Abstract
Objectives
Aortic intramural hematoma (IMH) is a rare disease. Thus far, only limited data is available and the indications for conservative and endovascular treatment are not well defined. The aim of this study was to investigate clinical presentation, course, CT imaging features and outcome of patients with type B aortic IMHs.
Methods
We included all patients with type B IMHs between 2012 and 2021 in this retrospective monocentric study. Clinical data, localization, thickness of IMHs and the presence of ulcer-like projections (ULPs) was evaluated before and after treatment.
Results
Thirty five patients (20 females; 70.3 y ± 11 y) were identified. Almost all IMHs (n = 34) were spontaneous and symptomatic with back pain (n = 34). At the time of diagnosis, TEVAR was deemed indicated in 9 patients, 26 patients were treated primarily conservatively. During the follow-up, in another 16 patients TEVAR was deemed indicated. Endovascularly and conservatively treated patients both showed decrease in thickness after treatment. Patients without ULPs showed more often complete resolution of the IMH than patients with ULPs (endovascularly treated 90.9% (10/11) vs 71.4% (5/7); conservatively treated 71.4% (10/14) vs 33.3% (1/3); P = .207). Complications after TEVAR occurred in 32% and more frequently in patients treated primarily conservatively (37.5% vs 22.2%). No in-hospital mortality was observed during follow-up.
Conclusions
Prognosis of IMH seems favourable in both surgically as well as conservatively treated patients. However, it is essential to identify patients at high risk for complications under conservative treatment, who therefore should be treated by TEVAR. In our study, ULPs seem to be an adverse factor for remodeling.
Keywords
Introduction
Aortic intramural hematoma (IMH) is one manifestation of acute aortic syndrome (AAS), which also includes aortic dissection and penetrating atherosclerotic ulcer (PAU). The Stanford classification is the basis to classify all 3 clinical conditions. 1 The writing committee of the European Association for Cardio-Thoracic Surgery (EACTS) and the European Society for Vascular Surgery (ESVS) has recently updated the classification, defining type A as any disease involving the ascending aorta and type B when only the descending thoracic aorta is involved. Arch involvement either by the most proximal tear or by retrograde extension is referred to as non-A-non-B. 2 The incidence of AAS is 3.5 to 6.0 per 100,000 patient-years and the in-hospital mortality derived from the International Registry of Acute Aortic Dissections (IRAD) is about 21%. 3 The IRAD data demonstrate a 5.7% prevalence of IMHs in AAS patients. Like classic aortic dissection, IMH is a highly lethal condition when it involves the ascending aorta (39.1% mortality), but far less when it is limited to the arch or descending aorta (8.3% mortality).
IMH has also been described as “aortic dissection with closed and thrombosed false lumen”. 4 The pathogenesis of IMHs is not well understood. The most common hypothesis is spontaneous rupture of the vasa vasorum, the blood vessels that penetrate the outer half of the aortic media from the adventitia and arborize within the media to supply the aortic wall, due to atherosclerotic processes and systemic hypertension. 5 It has been shown that atherosclerosis is more prevalent in the descending than in the ascending aorta, 6 which might explain why IMHs and PAUs are located in the descending aorta in 60%–70% of cases. 7 Another hypothesis is that IMHs originate from small entry tears in the intima followed by thrombosis of these tears, causing difficulty in detecting these on imaging studies. 8 In some cases, local defects of the intima, resulting in small saccular areas of intramural enhancement, called ulcer-like projections (ULPs), can occur. 9 They can proceed to classic dissections, if the IMH ruptures back into the true aortic lumen. 8 Furthermore, IMHs can also be provoked by traumatic thoracic contusion or percutaneous catheter manipulation. 7 Computed tomography (CT) is very helpful in the diagnosis, follow-up and management of IMHs. It is important to report the regions of the aorta involved by IMH, the overall aortic calibre, the thickness of the IMH, and any signs of complication, such as rupture, pericardial effusion, end-organ ischemia and ULPs. 9
Because of the high mortality of type A IMHs involving the ascending aorta, usually emergency surgical treatment is performed. 10 Type B IMHs tend to be medically managed by stringent blood pressure control and analgesia and observed with serial follow-up CT scans. 11 Aneurysm formation, conversion to classic dissection and rupture are potential complications of type B IMHs. Risk factors for aortic complications include ULPs, hematoma thickness greater than 10 mm, aneurysmal dilatation and increase in thickness of the hematoma at follow-up imaging and should trigger surgical treatment.4,12 Most IMHs are located in the descending aorta, and thoracic endovascular aortic repair (TEVAR) is the method of choice for treatment of complicated type B IMHs. 13
As mentioned above, aortic IMH is a rare disease. Since there is only limited data, the aim of this study was to investigate clinical presentation and course, CT imaging features and outcome after conservative and endovascular treatment of type B aortic IMHs.
