Abstract
Background
endovenous laser ablation (EVLA) represents the gold standard in treating both great and small saphenous veins (GSV and SSV) incompetence. To achieve a “no-scalpel” procedure in patients with chronic venous insufficiency (CVI, CEAP C3-C6), concomitant phlebectomies could be replaced by ultrasound-guided foam sclerotherapy (UGFS) into varicose tributaries. The aim of this study is to present a single-centre experience on EVLA + UGFS for patients with CVI secondary to varicose veins and saphenous trunk incompetence, analysing ling-term outcomes.
Methods
all consecutive patients with CVI and treated by EVLA + UGFS from 2010 to 2022 were included in the analysis. EVLA was performed using a 1470-nm diode laser (LASEmaR® 1500, Eufoton, Trieste, Italy), adapting the linear endovenous energy density (LEED) depending on saphenous trunk diameter. Tessari method was used for UGFS. Patients were evaluated clinically and by duplex scanning at 1, 3 and 6 months, and annually up to 4 years, to assess treatment efficacy and adverse reactions.
Results
5500 procedures in 4895 patients (3818 women, 1077 men) with a mean age of 51.4 years were analysed during the study period. A total of 3950 GSVs and 1550 SSVs were treated with EVLA + UGFS (C3 59%, C4 23%, C5 17% and C6 1%). Neither deep vein thrombosis nor pulmonary embolism were detected during follow-up, as well as superficial burns. Ecchymoses (7%), transitory paraesthesia (2%), palpable vein induration/superficial vein thrombosis (15%) and transient dyschromia (1%) were registered. Saphenous and tributaries closure rate at 30 days, 1 and 4 years were 99.1%, 98.3% and 97.9%, respectively.
Conclusions
EVLA + UGFS for an extremely minimally invasive procedure appears to be a safe technique, with only minor effects and acceptable long-term outcomes, in patients with CVI. Further prospective randomized studies are needed to confirm the role of this combined therapy in such patients.
Keywords
Introduction
Recent guidelines indicate endovenous ablation with laser (EVLA) or radiofrequency as the first choice in treating saphenous trunks (great and small saphenous veins, GSV and SSV respectively) incompetence. 1
EVLA mini-invasiveness, along with similar to better outcomes than surgery, are some of the main advantages in choosing this treatment for patients with saphenous vein disease, form CEAP class C2 to C6.2,3 In particular, for patients experiencing more advanced disease (chronic venous insufficiency, CVI), including oedema (C3), skin changes (C4) and/or venous ulcers (C5-6), the treatments should be less invasive as possible. Indeed, skin incisions for saphenous and/or varicose veins (VVs) removal/stripping could heal later and complicate/aggravate the disease in such patients. In other words, surgery should be lessened, to minimized skin complications, maintaining acceptable outcomes.
To achieve a “no-scalpel” EVLA procedure, concomitant surgical phlebectomies could be replaced by ultrasound-guided foam sclerotherapy (UGFS) injection into varicose tributaries. This approach allows to minimize skin traumas, particularly when skin pathological changes are present.
Although some experiences have been published in the last years regarding the concomitant use of EVLA and UGFS in the saphenous trunk and in tributaries, respectively,4-6 no specific analysis has been done on patients with CVI, which remain in a low percentage in published reports (<15–20%).
The aim of this study is to present a large single-centre experience (5500 treatments) regarding the use of EVLA + UGFS for patients with CVI secondary to VVs and saphenous trunk incompetence, analysing short and long-term outcomes, in terms of postoperative complications and occlusion rate.
Materials and Methods
All consecutive patients and underwent EVLA + UGFS in one centre from January 1, 2010 to December 31, 2022 for CVI secondary to GSV/SSV incompetence and associated VVs were included in the analysis. Data were prospectively recorded and retrospectively analysed. All patients were preoperatively evaluated through color Duplex ultrasound scan (CDUS) analysis and clinical assessment by a vascular surgeon. The CDUS exam was done according to European Society of Vascular Surgery guidelines recommendation published during the study period. 2 In particular, average saphenous trunk diameter (resulting from three measurement along the thigh in case of GSV or along the leg in case of SSV) was evaluated to adapt the linear endovenous energy density (LEED) needs to EVLA maximum efficacy. 7 Average VVs diameters were also detected for adjusting UGFS percentage and amount.
