD participants additionally completed the Beck Depression Inventory–II (BDI-II; Beck et al., 1961), to assess depressive symptoms, and the MMSE, to screen for global cognitive impairment.
Measure
The CSEQ (Toglia and Johnston, 2017): The CSEQ (available for free here: https://multicontext.net) includes four parts that can be used separately as well as together. In this study, three parts of the questionnaire were used to assess participants’ beliefs regarding their ability to recognize (Part 1) and manage (Part 2) cognitive symptoms and to perform cognitively complex functional activities (Part 4). Part 1 includes 16 items that assess confidence in the ability to recognize cognitive symptoms as they are occurring within an activity (e.g., “In mentally challenging activities or tasks I am sure that I can . . . Recognize when I forget something; Recognize when I become distracted; Recognize the first signs of mental fatigue”). Part 2 includes 19 items that assess confidence in the ability to deal with cognitive challenges (e.g., “If a task is mentally challenging, I am sure that I can . . . Think of methods to make the activity easier; Persist or keep going, even when things become difficult; Figure out ways to reduce distractions if needed”). Part 4 includes 23 items that assess confidence in the ability to perform everyday cognitively demanding tasks (e.g., “How certain are you that you can . . . Remember to pay your bills on time; Follow a conversation with a group of 5 people; Plan and organize a weekend trip”). Item scores range from 0 to 10 (0 = cannot do at all, 5 = moderately certain can do, 10 = highly certain can do) and are averaged within each Part for analysis. Each part had excellent internal consistency reliability (Cronbach’s alpha) in a sample of 200 healthy control participants (Part I = .93, Part 2 = .96, and Part 4 = .93; Toglia, n.d.).
Statistical Analysis