Methods
Study Cohort and Patient Characteristics
For this retrospective monocentric study, our radiology information system was searched for CT scans performed between January 2011 and March 2021 containing the words „intramural“ or „IMH“ within the radiology report. 622 matching reports were found. These reports were then assessed by a radiologist with 4 years of experience in CT angiographies for the presence of IMHs. Reports that contained phrases like „intramural air inclusions“ referring to bowel wall or „no proof of IMH“ were excluded. After this assessment, 321 CTs of 48 patients remained. 13 patients had type A IMHs and were excluded. Thus, 260 CTs of 35 patients with type B IMHs were included in this study.
Clinical Data
Clinical data from patient files, patient characteristics and radiological findings for each patient included in the final analysis were evaluated retrospectively. Pre-existing conditions, such as renal insufficiency, heart insufficiency, atrial fibrillation, arterial hypertension (blood pressure >140 mmHg 14 ), coronary heart disease, myocardial infarction, diabetes mellitus, hyperlipoproteinemia, tumor disease, nicotine abuse and COPD, were recorded. Chronic renal insufficiency was defined as an eGFR of less than 60 mL/min/1.73 m2. 15 Chronic heart failure was determined by restricted myocardial pumping function on echocardiography during the inpatient stay or pre-existing medical findings. Diabetes was defined as HbA1c greater than 6.5% in the laboratory or pre-existing medication with oral antidiabetics or insulin. Hyperlipoproteinemia was defined as >4 mmol/l total cholesterol or pre-existing statin therapy. 16 Furthermore, it was recorded if patients were symptomatic or asymptomatic at the time of initial diagnosis and if there was trauma at the time of onset of symptoms. In our final study cohort, there were no missing data.
Treatment Strategy
All patients were discussed at our interdisciplinary vascular board. We assessed if patients were treated surgically or conservatively. Conservative therapy at initial presentation in the intensive care unit included strict blood pressure control with target systolic blood pressure less than 120 mmHg. Initially, intravenous antihypertensives were administered, followed by oral application. The administration of a beta-blocker, if no contraindications were present, was an integral part. After reaching target blood pressure without intravenous medication for longer than 24 hours, transfer to a general ward was performed. CT angiography scans of IMHs were performed on day 3, 7 and before discharge. In case of surgery, we evaluated the indication, which might be early progress of IMH thickness, ULPs, late expansion >1 month after initial diagnosis, secondary occurring dissections or persistent pain. Time between initial diagnosis and surgery was assessed. We recorded the number of stents, landing zones and potential revascularization strategies for supra-aortic vessels. We assessed, if complete overstenting of the aortic pathology was performed or only spot stenting of an ULP. Mortality and all complications after surgery were analyzed, which could be surgical site infections, hematomas, kidney failures, myocardial infarctions, spinal cord ischemia or strokes. For follow-up after hospital discharge, IMHs were assessed by CT angiography at 6 weeks, 3 months, 6 months, 1 year, and annually thereafter.
TEVAR Technique
The TEVAR was performed via a sonographically guided percutaneous access of the common femoral artery. Intraoperative activated clotting time (ACT)-guided heparinization was performed with target ACT 200 to 250 s, starting with a bolus of 50 i.U. per kg body weight. The implant used was either a Conformable GORE® TAG® Thoracic Endoprosthesis (W. L. Gore and Associates GmbH, Newark, Daleware, USA) or a Valiant™ Captivia Stent Graft with the Captivia™ Delivery System (Medtronic, Dublin, Ireland) or a Zenith Alpha™ Thoracic Endovascular Graft (Cook Medical, Bloomington, Indiana, USA). Oversizing of the stent prosthesis was 10% after centerline measurement of the aorta in the preoperative CT angiography in 1 mm slices. A spinal catheter was not placed preoperatively, but rapid extubation and neurological assessment was performed postoperatively and a spinal catheter placed if indicated.
