Abstract
Objectives
Endovascular aortic repair may be complicated by type 2 endoleaks. Intervention is generally recommended when the native sac continues to grow more than 5 mm. Transcaval coil embolization (TCE) of the native aneurysm sac is an emerging technique for repair of type 2 endoleaks. The objective of this study is to report an institutional review of our experience with this technique.
Methods
11 patients underwent TCE during the study period. Data were gathered on demographics, size increase of native aneurysm sac, operative details, and outcomes. Technical success was defined as resolution of the endoleak during completion sac angiogram at end of the procedure. Clinical success was defined as no growth in the aneurysm sac at interval follow-up.
Results
Coils were the embolant of choice in all cases. Technical success was achieved in all cases except 1 resulting in a 91% technical success rate. Median follow-up was 25 months (range, 3-33). Of the ten patients that had technically successful embolization, 8 patients had repeat computed tomography (CT) scans which showed no further expansion of the native sac resulting in a 80% clinical success rate. No complications were noted immediately post-op or at interval follow-up.
Conclusions
This institutional retrospective review demonstrates that TCE is an effective and safe option for type 2 endoleaks after endovascular aortic repair (EVAR) in selected patients with favorable anatomy. Longer term follow-up, more patients, and comparison studies are needed to further define durability and efficacy.
Introduction
Endovascular aortic repair (EVAR) may be complicated by type 2 endoleaks, defined as persistent flow into the aneurysm sac from collateral vessels, such as the inferior mesenteric artery (IMA), lumbar arteries, or both. 1 This complication is common, occurring in 25% of EVARs and is often found at the time of surgery. Initially, type 2 endoleaks may be observed as approximately 50% will spontaneously resolve. However, if sac expansion is noted on interval follow-up, selective intervention may be indicated. 2 The Society for Vascular Surgery (SVS) guidelines recommend intervention with sac expansion >5 mm, while the European Society for Vascular Surgery (ESVS) guidelines have a higher threshold of >10 mm of sac expansion before recommending intervention.3,4
If a particular offending vessel is identified as the cause of a type 2 endoleak, such as the inferior mesenteric artery (IMA) or a lumbar artery, this can be targeted for embolization. However, this can be technically challenging if anatomy is not favorable, and studies have reported recurrent sac expansion rates ranging from 20 to 80%. 5 Alternatively, direct sac puncture via a translumbar approach has been found to have improved durability with clinical success rates of 70-90%. This improvement in clinical outcomes was thought to be due to the ability to deliver embolant directly into the native sac, causing thrombosis. However, this technique requires more difficult positioning, CT-guidance, and traversing multiple tissue planes, increasing the risk to para-aortic structures.5,6
Given these technical challenges, transcaval coil embolization (TCE) has been described as an alternative to the translumbar approach, while maintaining the same goal of native sac thrombosis. In this technique, the common femoral or internal jugular vein is cannulated, the aneurysm sac is accessed via the inferior vena cava, and embolant is delivered. The distal aortic sac must have direct apposition to the inferior vena cava with no visible tissue planes on imaging to safely access the aneurysm sac. Advantages of this technique include supine positioning, allowing access to both the venous and arterial systems, minimal anesthesia requirements, and reported clinical success rates of 75%.1,5–7 The objective of this study is to report a technical and institutional review of our experience with this technique in the management of type 2 endoleaks.
