Abstract
Introduction
Stentgraft limb occlusion (SLO) is a potential complication of endovascular aneurysm repair (EVAR). The purposes of this single centre study are to report the incidence of SLO after EVAR and to detect possible risk factors.
Methods
All patients who underwent EVAR between June 2001 and February 2020 were included in this retrospective study. Demographic data, cardiovascular risk factors, aneurysm characteristics, arterial anatomy, repair strategy, systemic and stentgraft-related complications, and in-hospital and late mortality were collected. Routine follow-up included duplex examination and/or CT angiography at 3 months, 12 months and annually thereafter. Logistic regression analysis was performed to detect predictors for SLO.
Results
A total of 221 patients (425 stentgraft limbs) were included; of whom 11 patients (5.0%) occluded. Median time to occlusion was 3.3 months and most of the patients presented ischemic signs. Two risk factors for SLO could be identified: symptomatic aneurysm (P .015, odds ratio 4.62, 95% confidence interval 1.35-15.86) and length of the infrarenal abdominal aortic aneurysm (AAA) (P .021, odds ratio 1.31, 95% confidence interval 1.04 - 1.64).
Conclusion
The incidence of SLO after EVAR is low, and most occlusions occur within the first year. Predictors for SLO are the symptomatic aneurysm and the length of the infrarenal AAA. Further research is necessary to pool all predictors and to assess the clinical impact of different follow-up strategies for high-vs low-risk patients.
Keywords
Introduction
Endovascular aneurysm repair (EVAR) is widely accepted as treatment of infrarenal abdominal aortic and iliac artery aneurysms. 1
However, despite an early mortality benefit over open surgery, EVAR is associated with a significantly higher re-intervention rate; hence the need for close and prolonged follow-up. 2
Complications related to EVAR can be categorized as systemic or device-related. Systemic complications include acute myocardial infarction, cerebrovascular events and postimplantation syndrome. 1 Device-related complications include endoleaks, infection, migration or kinking of the stentgraft. 3
Stentgraft limb occlusion (SLO) is another complication, characterized by thrombotic obstruction of the blood flow in 1 or both stentgraft limbs which can lead to clinical signs of lower limb ischemia. 3
The purposes of this single centre study are to report the incidence of SLO after EVAR and to detect possible risk factors.
Methods
Procedure
This study is a single centre retrospective study, approved by the ethical committee of the Universitair Ziekenhuis Brussel, Belgium.
All patients who underwent elective EVAR between June 2001 and February 2020 at the Department of Vascular Surgery were reviewed. Demographic data, cardiovascular risk factors, aneurysm characteristics, arterial anatomy, repair strategy, systemic and stentgraft-related complications, and in-hospital and late mortality were collected. Routine follow-up included duplex examination and/or CT angiography at 3 months, 12 months and annually thereafter.
We determined the incidence of SLO and described the location, time to occlusion, clinical presentation, diagnosis and treatment.
Further, we compared patient groups with and without SLO to detect predictors for this adverse outcome.
Patient characteristics included gender, age at time of EVAR, smoking (never, old or current), diabetes type 1 or 2 under therapy, dyslipidemia under therapy, arterial hypertension under therapy, ischemic heart disease, chronic kidney disease (eGFR <60 mL/min/1.73 m2 for ≥3 months), chronic obstructive lung disease, body mass index, and pre-operative kidney function (creatinine in mg/dL).
Type (infrarenal abdominal aortic aneurysm (AAA) with or without iliac artery aneurysm (IAA)), diameter, length, large diameter (diameter of infrarenal AAA ≥5.5 cm in men and ≥5.0 cm in women, or diameter of IAA ≥3.0 cm), fast growth (≥1.0 cm/year), morphology (fusiform or saccular) and clinical presentation of the aneurysm were noted. Presenting symptoms were abdominal symptoms, lower back pain, painful palpation localized to the AAA or radiating to the back or to the genitals, and peripheral embolization with symptoms of (sub)acute leg ischemia. Measurements were done by means of pre-operative CT angiography.
Arterial anatomy included the diameter, length, angulation and presence of circumferential calcification >50% of the infrarenal aortic neck and iliac arteries, the distal aortic diameter and the angle of aortic bifurcation. In addition, the presence of a hostile neck was noted. The infrarenal aortic neck was considered hostile if diameter >2.8 cm, length <1.0 cm, angulation ≥60° or circumferential calcification >50%. 4 Measurements were done by means of pre-operative CT angiography.