Imaging Analysis
Imaging analysis was performed on a radiology workstation using a 3D imaging tool (IMPAX EE R20, Agfa HealthCare N.V., Mortsel, Belgium) by a radiologist with 4 years of experience in CT angiographies. The affected aortic segments, as defined by the Society for Vascular Surgery/Society of Thoracic Surgeons,
17
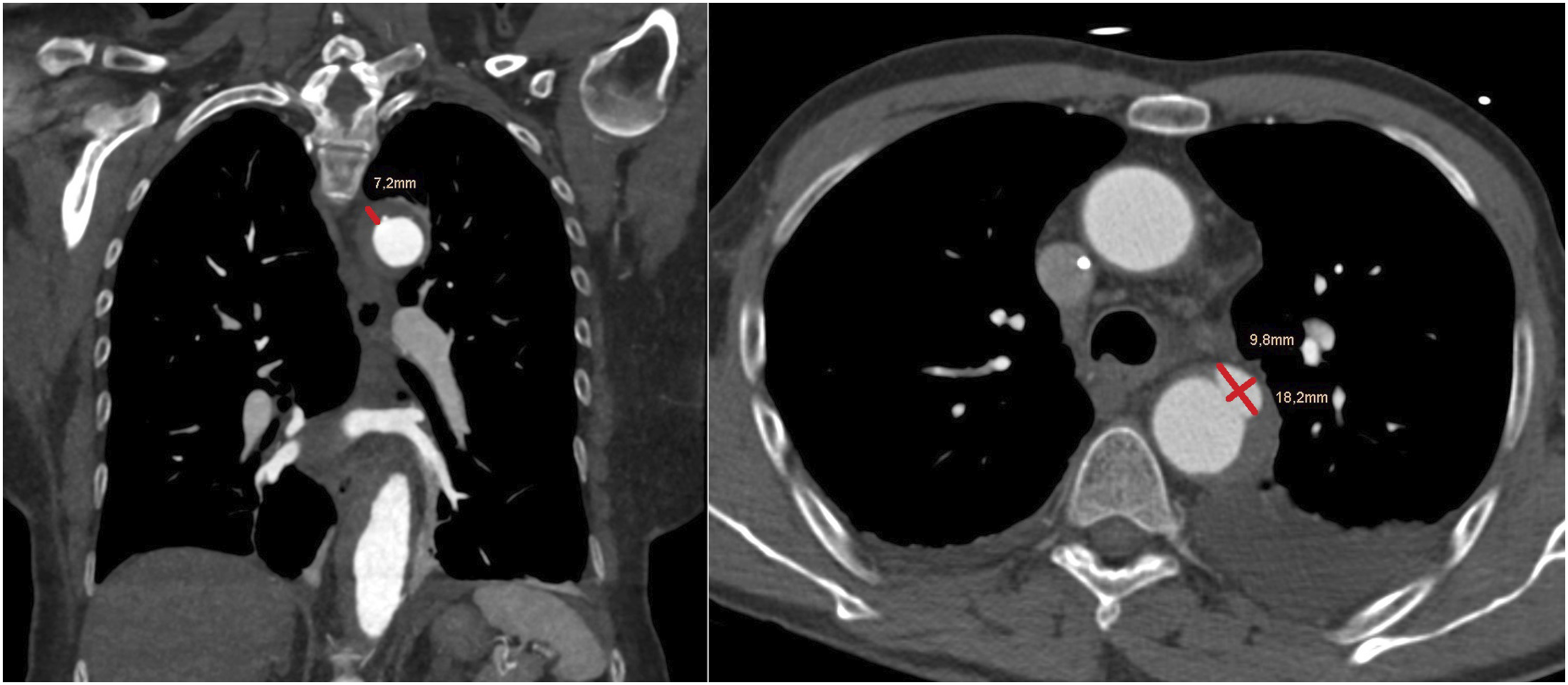
were determined in all 260 CTs (Figure 1) and the maximum thickness of the IMH was measured (Figure 2). Time course of hematoma thickness was evaluated for all patients. The number of ULPs was documented and, if present, location and size (width/depth) of the biggest ULP was measured (Figure 2). If pericardial or pleural effusion (right/left/both) was present, maximum thickness and density were documented. Aortic segments affected by IMHs. Measurement of maximum thickness of IMH in the aortic arch (coronal CT angiography image; left) and of ulcer-like projection in the descending aorta (axial CT angiography image; right).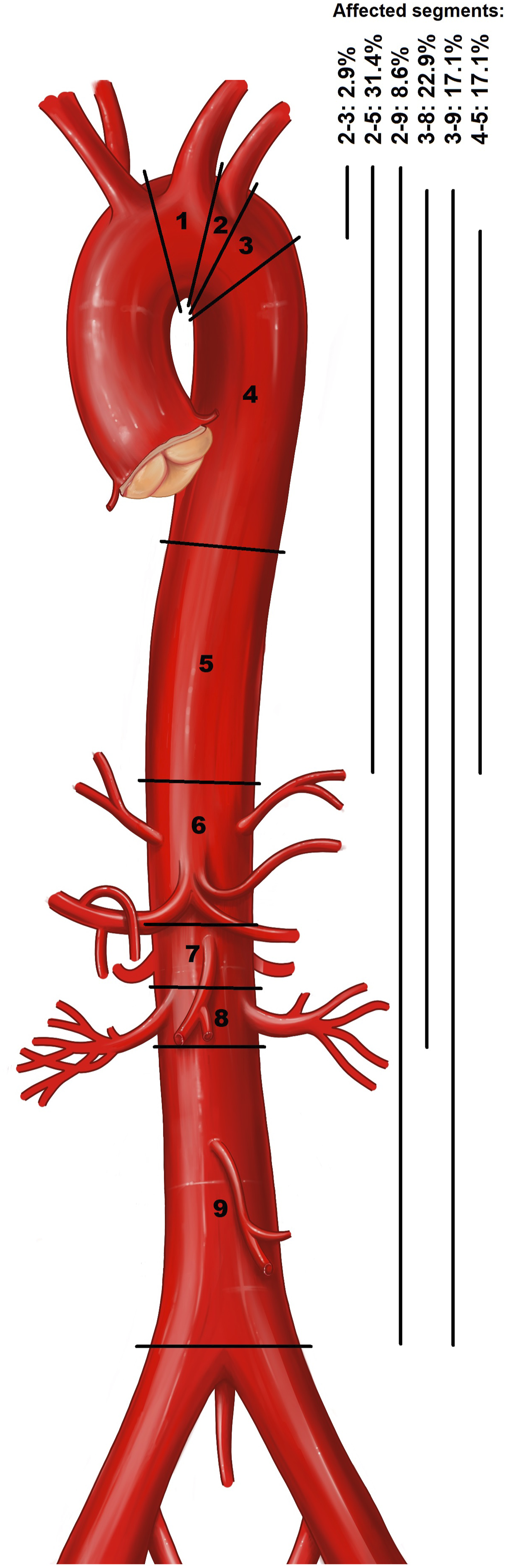
Ethical Approval
Approval was obtained from the local ethics committee at the Technical University Dresden (decision number BO-EK-401092022) and the study was conducted in agreement with the Declaration of Helsinki, according to the ICH Harmonized Tripartite Guideline for Good Clinical Practice. The ethics committee is registered as institutional review board (IRB) at the Office for Human Research Protections (OHRP) (registration number IRB00001473 and IORG0001076).