Methods
Study Cohort
All patients that underwent attempted TCE for type 2 endoleak during the study period (2018-2020) were included in the study cohort. The study was approved by the University of Louisville Institutional Review Board (approval number 22.0403) with a waiver of informed consent for the use of deidentified data. 11 patients were identified, and data were gathered on demographics, size increase of native aneurysm sac, operative technique details, and immediate and long-term outcomes. Each patient had imaging (Computed Tomography Angiography, CTA) that showed persistent increase in the native sac >5 mm after EVAR with confirmation of a type 2 endoleak of the aortic graft (Figure 1). Transcaval coil embolization was only considered technically feasible if the distal aortic aneurysm sac had direct apposition to the inferior vena cava and the aortic graft was far enough away from the planned puncture site to avoid an iatrogenic type 3 endoleak. Axial computed tomographic scan demonstrating type II endoleak with persistent contrast in the native aortic sac (red arrows). Note the close approximation of the inferior vena cava to the native aortic sac (yellow arrow) indicating favorable anatomy for transcaval embolization.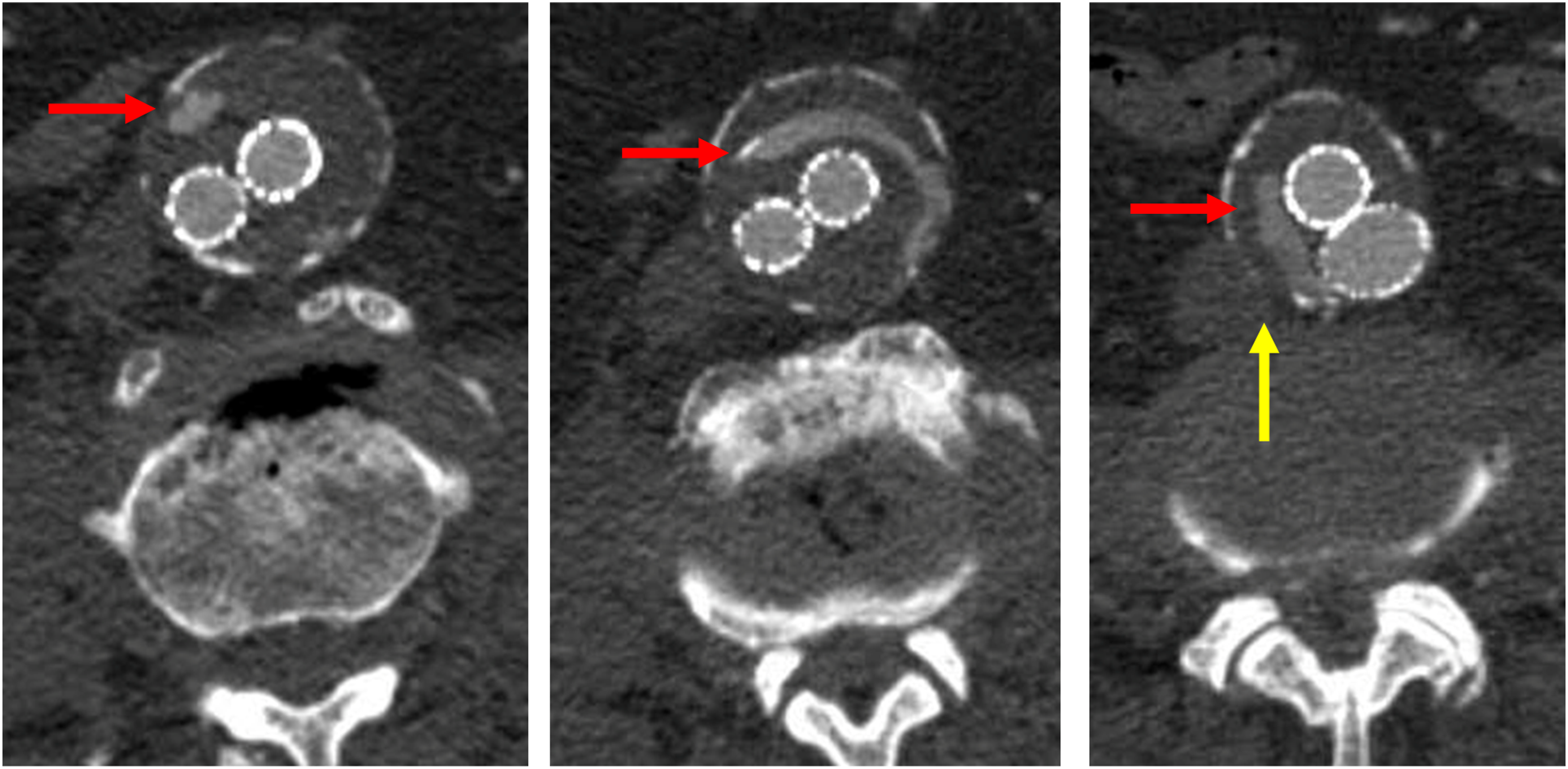
Technical Description
