Abstract
Objective
Endovascular aortic repair (EVAR) for elective and emergency infrarenal aortic pathologies is the primary approach for treatment nowadays. During such procedure, the suture-mediated closure device (SMCD) (Perclose ProGlideTM, Abbott Laboratories, Chicago, IL, USA) is commonly used. This study aimed to identify potential contributors for SMCD failure in a patient cohort of elective and emergency EVAR.
Methods
Archived medical records from patients who underwent EVAR for aortic pathologies in elective and emergency setting at the University Hospital Düsseldorf, Germany were included. Patient’s co-morbidities, access vessel morphologies and hemostasis-related blood parameters were evaluated on their association with SMCD failure applying different statistical methods.
Results
A total of 71 patients (139 femoral accesses) was included. The mean age was 73.5 ± 8.4 years. Overall SMCD failure rate was 4.3%, 4.1% for elective and 5.9% for emergency cases, respectively. Total procedure time was longer for the SMCD failure group (323 ± 117.8 min vs 171 ± 43.7 min). The calcification status of the common femoral artery (CFA), the diameter of the aortic bifurcation, and dual anti-platelet therapy (DAPT) on the medication plan prior to the procedure were associated with SMCD failure. Univariate binary logistic regression analysis nominated several potentially relevant predictors for SMCD failure who underwent subsequent multivariable binary logistic regression analysis. Here, DAPT on the medication plan was identified as being promising in predicting SMCD failure (OR 30.5), while anterior plaque formation in the CFA maintained as only statistically relevant determinant (OR 44.9).
Conclusions
This study confirms the CFA calcification status to be associated with SMCD failure. Although discontinued prior to endovascular treatment, DAPT was also found to be associated with SMCD failure. Our results may advocate to perform obligatory platelet testing prior to EVAR to maximize patient safety.
Keywords
Introduction
Abdominal aortic aneurysm (AAA) is defined as a local dilation of the aorta of greater than 50% compared to non-diseased adjacent aortic segments and predominantly located in the infrarenal aortic segment. 1 AAA prevalence is 1.6-7.2% in the general population aged 60 years or older while AAA-related mortality is estimated to account for 150,000-200,000 deaths worldwide per year.2,3
Current guidelines recommend invasive elective treatment for AAA exceeding 55 mm maximum diameter in men and >50 mm in women.4,5 Endovascular aortic repair (EVAR) has evolved over the last decades as first-line treatment for AAA with the majority of patients undergoing EVAR, while open aortic repair (OAR) is still considered beneficial for patients with extended life expectancy. 6 During EVAR, an arteriotomy is mandatory to gain access to the aorta and several methods for bleeding control following the procedure have been developed over time.
Manual compression was initially used for smaller arteriotomies (<8 French (F)) but necessitates close follow-up observation while causing significant patient discomfort. 7 Therefore, closure devices for total percutaneous access (TPA) were developed to minimize access side trauma without the need for surgical cut-down (SCD).8,9 Of interest, the use of TPA during EVAR procedures is reported to cause similar access-side complications when compared to SCD. 10 TPA devices are categorized as plug-based and suture-mediated closure devices (SMCD). 11
The implementation of SMCD into clinical routine facilitated TPA during EVAR. 12 Potential parameters that are associated with SMCD failure, such as the body mass index (BMI), a history of peripheral arterial occlusive disease (PAOD), the presence of common femoral artery (CFA) calcification, and the depth of the skin puncture site have been reported recently. 11 Comprehensively, SMCDs have reduced the necessity for SCD while reducing both, patient’s morbidity, and procedural costs. 13 Although the expertise in using SMCD has been increasing among users, their failure rates are still reported as being as high as 2-8% when using sheaths greater than 16F for percutaneous accesses. 11 That being said, there is still a need to expand the current knowledge in using SMCD during EVAR procedures to confirm established and identify potentially new parameters that predict SMCD failure aiming to increase patients’ safety.
This retrospective analysis evaluates patient characteristics, relevant morphological dimensions and hemostasis-related blood parameters regarding their association and potential in predicting SMCD failure during elective and emergency EVAR procedures.