Type and size of stentgraft, repair strategy (access, extensions, coiling of internal iliac artery, iliac branch device, endograft anchors), additional procedures and incidence of intra- or postoperative endoleaks were carefully noted.
All systemic and stentgraft-related complications were reviewed. Stentgraft migration was defined as >1.0 cm displacement between first postoperative and subsequent CT angiography. 5 Stentgraft kinking was defined as doubling of peak systolic velocity on duplex examination, 6 or as sharply localized angulation (>90°) of the limb. 7
Statistics
Data are presented as median with range, mean ± standard deviation, or n (%).
Statistical analysis was carried out using SPSS (version 28) for Windows.
With the help of logistic regression analysis, significant predictors for SLO were selected. This was done using a forward stepwise procedure by means of likelihood ratio testing. Only variables with significance level of α < .05 and without extreme standard error were retained in the final model.
Results
Between June 2001 and February 2020, 257 patients were eligible for endovascular repair of infrarenal AAA and/or IAA. Eleven patients with an isolated IAA and 12 patients treated with an aortic tube stentgraft were excluded. We also excluded 2 patients lost in follow-up, 10 patients with missing values in data collection and 1 patient with conversion to open surgery. This resulted in 221 patients who were eligible for analysis. All stentgrafts were placed within the instructions for use; 204 bifurcated and 17 aorto-uni-iliac leading to a total of 425 stentgraft limbs.
Patients with Stentgraft Limb Occlusion
Eleven limbs of the 221 patients (5.0%) occluded. Median time to occlusion was 3.3 months (day 1 - 89 months) and occlusion was unilateral in all patients. All patients manifested ischemic signs (subacute leg ischemia, intermittent claudication or rest pain), except 1 patient who remained asymptomatic (accidental find). Some patients identified a trigger that presumably elicited ischemia: prolonged knee flexion while working in the garden, or intensive cycling session that evoked gluteal pain.
Three patients died after several months to years, for reasons unrelated to the aneurysm repair.
Stentgraft Limb Occlusion Characteristics.

Risk Factors for Stentgraft Limb Occlusion
Patient Characteristics.
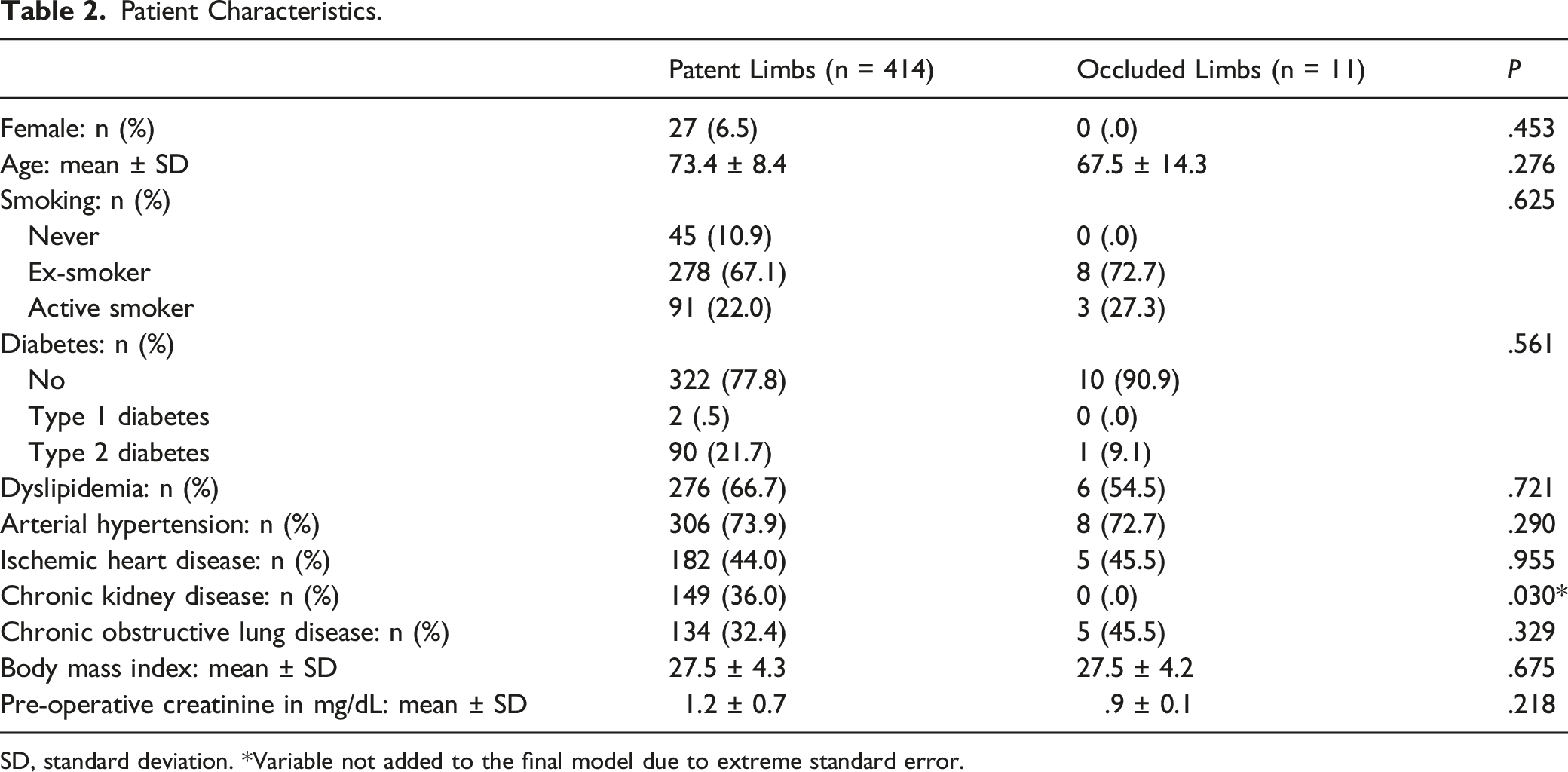
SD, standard deviation. *Variable not added to the final model due to extreme standard error.
Aneurysm Characteristics.
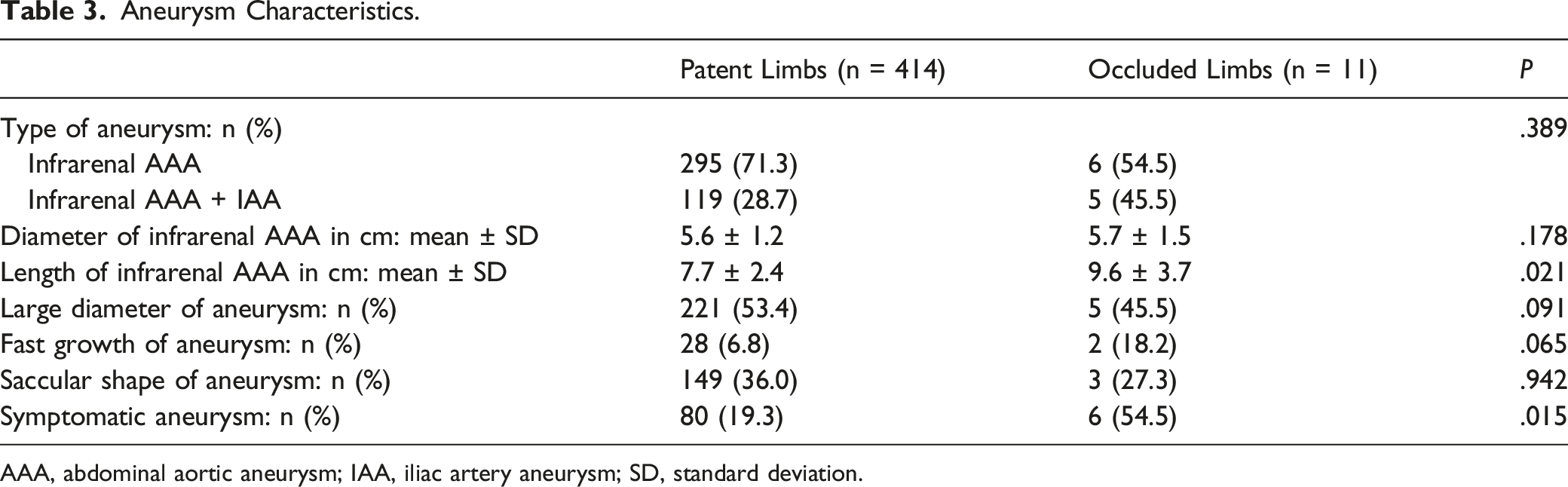
AAA, abdominal aortic aneurysm; IAA, iliac artery aneurysm; SD, standard deviation.