Under local anaesthesia, EVLA was achieved according to literature recommendation published over the study period.1-3 Percutaneous, CDUS guided, saphenous trunk access was preferred in all cases. A 1470-nm diode laser (LASEmaR® 1500, Eufoton, Trieste, Italy), was used for EVLA, controlled by a vascular surgeon skilled in phlebological endovenous technique. LEED has been adjusted as follows: - 150 J/cm in the proximal 3 cm of the target vein; - 80 J/cm in the venous segment to be treated having <10 mm in diameter; - 90 J/cm in the venous segment to be treated having >10 mm and <13 mm in diameter; - 100 J/cm in the venous segment to be treated having >13 mm and <16 mm in diameter; - 110 J/cm in the venous segment to be treated having >16 mm and <20 mm in diameter.
After EVLA but in the same operating room (OR) session, UGFS of VVs was carried out, according to guidelines recommendations published over the study period1-3,6 using the Tessari method. Sclerosing agent (Polidocanol or Sodium Tetradecyl Sulphate [TDS]) has been used as follows: - Polidocanol .5% or TDS .25% for veins 5 mm in diameter; - Polidocanol .6% or TDS .3% for veins 6 mm in diameter; - Polidocanol .7% or TDS .35% for veins 7 mm in diameter; - Polidocanol .8% or TDS .40% for veins 8 mm in diameter; - Polidocanol 1% or TDS .5% for veins of 10 mm diameter; - etc…
After the procedure, an elastic compression stocking (monolayer I class, 18–21 mmHg) was dressed in the OR and maintained for 5 days, day/night, and for others 35 days only during the day. Low molecular weight heparin (LMWH) with a dosage of 4000 IU/day for 7 days was prescribed. All patients were discharge in the operative day.
Postoperative complications have been evaluated and noted as follows:
8
- Ecchymosis: subcutaneous spot of bleeding from blood extravasation appearing along the saphenous ablated tract or treated VVs, more than 10 cm in diameter; - Superficial vein thrombosis (SVT): pain, redness and sense of stiffness along the course of the ablated/sclerosed vein. Also, VVs hardening after UGFS without pain and redness were counted, despite these were not properly SVT; - Skin burns: superficial propagation of thermal energy derived from EVLA.
Regarding CDUS assessment, EVAL and/or UGFS failure has been defined as follows: - Saphenous trunk partial recanalization: a recanalized tract (compressible and colourable during CDUS) less than 10 cm in length; - Saphenous trunk recanalization: a recanalized tract (compressible and colourable during CDUS) more than 10 cm in length; - VVs recanalization: a recanalized tract (compressible and colourable during CDUS) less than 5 cm in length.
Patients were evaluated clinically and by duplex scanning at 1, 3 and 6 months, and annually up to 4 years, to assess treatment efficacy and adverse reactions.
Each patient signed a written consent document for clinical and scientific purposes, according to Helsinki declaration. Due to the retrospective nature of the study, local ethics committee approval was not required.
Results
Demographic patients ‘characteristics.