Material and Methods
Archived medical records from patients who underwent elective and emergency EVAR using SMCD (Perclose ProGlideTM, Abbott Laboratories, Chicago, IL, USA) for infrarenal aortic pathologies at the Clinic for Vascular and Endovascular Surgery at the University Hospital Düsseldorf, Germany from 1 January, 2016 through 30 April, 2023 were screened for patients’ characteristics and procedural details. All procedures were performed by experienced endovascular consultants. During EVAR procedures, 2 SMCDs were placed in femoral accesses before the main body or contralateral leg were inserted, thus 2 SMCDs were used in femoral accesses with sheath size of ≥ 14F. Of note, for femoral accesses with sheath sizes of 6-8F during e.g. unibody stent grafts, only one SMCD was placed. Preoperative blood tests were analyzed. Computer-assisted tomography (CAT) scans of patients prior to EVAR were examined for morphological dimensions. Data is reported as mean ± SD or absolute frequency with percentages. Inclusion criteria were as follows: elective or standard EVAR with total percutaneous femoral access for AAA, penetrating aortic ulcer (PAU) and/or dissecting aortic aneurysm (DAA), >18 years of age. Exclusion criteria were as follows: endovascular treatment for thoracic aortic aneurysm (TAA) and/or paravisceral AAA, previous endovascular surgery with femoral vascular access, <18 years of age, planned SCD prior to surgery, no available CAT scan within a 6-month period prior to EVAR. Potential parameters were evaluated for their association with SMCD failure applying Mann-Whitney U test for continuous and Chi-squared test (Fisher’s exact test) for dichotomous variables following Shapiro-Wilk test for normality. Univariate binary logistic regression was conducted to predict SMCD failure. Here, variables with a P-value of < .1 were included in a subsequent multivariable binary logistic regression analysis. A P-value of P < .05 was considered statistically significant. The local Ethic Committee at the Medical Faculty of the Heinrich-Heine-University Düsseldorf approved the study (Number: 2022-2204).