Arterial Anatomy.

SD, standard deviation; CIA, common iliac artery; EIA, external iliac artery.
Repair Strategy.
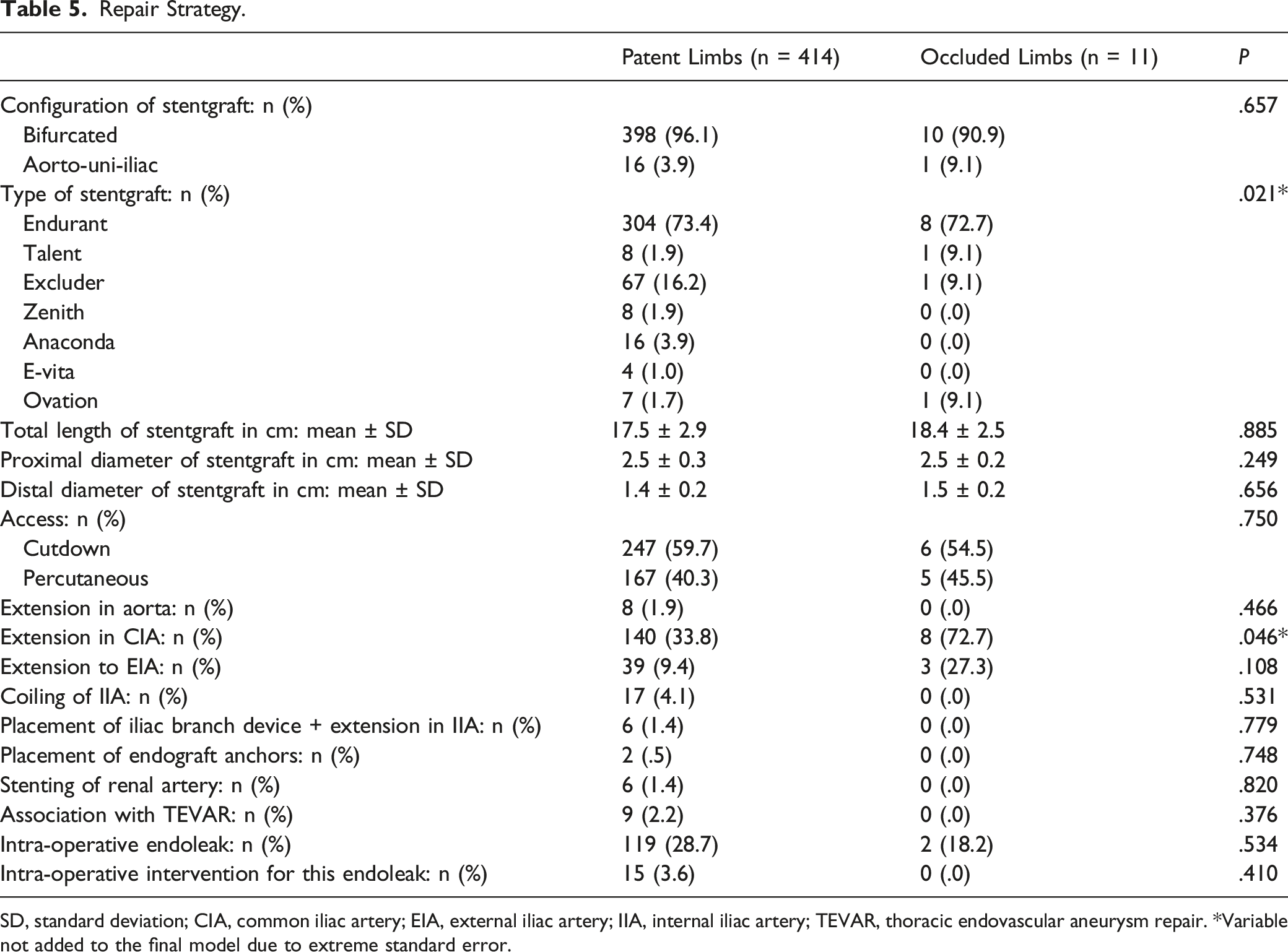
SD, standard deviation; CIA, common iliac artery; EIA, external iliac artery; IIA, internal iliac artery; TEVAR, thoracic endovascular aneurysm repair. *Variable not added to the final model due to extreme standard error.
