Abstract
Objectives
Colonic ischaemia is a rare but devastating complication of open aortic aneurysm repair and is associated with high morbidity and a mortality of up to 50%. The aim of this study was to determine the safety and effectiveness of using indocyanin green florescence (ICG) to interrogate colonic perfusion intra-operatively.
Design
Prospective observational study.
Methods
All elective open abdominal aneurysm repairs over a 6 month period underwent colonic perfusion interrogation with ICG according to a pre-defined protocol. Patient demographics and imaging findings were recorded prior to surgery. ICG was given just prior to laparotomy closure. Time to florescence was measured from the start of IV administration to surgeon defined maximal florescence of the sigmoid colon.
Results
Ten patients fulfilled the inclusion criteria. All patients were male with an average age of 69.7 years. Inferior mesenteric artery reimplantation was performed in 5 patients. Median colonic fluorescence time was 58 s. No complications related to ICG were identified. A single patient had clinical concern of colonic ischaemia and delayed perfusion (>3 min) on ICG; colorectal opinion advised not for immediate resection. At relook laparotomy, ischaemic colon at the area of demarcation was noted and a Hartmann’s procedure was performed. No other patients had delayed perfusion and no further episodes of colonic ischaemia were noted. IMA reimplantation did not show statistical difference in colonic ICG time (P = .81, 95% CI -1.98 to 2.45). There was no statistical difference between operating times between the cohort and all repairs performed 6 months before the data collection (P = .59, 95% CI -.73 to 1.24).
Conclusion
In this pilot study ICG appears to be a safe and useful adjunct in objective assessment of colonic perfusion during open AAA repair. Further research is required to fully determine its role in this cohort of patients.
Keywords
Introduction
One of the most devastating complications during open abdominal aortic aneurysm repair (OR) is colonic ischaemia. The incidence of colonic ischaemia in OR is reported to be between 1-3% but is associated with high morbidity and a mortality of up to 50%. 1 Patients will often require colostomy formation and very few patients that undergo emergency stoma formation go on to have reversal. 2 Intra-operatively, colonic perfusion is often judged on subjective clinical findings; warmth, appearance, and presence of mesenteric pulsation alongside intra-operative doppler interrogation of the mesenteric vessels. A variety of other methods are available to better assess perfusion including intra-operative angiography, duplex ultrasonography and transit-time flow measurement which will give more objective assessment. Most of these methods are invasive and are time consuming, resulting in prolonging operative time in a major procedure. With colonic ischaemia, the only intervention that re-establishes flow is reimplantation of the inferior mesenteric artery (IMA). This is still a controversial topic within vascular surgery as research into IMA reimplantation has not been shown to provide any reduction in the rate of perioperative mortality or morbidity as there is little high-quality evidence to base this on. 3 The most recent ESVS guidelines recommend re-implantation only in selected cases of poor colonic perfusion. 4
Indocyanin green (ICG) is a non-toxic cyanin dye which demonstrates near-infrared fluorescence at frequencies of 800-1000 nm. In circulation, ICG has a half-life of 150-180 s and is metabolized in the liver. Although it is non-toxic, a maximal dose of 2 mg/kg is advised; the typical administration dose of 7.5 mg used to assess bowel perfusion being far under this. ICG has been utilized in many other areas including opthalmology, neurosurgery and in colorectal surgery to determine colonic perfusion prior to anastomosis. 5 In vascular surgery, the use of ICG is still in its infancy. A small study by Perry et al demonstrated utility of ICG as a non-iodinated contrast agent to assess flow through peripheral arterial bypass grafts and demonstrate demarcation for area of surgical debridement.6,7 In aortic surgery, a single case describes the use of ICG to demonstrate perfusion of the colon during aortic aneurysm repair in 2 patients. 8 More recently, ICG has been used successfully to determine colonic perfusion after IMA ligation for type II endoleak management.9,10
Given the good safety profile and its utility in assessing perfusion, this case series aimed to determine whether ICG could be used safely to determine colonic perfusion during OR. To the best of our knowledge this is the first case series utilizing ICG in open aneurysm repair to interrogate colonic perfusion.
