Abstract
Over the last few decades, the use of covered stent grafts became increasingly popular; as it plays a pivotal role in the management of various atherosclerotic diseases that are rising in both incidence and prevalence. Subsequently, vascular stent infections, although rare, are becoming a well-recognized complication with possibly devastating consequences, owing to the difficulties associated with its diagnosis and treatment. This has prompted significant interest in the condition regarding its pathophysiology, modifiable and non-modifiable risk factors, diagnostic and therapeutic approaches, and the possible implementation of prophylactic measures. We herein present a case of a patient with an infected aortoiliac stent 4 weeks after endovascular revision with atherectomy and additional stent insertion. The patient initially developed nonspecific symptoms and later developed a life-threatening hemorrhage, which was urgently controlled using a percutaneously inserted covered stent at the infected site. Definitive treatment using extraanatomical bypass implantation and an explantation of the infected stents was performed with excellent clinical response.
Keywords
Introduction
Infection of vascular stents is a rare but serious condition that causes ambiguous and non-localizing presentation, making prompt diagnosis and management a difficult task. 1 Symptoms of vascular stent infection include constitutional symptoms such as headache, fatigue, malaise, arthralgias, and myalgias, as well as persistent fever of unknown origin, generalized and/or localized pain, and septic shock. 1 Although the exact cause of infection is unclear in most cases, violation of the sterile technique during the surgical procedure, repeated use of the same catheter insertion site, and multiple guidewire manipulations, are theorized to be major contributing factors. 2 Moreover, Staphylococcus Aureus, a highly virulent organism that can cause rapid and severe vascular stent infection, remains the most commonly implicated agent, being responsible for 76.6% of stent infections according to data extrapolated from the literature. 1 Due to the commonly delayed presentation of infected vascular stents, the development of complications such as perivascular abscesses, vascular perforation, and recurrent emboli frequently occur. 3 Unsurprisingly, cases of infected stents require surgical intervention, with stent explantation and vascular reconstruction being the primary end goal of the procedure.1,3 Surgical management in such cases is crucial; as the sole use of antibiotics is often insufficient and futile. We herein report a case of an infected aortoiliac artery stent 4 weeks after endovascular revision with atherectomy and additional stent insertion, presenting with signs and symptoms of persistent non-localized infection, vascular perforation, and hematoma formation.
Case Presentation
