Abstract
Many individuals post-stroke have difficulty identifying if or how they can continue performing meaningful daily life tasks. The objective of this study was to evaluate the feasibility of metacognitive strategy training (MCST) and transcranial direct current stimulation (tDCS) in chronic stroke. A case series design was used. Participants completed 12 intervention sessions over 4 weeks consisting of 20 min of tDCS and 45 min of MCST to address occupational performance goals. Feasibility outcomes of acceptability and safety/tolerability were evaluated and measures of occupational performance were administered pre- and post-intervention. Participants perceived the intervention to be highly acceptable and relevant to their needs. Large improvements were observed for performance and satisfaction with goals trained (Hedge’s g = 2.07 and 2.11, respectively) and untrained (Hedge’s g = 1.25 and 1.43, respectively) within the intervention. An intervention combining MCST with tDCS was feasible to administer and positively received by stakeholders; further research is warranted.
Keywords
Introduction
Despite the high incidence and economic impact of stroke, current rehabilitation methods fall short in effectiveness with almost 60% of meaningful life activities discontinued after stroke (Hartman-Maeir et al., 2007). Discontinued activities typically include basic self-care, instrumental activities of daily living, leisure, and work; collectively, these changes in participation contribute to decreased quality of life (Hartman-Maeir et al., 2007; Mayo et al., 2002; Singam et al., 2015). In contrast to commonly used impairment-based stroke interventions, metacognitive strategy training (MCST) can be used to directly address occupational performance. Cognitive Orientation to daily Occupational Performance (CO-OP), a type of MCST, uses a problem-solving approach to equip participants with the ability to apply cognitive strategies within everyday activities (Dawson et al., 2017). CO-OP focuses on learning of a global problem-solving strategy, Goal-Plan-Do-Check (GPDC). Use of a broadly applicable strategy increases likelihood of skill transfer to other environments and tasks. Skill transfer enables client independence in addressing the variety of occupational performance difficulties that arise post-stroke (Geusgens et al., 2007; Verberne et al., 2018).
Current evidence supports CO-OP as an effective intervention for improving occupational performance in the stroke population (Henshaw et al., 2011; McEwen et al., 2015; Polatajko et al., 2012). Prior CO-OP literature has demonstrated improvement on not only activities that were directly addressed within the intervention (i.e., trained goals), but also those not explicitly addressed (i.e., untrained goals) (McEwen et al., 2015). These observed improvements in untrained goal performance provide evidence of skill transfer. In alignment with the principles of neuroplasticity, CO-OP uses executive processes repetitively within meaningful occupations to drive new skill learning and neural reorganization.
There is strong evidence to support the use of CO-OP in chronic stroke (Henshaw et al., 2011; McEwen et al., 2015; Polatajko et al., 2012), yet there is theoretical support that neural stimulation may amplify the positive effect of CO-OP (Bolognini et al., 2009). Transcranial direct current stimulation (tDCS) is a non-invasive brain stimulation method of modulating cortical excitability through application of low currents through targeted scalp regions (Bolognini et al., 2009).
By affecting resting membrane potential, tDCS makes it more likely (i.e., toward depolarization) or less likely (i.e., toward hyperpolarization) that a neuron, or group of neurons, will initiate an action potential (Lefaucheur et al., 2017; Nitsche & Paulus, 2000). The ability to strategically affect neuronal activity may be used to influence neuroplasticity, and thus rehabilitation outcomes, in clinical populations such as stroke (Boonzaier et al., 2018; Kang et al., 2016; Yan et al., 2020). tDCS possesses high clinical utility as it is safe when applied within certain parameters, inexpensive, transportable, and relatively simple to apply with proper training (Bikson et al., 2016; Woods et al., 2016). Systematic reviews provide support for the use of tDCS for long-term improvement of motor and cognitive performance after stroke, but only when coupled with a behavioral training component (e.g., upper extremity motor training, computer-based cognitive training) (Kandel et al., 2012; Park et al., 2013). To date in stroke populations, tDCS has only been paired with impairment-based interventions that have a primary target of improving a specific process with an assumption that there will also be accompanied by observable improvements in occupational performance or life participation.
