Abstract
Purpose
To demonstrate the feasibility and outcomes of using the microvascular plug (MVP) for intentional occlusion of directional branches (DB) during complex endovascular aortic procedures.
Case Report
Two patients were treated with the off-the-shelf four-branched Zenith t-Branch thoracoabdominal stent-graft (Cook Medical, Bloomington, Ind). In both cases, the renal arteries (on one side in patient #1 and on both sides in patient #2, respectively) were occluded at time of index intervention. Preoperative planning included the intentional occlusion of each DB with one 7Q-MVP. Technical success was achieved in all cases and maintained at mid-term follow-up (12 months and 36 months, respectively).
Conclusions
Use of the MVP appears to be feasible and effective for intentional occlusion of DB during complex endovascular aortic repair. This novel and simple technique may present some unique technical advantages, allowing precise placement of the device while avoiding extension using a bridging stent and DB cannulation with larger sheaths.
Keywords
Introduction
The evolution of endovascular technology has expanded the indications and applicability to patients with extensive thoracoabdominal aortic aneurysms (TAAAs) and acute aortic syndromes (AASs), using multibranched stent-grafts (BEVAR) particularly in patients considered “unfit” for open surgical repair.1-4 The use of “off-the-shelf” endografts has the great advantage to avoid the waiting time for production and delivering of custom-made devices, thereby allowing treatment of patients in a shorter period of time and in urgency/emergency settings.5,6 However, in particular circumstances, there may be the need to voluntarily occlude one (or more) directional branch (es) (DB), due to clinical or anatomical reasons.
7
Failure to do so, when clinically indicated, would result in high-flow type III endoleak with sustained perfusion of the aneurysm sac. In this manuscript we report a novel technique for intentional occlusion of DB in two patients using the 7Q microvascular plug (MVP). The MVP (Medtronic, Minneapolis, MN, U.S.) consists of a self‐expanding nitinol frame with the proximal half covered in polytetrafluoroethylene (PTFE) and has been reported for embolization of different vascular territories.8-11 The unconstrained diameter of the MVP-7Q device is 9.2 mm and it is indicated for occlusion of vessels up to 7 mm in diameter. The unconstrained length of the MVP‐7Q is 16 mm and it can be deployed through a 4‐Fr catheter. In this case-series, we report a novel technique for occlusion of DB during BEVAR interventions, using the MVP device and describe the immediate technical results as well as the mid-term outcomes. A, intraoperative completion angiography showing complete occlusion of the right renal branch with patency of all remaining branches. B, CTA at 12 month showing good placement of the microvascular plug with absence of any type 1 or 3 endoleak.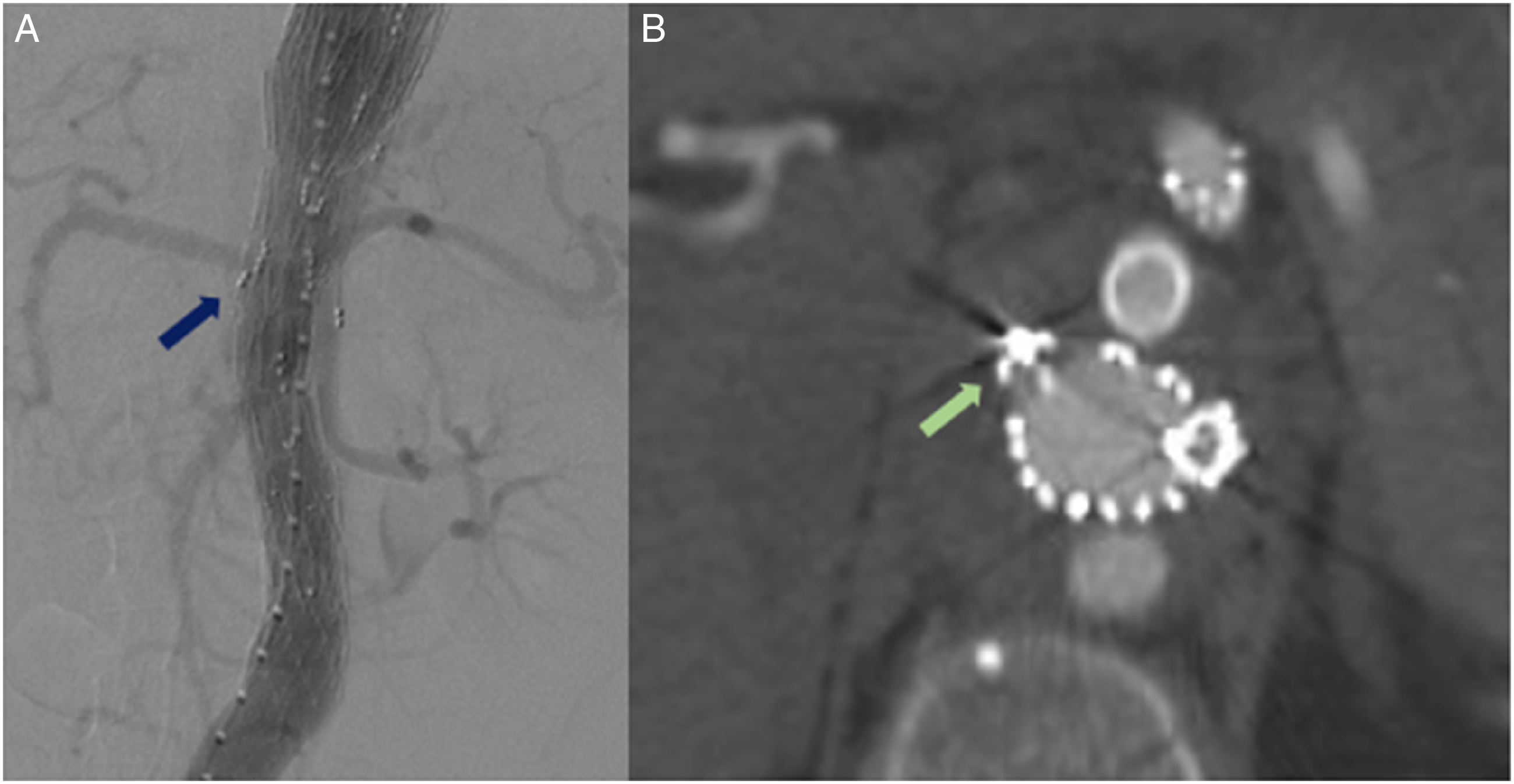
Case-Series