Results
We report 6 SMCD failures in 5 patients, thus the overall SMCD failure rate was 4.3% during 139 femoral accesses in a collective of standard and emergency EVAR procedures. There was a total of 61 AAA, 8 PAU, and 2 DAA. SMCD failure occurred during 2 right and 4 left femoral accesses. There were 62 elective and 9 emergency EVAR procedures. Within the study cohort, there was one aorto-monoiliac stent-graft placement for a ruptured AAA (rAAA) procedure, and two aortic tube stent graft placements for a ruptured/symptomatic infrarenal PAU. Such procedures required only one femoral access. Moreover, there were 5 unibody stent graft and 63 bifurcated stent graft placements. Of interest, SMCD failure occurred during femoral accesses in 5 elective and 1 emergency setting. In more detail there was 1 SMCD failure in 1 (right) of 17 a femoral access during emergency EVAR procedures, thus the SMCD failure rate was 5.9%. For elective EVAR procedures, we report an SMCD failure rate of 4.1% in 122 femoral access, of which 4 occurred during left femoral access and 1 during a right femoral access (p = non-significant).
Patient Characteristics
Patient Characteristics (n = 71). Dichotomous data is shown as total numbers with percentage [%]. Continuous variables are presented as mean ± SD.
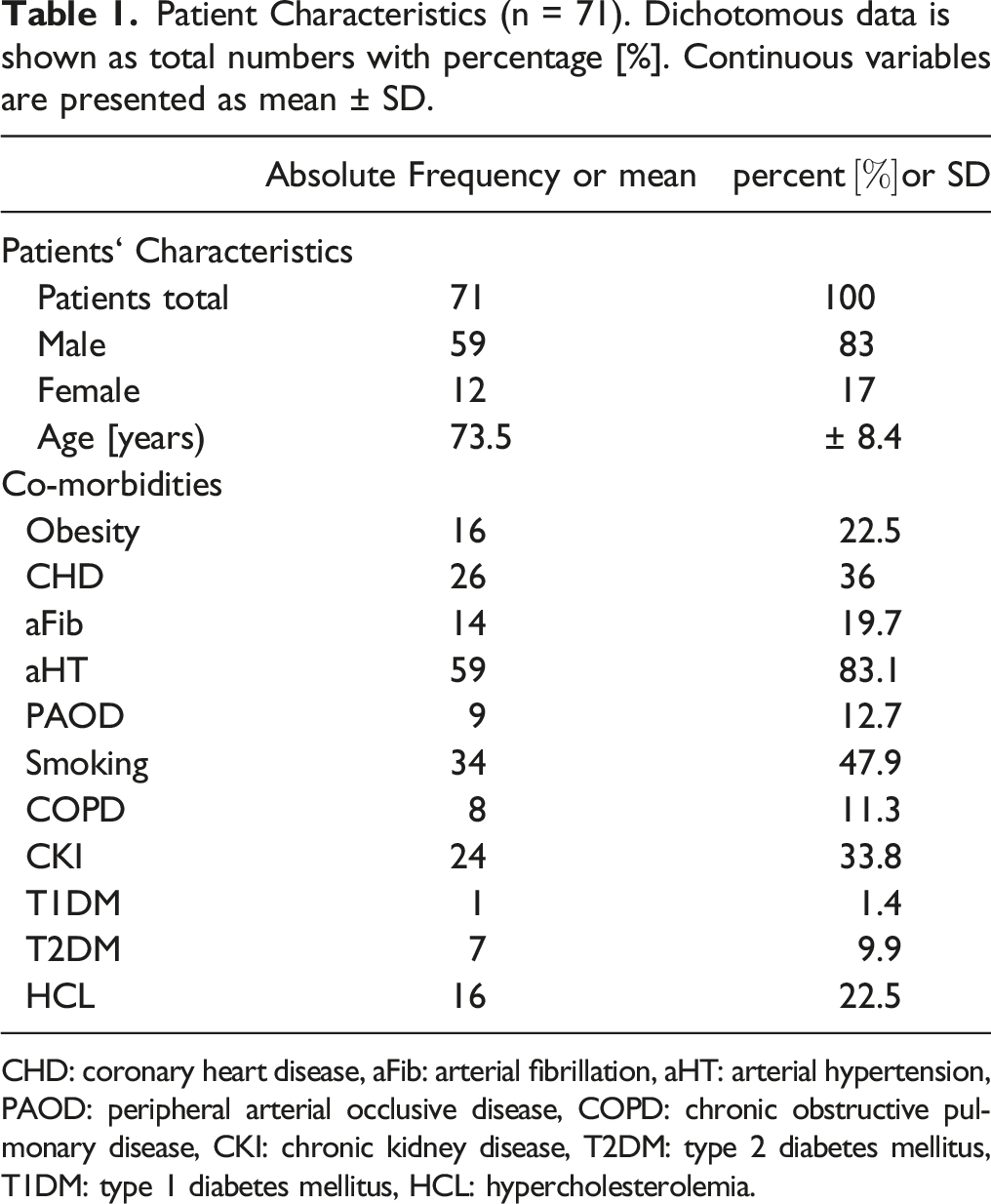
CHD: coronary heart disease, aFib: arterial fibrillation, aHT: arterial hypertension, PAOD: peripheral arterial occlusive disease, COPD: chronic obstructive pulmonary disease, CKI: chronic kidney disease, T2DM: type 2 diabetes mellitus, T1DM: type 1 diabetes mellitus, HCL: hypercholesterolemia.