Although there are known positive effects of CO-OP and tDCS to separately improve outcomes post-stroke, tDCS has never been used to augment a functional intervention such as CO-OP that directly addresses occupational performance. Neuroplasticity is known to be driven by repeated engagement in appropriately challenging experiences that are meaningful, salient, and highly specific to the learning goal, such as the training encountered in CO-OP (Kleim & Jones, 2008). Preliminary evidence for neuroplastic changes underpinning behavioral changes as a result of CO-OP exists in individuals with cancer-related cognitive impairment (CRCI). Specifically, in a single-group study in women treated for breast cancer experiencing CRCI, positive increases in neural connectivity were observed in cognitive control networks measured with resting-state functional magnetic resonance imaging (fMRI) (Wolf et al., 2016). Importantly, changes in functional connectivity were moderately correlated with changes in cognitive performance.
There have been repeated calls for multimodal rehabilitation interventions that may have an interactive and summative effect (Bolognini et al., 2009; Craig et al., 2013; Takeuchi & Izumi, 2015). When exploring a new intervention, particularly a multicomponent intervention, the first recommended phase of intervention development is feasibility evaluation (Bowen et al., 2009; Craig et al., 2013; Lawson et al., 2022). Use of a small-scale, feasibility design early in the research process can assist in determining if further research is pragmatic and inform intervention refinement prior to large-scale deployment. Intervention acceptability, a core component of feasibility, provides information such as if the intervention addresses pertinent needs of the target population and if the intervention format is appropriate. Although tDCS safety and tolerability is well documented in the literature, continuous evaluation of these important outcomes is recommended (Bikson et al., 2016). Therefore, the purpose of this study is to evaluate the feasibility of combining CO-OP with tDCS and specifically to evaluate acceptability and to complete limited-efficacy testing.
Method
This study used a prospective case series design (n = 7) to evaluate the feasibility of combined use tDCS and CO-OP in the stroke population. In addition to feasibility outcomes, the preliminary effect was measured through administration of an outcome battery pre- and post-intervention. This study was reviewed and approved by the University of Missouri Institutional Review Board, and all participants provided written informed consent. Each participant received three CO-OP + tDCS intervention sessions per week for 4 weeks (12 total sessions). All study procedures were completed in a research laboratory at the University of Missouri.
Participants
Participants with chronic stroke were recruited through the University of Missouri Stroke Registry and from the community in the central Missouri region. Individuals in the stroke registry meeting diagnosis and age requirements were contacted via telephone to determine interest and to complete a pre-screening survey as appropriate. Individuals were also recruited from the community via recruitment flyers and word of mouth. Participants met the following inclusion criteria: (a) at least 6 months post-ischemic stroke, (b) between the ages of 50 and 85, (c) not currently receiving occupational therapy services, (d) residing in the community, and (e) a minimum of four self-identified occupational performance goals on the Canadian Occupational Performance Measure (COPM; Law et al., 2014). Participants were excluded according to the following criteria: (a) severe depressive symptoms (≥20 on Patient Health Questionnaire (PHQ-9) (Kroenke et al., 2001), (b) dementia symptoms as indicated by a score of <24 on the Montreal Cognitive Assessment (Nasreddine et al., 2005; Sweet et al., 2011), (c) any additional self-reported neurological or psychological disorders other than stroke (e.g., major depressive disorder, Parkinson’s Disease), (d) moderate or severe aphasia as indicated by an National Institutes of Health Stroke Scale (NIHSS) (Brott et al., 1989) aphasia score of ≥2, or (e) tDCS contraindications (i.e., pregnancy, history of seizures, metallic implants above the chest, history of welding/metal work, or implanted medical devices).
Data Collection
Feasibility outcomes align with two key feasibility areas outlined by Bowen and colleagues (2009): acceptability and limited-efficacy testing. Furthermore, the outcome safety and tolerability were evaluated as feasibility outcomes in accordance with tDCS recommendations (Bikson et al., 2016). To evaluate feasibility outcomes of acceptability and appropriateness of the intervention, we used standardized assessments following intervention completion (i.e., Client Satisfaction Questionnaire-8 [CSQ-8], Acceptability of Intervention Measure, Intervention Appropriateness Measure, and Feasibility of Intervention Measure) (Attkisson & Zwick, 1982; Weiner et al., 2017). For feasibility outcomes of safety and tolerability, adverse events and experiences of the individual during brain stimulation were tracked during each intervention session using the tDCS Sensations Survey (Fertonani et al., 2015). Any adverse events experienced outside of intervention sessions were also tracked.