Statistical Analysis
The statistical analysis was performed using IBM SPSS Statistics (Version 25, SPSS Inc, Chicago, IL). All patient characteristics were grouped to build categorical or nominal variables. Continuous variables were presented by the mean and standard deviation or median and range. Complication rates for different therapeutic strategies and aortic remodeling depending on the presence of ULPs were analyzed using the Chi-square test. Diagrams were created to show the time course of IMH thickness in each patient. Growth rates were calculated with the “last-first/time” method. The difference in diameter and time was calculated by last minus first measurement and the growth rate was calculated as the according ratio. The average growth rates were calculated by a linear regression model where “diameter = growth rate * time”.
Results
Study Population and Clinical Findings
Pre-existing Conditions in Patients With IMHs.
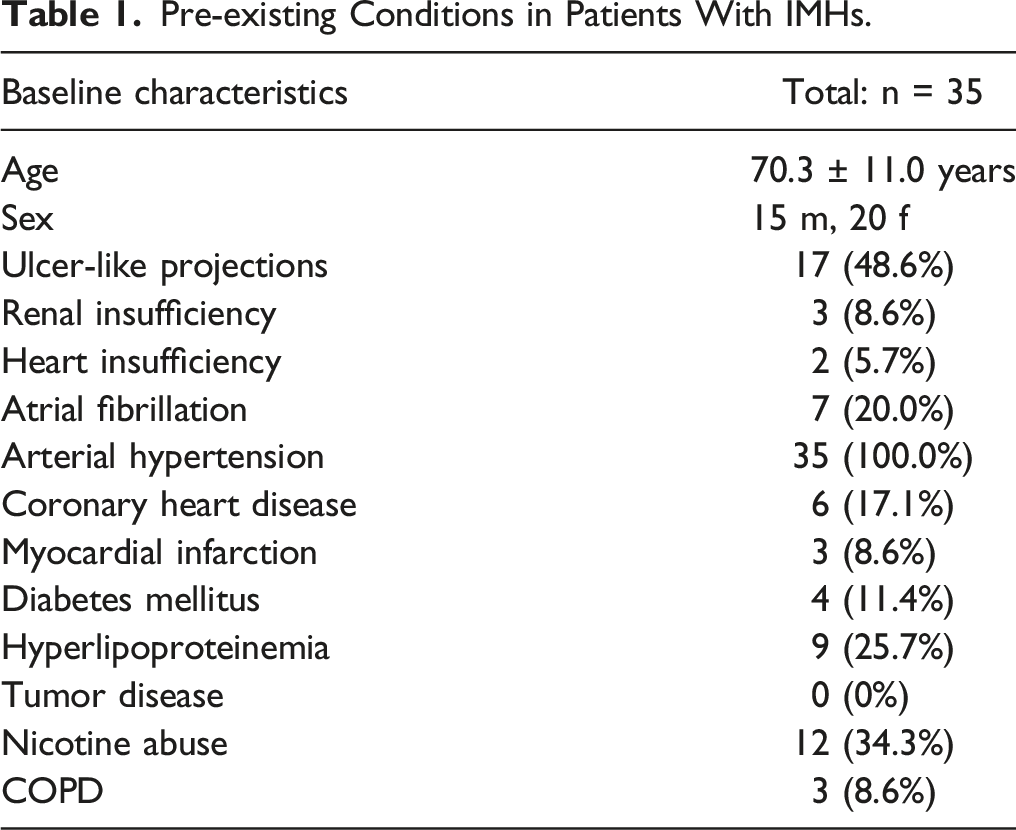
34 of 35 patients (97.1%) were symptomatic with back pain at the time of initial diagnosis. In 1 patient, the diagnosis was an incidental finding during tumour staging. The etiology was spontaneous in almost all cases (n = 34, 97.1%) and traumatic in 1 patient, due to a motor vehicle accident.