Patients were placed supine in the hybrid operating suite. Access to the right common femoral vein was obtained using a micropuncture needle and wire. A MicroSheath (Cook Medical, Bloomington, IN) was inserted and a Bentson wire (Cook Medical) advanced. A 10-French dilator was then introduced, followed by a 12-French dilator and sheath. A 10-French sheath with a metal cannula was then introduced under fluoroscopic guidance. A venogram was performed to confirm cannulation of the inferior vena cava and to identify bony landmarks for transcaval sac puncture (Figure 2A). A Rosch-Uchida Transjugular Liver Access Set (Cook Medical) needle was passed through the sheath and access was gained into the native aortic aneurysm sac using a combination of bony landmarks, venogram, and pre-operative imaging. The metal cannula was advanced and both fluoroscopy and arterial back bleeding from the catheter was used to confirm access to the aneurysmal sac. A Glidewire (Terumo Interventional Systems, Somerset, NJ) was then advanced through the sheath and a Glide catheter was advanced, replacing the metal cannula. Transcaval embolization technique. (A). Venogram is performed to confirm presence within the inferior vena cava (arrow denotes indentation of sac on IVC). (B). Transcaval placement of catheter into aneurysm sac with embolic coils delivered (arrow denotes transcaval catheter). (C). Completion of coil embolization of aneurysm sac after removal of catheter (arrow denotes embolized sac).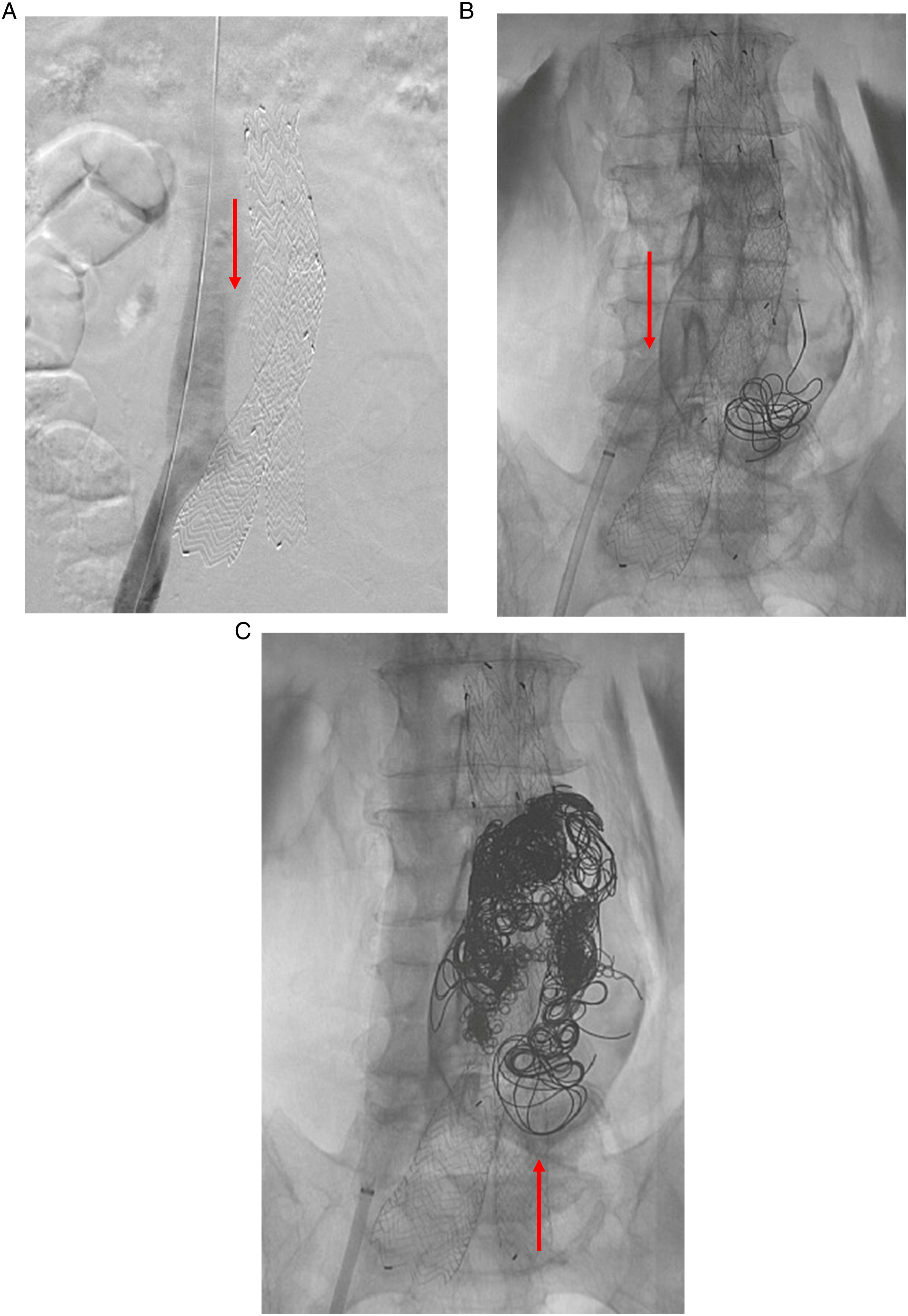