(1) Informed consent has been obtained from the patients for publication of the case report and accompanying images. (2) Ethical approval from the local board was not required for publication of case-reports. (3) Case 1. The first case describes a 61-year-old female patient, affected by arterial hypertension, and admitted to the emergency department for retrosternal, abdominal and back pain. A computed tomography angiography (CTA) detected cute an acute intramural hematoma (IMH), extending from the proximal descending thoracic aorta down to the iliac axes, with complete occlusion of the right renal artery (RRA). The patient was initially admitted at the Intensive Care Unit (ICU) and treated by medical therapy; she was discharged at home seven days later with adequate arterial blood pressure control and with plans for CTA in one month. However, subsequent CTA one month later showed significant enlargement of the paravisceral aorta. For this reason, an endovascular treatment was planned on urgent basis. Under general anesthesia and by right femoral artery surgical access, Zenith (Cook Medical, Bloomington, IN, USA) 36-36-159 thoracic endovascular aortic repair was deployed at Ishimaru zone 4, with a distal extension by Zenith t-Branch. From a left upper extremity approach, the DB for the superior mesenteric artery (SMA), celiac trunk (CT) and left renal artery (LRA) were sequentially catheterized and stented using self-expanding covered stents (Covera, Bard, Tempe, Arizona; 8 × 60 mm, 8 × 60 mm and 6 × 60 mm, respectively. Owing to the off-the-shelf design of the multibranched endograft and the absence of a target RRA in which to land a covered stent, the decision was taken to occlude the residual DB during the same operation. The MVP-7Q was introduced through a 4-Fr Bernstein catheter directly into the DB for the left renal artery (LRA) and deployed as the catheter was withdrawn from the DB. Technical success was achieved at the end of operation (Figure 1A). No perioperative complications were observed, without any MVP-related adverse events. Twelve months later, a new CTA showed the MVP in place, without signs of type I or III endoleak and reduced diameter of the IMH (Figure 1B). (4) Case 2. The second case describes a 51-year-old male patient, active smoker, with Bechet’s disease, who had undergone bilateral nephrectomy for renal tumor and previous abdominal surgery for bowel obstruction, actively receiving hemodialysis by left brachio-cephalic forearm arteriovenous fistula. The patient was admitted at the emergency department for acute lumbar and dorsoscapular pain. The CTA revealed a contained rupture of the supra-visceral aorta (his CTA from one year before was otherwise normal), and the patient was hemodynamically stable. Under general anesthesia, an off-the-shelf Zenith t-Branch was deployed at Ishimaru zone 4. From a right upper extremity approach, the DB for the CT and SMA were sequentially stented using balloon-expandable covered stents (8L-59 VBX stent-graft and 8L-79 VBX stent-graft, respectively; W.L. Gore, Flagstaff, AZ, USA). Owing to the urgent presentation and the concerns related to leaving a high-flow type III endoleak from the DB (as the patient did not have any residual target renal artery due to the prior bilateral nephrectomy), it was decided to perform the occlusion of both renal DB during the same operation. Therefore, selective catheterization of both renal DB was performed, with deployment of one MVP-7Q into each using a 4-FR Bernstein catheter. In order to close the branches. The procedure was then completed with a Zenith distal unibody extension and two iliac limbs. Technical success was achieved at the end of operation No perioperative complications were observed, without any MVP-related adverse events. A CTA, performed 36 months after the procedures, showed the MVP in place, without signs of type I or III endoleak and reduced diameter of the IMH (Figures 2A and 2B).