A 56-year-old woman presented to the emergency department complaining of generalized pain including the bilateral lower limbs, back, and hips, with difficulty in standing and ambulating. She also reported developing fevers and a burning sensation in the lower limbs. Her past medical history revealed long-standing asthma. Past surgical history was significant for Endovascular Reconstruction of Aortic Bifurcation surgery, which was done to treat aortoiliac occlusive disease 5 years prior to her current presentation. Furthermore, she underwent several peripheral vascular interventions for concurrent peripheral atherosclerotic disease and received percutaneous recanalization of stent restenosis in the right common iliac artery 4 weeks prior to her most recent presentation. On admission, a thorough physical examination revealed generalized pain and tenderness of the back and upper and lower extremities. Additionally, there were no signs of ischemia despite her history of extensive atherosclerotic disease. The peripheral pulses were bilaterally palpable, the ankle/brachial index (ABI) was normal measuring 1 (N ≥1), and there were no signs of inguinal hematoma. Duplex ultrasound on admission revealed no signs of lower limb ischemia bilaterally (Figure 1). Duplex Ultrasound on admission revealing no signs of vascular obstruction in the lower limbs bilaterally.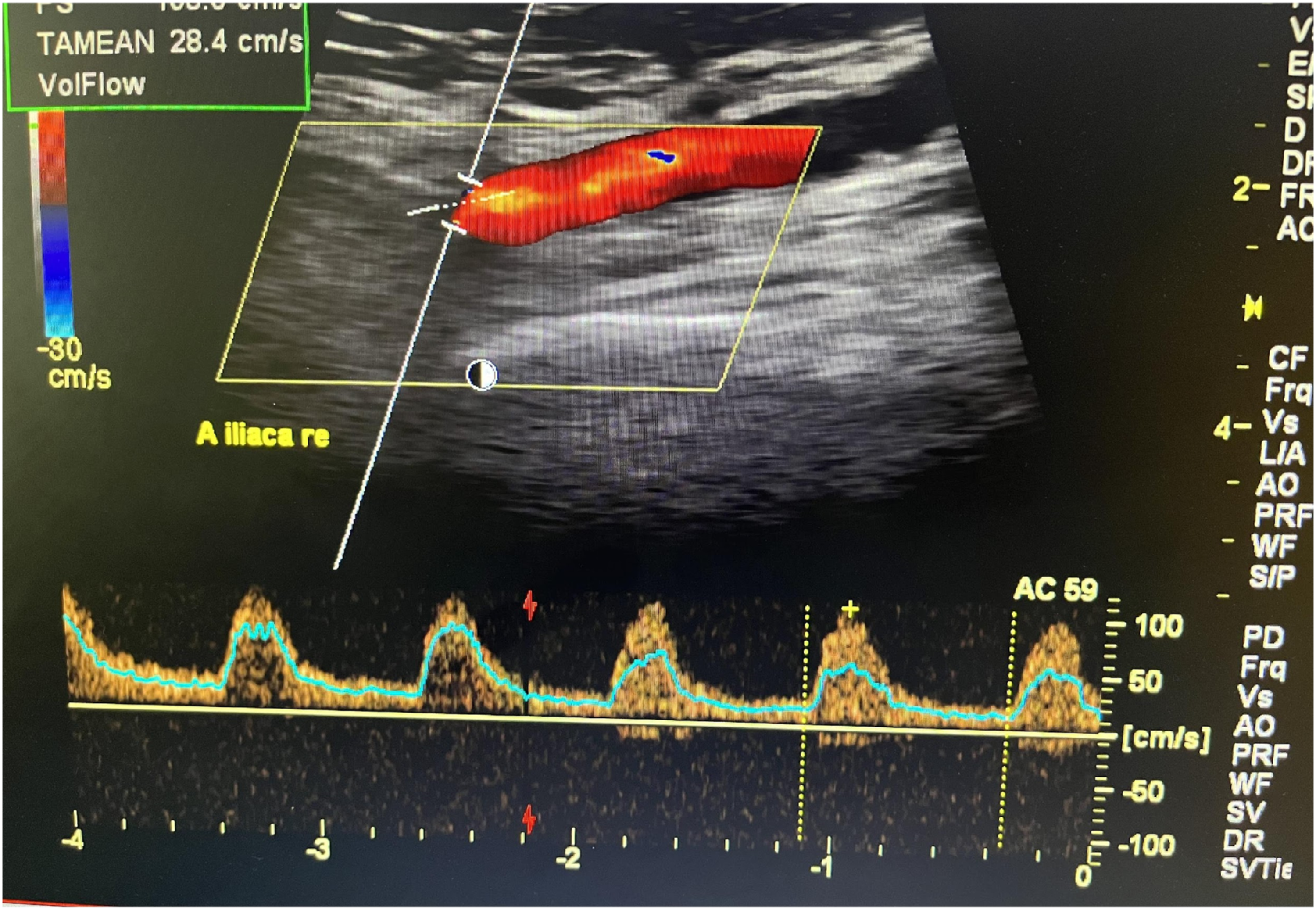
Upon further questioning, the patient was found to be a heavy smoker and had undergone multiple vascular interventions. In 2011, she underwent endovascular angioplasty by kissing-stenting technique for both common iliac arteries. In 2012, in-stent restenosis of the left common iliac artery occurred and was subsequently treated with percutaneous balloon dilation (POBA). In 2015, she was treated for aortic stenosis using an uncovered self-expanded stent in the aorta (Luminexx, BD Dickinson, USA) and POBA for both common iliac arteries. In 2021, she underwent a percutaneous atherectomy for recurrent stenosis in the right common iliac artery stent.
Subsequently, the patient was admitted to the hospital for further investigation. Laboratory testing revealed leukocytosis with neutrophilic predominance, and elevated inflammatory markers with C-reactive protein (CRP) measuring 15.54 mg/dl (Normal = less than 1 mg/d). Based on the patient’s clinical presentation and laboratory findings suggestive of a hyperinflammatory state, an infectious etiology was suspected as a cause of her symptoms. Thus, an extensive infectious work-up was done, which excluded endocarditis, pneumonia, urinary tract infection, and septic arthritis as possible causes. Further in-patient management included the administration of intravenous empiric antibiotics after blood cultures were obtained. Three days later, the patient showed persistence of leukocytosis with a progressively increasing white blood cell count. The blood cultures withdrawn earlier demonstrated the growth of Staphylococcus Aureus, which was sensitive to ampicillin-sulbactam. She was subsequently switched to a targeted antibiotic regimen.