Co-morbidities (n = 139 femoral accesses). The table illustrates patients' co-morbidities at baseline. Data presents the contingency table for (no) suture-mediated closure device (SMCD) failures for each variable. Pearson-Chi-Square test (Fisher’s exact test) was applied to evaluate the association between patients’ co-morbidities and SMDC failure.
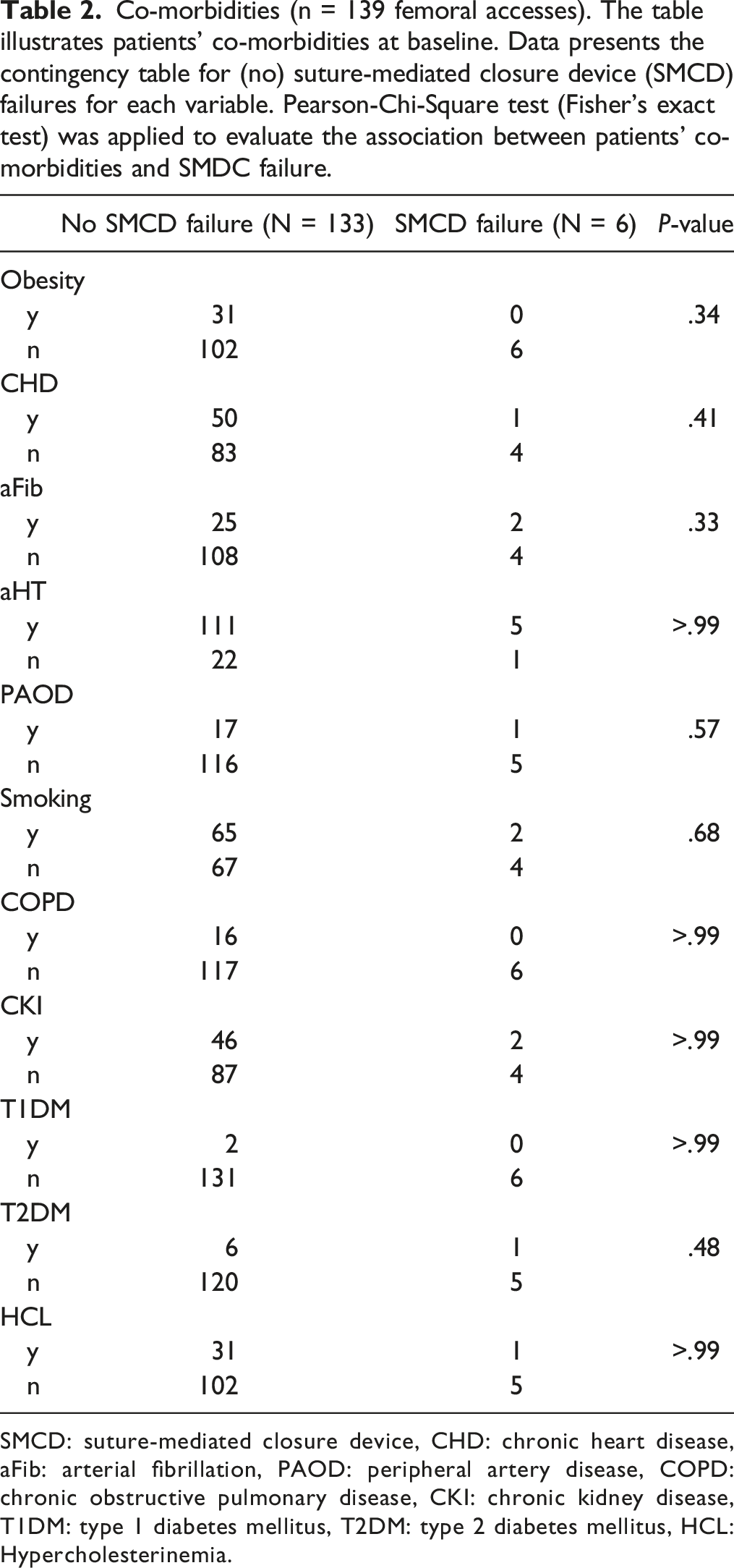
SMCD: suture-mediated closure device, CHD: chronic heart disease, aFib: arterial fibrillation, PAOD: peripheral artery disease, COPD: chronic obstructive pulmonary disease, CKI: chronic kidney disease, T1DM: type 1 diabetes mellitus, T2DM: type 2 diabetes mellitus, HCL: Hypercholesterinemia.
As expected, total procedure time was longer in the SMCD failure group (323 ± 117.8 min vs 171 ± 43.7 min) (P < .05). The dose area product (DAP) was 140 ± 55.2 Gycm2 in the SMCD failure group and 166 ± 139 Gycm2 in the non SMCD failure group (p = n.s.). Total fluoroscopy time was longer in the SMCD failure vs the no SMCD failure group (2355 ± 1016.4 min vs 1737 ± 990.8 min) (p = n.s.).
Morphological dimensions of access vessels were evaluated. The results suggest an association between the calcification of the CFA in general and/or anterior plaque formation in the CFA with SMCD failure (P < .05).
Morphological dimensions and anatomical determinants of access vessels (n = 139 femoral accesses). The table illustrates the morphological dimensions and key anatomical features for access vessels during elective and emergency EVAR procedures. Data is presented as mean ± SD with absolute frequency (N) or relative frequency (%) for cases with/without suture-mediated closure device (SMCD) failure. Pearson-Chi-Quadrat test (Fisher’s exact test) for dichotomous and Mann-Whitney-U Test for continuous variables was applied to analyze the association between SMCD failure and morphological dimensions/anatomical determinants.