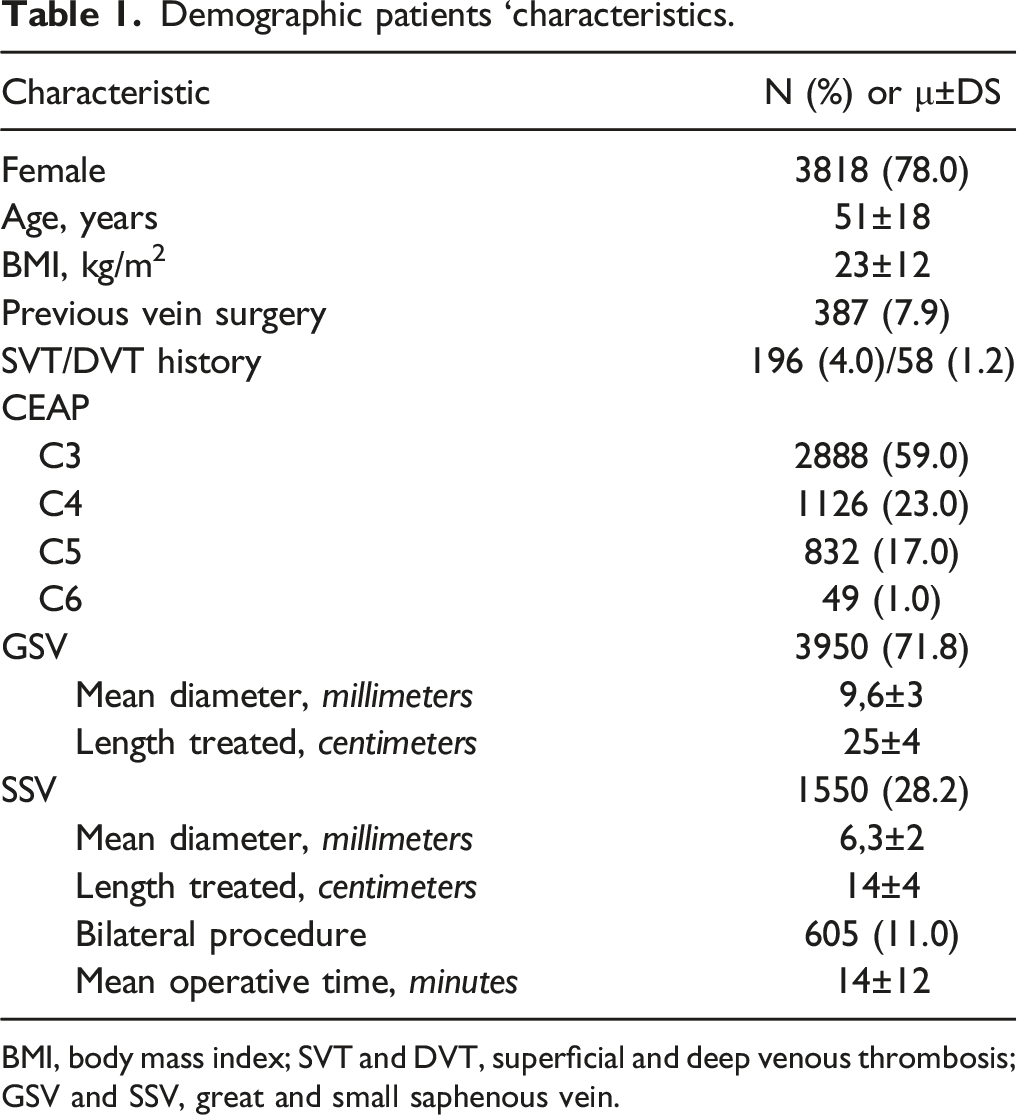
BMI, body mass index; SVT and DVT, superficial and deep venous thrombosis; GSV and SSV, great and small saphenous vein.
Neither deep vein thrombosis nor pulmonary embolism were detected during the perioperative period and the follow-up. No superficial burns due to EVLA were also described. Ecchymoses (7%), transitory paraesthesia (2%), palpable vein induration/SVT (15%) and transient dyschromia (1%) were listed. In particular, these minor complications were not yet detected 6 months after intervention. Paraesthesia occurred only after SSV treatment. Saphenous and tributaries closure rate at 30 days, 1 and 4 years were 99.1%, 98.3% and 97.9%, respectively. This included patients with complete closure or partial (<10 cm) recanalization. In particular, saphenous partial trunk recanalization was due to perforating veins (91%) or saphenous tributary (the so called “R3”, 9%). The reintervention rate was .6% in the first year and 1.8% from 1 to 4 years (cumulative rate at 4 years 2.4%), mostly due to anterior accessory saphenous veins incompetence with VVs (82%) and leg perforating veins incompetence with VVs (14%).