Descriptive statistics were calculated for all variables. The Kolmogorov–Smirnov test and visual inspection were used to verify normality. A mixed ANOVA (analysis of variance) with planned pairwise comparisons was used to compare CSEQ scores for each part within and across participant groups. Bivariate correlations (Pearson’s r) were used to assess the relationships between CSEQ scores and demographic and disease-related characteristics within the PD group.
Results
Participant Characteristics
The study included 99 total participants (47 PD, 52 HC). Demographic and clinical characteristics of the sample are in Table 1. The groups differed on sex and work status distributions, such that the PD group had proportionately more males and people who were not working, χ2 ≥ 4.34, p < .04.
Demographic and Clinical Characteristics of the Sample.
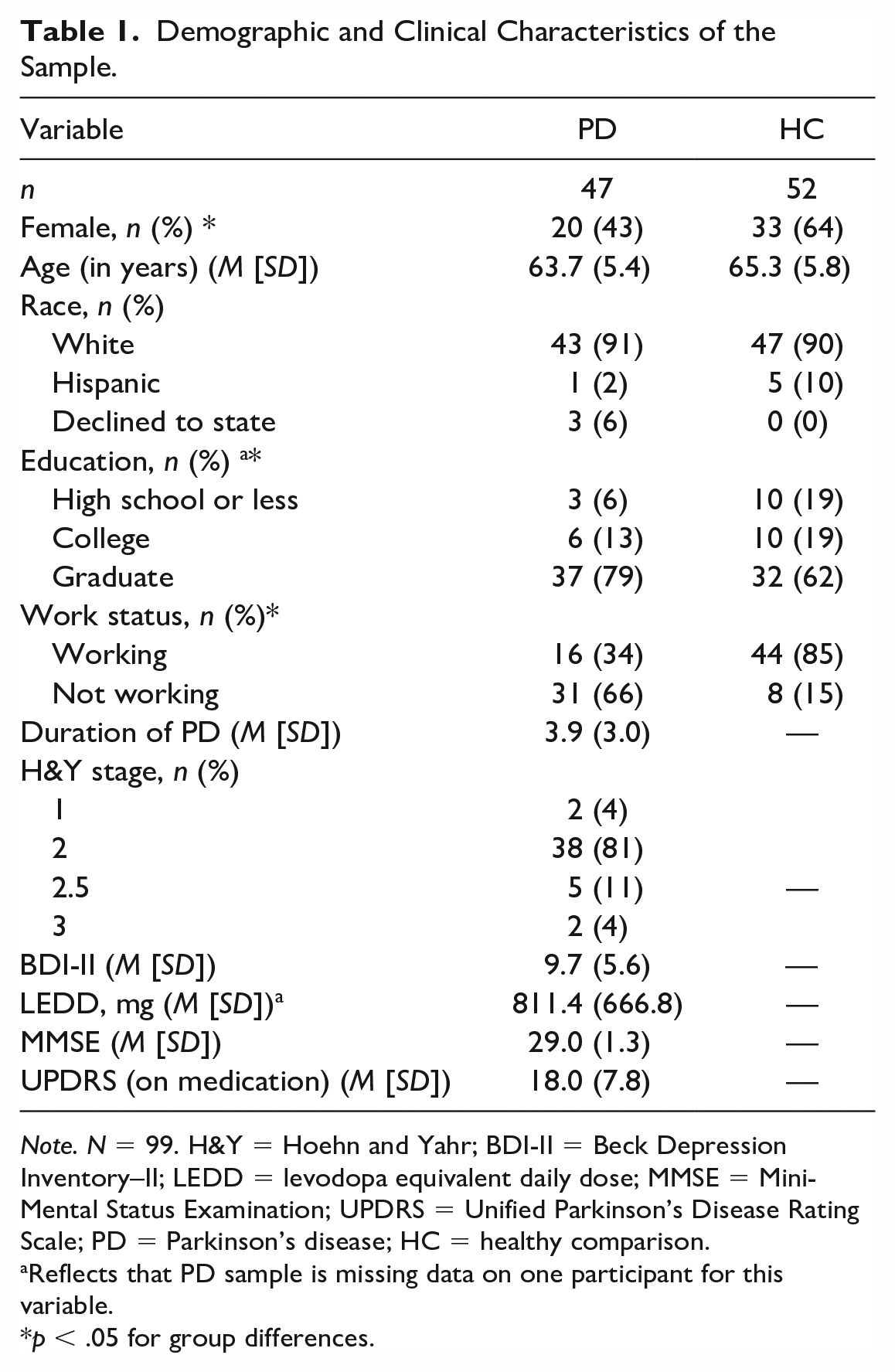
Note. N = 99. H&Y = Hoehn and Yahr; BDI-II = Beck Depression Inventory–II; LEDD = levodopa equivalent daily dose; MMSE = Mini-Mental Status Examination; UPDRS = Unified Parkinson’s Disease Rating Scale; PD = Parkinson’s disease; HC = healthy comparison.
Reflects that PD sample is missing data on one participant for this variable.
p < .05 for group differences.
Group Comparison of CSE
Group CSEQ scores are in Figure 1. Parts 1 and 2 were normally distributed, and Part 4 was slightly negatively skewed (p = .03). There was a main effect of group indicating that the PD group reported lower CSEQ scores than the control group on every part, F(1, 83) = 12.79, p = .001. There was also a main effect of CSEQ Part, F(2, 166) = 4.24, p = .016, such that Part 1 scores were higher than Part 2 scores, p = .002. However, this main effect was qualified by an interaction, F(2, 166) = 4.06, p = .019. Planned pairwise comparisons indicated that there were no differences between the CSEQ Parts in the HC group, but in the PD group, Part 2 was lower than Parts 4 and 1, p < .001 (which were equivalent to each other; Figure 1). After rerunning this analysis, controlling for work status and sex, the results remained the same.