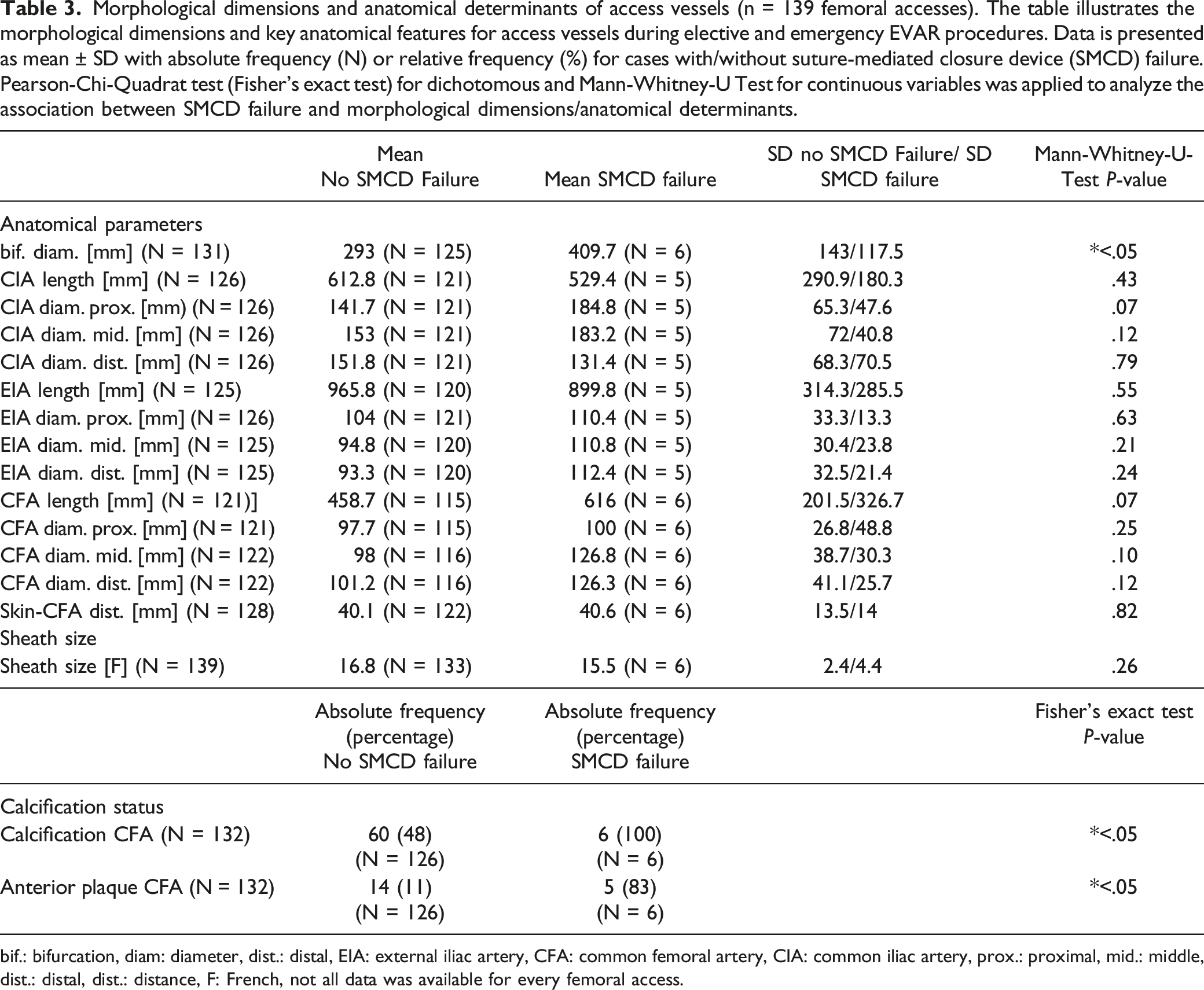
bif.: bifurcation, diam: diameter, dist.: distal, EIA: external iliac artery, CFA: common femoral artery, CIA: common iliac artery, prox.: proximal, mid.: middle, dist.: distal, dist.: distance, F: French, not all data was available for every femoral access.
Hemostasis-related blood parameters (n = 139 femoral accesses). The table illustrates hemostasis-related blood parameters. Data is presented as mean ± SD or relative frequency distribution for cases with/without suture-mediated closure device (SMCD) failure. Pearson-Chi-Quadrat Test (Fisher’s exact test) for dichotomous and Mann-Whitney-U Test for continuous variables was applied to analyze the association between SMCD failure and Hemostasis-related blood parameters.