For estimation of preliminary effect, objective (i.e., Performance Quality Rating Scale (Martini et al., 2015)) and self-report (e.g., COPM; Law et al., 2014) measures of occupational performance were administered pre- and post-intervention. See Table 1 for further details about assessments used. All screening, feasibility, and outcome assessments used in this study have been found to be reliable and valid with supporting citations provided in Table 1.
Study Measures.
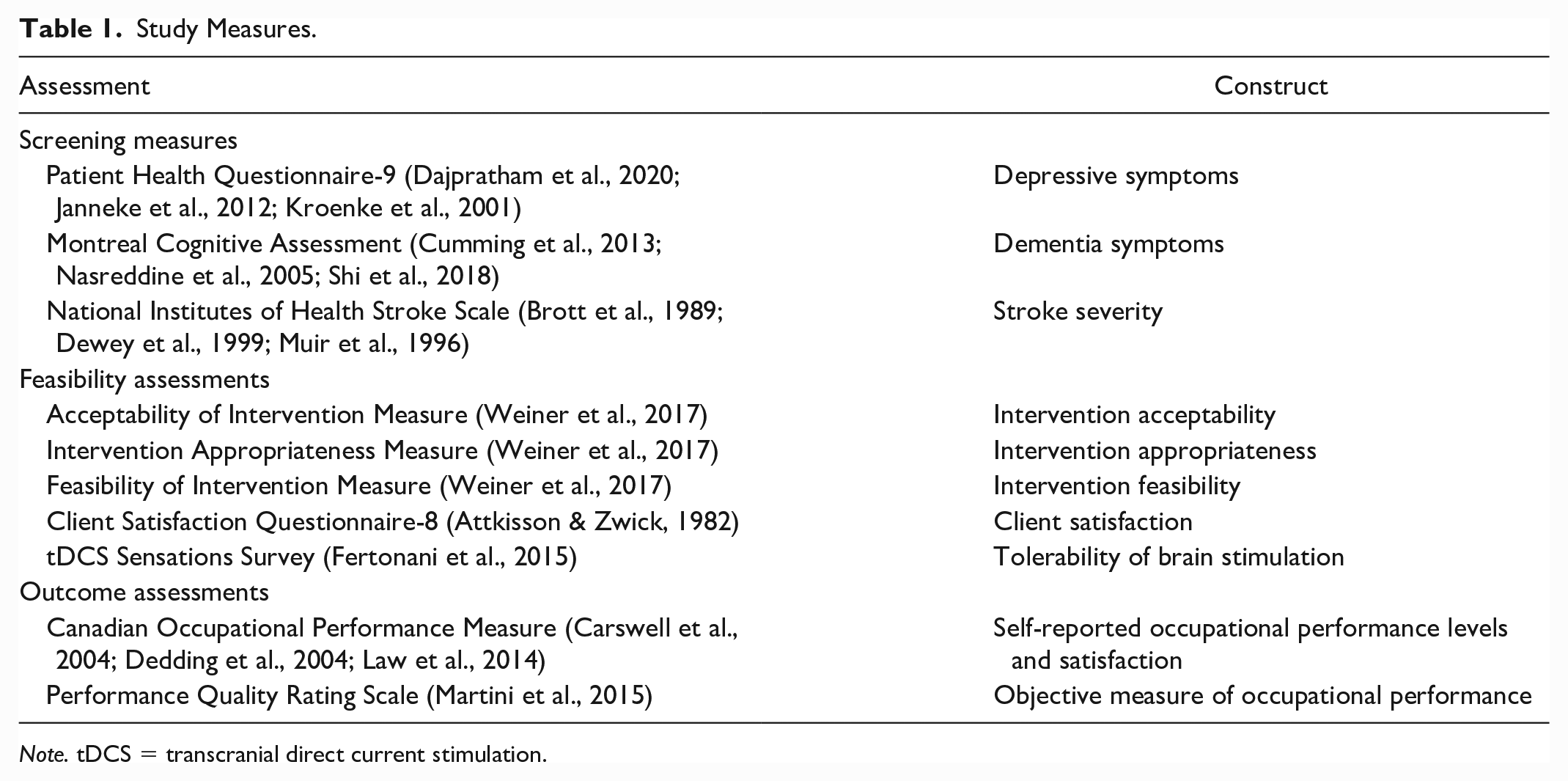
Note. tDCS = transcranial direct current stimulation.