Imaging Analysis
The maximum thickness of IMHs at primary imaging was 9.54 ± 3.11 mm (minimum 5 mm; maximum 17 mm). Initially, 17 patients (48.6%) had an aortic wall thickness >10 mm. Affected aortic segments, as defined by the Society for Vascular Surgery/Society of Thoracic Surgeons, 17 were as following: In 1 case (2.9%) segments 2-3, in 11 cases (31.4%) segments 2-5, in 3 cases (8.6%) segments 2-9, in 8 cases (22.9%) segments 3-8, in 6 cases (17.1%) segments 3-9 and in another 6 cases (17.1%) segments 4-5 (Figure 1). Six patients (17.1%) had bilateral pleural effusions and 6 patients (17.1%) a pleural effusion only on the left site. Two patients (5.7%) had a pericardial effusion, both of which were haemorrhagic. 16 patients (45.7%) had ULPs and 19 patients (54.3%) had no ULP at the time of diagnosis.
Treatment
At the time of diagnosis, TEVAR was deemed indicated in 9 patients (25.7%), whereas 26 patients (74.3%) were treated primarily conservatively. During the follow-up, in 16 (61.5%) of these 26 patients TEVAR was deemed indicated. Therefore, a total of 25 patients (71.4%) was treated by TEVAR and 10 patients (28.6%) remained under conservative therapy (see also Figure 3). The 2 patients with haemorrhagic pericardial effusions were assessed as unfit for cardiothoracic surgery after discussion at interdisciplinary vascular board, but during the follow-up developed early expansion and were then treated by TEVAR. Other indications for TEVAR were ULPs (n = 11, 31.4%), late expansion >1 month after initial diagnosis (n = 5, 14.3%), secondary occurring dissections (n = 3, 8.6%), persistent pain (n = 3, 8.6%) or renal ischemia (n = 1, 2.9%) (Table 2). 14 of the 16 patients (87.5%) with ULPs and 11 of the 19 patients (57.9%) without ULPs were treated by TEVAR. 14 patients (56%) were treated within the first 14 days after initial diagnosis. Median time between initial diagnosis and TEVAR was 13 days (0-622 days). Treatment strategy and outcome in our study cohort of patients with type B aortic IMHs. Indications for Treatment of IMH Patients by TEVAR.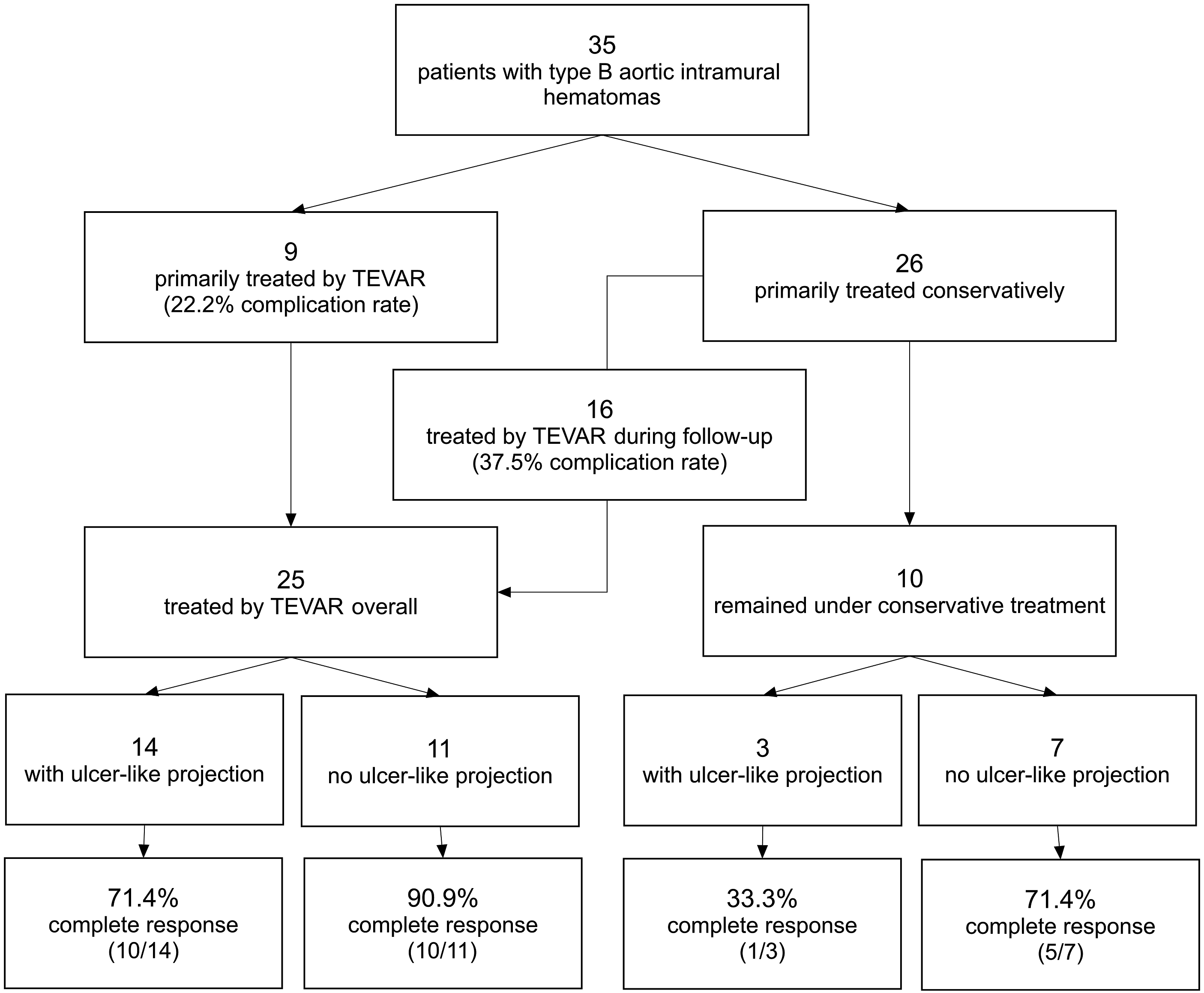

Technique of TEVAR.