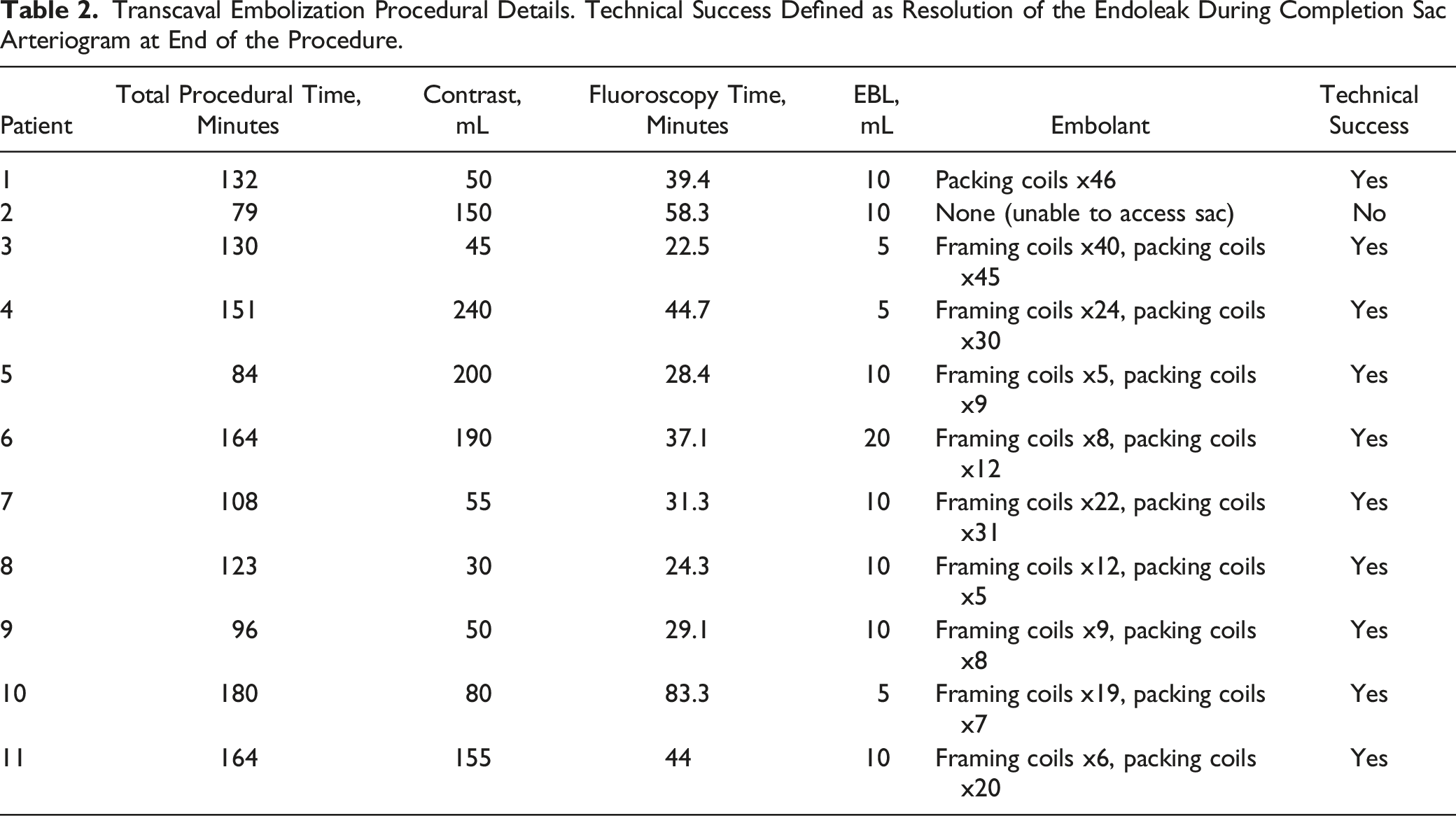
Sac angiography was then performed to identify the offending inflow vessel (lumbar or IMA) and patent areas in the native sac. Once this area was identified, a Lantern catheter (Penumbra Inc, Alameda, CA) was introduced and .020 detachable embolic coils (Penumbra Inc) were then delivered into the sac in an effort to thrombose the patent portions of the aneurysm sac (Figure 2B and C). A combination of framing and packing coils were used at the discretion of the operating surgeon. Sac arteriogram via the Lantern catheter was performed intermittently to check for cavity thrombosis. If other areas of sac patency or endoleak were discovered, the Glide and Lantern catheters were redirected to those areas and coils were delivered. Once sac thrombosis was confirmed, catheters were withdrawn back into the inferior vena cava and venogram was performed to check for aortocaval fistula or contrast extravasation. Wires and catheters were then removed from the right common femoral vein and pressure was applied to achieve hemostasis. Technical success was defined as resolution of the endoleak during completion sac arteriogram at end of the procedure.