Intervention
Participants completed three 1-hr 15-min sessions per week for 4 weeks for a total of 12 sessions. Each session began with 20 min of offline tDCS followed by 45 min of CO-OP. tDCS requires multiple sessions within a shorter time frame to enable cumulative effects and to ensure there is not a washout of effects between sessions (Lefaucheur et al., 2017); therefore, a higher rate of sessions was used in this study as compared with typical CO-OP format. Additional details related to CO-OP and tDCS are provided in upcoming sections.
Transcranial Direct Current Stimulation (tDCS)
Each session began with the 20 min of anodal tDCS with 1.5 mA current. Using the 10–20 EEG electrode system to identify location, the anode was placed on the ipsilesional dorsolateral prefrontal cortex (DLPFC) and the cathode was placed over the contralesional DLPFC. Electrodes were encased in two saline soaked sponges that were secured on the scalp. A Transcranial Direct Current Stimulator Clinical Trials (tDCS-CT) Model 1500 manufactured by Soterix was used. Although the functional networks engaged in executive functioning are known to be distributed throughout the brain, the prefrontal cortex generally and the DLPFC specifically are known to be hubs of this neural circuitry (Dosenbach et al., 2006, 2007). Bihemispheric tDCS over the DLPFC may help correct the interhemispheric imbalance that follows stroke in which it is thought that the contralesional hemisphere inhibits the already reduced activity within the ipsilesional hemisphere (Goodwill et al., 2016; Kubis, 2016; Perrotta et al. 2021). During the stimulation, all participants watched an animal documentary in effort to create consistency of environmental stimuli across participants.
Cognitive Orientation to Daily Occupational Performance (CO-OP)
CO-OP is a functional, problem-solving approach to improving occupational performance (Dawson et al., 2017). In this study, participants identified five participant goals using the COPM. Intervention directly addressed four of these goals and one was never discussed so that goal could serve as a measure of skill transferability. Therapists facilitated client learning with a guided discovery process through methods such as probing questioning (e.g., questions posed from broad to gradually more specific as needed) or modeling.
A key feature of CO-OP is the “Goal-Plan-Do-Check” process. First, the participant identifies a Goal to address. A Plan is established for improving performance of the goal and subsequently carried out (Do). Following the Do phase, the participant reflects on plan execution and determines plan success and whether further modifications are needed (Check). Participants applied the Goal-Plan-Do-Check process in the first session, and then therapists prompted participants to apply Goal-Plan-Do-Check to each trained goal with progressive levels of independence.
Analysis
Researchers used IBM SPSS Statistics (Version 27; IBM Corporation, Armonk, NY) for all analyses. For feasibility outcomes (e.g., acceptability, appropriateness, satisfaction), means and standard deviation of total scores were calculated. Frequency of adverse effects are reported using the tDCS Sensations Survey (e.g., tolerability, safety). For estimation of preliminary effect (i.e., occupational performance outcomes), researchers first checked the data for normal distribution with the Shapiro-Wilk test. For each variable, mean change and standard deviations are reported as well as Hedge’s g effect sizes with 95% confidence intervals. We used Hedge’s g as it is a more conservative estimate when using small sample sizes (Borenstein et al., 2021). Hedge’s g values are interpreted according to the following criteria: 0.2 = small, 0.5 = medium, and 0.8 = large (Cohen, 1988).