Systemic and Stentgraft-Related Complications.
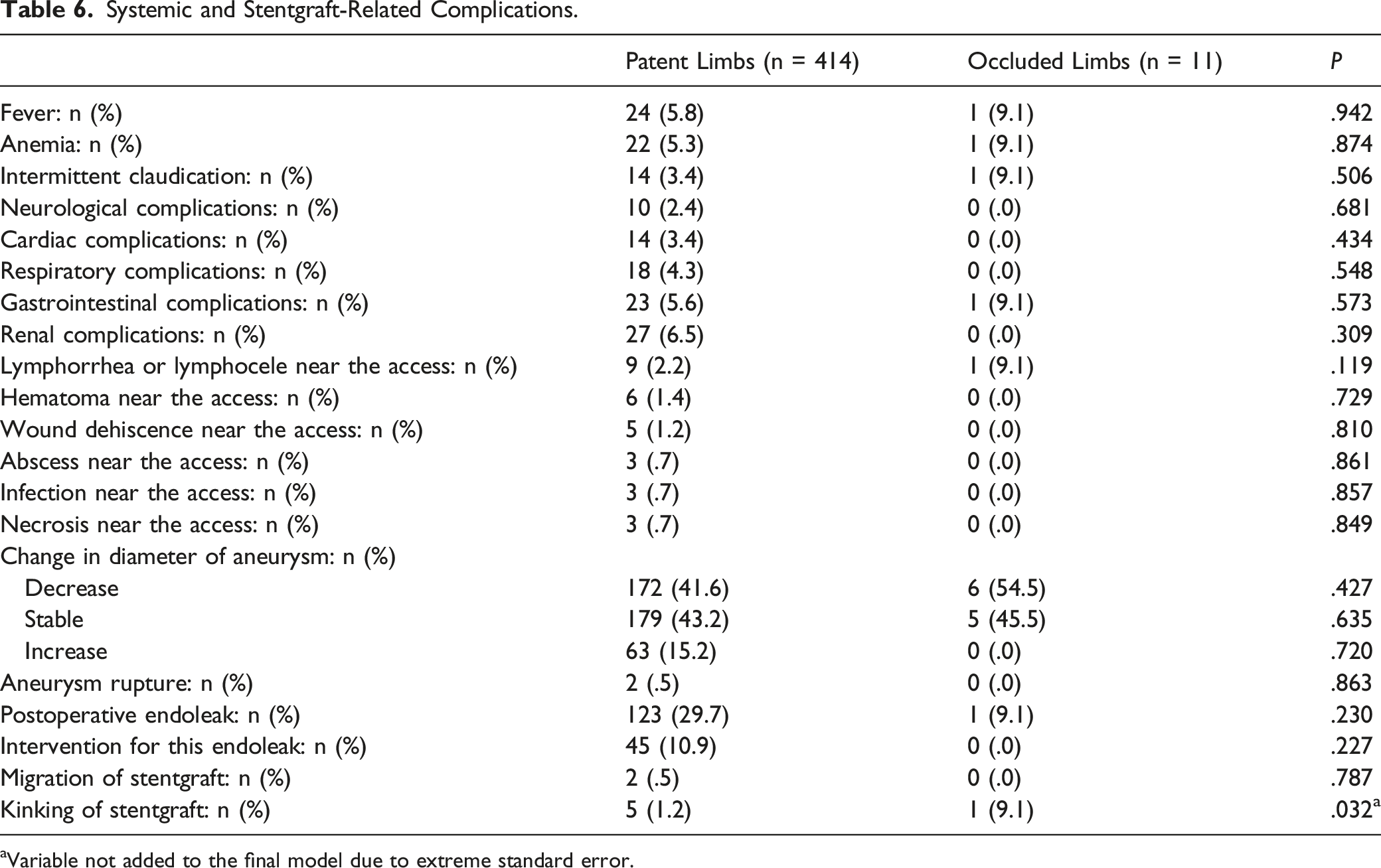
aVariable not added to the final model due to extreme standard error.
Final Model using Forward Stepwise Logistic Regression.