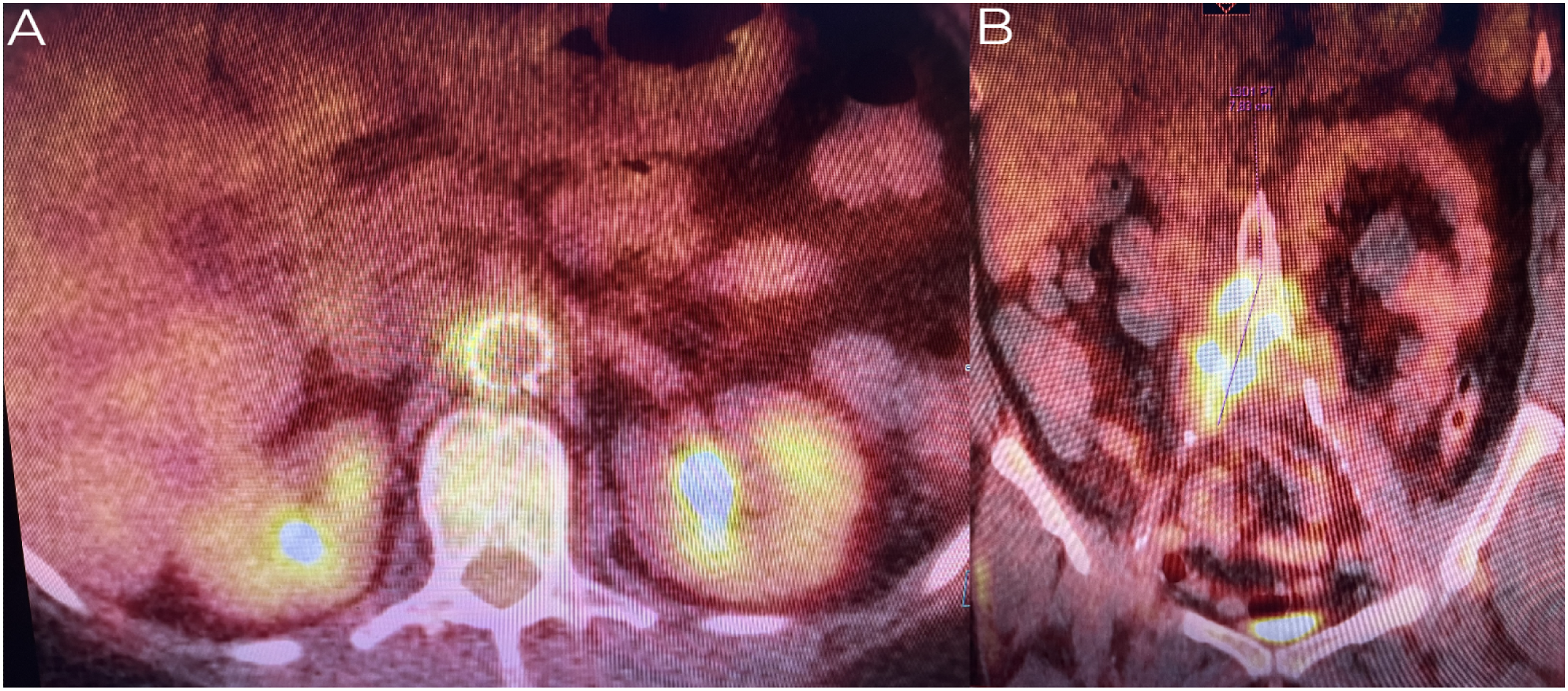
Blood, urine, and sputum cultures failed to localize the patient’s source of infection. Moreover, the patient’s fever of unknown origin continued to deteriorate, prompting the decision to perform an abdominal computed tomography (CT). However, the imaging findings were unremarkable and showed no signs of infection, with complete patency of aortic and bilateral iliac artery stents (Figure 2A). Additionally, a transesophageal echocardiography was done and excluded endocarditis. Abdominal ultrasound showed no internal organ abscesses and was not able to visualize any septic foci. Despite the extensive diagnostic interventions done, the septic focus was still not found. The patient continued taking her antibiotic course for a total duration of 2 weeks, which resulted in a decline in the inflammatory parameters without significant improvement in her pain. CT scan 2 weeks post-admission showed a mass around the right common iliac artery and increased enhancement (Figure 2B). A positron emission tomography (PET) CT scan showed extensive