The complication rate after TEVAR was 32% (n = 8). We observed 2 surgical site infections, 1 hematoma, 1 case of kidney failure, 2 non-ST-segment elevation myocardial infarctions, 1 spinal cord ischemia with permanent incomplete sensorimotor paraplegia and 1 minor, non-disabling stroke. Complications after TEVAR occurred more frequently in patients treated primarily conservatively (n = 6/16; 37.5%) compared with primarily surgically treated patients (n = 2/9; 22.2%), but the difference was not statistically significant (P = .432). Patients who remained under conservative therapy had no complications related to their aortic pathologies. In-hospital mortality was 0% and 30-day mortality was 0% as well. In the follow-up of in median 16 month (2-119 month) 2 patients died (5.7%) of causes unrelated to the aortic pathologies.
Aortic Remodeling
Patients, who were treated by endovascular surgery, showed on average an increase in IMH thickness of 2.3 mm/6 months before TEVAR and a decrease of 2.7 mm/6 months after TEVAR (Figure 4). There was no significant difference (P = .552) in aortic remodeling between patients with complete overstenting of the aortic pathology (-2.97 mm/6 months) and partial overstenting (-2.65 mm/6 months). 20 patients treated by endovascular surgery reached complete resolution of the IMH (10 of these (50%) with additional ULPs), 5 showed residual hematoma (4 of these (80%) with additional ULPs). Of these 5 patients, 2 had imaging available only from the day of surgery and 2 days after surgery, respectively. Time course of IMH thickness before and after endovascular treatment.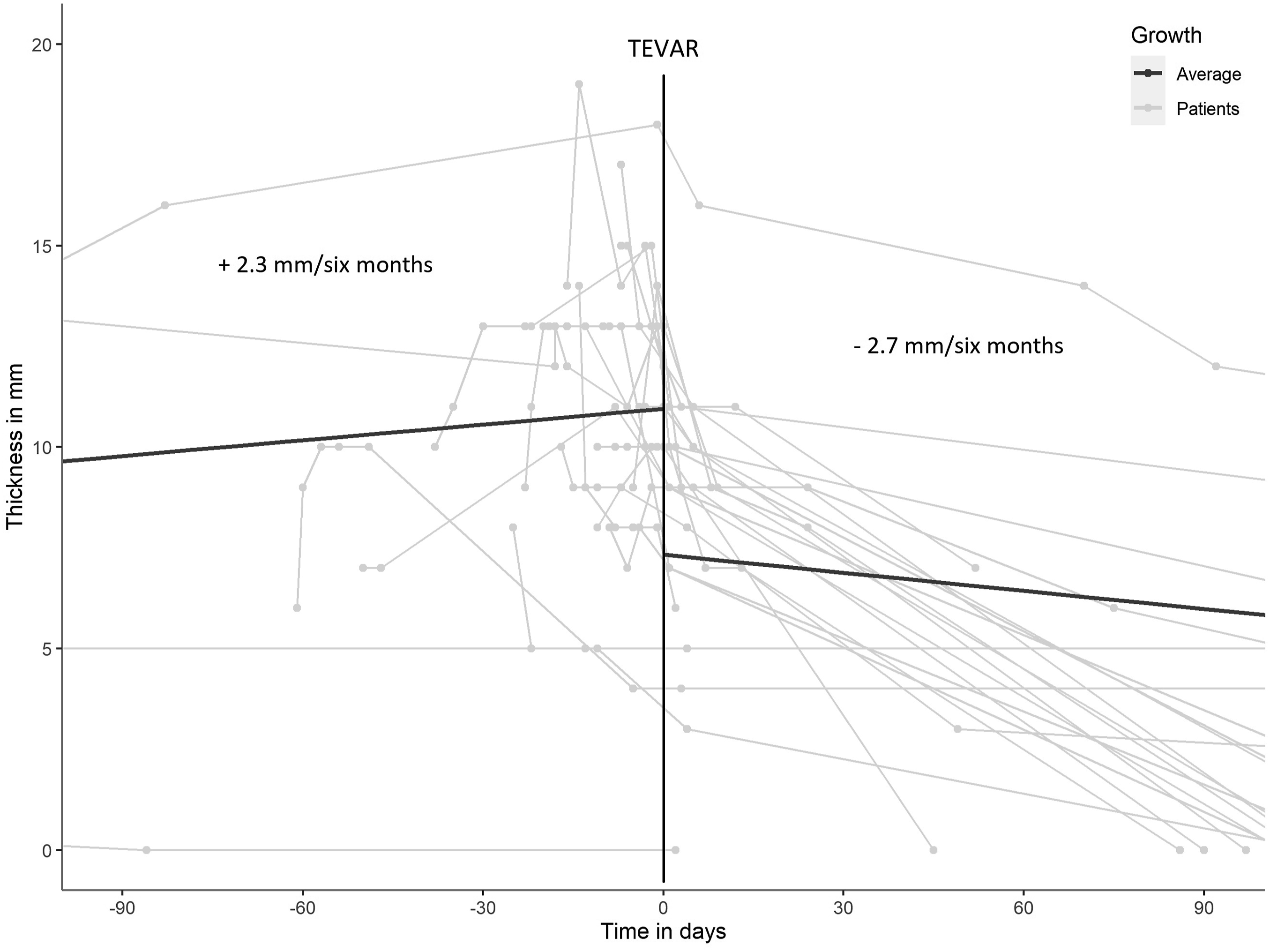
Conservatively treated patients on average showed decrease in IMH thickness of 4.7 mm/6 months (Figure 5). Six patients treated conservatively reached complete resolution of the IMH (1 of these (16.7%) with additional ULP), 4 showed residual hematomas (2 of these (50%) with additional ULPs). Follow-up imaging in these 4 cases was performed within a maximum of 42 days after diagnosis. Time course of IMH thickness in patients with conservative treatment.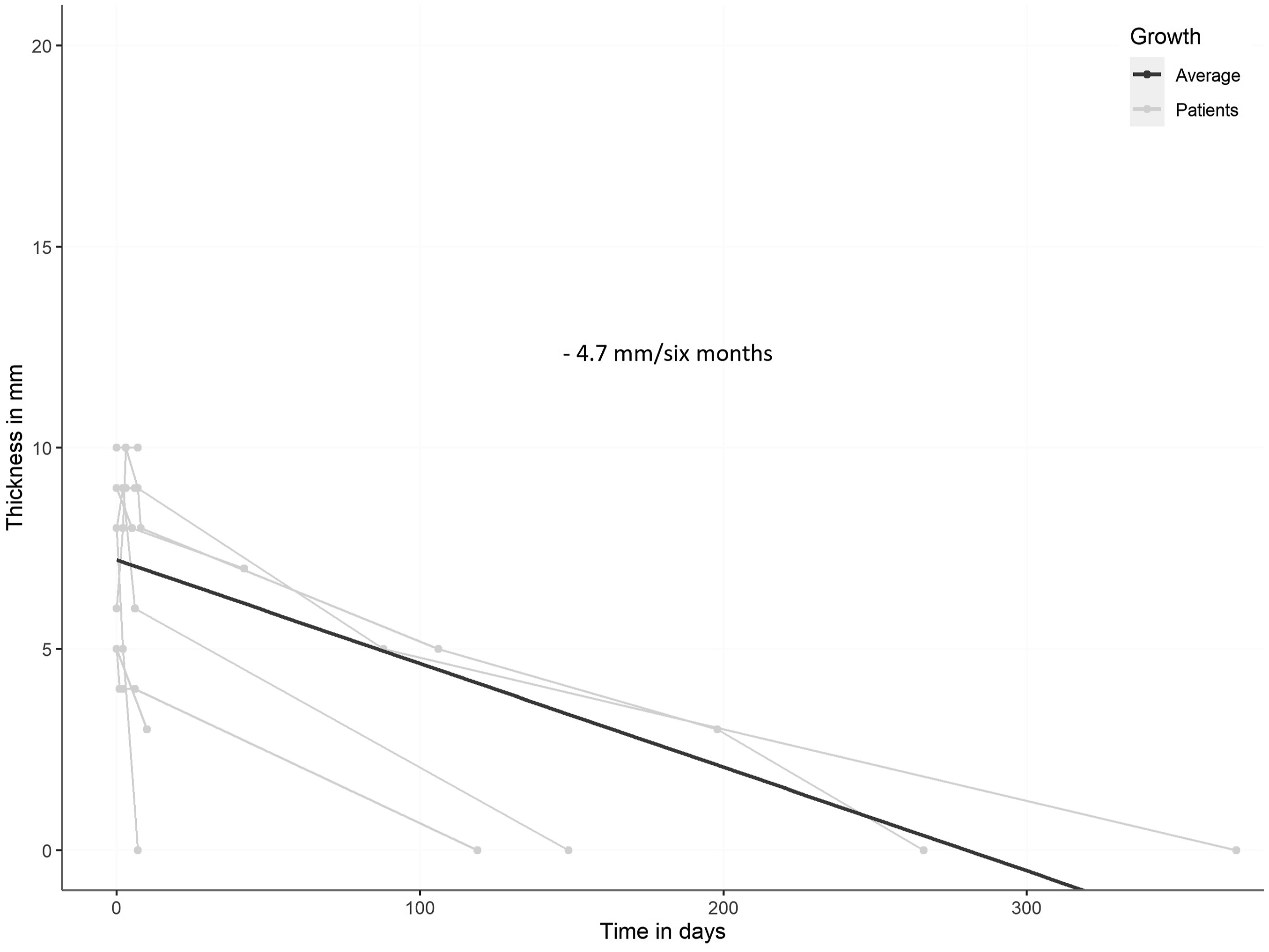
In summary, 90.9% (10/11) of the endovascularly treated patients without ULPs and 71.4% (5/7) of the conservatively treated patients without ULPs showed complete resolution of the IMH, while only 71.4% (10/14) of the endovascularly treated patients with ULPs and 33.3% (1/3) of the conservatively treated patients with ULPs showed complete resolution of the IMH (P = .207) (see also Figure 3).