Materials and Methods
All patient undergoing open aortic aneurysm repair during a 6-month period in the vascular unit of a tertiary referral centre from a prospectively collected database were included in the analysis. All patients, after reaching aortic measurement of 5.5 cm were discussed in a weekly multi-disciplinary team meeting and deemed suitable after fitness was assessed by a vascular anaesthetist and had no contraindications to open repair. Patients were excluded for the following criteria: rupture/perirupture AAA, previous anaphylaxis to ICG or iodinated contrast, previous colonic resection or clinical concern on the use of ICG intra-operatively (for example prolonged operating time and patient instability). The centre’s policy was to re-implant the artery during OR if the IMA was patent and of a calibre that amenable to anastomosis as determined by the operating surgeon at the time of surgery, with a 5 mm vessel generally deemed appropriate for re-implantation. Decision making was also guided by the imaging findings including the calibre ad patency of the other mesenteric vessels including the hypogastrics. Patients were individually consented for the use of ICG during the procedure.
ICG was given according to a protocol (see Appendix 1) and measured using SPY-PHI™ (Stryker, Mich, US). The ICG protocol utilized was developed by the centres colorectal surgery department and based on evidence in assessing colonic perfusion during colorectal anastomosis with amendments made for the context of OR. A dose of 3 mL of ICG was given intravenously by the anaesthetist at a standard concentration of 2.5 mg/mL followed by a bolus of 10 mL of saline. Timing was performed using a standard electronic stop-watch from the time of administration. ICG was administered after completion of both anastomoses prior to closure and at a point where the patient’s haemodynamics were stable as determined by the anaesthetic team.