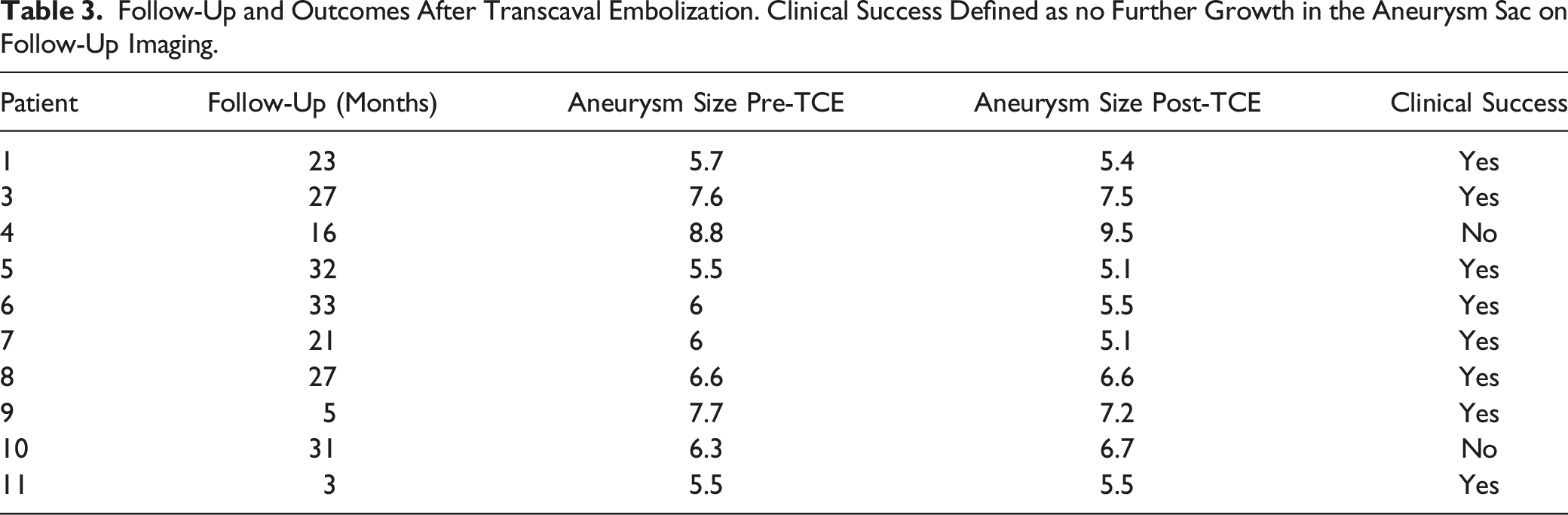
Patients were admitted to the intensive care unit overnight with a urinary catheter and arterial line for close hemodynamic monitoring. A non-contrasted CT was obtained on post-operative day 1 to check for development of a retroperitoneal hematoma. Patients were seen in the outpatient setting at 2 weeks for routine post-operative follow-up. Follow-up CT imaging was scheduled at