perivascular inflammation at the right common iliac artery, distal aorta, and the proximal part of the left common iliac artery, which was indicative of active stent infection and aortitis (Figure 3). CT scans comparing the patient’s radiological findings on admission (A) and 2 weeks post admission (B). The photographs show a significant increase in the mass size around the right common iliac artery in. Additionally, there is a notable increment in perivascular contrast enhancement. PET CT scan showing extensive perivascular inflammation at the distal aorta (A), right common iliac artery (B), and the proximal part of the left common iliac artery (B), which is indicative of active stent infection and aortitis.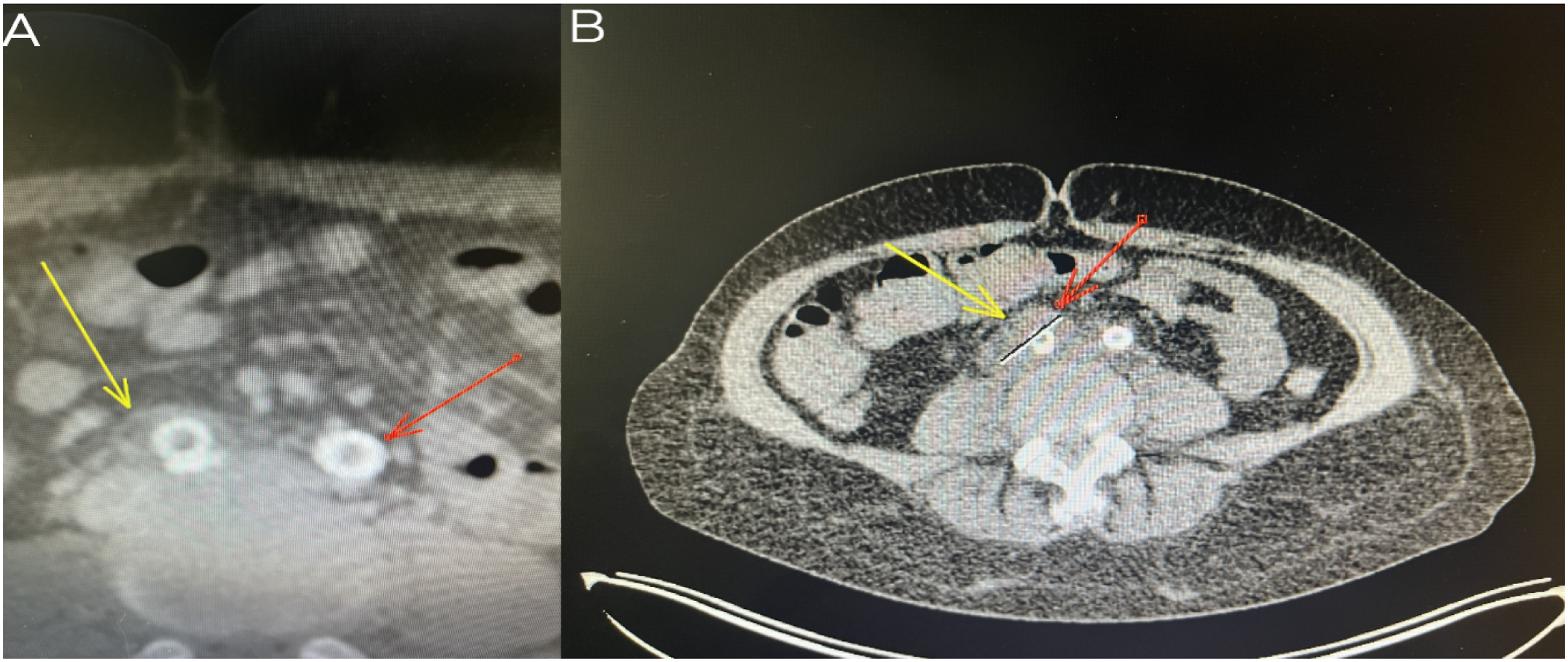
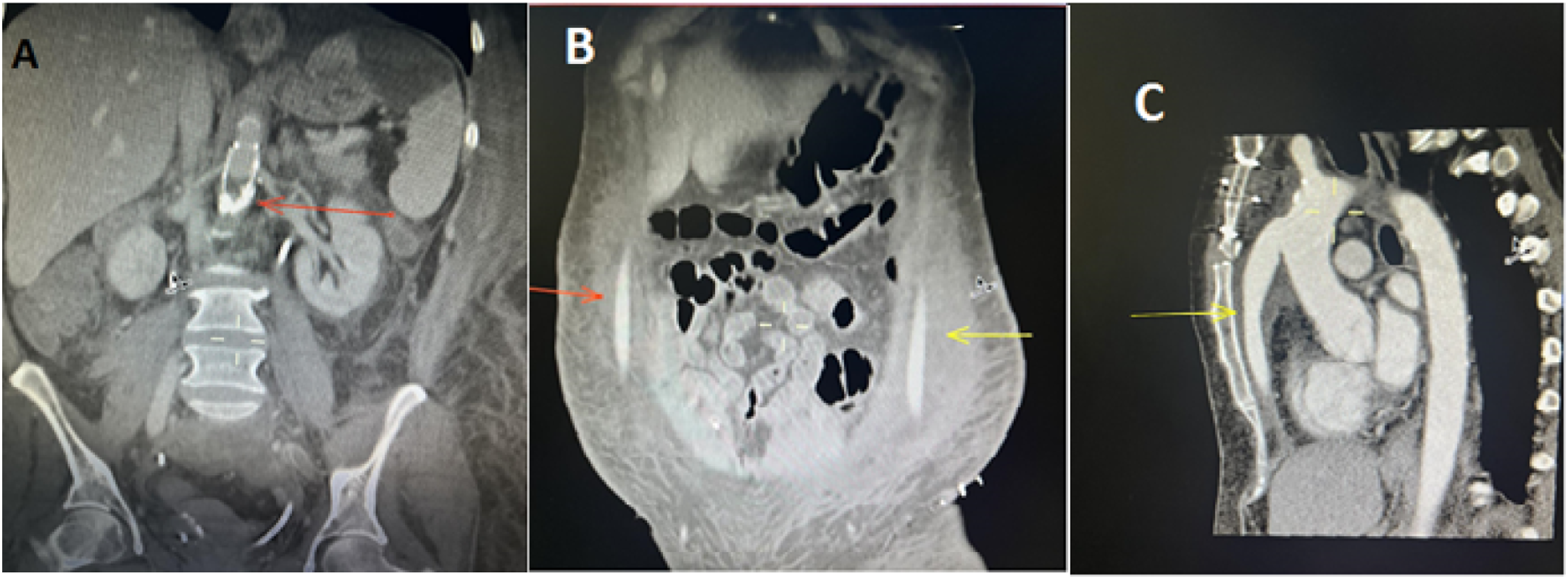
Shortly after, the patient started complaining of increasing abdominal pain. A repeat complete blood count (CBC) revealed a drop in the hemoglobin concentration compared to her baseline on admission. A repeat CT scan and digital subtraction angiography revealed an intra-abdominal hematoma with