Qualitative analysis by the operating surgeon was used to stop the timer; timing was stopped when the colonic fluorescence was determined by the operating surgeon and the surgical assistant to be of the same intensity as the small bowel.
Patient demographics and operating details were obtained from the electronic patient records as well as paper notes.
Ethical approval was previously granted by the institution for the use of ICG for assessment of colonic perfusion, so further ethical approval was not sought. It was established that the standard of colonic perfusion assessment was using ICG and therefore the study was registered and approved by the local audit department as a service evaluation (CA_2021-22-215).
Results
Patient Demographics and Co-Morbidities.

All were male, with average age of 69.7 years. Ischaemic heart disease and hypertension were the most common co-morbidities and 60% patients were ex-smokers. All patients had healthy coeliac and superior mesenteric as well as patent internal iliac arteries. There was no morbidity or mortality within 30 days of surgery. No patients experienced anaphylaxis from ICG.
Mesenteric Vessel Status, ICG Times and Outcomes.
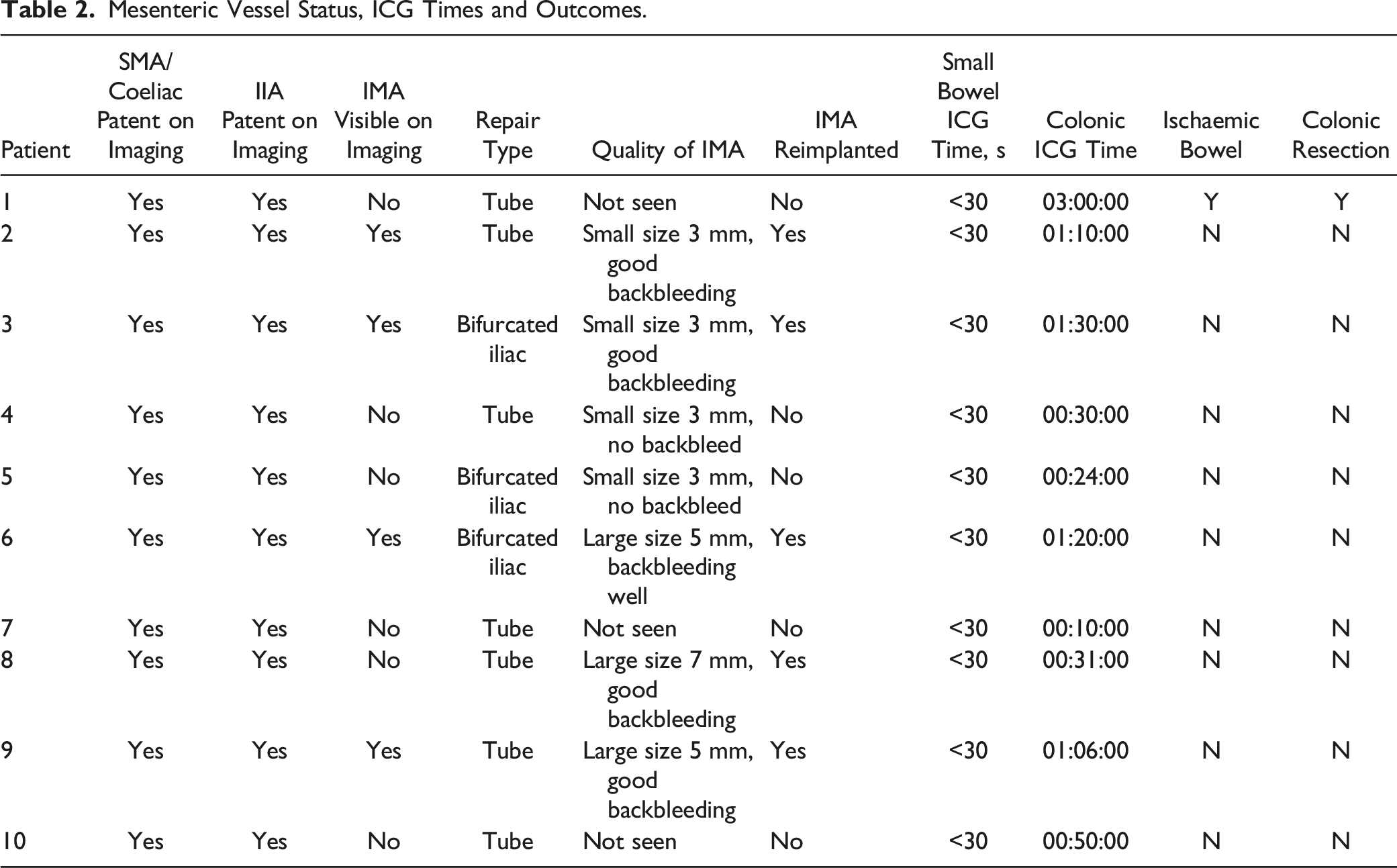
A single patient had clinical concern of colonic malperfusion. After completion of the anastomosis, the bowel appeared somewhat dusky, slightly cooler and no doppler signals were identified in the mesentery of the sigmoid segment of concern. Colorectal surgical opinion was sought. ICG interrogation demonstrated no perfusion in an area of demarcated sigmoid colon on initial administration (see Figures 1 and 2). Subsequent administration did not improve perfusion significantly. Two colorectal surgeons determined that although there was delayed