the discretion of the attending surgeon but was generally performed at 6 months for initial surveillance (Figure 3). Clinical success was defined as no further growth in the aneurysm sac on follow-up imaging. Axial computed tomography imaging after transcaval embolization.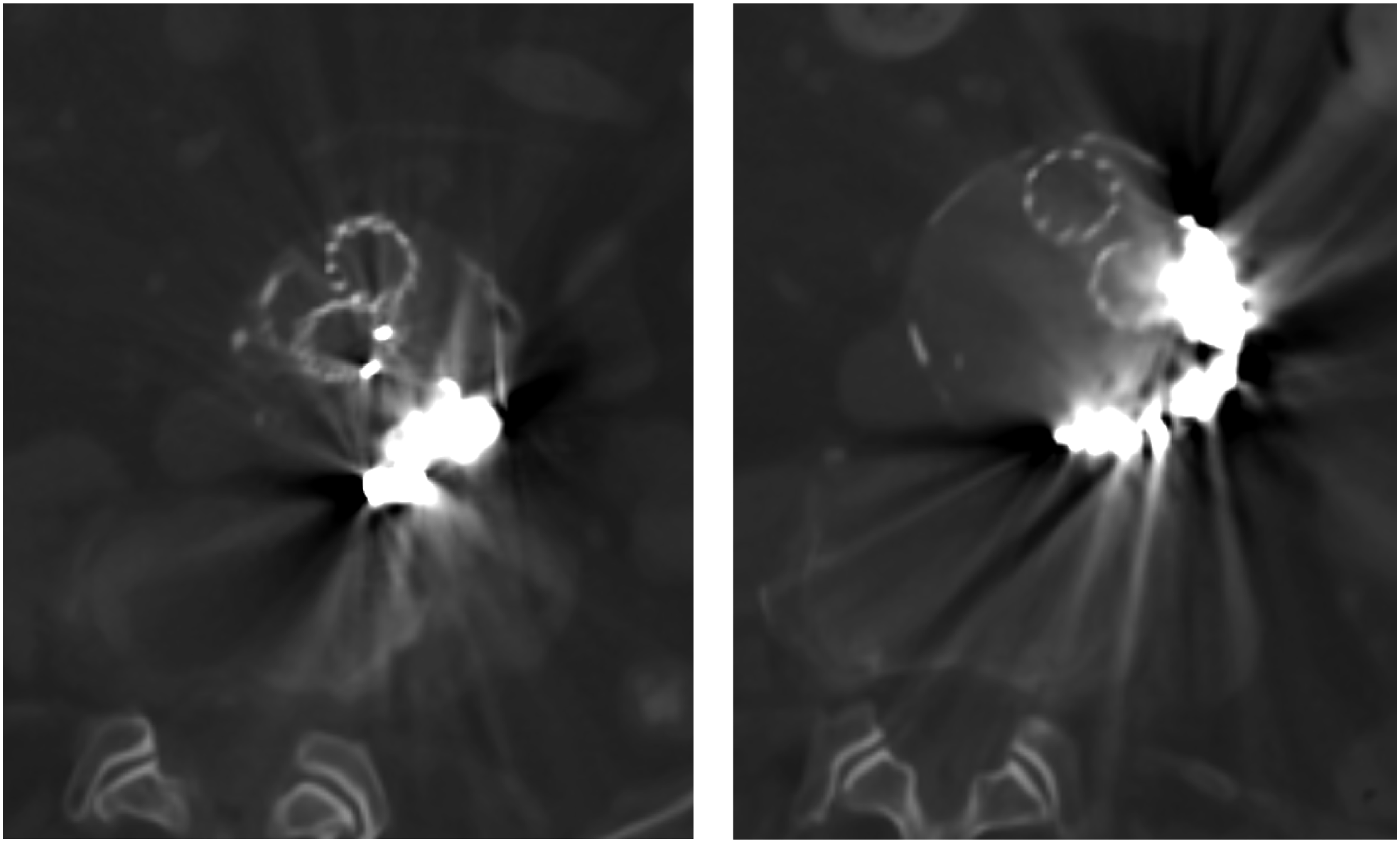
Results
Demographics
Patient Demographics, Time Since EVAR, Prior Embolization Attempts, and Aneurysm Growth.