Results
Seven individuals meeting inclusion/exclusion criteria participated in this study. On average, the sample was middle-aged and white with a few years of college education (Table 2). The group was approximately 2 years post-stroke, with a range of 8 to 89 months post-stroke. Participants demonstrated low levels of depressive symptoms and mild cognitive impairment.
Participant Characteristics.
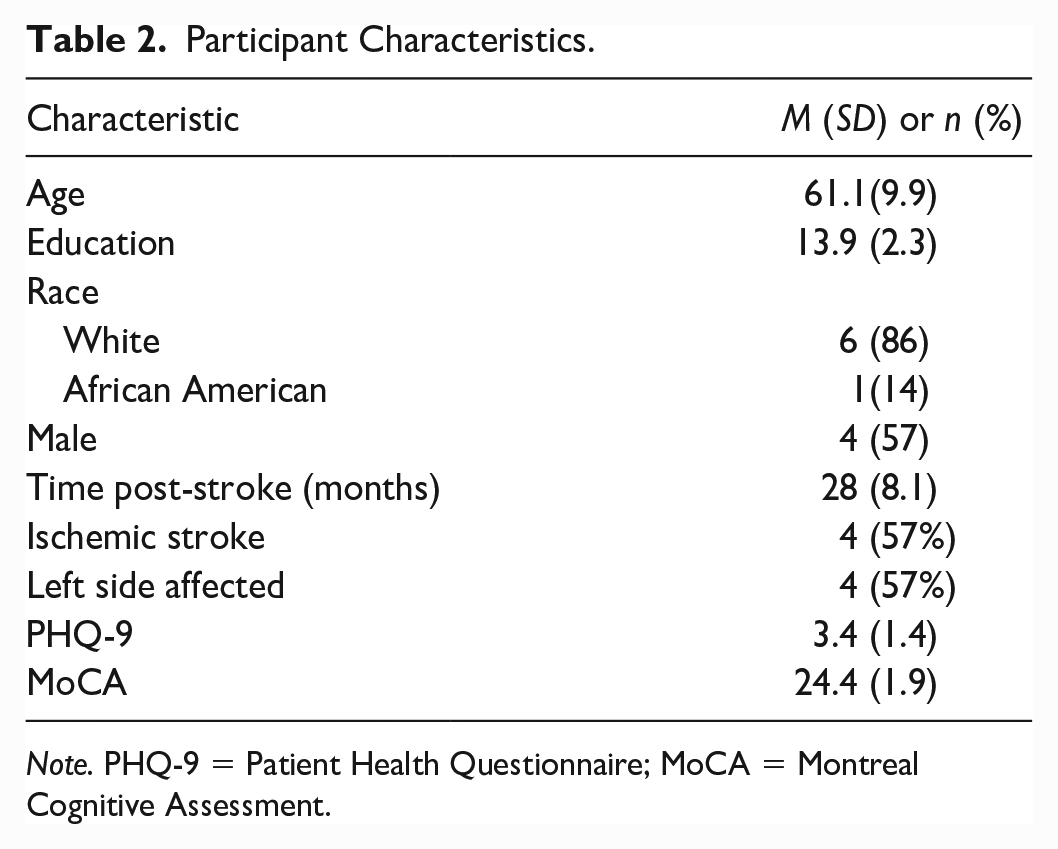
Note. PHQ-9 = Patient Health Questionnaire; MoCA = Montreal Cognitive Assessment.