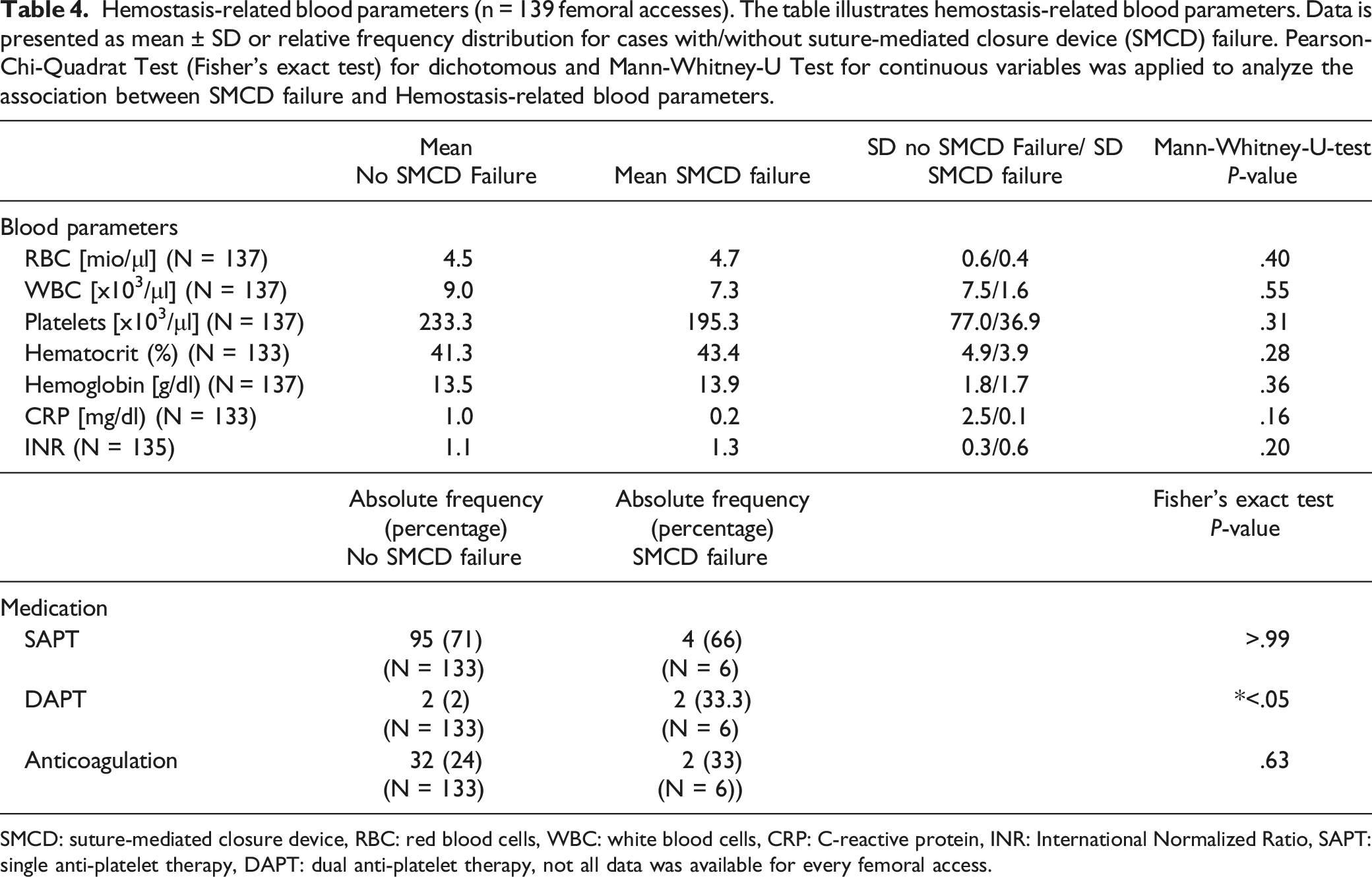
SMCD: suture-mediated closure device, RBC: red blood cells, WBC: white blood cells, CRP: C-reactive protein, INR: International Normalized Ratio, SAPT: single anti-platelet therapy, DAPT: dual anti-platelet therapy, not all data was available for every femoral access.