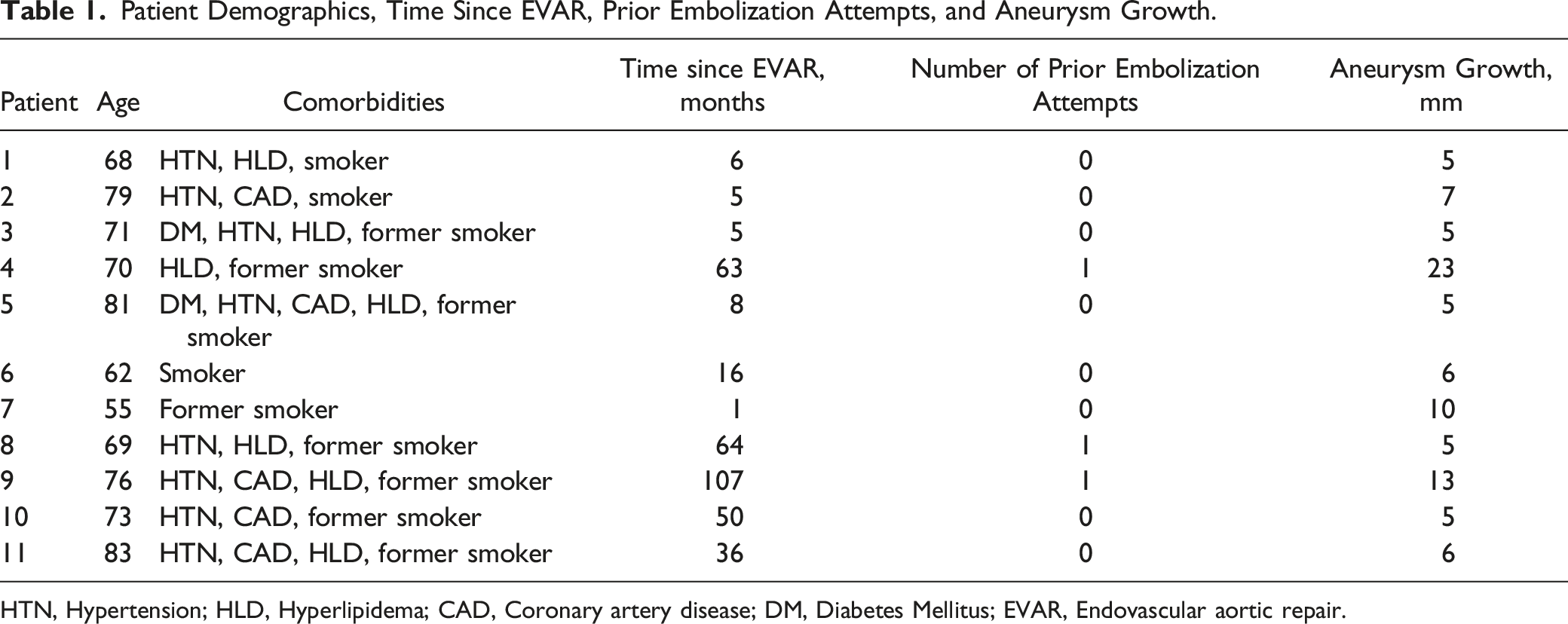
HTN, Hypertension; HLD, Hyperlipidema; CAD, Coronary artery disease; DM, Diabetes Mellitus; EVAR, Endovascular aortic repair.
Procedural Data
Transcaval Embolization Procedural Details. Technical Success Defined as Resolution of the Endoleak During Completion Sac Arteriogram at End of the Procedure.
Outcomes
Follow-Up and Outcomes After Transcaval Embolization. Clinical Success Defined as no Further Growth in the Aneurysm Sac on Follow-Up Imaging.
Discussion
Procedural options for the treatment of persistent type 2 endoleaks with native sac expansion reported in the literature consist of endovascular embolization (via transarterial, translumbar, and transcaval approaches), robotic or laparoscopic ligation of feeding arterial branches, or conversion to an open repair.2-4 At our institution, while we do perform transarterial embolization for type 2 endoleaks, we’ve found that TCE is often a technically easier first-line approach. In this study, we examined our technical and clinic outcomes of patients that underwent TCE over a 2-year period from when we first started employing this technique in 2018 until 2020. We report a good technical success rate (91%) with no immediate complications. The 1 technical failure was due to an inability to access the aneurysm sac through the inferior vena cava. This patient underwent successful transarterial embolization via a brachial artery approach the following day. We report an 80% clinical success rate, with no patients lost to follow-up. One patient had an increase in the size of the aneurysm sac from 6.3 cm to 6.7 cm on follow-up CTA at initial follow-up at 14 months. Given that the size increase was <5 mm, the patient was observed and brought back for clinic follow-up at 31 months from his initial embolization, which showed a stable native sac maximum diameter of 6.7 cm. The second patient had a stable aneurysm sac size at 8.8 cm at 11-month follow-up; however, at second follow-up at 16 months, the native sac increased to 9.5 cm. The patient was brought back for a second attempt at TCE and had stabilization of the native sac size at 9.4 cm at follow-up.