Feasibility
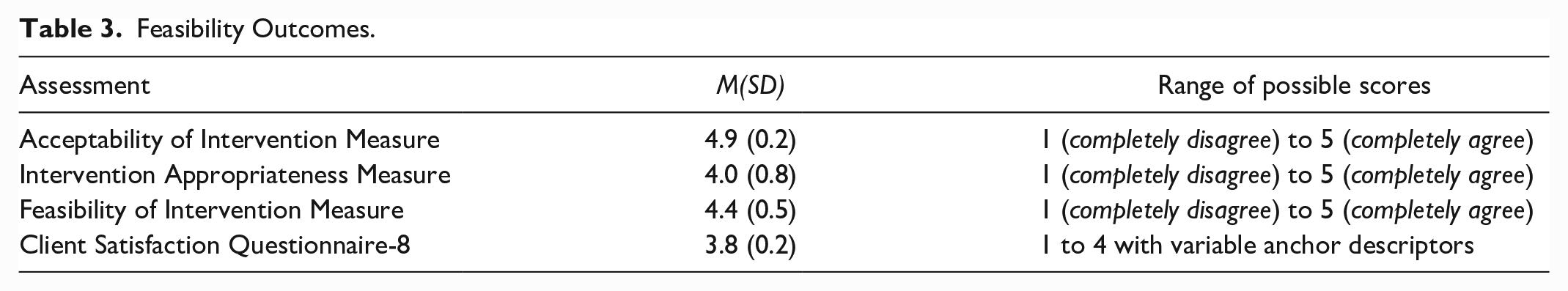
Participants perceived the intervention, specifically, the non-invasive brain stimulation component, as having good tolerability, and there were no adverse events. However, participants experienced mild tingling and itching sensations on the scalp during stimulation. Participants reported high levels of acceptability and appropriateness of the intervention as demonstrated by an average rating of 4.9 and 4.0 out of 5 on the Acceptability of Intervention Measure and Intervention Appropriateness Measure, respectively, and a score of 3.8 out of 4 on the CSQ-8. Furthermore, the intervention was perceived as highly feasible by participants with a score of 4.4 out of 5 on the Feasibility of Intervention Measure (Table 3).
Feasibility Outcomes.
Occupational Performance Outcomes
Table 4 contains means, standard deviations, and Hedge’s g values. Figure 1 illustrates these scores for pre- and post-testing. Participants experienced large improvements in performance (Hedge’s g = 2.07) and satisfaction (Hedge’s g = 2.11) with performance on goals directly trained within intervention per COPM measurements. For goals that were not trained or discussed over the course of the intervention, similar large effects occurred for performance level (Hedge’s g = 1.25) and satisfaction level (Hedge’s g = 1.43). A greater change was noted for trained goals as compared with untrained goals for perceptions of performance and satisfaction. A positive, moderate effect was shown for trained goals on the Performance Quality Rating Scale (PQRS) (Hedge’s g = 0.65) and a positive, small effect for untrained goals (Hedge’s g = 0.25).
Results of Outcome Assessments.
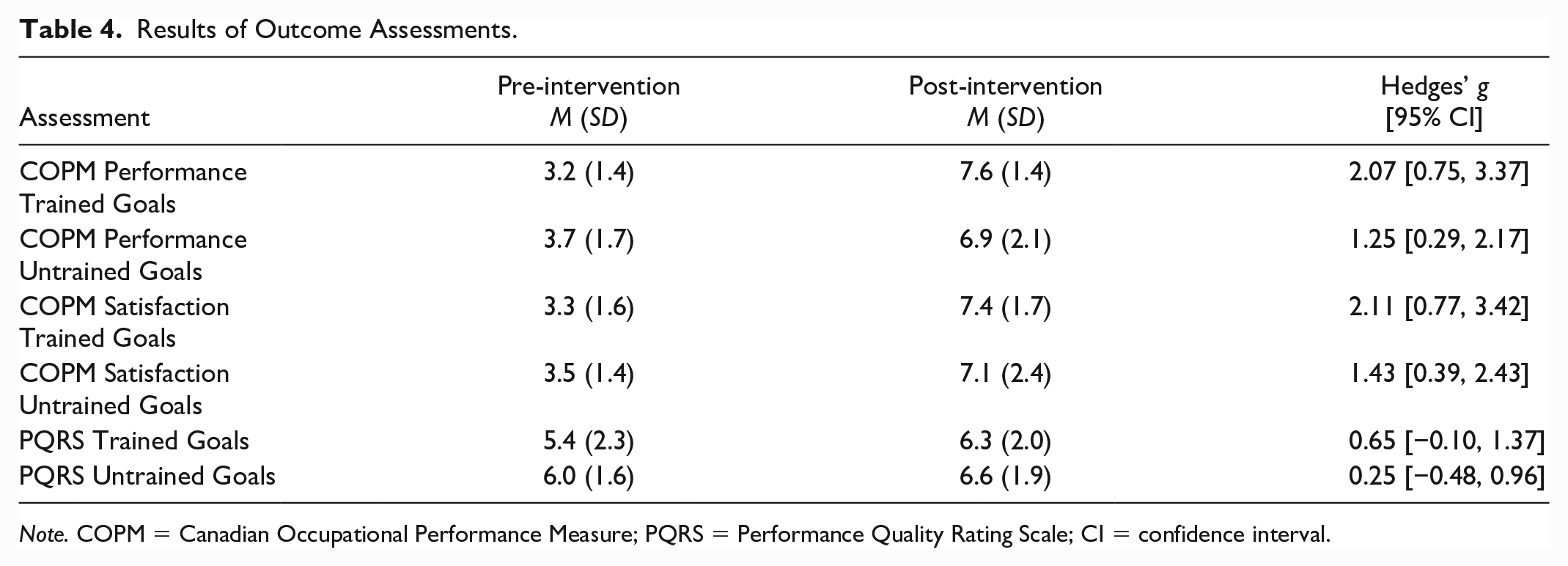
Note. COPM = Canadian Occupational Performance Measure; PQRS = Performance Quality Rating Scale; CI = confidence interval.