radiological signs suggestive of perivascular abscess formation, along with perforation of the common iliac artery due to infection (Figure 4). An urgent operative procedure was done to seal the perforation at the right iliac artery with a covered stent (6 × 59 mm Advanta). In addition, a decision to remove all stent material in the aorta and the iliac arteries was made. Subsequent management of the patient included 4 weeks of flucloxacillin, with the treatment of the infected aortic and iliac stents in a two-stage operation once the blood cultures showed no growth of organisms (Figure 5). Digital subtraction angiography (DSA) scan revealing a contained rupture of the right common iliac artery (red arrow) due to infection (left). CT Scan on the right. Postoperative CT scan months after the operation showing patent proximal anastomoses and the pathway of the bypass (B and C) with ruminants of Luminexx Stent (A).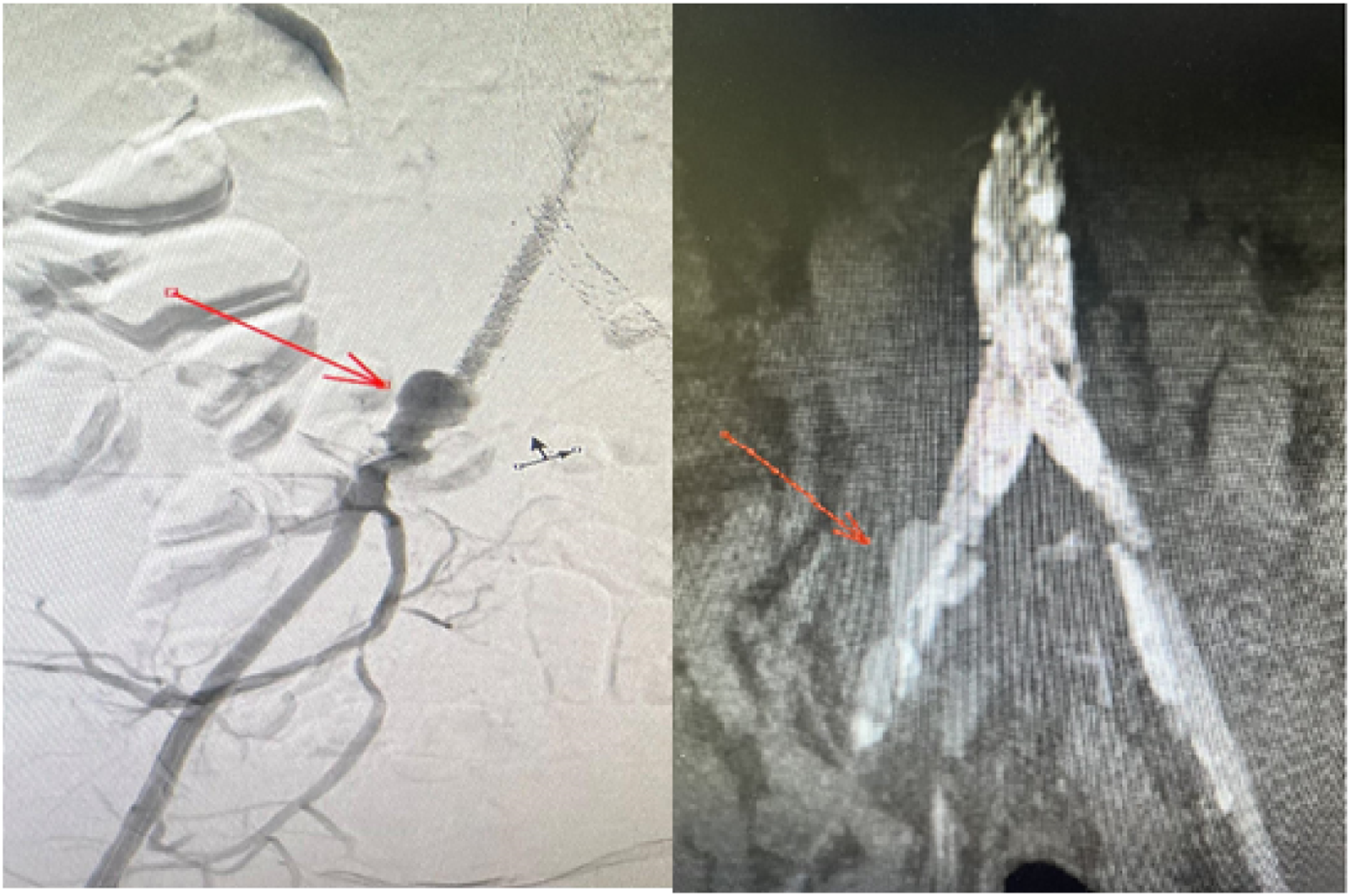
Subsequently, the patient underwent surgery. The surgical procedure consisted of two stages. The first stage included the implantation of an extra-anatomical ascending aortic-bi-femoral bypass. Through median semi sternotomy, the proximal anastomosis was performed in an end-to-side fashion with the ascending aorta. The bypass material was a Y-shaped prosthesis measuring 16 × 8 Dacron bonded with rifampicin. The bypass was intrathoracic and then subcutaneous tunneled to the femoral arteries. The distal anastomosis was attached to the femoral arteries in an end-to-side fashion.
One week later, the second stage of surgery was performed. This included a transabdominal explantation of the stents in the aortic and iliac arteries via medial laparotomy. The Luminexx stent, which was implanted in the aorta with supra-renal struts, was fixed to the supra-renal segment. Thus, the removal of the infra-renal segment of the stent was the only feasible option. This led to the decision of keeping the suprarenal parts; as the risks of injuring the renal and visceral arteries outweighed the benefits associated with its removal. The aortic stump was then closed, providing adjuvant sealing by using the greater omentum.