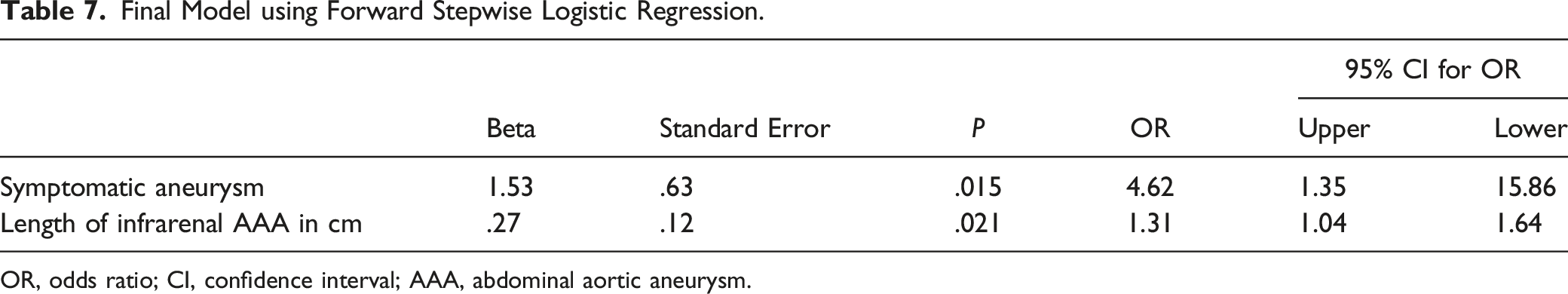
OR, odds ratio; CI, confidence interval; AAA, abdominal aortic aneurysm.
Four patients died in-hospital; all unrelated to the aneurysm. There were no late stentgraft-related deaths.
Using forward stepwise logistic regression, we detected 2 predictors for SLO: symptomatic aneurysm and length of the infrarenal AAA (Table 7). The risk of SLO is about 4.62 times as high if the aneurysm is symptomatic compared to when it is asymptomatic (P .015, odds ratio (OR) 4.62, 95% confidence interval (CI) 1.35-15.86). In addition, for every 1 cm increase in length of the infrarenal AAA, the odds of having an occlusion increases by 31% (P .021, OR 1.31, 95% CI 1.04-1.64).
Discussion
Patients with Stentgraft Limb Occlusion
The meta-analysis of Coelho et al, 6 which included 55 studies (27 509 patients), reported an incidence of SLO after EVAR ranging from .0 to 10.6%. Our study presented an incidence of 5.0%. Occlusion was unilateral in all patients; in literature this ranged from 91 to 100%.7-9 Median time to occlusion was 3.3 months (day 1 - 89 months) and 63.6% occurred within the first 6 months after EVAR. Cochennec et al 7 reported a median time to occlusion of 1.4 months (day 0 - 71 months) and 69.7% occurred within the first 6 months.
Clinical presentation of SLO was consistent with the meta-analysis of Coelho et al: 6 (sub)acute leg ischemia, intermittent claudication and rest pain. Our study also had 1 asymptomatic patient who had no femoral pulsation but a viable leg 1 day after EVAR. In literature, occlusions were sometimes found in asymptomatic patients during routine follow-up imaging.6,7,10
Three patients worked on their knees or cycled frequently before the moment of occlusion. This suggests that incorrect movements of the hip joint may influence the development of occlusion. These specific circumstances were not previously discussed in literature. Furthermore, our study showed that patients with occlusion (67.5 ± 14.3 years) were younger than those without occlusion (73.4 ± 8.4 years), however, not statistically significant (p .276). As such, younger patients who are more physically active may be prone to SLO.
In the meta-analysis of Coelho et al, 6 surgical treatment options consisted of thrombectomy and extra-anatomical bypasses. Endovascular treatment options included catheter-directed thrombolysis, mechanical thrombectomy, percutaneous balloon angioplasty, and additional bare metal stent (BMS) or stentgraft deployment. 6 In our study, treatment modalities consisted of surgical thrombectomy, femoro-femoral crossover bypass, and additional BMS or covered stentgraft deployment. Moreover, 2.4% of the occlusions in the meta-analysis of Coelho et al 6 were treated conservatively.
Risk Factors for Stentgraft Limb Occlusion
Our study identified symptomatic aneurysm as significant risk factor for SLO (P .015, OR 4.62, 95% CI 1.35-15.86). One hypothesis is that in symptomatic aneurysms, intervention takes place slightly faster, using stentgrafts available on the shelf. In addition, the morphology of symptomatic aneurysms may be unstable, which might predispose to stentgraft movement.