perfusion, resection was not warranted and the patient underwent planned relook laparotomy 48 hours later. Clinically during this period the patient was haemodynamically stable with a soft but mildly distended abdomen. The only clinical sign of concern was 1 episode of vomiting the morning of the re-look laparotomy. White blood cell count and lactate remained within normal limits. Re-look laparotomy revealed ischaemic colon in the area demonstrated by ICG fluorescence and a sigmoid colectomy and end colostomy was performed. The patient was discharged home 14 days after resection; the inpatient stay complicated by prolonged ileus lasting 5 days. Area of demarcation of ischaemic segment. Ischaemic colon at area demonstrated by delayed ICG florescence at relook laparotomy.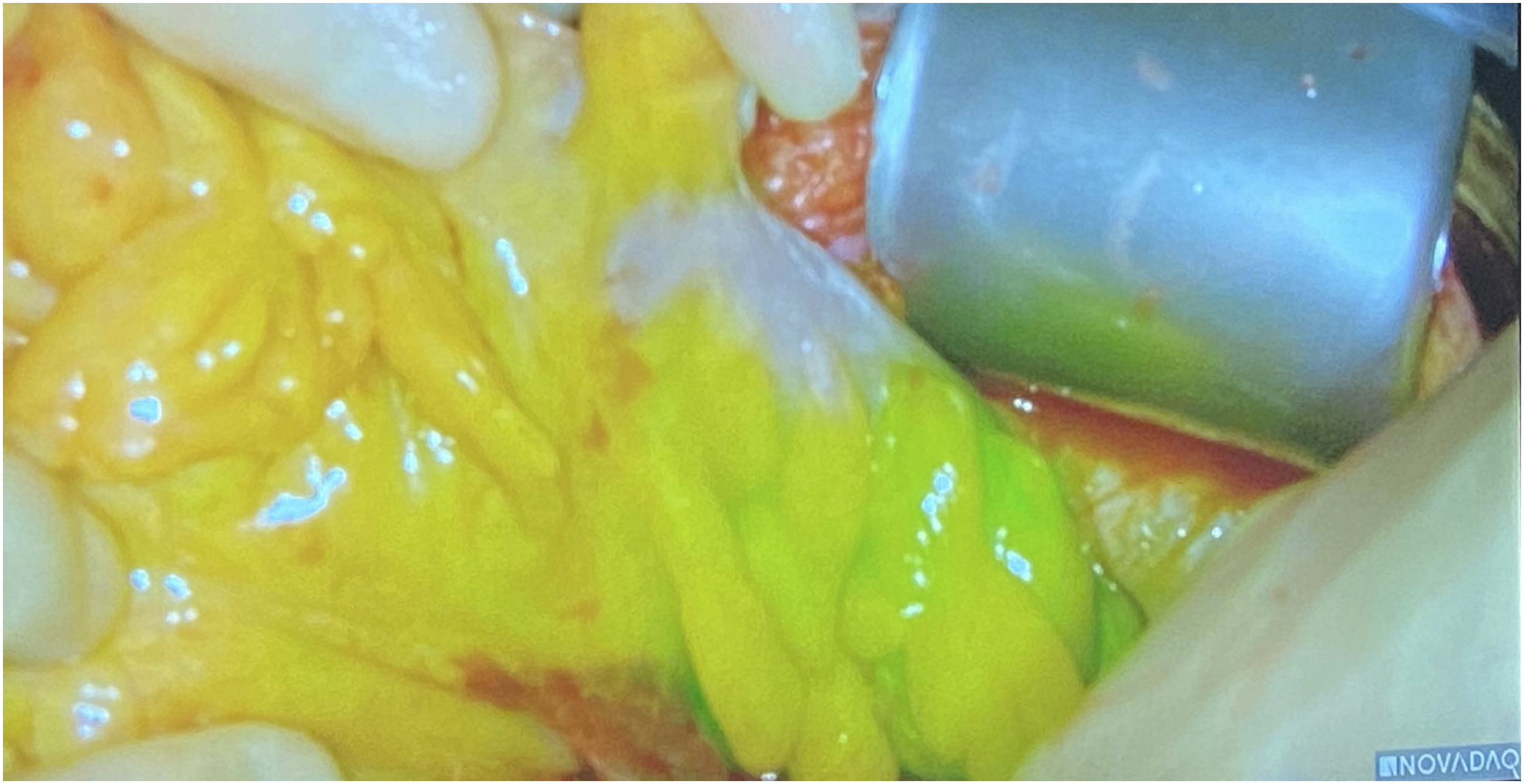
A single patient had technical failure of the initial ICG administration due to setup of the camera stack which led to a 15-min delay. However, once the camera stack issue was addressed further ICG administration demonstrated good uptake under 3 min. The patient was discharged home without any complications. No adverse cardiopulmonary events or renal failure requiring dialysis occurred in any patients.
The average operative time for the 10 cases was 4 h 29 min. When compared to all open AAA repairs done 6 months prior to the initiation of the study, no statistically significant difference was demonstrated (unpaired t test, P = .59, 95% CI -.73 to 1.24).
Discussion
Ischaemic colon is a devastating complication and results in high morbidity and mortality when it is presence is not recognised sufficiently early. There have been many previous efforts to isolate risk factors for colonic ischaemia and efforts to determine colonic perfusion, via pressure monitoring or other methods, have not provided sufficient evidence for their routine use.