Discussion
Contemporary reports of BEVAR have shown high technical success rates, with low mortality and morbidity that have contributed to expanding its indications and applicability.12,13 The technique of BEVAR invariably requires catheterization of DB and, through them, of their intended target vessels which may be accomplished using different technical options.
14
Variations of the normal renal and mesenteric arterial anatomy have been described and provide the rationale basis for off-the-shelf devices that are currently available or under investigations.
15
These devices avoid the time delay needed for custom-made stent-grafts, which can reduce the adverse events during waiting time and allow for expedite treatment or non-elective cases.
16
Although there is some variations in design between manufacturer, most apply the concept of four down-going cuffs or branches, which are intended for the CT, SMA, and renal arteries. CTA at 36 months with 3D volume-rendering A, and transversal view reconstructions B, showing patency of visceral branches and sustained occlusion of renal branches by the microvascular plugs.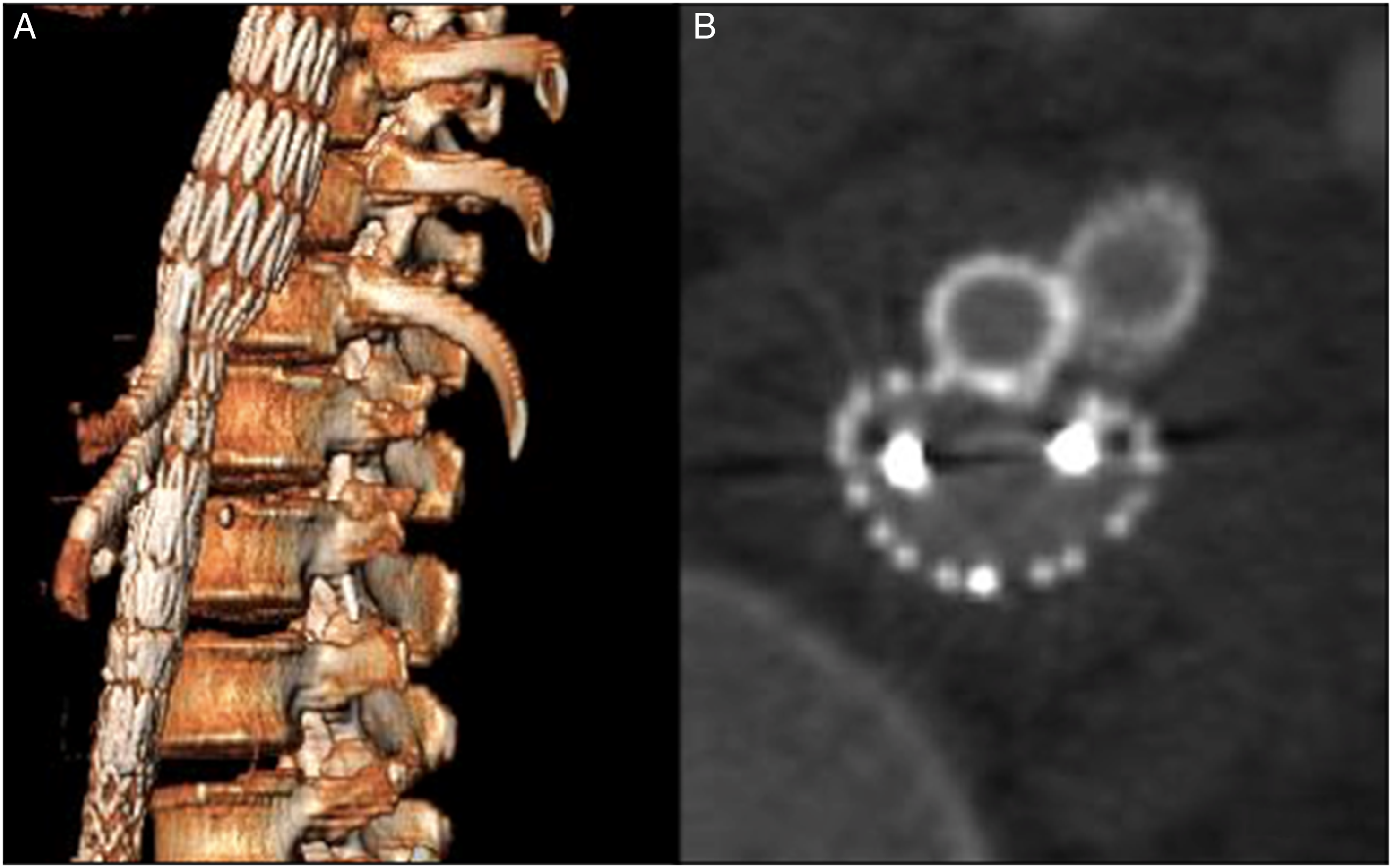
Intentional occlusion of one or more DB in patients with less than four TVs may be needed to accommodate variations in renal and mesenteric anatomy and has been previously reported using an accepted approach that entails stenting of the DB itself followed by its occlusion using the standard Amplatzer Vascular Plug (AVP). 7 Therefore, this technique may enable use of four-vessel multibranched off-the-shelf devices in patients who have chronic occlusion, surgical or congenital absence of one of the renal or mesenteric vessels, single functioning kidneys, pre-existing dialysis, or in patients with difficult anatomy in whom TV catheterization and stenting is not technically possible. The extension before placement of the AVP, as reported by Tenorio et al, intends to facilitate deployment and allow the full length of the plug into the branch cuff to be occluded, achieving maximum sealing length by avoiding the protrusion of the AVP discs outside of the branch cuff. 17 As the branch cuffs are usually 18 mm to 20 mm in length, most experts would recommend extending at least 20 mm beyond the end of the DB cuff (ie achieving a total length of about 40 mm) to ensure adequate sealing of the AVP.
By contrast, our technique only requires the use of a single MVP device owing to its shorter length that can allow precise placement into the full length of the target DB. The MVP device is currently approved for use in native venous and arterial vessels, but to the best of the authors’ knowledge its use has never been attempted within the environment of an endograft branch. The MVP device is currently available in four different sizes (3-5-7-9; the smaller two can be delivered via .021” and .0272 microcatheters, and can occlude vessels of 1.5-3.0 mm and 3.0-5.0 mm, respectively; the larger two require 4F and 5F angiographic catheters, and can occlude vessels of 5.0-7.0 mm and 7.0-9.0 mm respectively). The MVP device is attached to its own delivery wire and is fully retrievable/repositionable until detachment is accomplished by clockwise manual rotation of the distal end of the delivery wire.