The second risk factor for SLO identified in our study was length of the infrarenal AAA (P .021, OR 1.31, 95% CI 1.04-1.64). One hypothesis is that this gives the stentgraft more room to move.
These 2 predictors have never been described in literature before. Faure et al 8 found significant association between SLO and diameter of the infrarenal AAA <5.9 cm (P .001). In our study, there was no difference between patent (5.6 ± 1.2 cm) and occluded limbs (5.7 ± 1.5 cm) (p .178).
Our final model failed to show any significant association between SLO and different patient characteristics. Cochennec et al 7 identified younger age as predictor (p .007). In our study, patients with occluded limbs were also younger, but the difference was not statistically significant (73.4 ± 8.4 years in patent limbs vs 67.5 ± 14.3 years in occluded limbs, P .276). Taudorf et al 11 found significant association between SLO and higher body mass index (p .007). In our study, there was no difference between patent (27.5 ± 4.3) and occluded limbs (27.5 ± 4.2) (P .675).
Similar to Cochennec et al, 7 our study could not identify any characteristic of the arterial anatomy as predictor for SLO. Coelho et al 6 identified narrow distal aorta as risk factor. This is in contrast to our study, in which distal aortic diameter was larger in occluded limbs (3.3 ± 1.1 cm) than in patent limbs (2.7 ± .8 cm), however, not statistically significant (p .312). Several studies demonstrated iliac artery tortuosity and calcification as risk factors for SLO;6,11,12 in our study, there were no major differences. Faure et al 8 noted that if the diameter of the external iliac artery (EIA) was ≤1.0 cm, there was a higher risk of occlusion (P .001). In our study, diameter of the EIA was .8 ± .1 cm in patent limbs and .7 ± .1 cm in occluded limbs (P .110).
Neither type and size of stentgraft, nor repair strategy, nor additional procedures were found to be related to SLO in our study. It seemed that the Talent devices showed a higher risk to occlude (1.9% in patent limbs versus 9.1% in occluded limbs), but too few patients received such a device to draw any significant conclusions. Talent devices are first-generation devices. This was also the case for the Ovation devices (1.7% in patent limbs versus 9.1% in occluded limbs). However, we have no explanation for these devices. Carroccio et al 13 noted that if the distal diameter of the stentgraft was ≤1.4 cm, there was increased risk of occlusion (P .03). In our study, there was no major difference between patent (1.4 ± .2 cm) and occluded limbs (1.5 ± .2 cm) (P .656). Extension to EIA was a predictor for occlusion reported in many studies.6,8,9,13 In our study, there were also more extensions to the EIA in occluded limbs (27.3%) than in patent limbs (9.4%), however, not statistically significant (P .108).
No systemic or stentgraft-related complications were associated with SLO in our study. Faure et al 8 showed that postoperative intervention for endoleak was associated with higher risk of SLO. This was not the case in our study (10.9% in patent limbs vs .0% in occluded limbs, P .227). Coelho et al 6 noted that stentgraft migration was a risk factor for stentgraft kinking and occlusion. Stentgraft kinking was a risk factor for stentgraft thrombosis and occlusion reported in many studies.6-8 However, our study could not demonstrate association between SLO and stentgraft migration, nor stentgraft kinking. Migration and kinking were probably underestimated in our study.
Limitations
This study has several limitations. It was a retrospective single centre study and the incidence of SLO was low (5.0%). As previous studies also did not work with many cases of SLO, this may explain the differences in significant predictors between studies. Coincidence may play a role. Large multicentre studies and meta-analyses are needed to overcome this problem.
Conclusion
After EVAR for infrarenal abdominal aortic and iliac artery aneurysms, the incidence of SLO was low (5.0%).
Occlusion was unilateral in all patients. Median time to occlusion was 3.3 months and most of the patients presented signs of lower limb ischemia. Incorrect movements of the hip joint may elicit SLO but further investigation is needed. Diagnosis of SLO was mostly made by CT angiography in combination with duplex examination. Surgical treatment options included thrombectomy and femoro-femoral crossover bypass. Endovascular treatment options consisted of additional BMS or covered stentgraft deployment.
Logistic regression analysis identified 2 predictors for SLO: symptomatic aneurysm (P .015, OR 4.62, 95% CI 1.35-15.86) and length of the infrarenal AAA (P .021, OR 1.31, 95% CI 1.04-1.64). Further research is necessary to pool all predictors and to assess the clinical impact of different follow-up strategies for high-vs low-risk patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