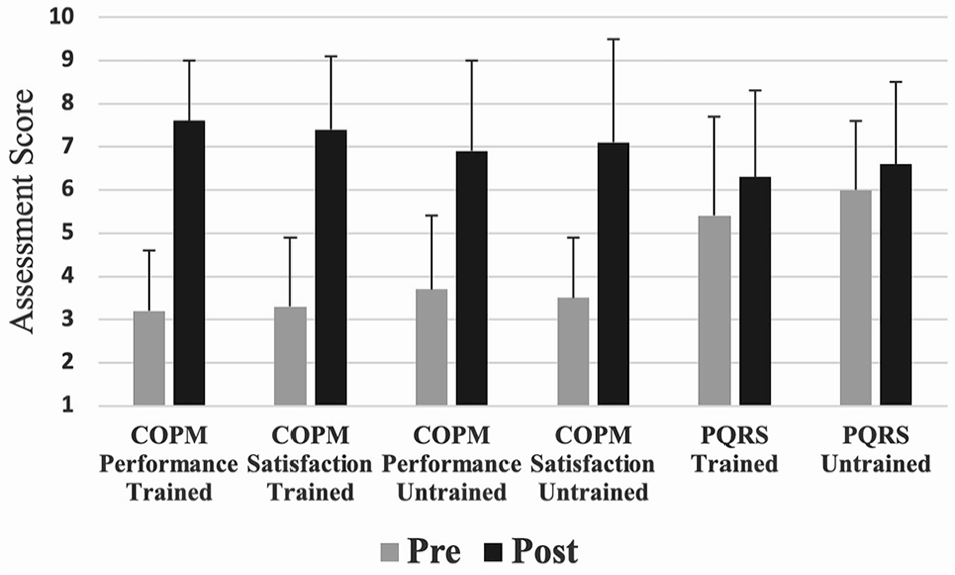
Change in Outcome Assessments Pre- to Post-Intervention.
Discussion
The negative effects of stroke on life participation continue well after the initial rehabilitation period (Kapoor et al., 2017). Singam and colleagues (2015) report that at 6 years post-stroke, as many as 65% have not returned to pre-stroke participation levels (Singam et al., 2015). CO-OP is becoming well-established as an evidence-based method for improvement of these functional-level outcomes; however, questions on how to best optimize this intervention still remain. The positive effects observed from CO-OP may be greater when paired with an additional intervention component. Unanswered questions remain in general CO-OP literature and, specifically with CO-OP augmented by tDCS, regarding maintenance of effects, appropriate dosing, or whether “booster” sessions would be beneficial.
The current study sought to determine the feasibility and preliminary effect of augmenting CO-OP with tDCS in the chronic stroke population. On the primary outcome of feasibility, the combined intervention was found to be highly acceptable and feasible. Although necessary to be interpreted with great caution due to the preliminary nature of this study (e.g., lack of control group, small sample), current findings also suggest a positive effect on occupational performance outcomes with a greater effect present for goals directly trained during the intervention.