Finally, we applied multivariate binary regression analysis to evaluate the potential of nominated variables in predicting SMCD failure. Here, anterior plaque formation in the CFA and DAPT on the medication plan were found to be of relevance in predicting SMCD failure (OR 44.91 and 30.51, respectively). Of note, only anterior plaque formation maintained it statistical significance (P < .05) (Figure1). Forrest plot with odds ratio (OR) and 95 confidence intervals (CI) for suture-mediated device (SMCD) failure prediction (N=139 femoral accesses). Following univariate binary logistic regression analysis relevant parameters (p <.1) were further analyzed applying multivariate binary logistic regression analysis. Dual anti-platelet therapy (DAPT) on the medication plan prior to EVAR procedure (n.s.) and anterior plaque formation (p<.05) were identified as promising in predicting SMCD failure. Data is presented with OR and 95% CI. Bif: bifurcation, CFA: common femoral artery, diam.: diameter, mid.: middle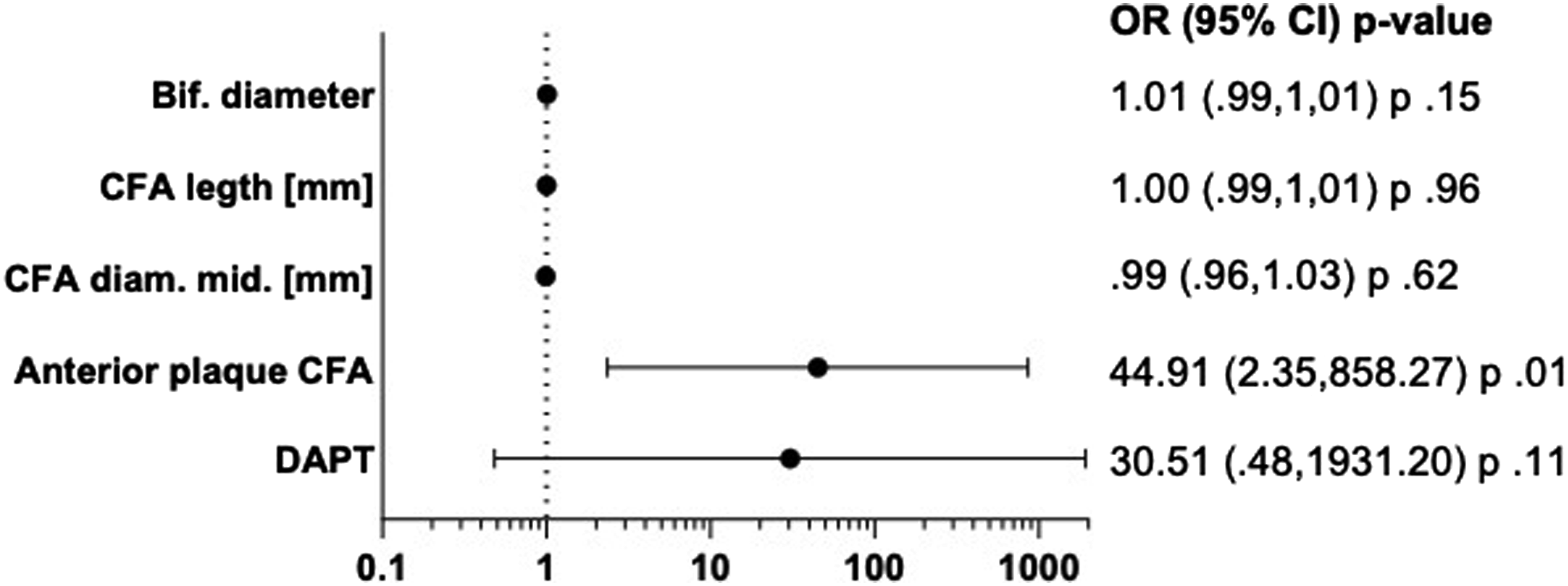
Discussion
This retrospectively single-center analysis aimed to evaluate the association of different potential contributors to SMCD failure and further analyzed their predictive value for SMCD failure in standard and emergency EVAR procedures. The study confirmed calcification and anterior plaque formation in the CFA to be associated with SMCD failure and further found discontinued T/DAPT prior to surgery potential contributor to SMCD failure. The calcification status and DAPT on the medication plan might also serve as predictors of SMCD failure prior to EVAR procedures.
The safety and efficacy of using SMCD in total percutaneous EVAR have been reported in several multicenter randomized studies and meta-analysis.14-18 To this end, the SMCD failure rate of the present study adds to this experience and reports a similar overall SMCD failure rate. That said, the use of SMCD is safe at low conversion rates.