Postoperatively, the patient was transferred to the intensive care unit for close observation with continued improvement of her condition. Consequently, she was transferred to the peripheral vascular suite 4 days later. During that time, she displayed no signs of infection, ischemia, or inflammation. Three weeks postoperatively, the patient developed signs of transient ischemic attack with ocular manifestations. In response, a CT angiography was ordered, revealing luminal obstruction of the left internal carotid artery. However, the patient’s neurological abnormalities resolved within hours with no residual deficit. Thus, a mutual decision including the neurology department was made not to intervene. Over the course of the next few days, the patient continued to be stable with no subsequent complaints or complications. She was then discharged on continued flucloxacillin therapy for additional 6 weeks. CT scan was done 1 year later and showed no signs of infection at the aortic stump, with a completely patent aorto-bifemoral from ascending aorta bypass.
Discussion
The utility of percutaneous transluminal angioplasty and stent-graft implantation has been deemed prevalent and efficient in the management of vascular atherosclerotic disease. However, it is essential to acknowledge the potential complications associated with these procedures, including vessel thrombosis, recurrent stenosis, septic embolization, arterial dissection, rupture, as well as issues related to stent expansion, placement, and positioning. 4 Vascular graft infection is another potential complication that is uncommon, yet associated with considerable rates of morbidity and mortality if not managed appropriately. 5 The incidence of aortoiliac stent infection has been reported to range from 2%–6% with a mortality rate of approximately 48%, possibly owing to the commonly delayed diagnosis related to its nonspecific initial presentation.6,7
It’s important to note that the risk of stent infection is significantly increased with a history of diabetes mellitus and smoking. Other noteworthy risk factors include a break in sterility measures, other sources of coincident bacteremia, lack of antibiotic prophylaxis, multiple stent implantation, and multiple procedures in the same surgical site. 8 Melissa et al 9 reported a case of a 58-year-old patient who underwent Redo vascular interventions for diffuse atherosclerotic peripheral vascular disease, that was complicated by a severe stent graft infection likely due to repeated access through previously placed stents. In our case, we postulate that our patient’s age, history of smoking, and recurrent endovascular, same-site interventions have all contributed to the development of the reported clinical manifestations.
Additionally, the exact pathophysiology of stent graft infection is still not fully understood. It is suggested that endovascular intervention may damage the arterial endothelial lining, facilitating further damage and potentially inciting an inflammatory process in the stents. This can constitute an optimum condition for bacterial colonization and invasion, which can then yield infectious manifestations, necrotic tissue, and even perforation. 10
The clinical manifestations of stent infection are variable and consist of nonspecific symptoms subject to multiple factors making the diagnostic process challenging, warranting a high index of suspicion under the right circumstances. According to an exhaustive review of literature that included 76 cases of infected stents, patients would most commonly present with fever, malaise, chills, leukocytosis, local pain, edema, and potentially skin symptoms including petechial rash, or cellulitis. Given the vague nature of these symptoms, there is a consequent delay of diagnosis with an average duration of 16.4 days to definitive treatment. 1 Shukuzawa et al 11 have reported a median period of 30 months (14 days - 86 months) from the time of endovascular intervention to stent infection. According to the timing of endovascular intervention, stent infections can be classified into early (presenting within 2 months of the stent insertion) or late (presenting 2 months after the stent insertion). The early presentation is characterized by the above-mentioned systemic, nonspecific symptoms which necessitate the exclusion of other causes of sepsis. On the contrary, the chronic type would usually yield more localized manifestations directly related to the site of the implanted stent; abscess formation, cutaneous fistula formation, and pseudoaneurysms in addition to localized pain and edema. 12 As evident by our case and two other literature reviews assessing approximately 90 stent graft infection cases, most patients present within the first month following stent placement. 1 Our patient presented with nonspecific and systemic symptoms, which prompted an infectious workup for endocarditis, osteomyelitis, UTI, and pneumonia, all of which were excluded resulting in a significant delay that eventually led to a life-threatening arterial rupture.
As highlighted previously, stent infection is a diagnosis of exclusion once all other potential causes of bacteremia are ruled out. Detecting an infected endograft involves a combination of clinical symptoms, microbial cultures, and radiologic imaging. 13 The diagnosis and exclusion of various differentials often rely on several commonly employed imaging modalities, including ultrasonography as an initial step, followed by CT scans, angiography, and tagged WBC scans. In cases of early stent graft infections, a CT scan may not reveal any indications but, as the infection progresses, may show abscess formation, perivascular stranding, or false aneurysms. The presence of a pseudoaneurysm is an important radiological sign of stent infection. 9 Out of 76 cases reported in a literature review by Bosman et al 1 , 77.9% were found to have a pseudoaneurysm. PET CT scan has also proven to be a useful modality for identifying stent infections. 14 In our patient, the initial CT scan did not demonstrate any remarkable findings. As a part of planning, a subsequent CT Angiography revealed a pelvic hematoma with active bleeding. This was followed with a PET-CT scan showing extensive perivascular inflammation at the right common iliac artery, distal aorta, and the proximal part of the left common iliac artery indicating a stent graft infection.