Use of a feasibility design is highly encouraged in early phase intervention studies to conserve finances, resources, and researcher/participant effort. As feasibility studies aid in determining which interventions warrant efficacy testing and what intervention modifications are required before a larger trial. As the inherent risk of non-invasive brain stimulation is greater than most OT interventions, there is an increased responsibility to conduct feasibility testing in clinical populations such as stroke (Bowen et al., 2009; Craig et al., 2013; Lawson et al., 2022). The tolerability and absence of adverse effects mirrors a large amount of existing research on the use of tDCS. Several recent reviews suggest that when operated within established parameters (e.g., <4 mA, <40 min), tDCS has not led to any serious adverse event across all healthy individuals and patient populations (Bikson et al., 2016; Nikolin et al., 2018; Russo et al., 2017). Participants did experience other mildly agitating sensations on the scalp such as tingling, itching, or a mild burning sensation such as that encountered in prior studies; however, these sensations were not noxious enough to end study participation or any single session of stimulation.
As demonstrated in this study, participants in prior acceptability evaluation of metacognitive interventions including CO-OP perceive it to be meaningful and relevant to their needs and situations (Allin et al., 2020; Ash Routen et al., 2020; Peny-Dahlstrand et al., 2020; Piamjariyakul et al., 2013; Zera et al., 2022). For example, Zera and colleagues implemented CO-OP in an adult day rehabilitation setting with individuals with chronic stroke; participants identified the metacognitive structure, motivating nature, and selecting their own goals as positive intervention attributes (Zera et al., 2022). Although the combination of CO-OP and tDCS required a longer session (i.e., 1 hr 15 min) and a higher rate of session attendance (3×/week for 4 weeks) as compared with the standard CO-OP protocol, feasibility ratings suggest that this was not a barrier for the current sample. However, researchers were flexible with scheduling of sessions, so this may not be generalizable. A high rate of sessions may be beneficial for early mastery of the problem-solving strategy, but it also could prohibit a complete opportunity to implement established homework plans between sessions. The necessary frontloading of intervention sessions may also be better complemented by booster sessions in future studies.
A secondary aim of this study was to evaluate the preliminary effect of CO-OP and tDCS on activity performance outcomes. Performance and satisfaction of participant goals that were directly trained within the intervention improved a large amount. Similar results were reflected by a moderate improvement in trained goals as measured objectively by the PQRS. The discrepancy in these ratings may be the result of an overall improvement in participant self-efficacy found in prior work (McEwen et al., 2015). Alternatively, it could be that the COPM and other self-report measures capture activity performance in a real-world context, whereas the PQRS only reflects a singular simulation of activity performance.
Use of a global metacognitive approach for learning increases likelihood of skill transfer by teaching broad strategies that are applicable over a range of potential activities as opposed to teaching highly activity-specific information that is limited in use. Current results provide some evidence to this effect with large effects on performance of goals not directly trained within the intervention (e.g., never discussed) as measured by the COPM and a small effect by the PQRS. These findings are promising given the notoriety of skill transfer being both of the utmost importance and very difficult to achieve. Polatajko and colleagues (2012) compared the use of CO-OP against usual care occupational services in the stroke population and similarly demonstrated improvements of the CO-OP group for untrained goal performance albeit to a lesser extent than directly trained goals.
This study has several limitations. The sample was predominately White and may not reflect the needs and perceptions of the diverse stroke population. Although feasibility findings are not intended to be generalized to a population, the small sample size further limits generalization. Finally, the lack of long-term follow-up assessment precludes knowledge of retention of intervention gains.
Conclusions
Our study findings indicated that a complex intervention combining MCST with non-invasive brain stimulation is acceptable, feasible, and tolerable in individuals with chronic stroke. These feasibility outcomes suggest a future larger scale trial is warranted. The sample experienced improvements in occupational performance outcomes with greater progress observed for directly trained goals.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the University of Missouri Research Council for funding of this work and our participants for their engagement in this study.
Clinical Trials
#NCT04617548
Ethical Approval
This study was approved by the University of Missouri Institutional Review Board (#2009451).