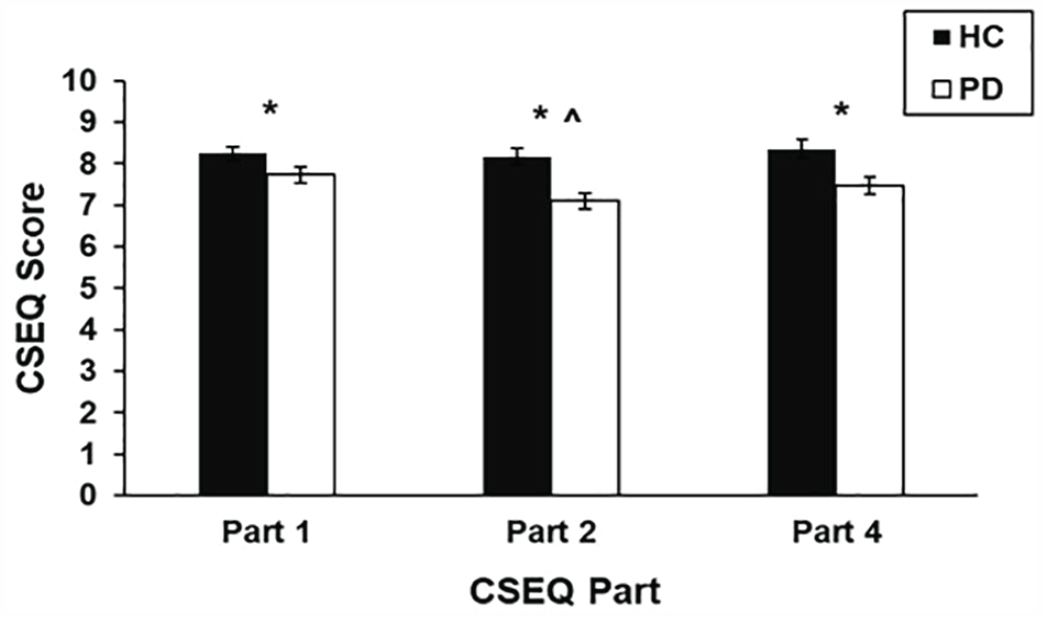
Group Scores (Estimated Marginal Means) for Each Part of the CSEQ
Factors Associated With CSE in PD
Within the PD group, BDI-II correlated with each of the CSEQ parts, r < –.30, p < .04, such that higher depression scores (worse depressive symptoms) related to lower CSE. LEDD correlated with CSEQ Part 4, r = –.44, p = .003, such that higher daily dopaminergic medication dosage related to lower confidence in the ability to perform cognitively complex functional tasks. There were no correlations between any of the other characteristics (e.g., age, education, motor dysfunction, and cognition) and the CSEQ, p > .10.
Discussion
The purpose of this study was to understand how PD affects CSE. To this end, we used the CSEQ to compare the CSE of participants with PD without dementia with age-matched healthy older adults and to examine relationships of CSE with PD-related characteristics. Both groups had relatively high CSE, and the PD group’s scores were only slightly below average compared with normative data (Toglia, n.d.). These findings are not surprising, given that the study sample comprised independent community-dwelling adults with normal cognition. However, despite overall high CSE, there were significant differences between the groups in CSE and among aspects of CSE within the PD group, and significant relationships between CSE and PD-related characteristics in the PD group. The findings can help guide future studies on cognition in PD as well as clinical assessment and intervention for this population.
We found that, even with generally intact cognition (as per the MMSE), non-demented people with PD have lower self-efficacy for their ability to recognize cognitive symptoms, manage cognitive symptoms, and perform cognitively complex daily activities compared with their healthy counterparts. Although the MMSE may lack some sensitivity to cognitive decline in PD (Mamikonyan et al., 2009), these findings are consistent with other studies demonstrating reduced functional cognition despite intact cognitive test performance in non-demented individuals with PD (Davis et al., 2019; Foster et al., 2014, 2022). Our study reinforces this pattern and extends it to CSE. Low CSE may be an outcome of cognitive decline in PD and, similar to performance-based assessments of functional cognition or measures of subjective cognitive decline, may be more useful in the early detection of cognitive decline in this population compared with traditional cognitive tests. Alternatively, low CSE may contribute to cognitive decline through its negative effects on the initiation, effort, or persistence of cognitively challenging tasks. This study was not designed to determine the causal relationship between cognitive decline and CSE; however, it does suggest that CSE is important to assess in those who are at risk of or who may have subtle cognitive difficulties. Furthermore, it contributes to establishing the validity of the CSEQ for this purpose by showing that it can detect differences in CSE between known groups.