Although the feasibility of a total percutaneous femoral access in standard and emergency EVAR procedures is not questioned nowadays, surgeons worldwide are aiming in minimizing SMCD failure rates. To do so, identifying determinants that contribute to SMCD failure is vital. Here, calcification of access vessels of >50% has been reported as a relevant predictor for SMCD failure. 19 Manunga et al also mentioned a safe use of SMCD during EVAR procedures in patients with <50% anterior wall calcification, irrespective of access sheath size or patient’s body mass index. In this study however, femoral artery calcification was still reported to be a major predictor for SMCD failure. 20 Also, the Italian Percutaneous EVAR (IPER) Registry validated in a multicenter trial that the only factor that predicts SMCD failure during percutaneous access in endovascular procedures is the presence of calcification in the CFA. 21 The experience of the present study confirmed these previous findings since the calcification status of the CFA and anterior plaque formation in the CFA were found to be associated with SMCD failure and further may serve as a relevant predictor for SMCD failure. When considering this accumulated evidence, a surgical incision with local thromboendarterectomy may be considered as beneficial in patients with an anterior plaque formation in the CFA to avoid SMCD failure. Nevertheless, such open surgical approach also comes with a cost since longer procedure time, prolonged in-hospital stay, and impaired wound healing has been described in previous studies.17,22
There is still a need to identify potential other relevant parameters considering the increase in endovascular aortic procedures in general. To this end, potential determinates have been identified recently. That said, clinical trials found that the successful use of SMCD was heavily dependent on the sheath size.23-26 Although somewhat obvious, the present study failed to find an association between SMCD failure and sheath size most likely due to the limited cohort size. In regard to this endpoint, the ratio between CFA diameter and sheath size may be more important than the sheath size alone. 24 Also, other parameters such as age and female gender, that have been introduced by various studies failed to be confirmed by the present study most likely due to the limited number of femoral access.27-30
This study introduces DAPT on the medication plan as a more novel parameter that may be associated with SMCD failure. Although discontinued for at least 5 days prior to the endovascular procedure, DAPT remained to be associated with SMCD failure during EVAR which requires sheath sizes being as big as 15-17 F. The significance of anti-platelet therapy as potential contributor to SMCD failure seems poorly elaborated in the current literature since recent meta-analysis also failed to control for anti-platelet therapy. 31 Notably, the significance of DAPT in predicting SMCD failure may heavily depend on the combination of substances, although we observed a relevant odds ratio (OR) for DAPT in predicting SMCD failure. Clopidogrel is a second-generation thienopyridine that differs in its substance specific effect profile from similar substances that target the same receptor. Prasugrel and ticagrelor are more potent P2Y12 inhibitors since both substances are associated with a 26-27% increase in major bleeding rate when compared to clopidogrel. 32 When considering these significant differences, it should be emphasized that all SMCD failure in this study occurred in femoral accesses with prasugrel on the medication plan prior to EVAR. That being said, our data may advocate for obligate platelet testing applying e.g. vasodilator-stimulated phosphoprotein phosphorylation (VASP) assay since recent meta-analysis described a significant impact of clopidogrel resistance in vascular and endovascular surgery. 33 Such approach may further minimize the risk for SMCD failure in patients with DAPT.
It should be mentioned that adjunct procedures may heavily impact the SMCD failure rate. One of these procedures may be the ultrasound-guided puncture of the CFA for arterial access. Such approach has been shown to reduce vascular complications and bleeding risk in percutaneous endovascular procedures and may of particular interest in antegrade procedures.34,35 In the present study, all femoral accesses were performed applying ultrasound-guided puncture, thus outcomes are controlled for this endpoint. Also, the use of the next generation of SMCD (ProStyle, Abbott Laboratories, Chicago, IL, USA) with an increased suture capture area may generate superior results in similar procedures and patient cohorts when compared to the SMCD used in the present study.
This study has major limitations. First and foremost, the study has a retrospective single-center design which compromise our findings. Also, the limited sample size may have heavily biased the results.
In summary this study confirms previous findings in similar patient cohorts that found the calcification status of CFA as main predictor for SMCD failure. Further, DAPT is reported to be associated with SMCD even when discontinued prior to the endovascular procedure. Such advocates for obligatory platelet testing prior to EVAR procedures whenever possible to increase patient safety.
Footnotes
Author Contributions
WG, AA, LM, JM and JS collected and analyzed the data. MD, HS and MUW interpretated the results and revised the manuscript’s intellectual content. WG, AA, and MUW drafted the manuscript. LM, JM, JDR, MD, JS, HS and MUW drafted the tables. All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