Of the four available sizes, the MVP‐7Q is suitable for occlusion of vessels up to 7 mm in diameter and was therefore selected in the described cases for occlusion of 6 mm DB. In addition to potential cost-savings (owing to avoidance of stent extension into the DB before plug placement), there are several technical aspects that may prove particularly helpful and deserves further discussion. First and foremost, the MVP-7Q is deliverable through a 4‐Fr catheter, thereby avoiding the need for using larger diameter sheaths that are needed for placement of vascular stents. Also, the MVP has relatively less nitinol in its framework compared to the AVP, which makes the device less stiff and easier to track and navigate. Indeed, it is usually advised to oversize the Amplatzer plugs with 20% to 50% in comparison to the vessel diameter in order to achieve dense packing of the nitinol wires and subsequently to induce a rapid thrombosis 18 ; in contrast, the MVP does not require much oversizing, and occlusion is almost immediate.
Another potential advantage of the MVP compared to the AVP is the potential to induce an immediate and complete occlusion irrespective of the hemostatic status of the patient, as the thrombogenic effect is related to the PTFE layer which completely and immediately occludes the vessel, even when the landing zone is short. Therefore, a single device would be sufficient in most cases, as described in our experience. Lastly, despite the lack of retention discs, we had no cases of device embolization with excellent occlusion rates up to 12 and 36 months. However, from a technical standpoint, it is crucial that the device be placed in a straight segment as placing it across a curvature may entail poor apposition of the PTFE layer to the inner wall thereby leading to potential risks for dislodgment and migration.
The major limitation of the study is the retrospective nature and limited number of cases available, although this may be considered acceptable owing to the newly introduced device for this particular procedure. Also, the advantages listed above are based on the operators' experience and may reflect personal preferences and skills.
Conclusions
Intentional occlusion of DB using the MVP is feasible and effective thereby allowing use of a four-vessel off-the-shelf multi-branched stent-grafts in patients with need to cannulate less than four renal-mesenteric TV. There were no endoleaks or secondary interventions associated with the occluded DB. This novel technique requires larger experience with longer follow-up to ascertain its durability, as well as comparative assessment of its outcomes vs other available technical options.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