Discussion
Endovascular techniques in chronic venous disease (CVD) have been confirmed as the gold standard in the treatment of both GSV and SSV insufficiency. For VVs removal, robust and univocal data have not been provided in the literature, in terms of the choice of technique to adopt. 1 The 2022 Society for Vascular Surgery, American Venous Forum, and American Vein and Lymphatic Society clinical practice guidelines for the management of varicose veins of the lower extremities recommend ablation of the refluxing venous trunk and concomitant phlebectomy or UGFS of the varicosities with physician-compounded foam or commercial prepared polidocanol endovenous microfoam. 2 This suggestion achieved the highest level of recommendation, despite the fact that the guidelines did not address the best choice between surgery or UGFS. The European Society for Vascular Surgery (ESVS) 2022 Clinical Practice Guidelines on the Management of Chronic Venous Disease of the Lower Limbs reserved a section on tributaries treatment. 3 Despite it presented pro and cons of phlebectomy or UGFS, no recommendations – again – were proposed in selecting the appropriate technique.
While the removal of VVs (irrespective of their saphenous or non-saphenous origin) remains a topic of utmost importance in recent guidelines, no one discusses in detail the fact that UGFS further decreases the invasiveness of the operation, particularly in patients with advanced disease (CVI, C3-C6). Indeed, these patients could benefit from a no-scalpel approach, to avoid serious complications caused by the state of their skin (lipodermatosclerosis, inflammation, corona phlebectatica, active or healed ulcers). In this scenario, it should be possible to choose only percutaneous approach, for both saphenous trunk and tributaries treatments.
In our experience, the combined use of EVLA + UGFS for patients with CVI proved to be a safe, effective and durable choice at 4 years of follow-up. Only transitory minor complications occurred, particularly is SSV was treated. Postoperative occlusion rate is in line with other populations treated with EVLA for C2-C6 disease.9-11
Similar experience published by Hauzer and collaborators on 200 cases (C2-C6 classes) reported a 98% of efficacy 12 months after EVLA + UGFS, using Leonardo laser at 1470 nm, with a linear fiber and continuous wave at 80-100 J/10W, depending on the diameter of the vessel. 12 In this population, the sclerosing agent was injected through the lateral port of the introducer into the inefficient collateral veins extending from the main trunk of the GSV in the femoral segment, after the ablation of the proximal GSV segment.
Despite Yilmaz and coworkers described that concomitant UGFS has been technically successful in 504 EVLA patients, supplementary sclerotherapy sessions were necessary in more than one third of patients, due to residual VVs. 4 The Authors reported a 12-months ablation rate of 98.3%.
Despite UGFS has been widely used for treating tributaries VVs, also cyanoacrylates have been used for this purpose alone or in combination with thermal and non-thermal ablation techniques. 13 According to the Consensus document of the Australasian College of Phlebology, 14 VenaBlock™ and Veinoff™ (Invamed, Ankara, Turkey) can be directly injected for the treatment of tributary veins and perforators. Moreover, off label use of Venaseal™ (Medtronic Inc., Minnesota, USA) via direct injection has been reported. Although this consensus document proposed this statement, no data have been yet provided to validate it. Consequently, although the invasiveness of cyanoacrylates is similar to UGFS, we prefer the latter for the treatment of tributaries in C3-C6 patients.
This study has some limitations. First, no detailed analysis and comparisons was carried out on CEAP C6 patients, analyzing if this cohort was more prone to perioperative or long-term complication rate if compared to C3-C5 cohorts; second, no detailed description of the VVs was provided (thigh and/or tributaries, number treated) at baseline; lastly, no scales were used to assess quality of life modifications after the treatment. Despite this, we present a large cohort of patients with acceptable long-term results. Moreover, despite some weaknesses, the paper could be useful in in validating and supporting the use of this minimally invasive and curative technique in patients with CVI.
Conclusions
The utilization of EVLA + UGFS as an ultra-minimally invasive procedure seems to be a secure method, exhibiting only minor impacts and satisfactory long-term results, for patients experiencing CVI. Nevertheless, additional prospective randomized investigations are necessary to confirm and validate the efficacy of this combined therapy for such patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author CB has served as speaker for Eufoton; Eufoton didn’t provide any feedbacks or opinion about data, results and considerations expressed in this manuscript and didn’t take part to both proof writing and final revision. The authors certify that there is no other conflict of interest with any financial organization regarding the material discussed in the manuscript.
Funding
the author(s) received no financial support for the research, authorship, and/or publication of this article.