Discussion
Our study shows favourable prognosis of IMH, as a rare manifestation of acute aortic syndrome. Endovascular therapy leads to remodeling of the aorta over time, but IMHs can regress under conservative therapy as well. It is important to identify type B aortic IMH patients, who should be treated by TEVAR and those, who can be treated conservatively.
In our study, ULPs seem to be an adverse factor for remodeling. Other high-risk features, that often indicate TEVAR, are early expansion or late expansion >1 month after initial diagnosis, secondary occurring dissections, persistent pain or renal ischemia. The majority of patients with type B aortic IMHs is symptomatic at the time of initial diagnosis, so there are virtually no incidental findings of IMHs. The etiology of IMHs seems to be spontaneous in almost all cases due to cardiovascular risk factors, mainly arterial hypertension. This is in keeping with previous studies.18,19 Traumatic genesis is rare.
CT imaging plays a vital role in identifying patients with high-risk features. About 25% of patients in our study were indicated for TEVAR at the time of initial diagnosis. Furthermore, CT is an important tool to determine further treatment and monitor disease progression. A high proportion of our initially conservatively treated patients showed high-risk features in the follow-up that required treatment by TEVAR, which is an effective treatment and can delay the progression of IMHs and promotes regression. 20
In our cohort, in about 50% of the endovascularly treated patients only spot stenting of an ULP was performed without covering the entire pathology. With both techniques, endovascular therapy led to comparable remodeling of the aorta over time. But IMHs can regress under conservative therapy as well. Therefore, it is essential to identify patients, who have to be treated surgically, and those, who can be treated medically. A study by Luo et al. showed that patients could benefit from endovascular treatment when the initial maximum aortic diameter is ≥ 50 mm or the hematoma thickness is ≥ 11 mm. 21 Besides the maximum aortic diameter, current studies found ULPs as an independent risk factor for patients with type B IMHs.22,23 Accordingly, in our study, the proportion of patients who showed complete resolution of the IMH was higher in patients without ULPs than in patients with ULPs, irrespective of the treatment strategy. A meta-analysis by Chakos et al. found no statistically significant difference between endovascular and medical treatment for aortic related death or regression of IMHs at a mean follow-up of 37 months, while TEVAR was found to be associated with lower risk of dissection or rupture during follow-up. 24 Another study by Tian et al. shows a higher survival rate and less frequent late progressions in aortic dissections in patients who were treated endovascularly vs medically. 25
Despite good results regarding remodeling after TEVAR, major complications have to be considered. Furthermore, re-interventions are common, either due to progression of proximal or distal aortic disease or stent graft related complications. 26 In the acute phase, TEVAR has limitations due to mechanical stress on the device, which can lead to secondary endoleaks, intimal ruptures, distal new entry tears and pseudoaneurysm formation.13,27,28 Furthermore, TEVAR within 24 hours of admission is associated with less aortic remodeling and higher occurrence of late aortic-related adverse events and mortality. Therefore, delaying TEVAR could improve aortic remodeling and aortic-related outcomes. 29 Another essential factor for the success of TEVAR is accurate sizing of the endograft, as both under- and oversizing can lead to complications. Especially in emergency TEVAR, sizing can be difficult due to diameter changes of the aorta, because of haemorrhagic shock or need for resuscitation. 30 In our study, we determined a complication rate of 32% after TEVAR. Interestingly, complications occurred more often in patients with delayed TEVAR after initial conservative treatment than in patients who were primarily treated by TEVAR (37.5% vs 22.2%), but the difference was not statistically significant, which could be due to the small sample sizes.
In-hospital mortality and 30-day mortality were 0%. In the median follow-up of 16 months (2-119 months), 2 patients died (5.7%) of causes unrelated to the aortic pathologies. In our study, patients with residual hematoma had a high percentage of concurrent ULPs, thus ULPs seem to be an adverse factor for remodeling.
One limitation of our study is the selection bias, because of the high portion of patients with indications for operative therapy in the early phase after diagnosis and the confounding by indication. Other limitations are the relatively low case number, which lead to high risk of type II errors, and the retrospective, monocentric study design, which limits generalizability. Because of the rarity and the heterogeneous patient population, prospective studies about IMHs are not easy to perform.
Conclusion
Prognosis of IMH seems favourable in both surgically as well as conservatively treated patients. However, it is essential to identify patients at high risk for complications under conservative treatment, who therefore should be treated by TEVAR. In our study, ULPs seem to be an adverse factor for remodeling.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