This case series demonstrates the safe use of ICG to interrogate colonic perfusion during in open AAA repair. ICG was noted to be easy to use with no statistically significant difference in the operating time. Delayed fluorescence appears to be a marker of poor colonic perfusion and was a useful adjunct in addition to clinical suspicion.
We acknowledge this study has several limitations. Ischaemic colon is a rare complication of OR and the sample size is small. The sample was also exclusively male and although there is a typical male predisposition for aneurysmal disease, the lack of female patients limits the generalizability of this study. The other limitation is that there was no quantitative analysis of ICG fluorescence and was qualitatively assessed by surgeon, which limits the reproducibility of the findings. There is some evidence in colorectal surgery that the time to maximal fluorescence is an important determinant of perfusion and quantitative analysis would have eliminated this potential bias. 11 There is also no consensus in colorectal surgery regarding ICG parameters, with acceptable times to qualitatively determined florescence ranging from 30 s to 4 min. 11 It must be appreciated however, that the haemodynamic changes in patients undergoing OR comparative to colorectal resection is vastly different. In our study, a single patient who had delayed sigmoid perfusion of greater than 180 s had ischaemic complications whereas the rest of the cases with sigmoid perfusion under 90 s did not have any complications. It appears that sigmoid fluorescence under 90 s indicates good perfusion and low risk of ischaemic colon. However, it is still unclear as what is an adequate time to demonstrate perfusion although delayed perfusion of more than 180 s should give the clinician suspicion of the risk of colonic ischaemia. Another significant limitation is that haemodynamic parameters were not included in this initial study, given the frequent haemodynamic changes during OR. However, as there was rapid florescence within the small bowel initially after ICG administration any volume depletion or vasopressor usage should have been evident from this. The final limitation is no comparator was utilized in subsequent patients without clinical concern of ischaemia; hand-held doppler signals being a straight-forward and useful adjunct for assessing perfusion. However, the utility of ICG allows assessment of the actual colonic perfusion and in the case of malperfusion, the extent of the ischaemic segment.
We recognize that there is an important subgroup missing from this study as patients with ruptured aneurysms are more likely to have colonic ischaemia. 1 The higher utilisation of vasopressors and transfusions in ruptured AAA currently have unknown effects on colonic florescence. Overall given the sparsity of evidence of the use of ICG within patients undergoing OR, it was felt that a pilot study assessing the safety and feasibility of the technique was safer and further research is warranted in this high risk group of patients.
There are many questions raised by this study. The unit’s policy was to re-implant the IMA if this was an adequate vessel; the rationale behind IMA reimplantation was due to prior institutional experience of several cases of significant colonic ischaemia without reimplantation. This study however was not aiming to determine the role of IMA reimplantation and as reimplantation was done prior to ICG administration, it is not clear whether perfusion was improved by this procedure. However, ICG may in the future, allow for selective reimplantation in those that show evidence of poor perfusion. In this series, ICG gave further objective evidence that was concerning for ischaemia, prompting a planned relook and resection. The utility of ICG may also lie in cases where the patient is high risk for ischaemia or borderline cases, giving further objective evidence to plan a relook and planned resection rather than an emergency resection in a sick patient. Further evidence is required to address these 2 questions. Given its safe clinical profile and not requiring ionizing radiation, ICG may be an answer to the challenge managing ischaemic colon in open aneurysm repair.
Conclusion
ICG appears to be a safe and useful adjunct in objective assessment of colonic perfusion during open AAA repair. However further research is required to determine its role in determining colonic perfusion and IMA re-implantation.
Supplemental Material
Supplemental Material - Indocyanin Green Fluorescence Evaluation of Colonic Perfusion During Elective Open Abdominal Aortic Aneurysm Repair
Supplemental Material for Indocyanin Green Fluorescence Evaluation of Colonic Perfusion During Elective Open Abdominal Aortic Aneurysm Repair by Adam Tam, Ahmed Abdel-Rahim, Francis Dix, Jamie Barwell, and Devender Mittapalli in Vascular and Endovascular Surgery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