On the other hand, a comprehensive microbiological assessment is crucial for guiding treatment. Commensal skin bacteria were the most identified organisms in reported cases of stent infections (87%), among these, staphylococcus aureus (including MRSA) constituted 76.6% of cases. The remaining pathogens were non-bacterial mostly attributed to immunocompromised and diabetic patients. 1 This microbial epidemiology reiterates the significance of abiding by correct sterility techniques peri-operatively. The blood culture results in our case grew staphylococcus aureus, supporting the conventional biological distribution and justifying the commonly used empirical antibiotic regimen.
The treatment of stent graft infection requires a multidisciplinary decision, and choosing the best management approach should be tailored individually for each case. It involves a combination of medical and surgical interventions, with the ultimate goal of eliminating the underlying infection. This involves replacing the affected vascular material and administering systemic antibiotics. 15 The surgical approach involves addressing the source of infection, stent explanation, and vascular reconstruction if needed.1,3 The conservative therapy option may only be considered for patients who are poor surgical candidates.16,17 Daptomycin and rifampin are usually administered given their ability to diffuse through the stent biofilm and potentiate their bactericidal effect. 18
In our case, the decision to opt for an ascending aortic to bilateral femoral artery bypass over alternative methods such as axillary bifemoral artery bypass or a descending thoracic to bifemoral artery bypass was deemed more suitable due to factors including the patient's anatomy, the extent of the vascular involvement, the surgeon’s expertise, and the urgency of the situation. It's essential to note that the selection of the revascularization technique should be individualized to each patient based on their specific clinical presentation and anatomical considerations. Regarding the technical aspects of the procedure, side-biting clamp was used to avoid full cardiopulmonary bypass. This technique allows for aortic clamping without completely stopping the heart and initiating cardiopulmonary bypass. This method was employed to minimize the risks associated with full bypass, such as potential clot formation, systemic inflammatory response, and other cardiopulmonary complications. However, it’s worth acknowledging that the choice of this particular technique, while suitable in this case, does come with certain inherent risks. For instance, if a future infection of this graft develops, this might necessitate hypothermic circulatory cardiopulmonary arrest to address the issue. This aspect should be considered in long-term management and follow-up planning for the patient. On the other hand, the choice between in-line and extra-anatomic bypass is often determined by the specific clinical circumstances and the risks and benefits for each patient. While in-line bypasses are commonly performed, in cases of severe and extensive vascular infections such as in our case, extra-anatomic bypass procedures can be a viable option to control the infection and maintain blood flow while minimizing surgical risks.
As for prevention, the European Society for vascular surgery (ESVS) recommends administering prophylactic antibiotics at least 30 min before performing a dental procedure that involves gengival manipulation in patients with vascular stents. According to Bosman et al, 1 some conditions warrant the use of antibiotic prophylaxis, and these include immunocompromised patients of different etiologies, complicated protracted surgical procedures, high-risk patients in which multiple punctures are expected, prolonged indwelling catheter use, stenting procedures in challenging anatomical locations with a high risk of restenosis, and patients with known pre-procedure colonization. The optimal duration for administering prophylactic antibiotics is still not clear. Hogg et al 9 proposed a life-long regimen similar to that prescribed following prosthetic valve operations.
Conclusion
Vascular stent infection is a life-threatening condition that should be investigated in patients with a history of vascular stent insertions presenting with vague non-localizing symptoms of unknown etiology. A combination of clinical, radiological, and laboratory investigations are often required for a definitive diagnosis. The surgical approach remains the definitive treatment modality for the majority of cases. Importantly, an iatrogenic component has been strongly linked to the development of stent infections, specifically in instances where strict adherence to aseptic surgical techniques was compromised. This showcases the importance of developing and implementing evidence-based guidelines for surgical procedures on both the institutional and individual levels. Further studies are required to decipher the specific causes, course, and long-term prognosis of this condition.
Footnotes
Acknowledgments
The authors thank the patient and his family for their great cooperation in the development of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