The PD participants had particularly low self-efficacy for their ability to manage cognitive symptoms (CSEQ Part 2). These differences were not observed in the healthy control sample. Our findings suggest that, although non-demented people with PD may recognize cognitive problems, they are not confident in their ability to prevent them from occurring or to deal with them when they occur. Recognizing a problem, yet believing you have little control over managing it, can contribute to emotional distress (Bandura, 2012) as well as lower self-esteem and decreased sense of hope (Tonga et al., 2020). In addition, it can render one less likely to seek treatment or more likely to avoid cognitively challenging tasks (leading to further decline) or quickly give up when difficulty is experienced. Self-efficacy for managing cognitive symptoms is a strong independent predictor of quality of life and life satisfaction (Cicerone & Azulay, 2007). Critically, this highlights the need for interventions that target CSE for people with PD and focus on improving people’s confidence and sense of control in their ability to manage daily cognitive challenges. It also emphasizes the need to assess CSE when treating clients at risk of cognitive deficits and supports the usefulness of the CSEQ in doing so.
Among the PD participants, higher depressive symptoms related to poorer CSE. These findings are consistent with prior studies in people with chronic diseases, including PD, in which lower self-efficacy correlates with higher depression levels (Brands et al., 2019; Gruber-Baldini et al., 2017; Shulman et al., 2019; Tonga et al., 2020). In addition, higher dopaminergic medication dosage related to poorer self-efficacy in the ability to perform cognitively complex functional tasks. Dopaminergic medication may be an indicator of disease severity, so this finding could reflect a true decline in functional cognitive performance as the disease progresses, a decline only in CSE, or both. Future studies are needed to disentangle the relationship between functional cognitive decline and CSE.
Due to its exploratory nature, this study has several limitations that constrain the interpretation and generalizability of our results and which must be addressed in future work. Due to the lack of assessment of functional cognitive performance, we do not know whether participants’ CSE is consistent with their actual cognitive performance. As mentioned previously, the MMSE may not detect subtle cognitive deficits in PD, so we cannot be completely confident that our PD group had no cognitive decline; however, the high standard for study inclusion (MMSE score ≥27) and high average score in our sample rule out dementia and likely also PD-related mild cognitive impairment (PD-MCI; Litvan et al., 2012). In addition, although the group differences in sex and work status distributions did not appear to influence the results, future studies should ensure that groups are matched on these characteristics. Finally, future work should include PD participants across the spectrum of disease severity and with MCI to gain a more comprehensive understanding of the effect of PD on CSE.
Cognitive impairment is now understood to be critical to the function and quality of life of people with PD, and there are increasing calls for cognitive rehabilitation interventions to address it (Foster et al., 2021; Kalbe et al., 2018; Toglia & Foster, 2021; Walton et al., 2017; Wood et al., 2022). To develop effective cognitive interventions for people with PD, we need a thorough understanding of factors that affect not only functional cognitive performance but also the process of cognitive rehabilitation in this population. CSE is one such factor that may play a central role in both of these outcomes as it may influence whether someone engages or persists in cognitively challenging tasks in their daily life and whether they seek out or actively participate in cognitive rehabilitation. Interventions that allow a person to experience success and mastery and that focus on understanding methods to increase self-management of cognitive performance (e.g., the Multicontext Approach) may be particularly helpful in building CSE and should be considered and explored with this population (Toglia & Foster, 2021).
To summarize, we found that non-demented people with PD have lower self-efficacy than healthy older adults for the ability to recognize and manage cognitive symptoms and to perform cognitively complex daily activities. Self-efficacy for the management of cognitive symptoms is particularly low among non-demented people with PD. In addition, CSE is associated with other PD-related characteristics, such as depressive symptoms and disease severity. This study represents an initial step in understanding the effect of PD on CSE. Future studies are needed to better understand the relationships among CSE, functional cognitive performance, and other disease-related characteristics in this population. Ultimately, this work will inform the use of assessments and interventions that address CSE to improve or maintain functional cognitive performance and participation.
Footnotes
Research Ethics and Patient Consent
Approval was granted by Mercy College Institutional Review Board in Reference No. 10-72; Washington University in St. Louis Human Research Protection Office Nos. 201103176, 201109224, and 201403072.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: P.M.J. and E.R.F. report no conflicts of interest. J.T. is a coauthor of the Cognitive Self-Efficacy Questionnaire used in this study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