While this study does not directly compare this technique to others, review of the literature has shown transarterial embolization technical and clinical success rates of 82% and 60%, respectively, and translumbar technical and clinical success rates of 100% and 79%, respectively. Transcaval coil embolization technical and clinical success rates ranged from 90-100% and 66-95%, respectively.1,5-9 Thus, our data are similar to other studies and continue to support TCE as a safe and effective method for treatment of persistent type 2 endoleaks in selected patients with favorable anatomy.
While there is no consensus yet on the preferential method for treating type 2 endoleaks with persistent expansion, the clinical success rates seem to be more in favor of a transcaval or translumbar approach compared to a transarterial approach. Prior studies have found that long term recurrence of type 2 endoleaks after transarterial embolization was due to either multiple communicating vessels with the native sac, persistent flow through the coils of the embolized artery, and/or recruitment of collateral arteries to fill the native sac.10,11 This would suggest that type 2 endoleaks behave as arterial malformations and both TCE and translumbar embolization have improved clinical success rates due to embolization of the central nidus of the malformation, or the native sac in the case of type 2 endoleaks.5,7,11 Therefore, in our technique, our primary goal was to identify patent areas of the native sac to target for embolization. If we were able to identify inflow artery origins (lumbar or IMA) into the native sac, we did try to target these for embolization to treat the inflow of the endoleak. However, given the pathophysiology that type 2 endoleaks behave as arterial malformations, we felt that even if we embolized a single inflow artery, we still needed to embolize the remaining patent areas of the native sac to reduce the risk of recurrence. Our data support this method as our recurrence and clinical success rates are in line with similar studies from other institutions.1,5,7
Despite this reported efficacy, widespread adoption of TCE in the treatment of type 2 endoleaks remains limited given the anatomical requirements needed for technical feasibility. The vena cava and aortic sac must have direct apposition with no tissue planes visible between on pre-operative imaging; lack of this apposition would risk retroperitoneal hemorrhage. If the vena cava and the sac are appropriately apposed, the next anatomical challenge is the position of the EVAR graft itself in the sac; if the limbs or the main body are too close to the puncture site, there is risk of damage to the graft, resulting in a possible iatrogenic type 3 endoleak.1,5,6,8,9 Given these constraints, a patient-specific approach is often required for the treatment of type 2 endoleaks with persistent native sac expansion with TCE being complimentary in nature to other techniques to ultimately reduce the risk of sac rupture. At our institution, transcaval embolization has gained popularity given its high technical and clinical success rates and, thus, is attempted first if anatomy is favorable. If not, a transarterial approach is attempted if the offending IMA or lumbar artery can be easily identified and accessed.
In this study, detachable coils were the embolant of choice for all patients. Other studies have used other embolants, such as N-butyl cyanoacrylate, Onyx, thrombin, or combinations of such embolants, all with similar technical and clinical success rates.1,5,7,8,12,13 Our institution has preferred the use of detachable coils to allow for precise delivery and the ability to easily retrieve the coils if they do not end up in the desired location. However, exclusive use of coils is expensive and can be time consuming given the volume needed for aortic sac embolization. While we report a similar overall clinical success rate compared to other similar studies, we acknowledge that use of a different embolant or combining other embolants with coils may improve both overall cost and procedural time.
Limitations of this study include that it is a single-institution retrospective review with a small patient population, possibly leading to selection bias and type II error. Additionally, the clinical success of 80% was based on CT follow-up imaging that showed a stable native aneurysm sac size when compared to the pre-procedural CTA imaging. Given that coils were the embolant of choice in all cases, this did create significant artifact on interval imaging, sometimes making it difficult to determine if the endoleak had resolved. While we do feel confident in the repeat imaging measurements of the native sac, other studies have employed abdominal duplex US and dynamic MRI to help minimize artifact and may be a better option for follow-up. 8 Finally, while this study found no complications with this technique, other reported complications include inadvertent intracaval embolization of coils, retroperitoneal hemorrhage, unintentional puncture of the aortic graft leading to type 3 endoleak, injury to periaortic and pericaval structures, and aortocaval fistulas. 5 As use of this technique becomes more widespread, reporting and studying of these potential immediate and long-term complications remains important.
Conclusion
While there remains to be a gold standard for the management of type 2 endoleaks with persistent sac expansion after EVAR, this study supports a transcaval approach to embolization of the native aneurysm sac in selected patients with favorable anatomy. While this is a small, single institution retrospective review, we still report a good technical and clinical success rate with no complications, adding to the current available data that this technique is both safe and effective.
Footnotes
Author’s Note
Presented at the Society for Vascular Surgery Vascular Annual Meeting 2021 in the Interactive Poster Session.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
